Introduction
In order to use the high genetic potential of modern broiler strains, nutrient requirements, and nutritional management of poultry have changed (Ravindran, 2013a). Feed represents the most significant component of poultry production costs. Poultry diets are primarily based on corn and soy. The continuous rise of corn and soy prices worldwide generated a growing use of alternative cereals in broiler diets, such as wheat and barley. However, the cell wall of these cereals are rich in insoluble and soluble polysaccharides other than starch. These complex non-starch polysaccharides (NSP) are a heterogeneous group of compounds that include cellulose, pectins, β-glucans, pentosans, heteroxylans, and xyloglucan, which cannot be hydrolyzed by the endogenous digestive enzymes of humans and monogastric animals (Kumar et al., 2012).
Soluble NSP increase digest viscosity due to their high capacity to absorb water and gelatinize the intestinal tract contents of birds (Choct et al., 2010). This phenomenon disturbs digestion with consequences for feed energy, feed efficiency (Choct, 2006), and broiler performance (Chandra Shekhar et al., 2014). Hence, cereal grains high in non-starch polysaccharides (NSP) such as wheat (cellulose, and therefore largely insoluble) and barley (largely soluble) exhibit poor nutritional value (low apparent metabolizable energy, AME). Along with this, high dietary levels of NSP lead to high nitrogen and phosphorus excretion, which is a major environmental concern in densely populated regions.
Wheat and corn are rich in arabinoxylan, whereas barley has high levels of β-glucan (Knudsen, 2014). The anti-nutritional properties -expressed by water- soluble NSPs, such as pentosans (arabinoxylans) and β-glucans- can be reduced by including NSP degrading enzymes (xylanase-glucanase and cellulase) in broiler diets. The NSP degrading enzymes (NSPases) are known to increase the digestibility of raw materials for monogastrics and a combination of NSPases, amylases and proteases help digesting feed ingredients, thereby increasing the use of nutrients and the energy available for growth (Oluyinka et al., 2008) and production (Friesen et al., 1992; Zanella et al., 1999; Lázaro et al., 2003).
Although the effect of enzyme supplementation in wheat and barley-based diets for broilers has been studied for the last decades, controversial results have been published (Mathlouthi et al., 2002; 2003a; 2003b). Therefore, the objective of this study was to evaluate the effect of different levels (0, 0.025, and 0.05%) of two commercial enzyme mixtures (Kemin®, glucanase, amylase, xylanase, and bacillolysin; Kemin Industries, Inc., EMEA, Herentals, Belgium, and Rovabio®, glucanase and xylanase; Adisseo France S.A.S., Antony, France) on performance, blood constituents, and carcass characteristics of broiler chicks fed corn and wheat/ barley diets during a 6-wk rearing period. Birds were fed the experimental diets from hatch to 42 d of age.
Materials and methods
Ethical considerations
This experiment was conducted during July and August of 2013 in the poultry facility of the Islamic Azad University, in Rasht, Iran (37°14’37.15’’N and 49°34’39.42’’E). All procedures were approved by the Scientific Board of the Islamic Azad University and the study was conducted according to the International Guidelines for research involving animals (Directive 2010/63/EU).
Housing and management
A total of 300 male Ross 308 (Aviagen, Newbridge, Scotland, UK 35805) chicks were randomly allocated to 10 dietary treatments with 3 replicate pens/diet and 10 broiler chicks/pen. Birds were housed in an environmentally controlled system and growing conditions were similar in all treatments. Birds were vaccinated against infectious bronchitis virus (IBV), Newcastle disease virus (NDV) and Gumboro virus (GV) on varying time intervals. The infectious bronchitis virus strain H120 was provided at d 1 and d 19. The Newcastle disease virus strain Viscerotropic velogenic was provided at d 1, 11, and 19. The Gumboro virus was provided at d 14 and 25. Vaccines were prepared per vendor recommendation and were supplied via drinking water after a period of water removal for 3 h.
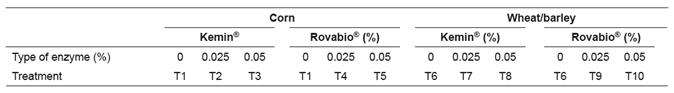
Dietary treatment
The ingredients and nutritional composition of the diets used are presented in Table 1.
