Introduction
Lernaea cyprinacea (Crustacea, Lernaeidae) is an ectoparasite, which has been reported in Africa, central Asia, southwest Siberia, Europe, Japan and Israel (Plaul et al., 2010). Originally, L. cyprinacea was not present in South America, but it has been introduced to many countries by translocation of cyprinids (Piasecki et al., 2004). Currently, L. cyprinacea is a common parasite infecting aquaculture species in Brazil, and is also very common in wild fish in main drainage basins throughout the country (Piasecki et al., 2004). Reports of lernaeid parasites infecting freshwater fish in Argentina are very scarce (Plaul et al., 2010); the first record dates from 1993, parasitizing Odontesthes bonariensis captured in San Roque Reservoir, Córdoba (Mancini et al., 2008b).
L. cyprinacea has great morphological plasticity depending on environmental characteristics, host species, and even the insertion site on the host (Moreno et al., 1986). It has a high pathogenic potential and is rapidly disseminated, posing a serious health threat to the fish farming industry. It produces skin lesions in fishes, generating direct damage and facilitating the occurrence of secondary infections (Fischer et al., 2003). The present study reports the presence of L. cyprinacea parasitizing Rhamdia quelen (Pisces, Heptapteridae) under captivity in the Santa Fe province. A detailed description and previous records in the region, are also provided.
Materials and Methods
Bioassay
In May 2014, a bioassay was performed with thirty samples of Rhamdia quelen (“bagre sapo”) in the Chronobiology Laboratory of the, Veterinary Sciences School at, Universidad Nacional del Litoral, Esperanza (Santa Fe, Argentina). Approval was obtained from the CAES-FCV-UNL 268 protocol, following recommendations from the National Research Council (2011). Average length of fishes was 12.461 cm (± 0.280) and average weight was 30.88 g (± 11.72). One month before the bioassay with R. quelen and in order to cycle the water of two aquariums, two samples of Prochilodus lineatus (Pisces, Prochilodontidae) were incorporated in each aquarium together with a sponge filter from ponds stabilized with fishes captured in the Salado River (Esperanza, Santa Fe). Their faeces provided the substrate for the growth of the bacteria supplied by the filter, thus securing the biological cycle of nitrogen to develop in the culture system. Prior to incorporation of R. quelen, the P. lineatus samples were removed and returned to their original ponds. Male and female specimens of R. quelen from the city of Paraná (Entre Ríos) were distributed in 100 L ponds, three specimens per pond, and kept under isolation conditions at constant temperature.
Separation and identification of ectoparasites
To extract crustaceans, fish were subjected to immersion for 10-30 minutes with 1% sodium chloride. Parasites that did not detach on their own were removed with dissecting forceps. Once removed, the affected area was treated with a mild disinfectant.
Ectoparasites were preserved in 70% ethanol for subsequent identification in the Laboratory of Natural Sciences Department, School of Humanities and Sciences (UNL, Santa Fe). The taxonomic determination was based on published references (Yamaguti, 1963; Demaree, 1967; Moreno et al., 1986; Thatcher, 2006). Measurements are expressed in mm.
Results
A detailed description of L. cyprinacea is presented, including the corresponding measurements (Figure 1, Table 1). Representative samples were deposited in the Florentino Ameghino Provincial Museum of Natural Sciences (Santa Fe), under N° MFA-ZI 579.
Lernaeidae Family
Lernaea cyprinacea (Linneaus, 1758)
Description: General morphology, post metamorphic females: elongated body segmented into cephalothorax, neck and genito-abdomen. Total Length (TL) cephalothorax 0.965 (0.594-1.752; N=9), TL neck 8.527 (6.528-11.184; N=10), TL genito- abdomen 1.022 (0.600-1.656; N=10), TL body 10.666 (8.181-12.810; N=9).
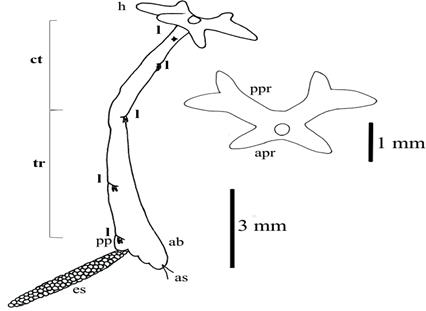
Figure 1 Lernaea cyprinacea (to scale, modified according to Demaree, 1967). (A) Side view: ab=abdomen; as= anal setae; ct= cephalothorax; es= egg sac; h= head; pp= pregenital prominence; l= legs; tr=trunk. (B) Detail of cephalic structures: apr= anterior protuberance; ppr= posterior protuberance.