The two basal diets used were corn-based and wheat/barley-based. These diets were formulated to be isoenergetic and isonitrogenous and met or exceeded Ross 308 catalogue recommendations (Aviagen, 2007). Starter diets were offered from d 1 to 14, grower diets from d 15 to 28 and the finisher diets from d 29 to 42. The experimental design was completely randomized in a 2×2×2 factorial arrangement with the two basal diets (corn-based or wheat/barley-based), two commercial enzymes (Kemin ® or Rovabio ® ) and two enzyme levels (0.025 or 0.05%). Thus, broilers were assigned to 10 dietary treatments (Table 2): Two control diets [i.e. without any enzyme supplementation (T1, diet based on corn without enzyme and T6, diet based on wheat-barely without enzyme)], a corn-based diet in which 0.025% Kemin ® was added (T2), a corn-based diet in which 0.05% Kemin ® was added (T3), a corn-based diet in which 0.025% Rovabio ® was added (T4), a corn- based diet in which 0.05% Rovabio ® was added (T5), a wheat/barley-based diet in which 0.025% Kemin ® was added (T7), a wheat/barley-based diet in which 0.05% Kemin ® was added (T8), a wheat/barley-based diet in which 0.025% Rovabio ® was added (T9), a wheat/barley-based diet in which 0.05% Rovabio ® was added (T10).
Table 1 Feed ingredients and nutrient analysis of diets used during the experiment.

1 Vitamin A: 5000 IU/g; Vitamin D3: 500 IU/g; Vitamin E: 3 mg/g; Vitamin K3: 1.5 mg/g; Vitamin B2: 1 mg/g. 2 Calcium pantothenate: 4 mg/g; Niacin: 15 mg/g; Vitamin B6: 13 mg/g; Cu: 3 mg/g; Zn: 15 mg/g; Mn: 20 mg/g; Fe: 10 mg/g; K: 0.3 mg/g.
Enzyme mixtures
The exogenous enzymes mixtures used were the two commercial multi-enzyme preparations, Kemin® (Kemin Industries, Inc., EMEA, Herentals, Belgium) and Rovabio®(Adisseo France S.A.S., Antony, France). The Kemin® mixture includes five active substances: Endo-1,3(4)-ß-glucanase, endo-1,4-ß-glucanase (cellulase), α-amylase, endo-1,4- ß-xylanase and bacillolysin (protease) with a guaranteed minimum enzyme activity of 2,350, 180,00, 400, 35,000, and 1,700 U/g, respectively. The Rovabio® mixture includes endo-1,3(4)-ß-glucanase (30,000 U/g) and endo-1,4-ß-xylanase (22,000 U/g) produced by a non- genetically modified strain of Penicillium funiculosum Pf 8/403 (International Mycological Institute, under the number IMI 378536).
Performance traits
Feed intake (FI, g/bird/period) and body weight gain (BWG, g/bird/period) were recorded for the period at the beginning of the experiment (d 1) until the end of the starter period (14th d of age), the grower period (15th-28th d of age) and finisher period (29th-42nd d of age). Feed efficiency (referred to as feed conversion ratio -FCR) was calculated by dividing feed intake by body weight gain. On the final day of the experiment, at 42 d-of-age, one bird from each replicate (three from each treatment) was randomly selected and slaughtered, plucked and eviscerated. Economically valuable carcass and gastrointestinal segments were removed and weighted. Organ weights were used to calculate percent of total body mass associated with each organ. Liver, pancreas, gizzard, and abdominal fat were removed to obtain the eviscerated carcass. The pectoralis major and peroneous longus muscles were excised from breast and drumstick for analysis.
Sampling and blood parameters
At the end of the experiment, at 42 d-of-age, 1 bird/replicate, totaling 3 birds/treatment, was randomly selected for blood collection. Blood samples (1.5 mL/bird) were collected from the wing vein using a 2 mL syringe and left to stand at 30 °C to allow clotting and clot retraction. The serum that remained after clotting was centrifuged at 3000 rpm for 10 min at room temperature. Samples were chilled until analyzes.
Blood parameters analyzed in serum were glucose (GLU), total triglycerides (TG), total cholesterol (TC), very low density lipoprotein (VLDL), high density lipoprotein (HDL), total protein (TP), albumin (Alb), uric acid (UAc) and alkaline phosphatase (ALP). The concentrations for these parameters were determined by routine methods using commercial laboratory kits (Pars Azmoon Co., Tehran, Iran), according to the manufacturer’s instructions.
The GLU was measured by a glucose-oxidase photometric assay, based on the combined action of glucose oxidase (GOD) and peroxidase (POD; COD- POD assay; Barham and Trinder, 1972). In this assay glucose is oxidized to gluconic acid and hydrogen peroxide in the presence of glucose oxidase. Hydrogen peroxide reacts with phenol and 4-aminoantipyrine in the presence of peroxidase to form a quinoneimine dye (Trinder’s reaction). Intensity of the pink color formed is proportional to glucose concentration.
The levels of serum TC, HDL, VLDL, and TG were determined using enzymatic methods (Teif AzmoonPars, Co., Tehran, Iran). The colorimetric determination of cholesterol in blood serum samples by the CHOD-PAP assay involved the hydrolysis of cholesterol esters to free cholesterol by cholesterol ester hydrolase. The free cholesterol produced is oxidized by cholesterol oxidase to cholest-4-in-3-one with the simultaneous production of hydrogen peroxide, which oxidatively couples with 4-aminoantipyrine and phenol in the presence of peroxidase to yield a chromogen with maximum absorption at 500 nm (Allain et al., 1974). High-density lipoprotein (HDL) and low-density lipoprotein (LDL) was measured directly after VLDL and/or LDL precipitation by the action of a precipitating reagent (heparin, for VLDL determination, and polyethylene glycol (PEG), for HDL determination) on the serum. When heparin was used as the precipitation reagent all the supernatant contained VLDL and HDL, which were measured by cholesterol CHOD/ PAP method. Low-density lipoprotein was equal to the difference between total cholesterol (TC) and cholesterol in the supernatant. When PEG was used as the precipitation reagent, all the VLDL and the LDL were precipitated. The HDL remained in the supernatant and was then assayed as a sample for cholesterol (CHOD/ PAP method).
Plasma triglycerides (TG) were measured using a series of coupled reactions in which triglycerides are hydrolyzed by lipase, and the released glycerol is assayed in a reaction catalyzed by glycerol kinase and L-alpha-glycerol-phosphate oxidase in a system that generates hydrogen peroxide. The hydrogen peroxide is monitored in the presence of horseradish peroxidase with 3,5-dichloro-2-hydroxybenzenesulfonic acid/4- aminophenazone as the chromogenic system (Fossati and Prencipe, 1982).
Blood serum albumin (Alb) was determined based on the bromocresol green method (Doumas et al., 1971), whilst UAc was determined by enzymatic methods using the uricase-TOOS method (Kayamori et al., 1997), TP was assayed by the Biuret method (Gornall et al., 1949). The serum activity of ALP was determined enzymatically using commercial kits (Teif Azmoon Pars, Co., Tehran, Iran) according to the method described by Bessey et al. (1946).
Statistical analysis
Data were analyzed by a General Linear Model using a 2×2×2 factorial arrangement (Statistical Package for the Social Sciences, SPSS, 1997; Base 7.5 for Windows. Chicago, IL). The statistical model used was as follows:
Y ijk 1= μ+a i +b j +c k +ab ij +ac ik +bc jk +abc ij k+e ijkl ,
In which the value of each observation (Y) relies on the population effects average (μ) plus the diet effect (α i , basal diet; corn or wheat and barley-based), the type of enzyme effect (β j , commercial feed enzyme type; Kemin® or Rovabio®), the level of enzyme effect (c k , commercial feed enzyme level; 0.025 or 0.05%), their interactions [(aβ) ij , (αχ) ik , (βχ) jk , (αβχ) ijk ], and the experimental error (ε). The Duncan post hoc test was used if the initial test result was significant at p ≤ 0.05.
Results
Feed efficiency
Results of feed intake (FI), body weight gain (BWG), and feed conversion ratio (FCR) are summarized in Tables 3, 4, and 5, respectively.
Concerning birds fed corn-based diet supplemented with 0.025 and 0.05% Kemin® or 0.025 and 0.05% Rovabio® enzymes, the results showed that during starting and growing periods FI was significantly lower (p<0.05) than in birds of the corn-fed control group. This was in contrast with the significantly (p<0.05) increased FI of birds fed wheat/barley- based diet supplemented with 0.05 Kemin® or 0.05% Rovabio® enzyme mixtures (Table 3).