Cephalothorax composed of head, mouth structure, four cylindrical projections, lobes or horns and first pair of legs. Anterior lobes are simple and smaller [L 0.980 (0.696-1.368; N=11) and Maximum Width (MW) 0.533 (0.306-0.861; N=9)]. Posterior lobes are larger, with a smaller ramification each [L 1.899 (1.445- 2.267; N=11), MW 0.565 (0.297-0.720; N=6), TL of the smaller ramification 0.499 (0.346-0.792; N=6) and TL of the larger ramification 0.794 (0.574-0.999; N=6)]. Mouth structure with bi-segmented maxillipeds; the basal segment is longer and presents one horn, while the terminal segment presents five unequal horns.
First pair of three-segmented antennae or antennules, second pair of bi-segmented antennae, with a horn in the second segment and a stout terminal claw. TL 0.307 (0.240-0.405; N=10) and MW 0.249 (0.217-0.316; N=10). Swimming legs, typical of copepods, reduced in size.
Slim, cylindrical, long neck, gradually enlarging towards the rear end, TL 8.527 (6.528-11.184; N=10) and MW 1.305 (1.008-1.488; N=10), in which 2 to 5 biramous legs are arranged, which are reduced in size, typical of copepods: Distance (D) between 1st and 2nd is 1.185 (0.720-2.496; N=8); D between 2nd and 3rd is 2.337 (1.440-2.928; N=8); D between 3rd and 4th is 3.056 (2.424-3.600; N=9); and D between 4th and 5th is 2.179 (1.008-4.488; N=10).
Cylindrical conical genito-abdomen, TL 1.022 (0.600-1.656; N=10) and MW 0.913 (0.600-1.108; N=10), with 2 terminal setae. Gonopores with a pair of egg sacs with rows 3-4-serial eggs, always with and odd ending, TL 4.088 (2.740-5.090; N=13) and MW 0.381 (0.310-0.576; N=14). Eggs, D 0.124 (0.089-0.264; N=10).
Infestation location: on the sides of the body and at the insertion of the fins.
Prevalence: 53 %
Site: Laboratory of Chronobiology FCV-UNL, Esperanza (Santa Fe), (31º26´32´´ S, 60º 56´28´´ W).
Table 1 Previous records of hosts for Lernaea cyprinacea in Argentina.
| Province | Location (original names in Spanish) | Hosts | References |
| Buenos Aires | Four lakes without name, Bahía Blanca (38°64´S, 62°16´W) | Rhamdia sapo (= Rhamdia quelen) | Vanotti & Tanzola, 2005 |
| Lagoon without name, zBahía Blanca (38°44´S, 62°15´W) | Rhamdia quelen | Mancini et al., 2008b | |
| Arroyo Napostá Grande, Bahía Blanca (38°48´S, 62°14´W) | Oligosarcus jenynsii | Garibotti & Guagliardo, 2004; Mancini et al., 2008b | |
| Lagoon without name, Monte Hermoso (38°59´S, 61°15´W) | Oligosarcus jenynsii | Mancini et al., 2008b | |
| Río Luján y valle de inundación, Tigre (34°26´S, 58°32´W) | Astyanax bimaculatus Odontesthes bonariensis | Plaul et al., 2010 | |
| Río de La Plata, Punta Lara (34°49´S, 57°59´W) | Cyprinus carpio | Plaul et al., 2010 | |
| Laguna del Burro, Río Salado (35°41´S, 57°55´W) | Rhamdia quelen | Plaul et al., 2010 | |
| Lagoon without name, La Plata (34°55´S, 57°57´W) | Carassius auratus Corydoras paleatus Hypostomus plecostomus | Plaul et al., 2010 | |
| Córdoba | Embalse La Viña (31°17´S, 65°01´W) | Odontesthes bonariensis | Mancini et al., 2008a |
| Río de los Sauces, Nono (31°47´S, 65°01´W) | Odontesthes bonariensis | Mancini et al., 2008b | |
| Arroyo, Tanti (31°21´S, 64°35´W) | Astyanax sp. Jenynsia sp. | Mancini et al., 2008b | |
| Embalse Río Tercero (32°14´S, 64°25´W) | Cyphocharax voga Odontesthes bonariensis Oligosarcus jenynsii Oncorhynchus mykiss Rhamdia quelen | Mancini & Grosman, 1998; Mancini et al., 2008b | |