The increase in FI observed in birds fed wheat/ barley-based diet supplemented with 0.05 of Kemin® or 0.05% of Rovabio® enzyme mixtures was accompanied by an increase in BWG (p<0.05), while with corn diets the lower FI also resulted in lower BWG, which was particularly evident during the starter period (Table 4).
During the starter and grower periods, the FCRs for the wheat/barley-based diets supplemented with 0.05% enzyme mixture were significantly lower (p<0.05) than for the wheat/barley control diet. For birds fed corn-based diets supplemented with NSPases, the FCRs were significantly higher (p<0.05) than in the corn-based control diet during the first 14 d period (Table 5). Nevertheless, when compared with the corn-based control diet, the differences in the FI and the BWG observed in birds fed corn-based diets supplemented with commercial enzymes disappeared with age, as did the differences in the FCR. These results suggested a small bio-efficacy of exogenous xylanases and glucanases in corn diets during the earlier stages of bird’s life.
Table 3 Effects of supplementing corn and a wheat/barley-based diet with enzyme mixtures (Kemin® and Rovabio®, at two dose levels each) on feed intake (FI; g/chick/period; mean ± sem).
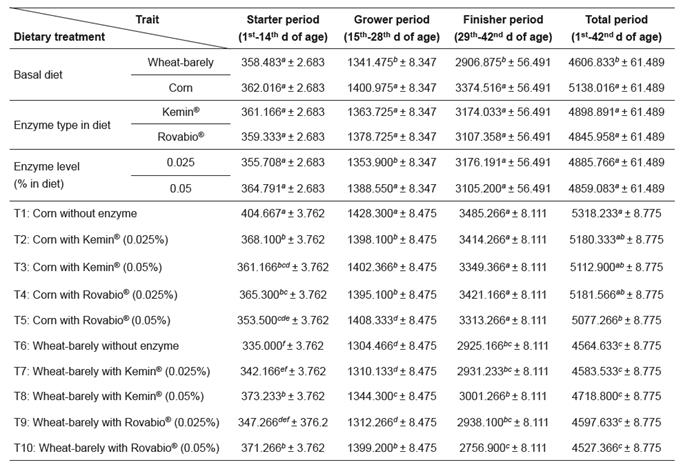
Growth period values (within columns) with different superscripts letters ( a, b, c, d, e, f ) differ significantly (p<0.05).
The birds fed corn-based diets showed the highest BWG throughout the experiment (Table 4). In wheat/barley-based diets, supplementation levels of 0.05% Kemin® (T8) and both 0.025 and 0.05% Rovabio® significantly (p<0.05) improved the BWG when compared with the wheat/barley-control group (p<0.05). Furthermore, both 0.025 and 0.05% Kemin® supplemented chicks showed a BWG that was not different (p>0.05) from birds supplemented with 0.025 or 0.05% Rovabio®.
At d 42, results showed that birds fed corn-based diets with Kemin® and Rovabio® had no improvement in feed efficiency (Table 5). Indeed, FCR appeared to be increased, but the two enzyme mixtures did not cause significant changes (p>0.05). Overall, birds fed the wheat/barley-based diets showed greater FCR than the birds fed corn-based diets. Significant differences were observed for birds fed wheat/barley-based diets with 0.025% of Kemin® or Rovabio® when compared with corn-based diets. Birds fed wheat/ barley-based diets with 0.05% exogenous NSPases supplementation presented decreased FCR when compared with the wheat/barley-based control diet (Table 5).
Carcass characteristics
The economical characteristics of the carcass were not significantly affected by diet (p>0.05). Nevertheless, the best performance for the eviscerated ready-to-cook carcass was observed in birds fed corn- based diets supplemented with commercial enzyme mixtures (66%). Birds fed wheat/barley-based diet with 0.05% NSPases showed the highest relative weight of both breast (21%) and drumsticks (25%), which was equal to the results of birds fed corn-based diet without enzyme supplementation. Additionally, birds submitted to wheat/barley-based diets with 0.025 or 0.05% of Kemin® or Rovabio® mixtures showed the same relative weight of liver (3%) and bile (3%) than corn and wheat/barley-control diets. Regarding gizzard, birds submitted to wheat/barley-based diets with 0.025% Kemin® or Rovabio® presented the same relative weight as birds on the corn and wheat/barley- control diets (3%).
Table 4 Effects of supplementation of corn and a wheat/barley-based diets with enzyme mixtures (Kemin ® and Rovabio ® , at two dose levels each) on body weight gain (g/chick/period; mean ± sem).
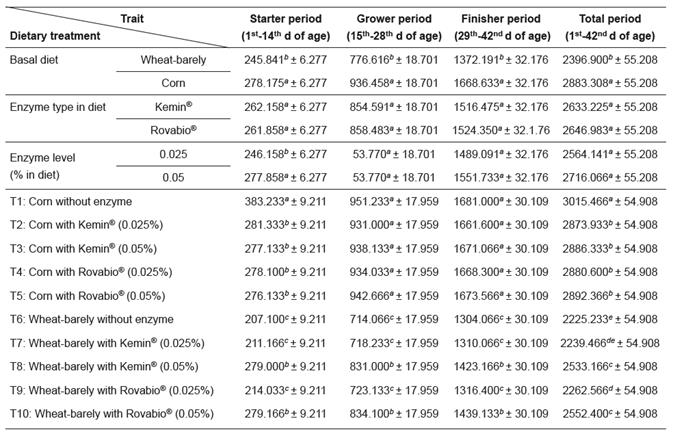
Growth period values (within columns) with different superscripts letters ( a, b, c, d, e, f ) differ significantly (p<0.05).
Blood parameters
The blood parameters determined in serum were not significantly affected by feed treatment except for GLU, VLDL and HDL (p>0.05; Tables 6 and 7). The highest and the lowest levels of serum GLU were observed in birds fed corn and wheat/barley- based diets without exogenous enzymes. Despite not significant (p>0.05), the inclusion of NSPases in corn and wheat/barley-based diets resulted, respectively, in low and high GLU mean values. Yet, serum GLU concentrations were lower in wheat/barley-based diets supplemented with 0.025 Kemin® or 0.025% Rovabio® (Table 6). In the groups submitted to these treatments, both serum VLDL and HDL levels did not differ from controls (p>0.05), while all other feed treatments resulted in significant decrease of VLDL and increase of HDL concentrations when compared with controls (p<0.05; Table 6). When data were analyzed independently of enzyme type and level, serum concentration of TC was higher in birds fed wheat/barley-based diets (p<0.05).
The BWG, FCR, and serum blood VLDL and HDL levels were clearly affected (p<0.05) by the interaction between basal diet and enzyme type (Table 8).
Discussion
Even though the diets were formulated to be isoenergetic and isonitrogenous, birds fed corn-based diet consumed more feed over the entire experiment, grew better and had a lower feed conversion ratio (FCR) compared with wheat/barley-based diets. It appeared that the FCR for birds fed corn-based diets could not be further improved by supplementation with NPSases while BWG and the FCR in birds fed wheat/barley-based diets can be significantly improved with the incorporation of 0.05% mixture of exogenous xylanases and glucanases. If 0.05% exogenous NSP-degrading enzymes are added to the wheat/barley-based diets, the final FCR were similar to those observed in birds fed corn. This result suggests that the digestibility of wheat/barley-based poultry diets can be improved by adding exogenous enzymes. Our results are consistent with previous findings (Mathlouthi et al., 2003a; 2003b; Shakouri et al., 2009). In a study conducted by Kocher et al. (2015), an enzyme product containing protease and xylanase activities was included in wheat-soybean meal diets fed to broilers from 1 to 42 d of age. The enzyme product improved average daily gain and feed conversion ratio, especially in birds up to 21 d of age. The simultaneous inclusions of phytase with α-galactosidases, protease, β-glucanase, and xylanase in corn-, barley-, or wheat-based broiler diets has additive effects in nutritionally marginal broiler diets. It appears that the activity of one type of feed enzyme may be facilitated by the other, possibly in a reciprocal fashion, by providing greater substrate access, and also by reducing the anti-nutritive effects of the substrates (NSP and phytate) on nutrient utilization (Ravindran, 2013b).
Table 5 Effects of supplementation of corn and a wheat/barley-based diet with enzyme mixtures (Kemin® and Rovabio®, at two dose levels each) on feed conversion ratio (FCR; g/chick/period; mean ± sem).