| Embalse Los Molinos (31°50´S, 64°30´W) | Odontesthes bonariensis | Mancini et al., 2008b | |
| Embalse Piedras Moras (32°10´S, 64°14´W) | Astyanax eigenmanniorum Bryconamericus iheringii Odontesthes bonariensis Oligosarcus jenynsii | Mancini et al., 2008b | |
| Córdoba | Urban lake, General Levalle (34°00´S, 63°55´W) | Odontesthes bonariensis | Mancini et al., 2008b |
| In aquarius / pisciculture, Río Cuarto (33°06´S, 64°20´W) | Botia macracantha Carassius auratus Cyprinus carpio Poecilia sp. Xiphophorus helleri | Mancini et al., 2008b | |
| Lagoon without name, Ucacha (33°02´S, 63°30´W) | Cyphocharax voga Cyprinus carpio Odontesthes bonariensis Oligosarcus jenynsii Rhamdia quelen | Mancini et al., 2008b | |
| Lago San Roque (31°22´S, 6°27´W) | Odonthestes bonariensis | Mancini et al., 2008b | |
| Río Cosquín (31°18’ S; 64°27’W) | Astyanax hermosus | Ramallo & Terán, 2014 | |
| Corrientes | Río Paraná, Itá Ibaté (27°26´S, 57°20´W) | Prochilodus lineatus | Roux et al., 2000 |
| La Rioja | Dique de Olta (30°38´S, 66°16´W) | Odontesthes bonariensis Odontesthes jenynsii | Mancini et al., 2008b |
| Mendoza | Río Atuel, Embalse Valle Grande (34°53´S, 68°40´W) | Odontesthes bonariensis | Mancini et al., 2008b |
| Río Atuel, Embalse El Nihuil (35°04´S, 68°44´W) | Odontesthes bonariensis Oncorhynchus mykiss Percichthys trucha | Mancini et al., 2008b; Plaul et al., 2010 | |
| Río Negro | Río Negro, Isla Choele-Choel (39°22´S, 65°43´W) | Cheirodon interruptus | Plaul et al., 2010 |
| San Luis | Pisciculture and Embalse La Florida (33°06´S, 66°00´W) | Odontesthes bonariensis Oncorhynchus mykiss | Mancini et al., 2008b |
| Embalse San Felipe, Renca (32°49´S, 65°28´W) | Odontesthes bonariensis | Mancini et al., 2008b | |
| Embalse Río del Rosario, La Toma (33°02´S, 65°39´W) | Rhamdia quelen | Mancini et al., 2008b | |
| Santa Fe | Hatchery conditions, Esperanza (31°26´S, 60°56´W) | Rhamdia quelen | This work |
Discussion
The morphological characteristics observed here are typical of L. cyprinacea (Demaree, 1967; Thatcher, 2006). There are precedents of learneaosis associated with R. quelen and other hosts, both in wild and hatchery fish species from Argentina (Table 1). However, even though R. quelen had already been reported as host of L. cyprinacea, there were no records from the province of Santa Fe. Therefore, this paper contributes to building knowledge about its distribution in the region.
Lernaea cyprinacea was introduced into South America in the beginning of the 20th century via importation of the common carp, Cyprinus carpio (Piasecki et al., 2004). Whether accidental or deliberate, the introduction of exotic species is one of the major reasons for the loss of biological diversity, habitat alteration, and overexploitation of natural resources. About 40% of aquatic species extinctions is caused by introduced species (Agostinho et al., 2007).
Exotic species can have a negative impact by transmitting diseases or etiological agents into native communities. Therefore, colonization of exotic pathogens in aquatic systems constitutes a permanent threat to the integrity of natural environments (Agostinho et al., 2007). It is suspected that this ectoparasite has colonized native fish species and is dangerously invasive when environmental conditions are favorable, as reported in this work. Further research efforts are needed to elucidate the ecological role currently played by L. cyprinacea in this region.