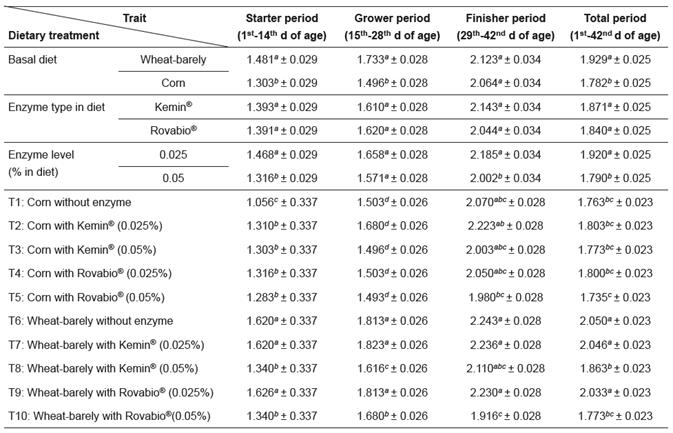
Growth period values (within columns) with different superscripts letters ( a, b, c, d, e, f ) differ significantly (p<0.05).
The magnitude of response in supplemented corn- based diets is generally lower than would be expected for diets based on wheat (Rosen, 2002). The main reason for this difference is the considerable different concentration of NSPs in wheat and barley compared with corn. Poor nutritional value of the latter two has been recognized for decades due to the presence of arabinoxylans in wheat and arabinoxylans and β-glucans in barley (Bedford, 1995; Saulnier et al., 1995). The mechanisms involved in poor digestibility are not fully understood but it is recognized that viscosity reduces the intestinal passage rate (van der Klis et al., 1993), reduces the diffusion of digestive enzymes and stimulates bacterial proliferation in the small intestine (Choct, 2006; Rodriguez et al., 2012). Moreover, the water-holding capacity brought about by soluble NSP markedly influenced voluntary feed intake. The breakdown of these NSPs via enzyme addition contributes to decrease viscosity of the gut contents (Bedford and Classen, 1992), improving feed intake and efficiency of nutrient utilization (Mathlouthi et al., 2002; Choct et al., 2004; Shakouri et al., 2009; Bedford and Partridge, 2010; Rodriguez et al., 2012).
Table 6 Effects of supplementation of corn and a wheat/barley-based diets with enzyme mixtures (Kemin® and Rovabio®, at two dose levels each) on the blood parameters of broilers (mean ± sem).
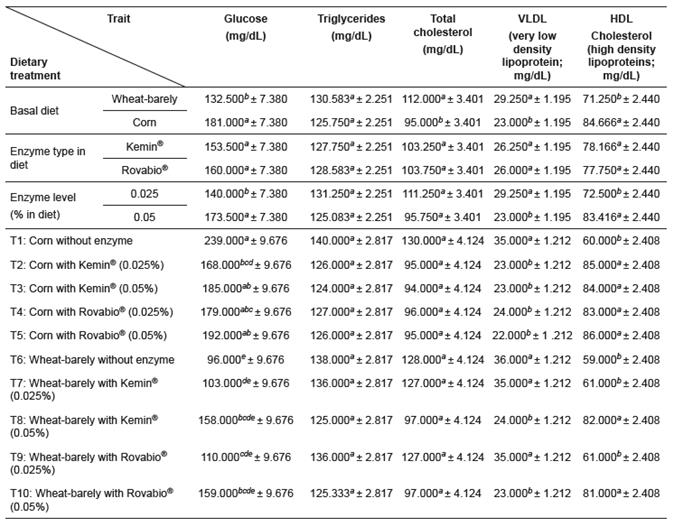
Growth period values (within columns) with different superscripts letters ( a, b, c, d, e, f ) differ significantly (p<0.05).
Plasma GLU, protein and lipid levels reflect the rate of digestion. Starch is the major dietary carbohydrate in poultry diets. Fecal starch digestibility (% of dry matter) in cereal grains is almost total (Yutste et al., 1991), but the dynamics of starch digestion can affect plasma glucose levels, which may have consequences for protein utilization (Weurding et al., 2003). Despite differences between cultivars (Mollah et al., 1983; del Alamo et al., 2009), starch digestion coefficients of wheat (93.8%) are lower than those of barley (98.3%), and corn (97.4%; Weurding et al., 2001). In the results presented here, VLDL decreased and HDL increased when 0.05% enzymes were included in the wheat/ barley-based diet. Serum protein levels, on the other hand, were not affected. The variations observed in serum GLU concentrations found in birds fed corn- based diets supplemented with enzymes seems to be due to the lower FI in the finisher period.
Table 7 Effects of supplementation of corn and a wheat/barley-based diets with enzyme mixtures (Kemin ® and Rovabio ® , at two dose levels each) on the blood parameters of broilers (mean ± sem).
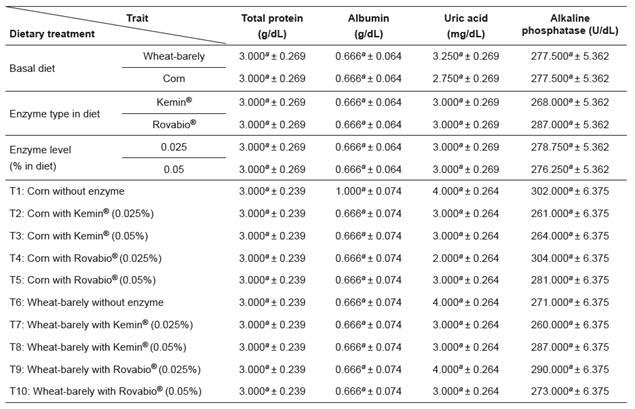
Growth period values (within columns) with different superscripts letters ( a, b, c, d, e, f ) differ significantly (p<0.05).
Table 8 Effect of basal diet (corn or wheat/barley-based), enzyme type (Kemin® or Rovabio®), and enzyme level (0.025 or 0.05%) on feed intake (FI), body weight gain (BWG), feed conversion ratio (FCR), glucose (GLU), very low density lipoprotein (VLDL), and high density lipoprotein (HDL).
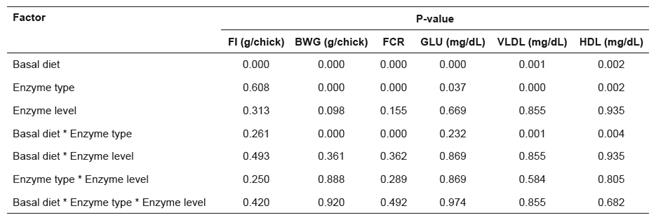
Viscous NSP can modify bacterial activity decreasing fat digestibility (Smits and Annison, 1996). Protein digestibility is also altered by increased intestinal viscosity (Wang et al. 1992). In this study we could not find significant effects of diets and supplements on the lipid and protein profiles in plasma. Additionally, no statistical significant changes were found for TG, TC, TP, Alb, UAc, and ALP plasmatic concentrations between dietary treatments.
Rodrıguez et al. (2012) found that a wheat/ barley-based diet supplemented with xylanase plus β-glucanase significantly improved the apparent digestibility of dietary fat in broiler chickens. In our work, the statistical changes found for blood VLDL and HDL profiles in birds fed enzyme-supplemented diets indicate that the absorption of these metabolites in the intestine is modulated by exogenous enzymes possibly in virtue of intestinal microflora.
It is commonly accepted that fat deposition is positively correlated with VLDL, HDL, and cholesterol blood levels, and that fat deposition depends mainly on available VLDL transported triglycerides. In our study, looking towards the overall changes in the serum VLDL and HDL concentrations in birds fed enzymes, it can be concluded that enzymes cause a lower availability of lipids for fat deposition, even though HLDL levels are higher than those of control diets. Since these kinetic parameters are nearly equal, it is possible that also abdominal fat deposition in the carcass was not different.
In this study, it was shown that body weight gain in birds fed wheat/barley-based diets can be improved by supplementation with 0.05% NSP enzyme mixtures (Kemin® and Rovabio®) such that birds reached the same FCR as birds fed supplemented and non- supplemented corn-based diets. Moreover, these birds maintained good carcass characteristics, supporting the use of exogenous NPSases enzymes in wheat/ barley-based diets.
In conclusion, the results showed the beneficial effect of NSPase supplementation on diets based on a mixture of wheat and barley, rich in NSP and a high fiber content, which could translate into economic benefits.