Introduction
Production of monosex fish is highly desirable for many species of commercial interest, such as Oreochromis and some salmonids, mainly due to size differences between sexes at maturity or to undesired reproduction during the production cycle (Dunhan, 2004). Monosex fish populations are typically obtained by sex reversal, which can be induced by temperature (Baroiller et al., 1999) or by hormonal administration. Hormones can be applied via the diet, by bath immersion, and injections or implants (Pandian, 2013). Dietary supplementation with hormones is a low-cost method for sex reversal, and is widely used in species with a labile period (period in which sex cells are totipotential) after the first feeding (Pandian, 2013).
However, the use of hormones, such as steroids, is controversial for human and environmental health, and many European countries have banned it for farmed fish (Desprez et al., 2003; El-Sayed, 2006). Consequently, monosex populations may also be induced indirectly by the combined use of hormones and mating strategies. This two-stage method of sex reversal has been implemented for the commercial culture of fish, such as salmonids (Hunter et al., 1983; Feist et al., 1995), to avoid direct application of hormones to species raised for human consumption.
The stages to produce hormone-free female offspring involve using hormones to directly masculinize females, followed by performance tests to identify masculinized females, known as neomales.
Subsequent crosses between neomales and normal females produce offspring that are theoretically 100% female (Donaldson and Devlin, 1996).
Fish farming of Rhamdia quelen, a gonochoristic fish species, has shown steady increases in the South of Brazil during recent years (Silveira et al., 2014), encouraging the development of new technologies to improve productivity (Poli et al., 2015; Garcia et al., 2017). In this species, both sexes present similar growth rates in the early stages from larval-fry to juvenile. However, at the time of harvest, females are larger than males (Fracalossi et al., 2004; Ghiraldelli et al., 2007), thus commercial productivity could be increased by using monosex cultivation of females.
As R. quelen larvae are capable of digesting artificial diets before yolk consumption (Silveira et al., 2013), dietary administration of hormones is a viable method to reverse sex in this species. Here we report on the masculinization of R. quelen adding 17α-methyltestosterone hormone to the diet as a first step toward production of neomales that can be used to produce all-female offspring free of exogenous hormones.
Materials and Methods
Ethical considerations
The study was conducted at the Laboratory of Biology and Cultivation of Freshwater Fish (Departamento de Aquicultura, Universidade Federal de Santa Catarina, Brasil), using experimental procedures approved by the Animal Ethics Committee (Protocol PP00788 - Development of Farming Technology to Native Fish) for the period of October, 2013 to January, 2017. The fish were kept in appropriate conditions throughout the trial period and euthanized with 100 mg/L of eugenol (Iodontosul® Eugenol, Porto Alegre, RS, Brazil) during 5 minutes prior to gonads removal.
Fish
R. quelen larvae were obtained by artificial breeding of wild fish captured in the Uruguay River Basin. A voucher specimen of the species was deposited in the museum at the State University of Londrina (MZUEL 10549), Brazil.
Spawning was induced according to Woynarovich and Horvath (1980), using a pool containing equal volumes of semen from two males and equal volumes of eggs from two females. Fertilized eggs were incubated in a conical cylinder incubator connected to a controlled water recirculation system that supplied water at 24.8 (± 0.35) °C.
Masculinizing test
R. quelen larvae at 2 days post-hatching (DPH), i.e., at the beginning of exogenous feeding, were randomly distributed in circular tanks (60 L) at a density of 6 larvae/L. The tanks were connected to a controlled water recirculation system with 800% replacement rate per day, and the following characteristics (mean ± SD): temperature: 24.80 ± 0.85 °C; pH: 7.77 ± 0.15; electrical conductivity: 242.63 ± 27.69 mS/ cm; dissolved oxygen: 7.54 ± 0.52 mg/L; un-ionized ammonia: 0.17 ± 0.09 mg/L; nitrite: 0.11 ± 0.09 mg/L; total alkalinity: 31.50 ± 5.08 mg/L CaCO3; and total hardness: 152.50 ± 23.95 mg/L CaCO3.
For 21 days, R. quelen larvae were fed to satiation five times per day with artificial diet supplemented with 17α-methyltestosterone (MT) (Sigma, St Louis, MO, USA) at 0 (control), 60, 80, or 100 mg/kg of feed, with three replicates per treatment group.
The MT was incorporated into commercial feed (Guabi® Pirá alevinos 55, Campinas, SP, Brazil) using ethanol evaporation (Shelton et al., 1981). Feed was sieved (0-250, 250-650, and 650-850 µm), individually packed, and refrigerated in preparation to be offered in the first (0-250 µm), second (250-650 µm), and third weeks (650-850 µm) of the masculinizing period. Feed remnants and feces were removed from the experimental units through bottom siphoning prior to the first feeding of each day.
Following the masculinizing period, the water volume of each tank was increased by 40 L to begin the growth phase, during which all fish received a commercial diet free of hormones (Guabi® Pirá 40, Campinas, SP, Brazil) until 150 DPH.
Histological procedures
The effects of the hormone on gonadal differentiation in R. quelen were assessed using histological techniques. Thirty fish from each masculinizing treatment were assessed at 150 DPH. After gonadal removal, the middle third of each gonad was fixed in Karnovsky solution (paraformaldehyde 2.0%, 2.0% glutaraldehyde, and phosphate buffer 0.1 M, pH 7.4) for histological preparation: paraffin embedding, microtome (serial sections 5.0 µm), staining, and slide mounting. Sections of 5.0 µm were stained with hematoxylin/eosin (HE) or Schiff’s periodic acid + iron hematoxylin + Metanil Yellow (MY) (Quintero-Hunter et al., 1991), and observed with a light microscope (LED Leica® DM 3000, Bannockburn, IL, USA).
For those fish with gonads that were too small to separate, the tissue around the most probable location of the gonads was removed and fixed for histological analysis. Ovaries and testes were classified according to Brown-Peterson et al. (2011) and Quagio- Grassiotto et al. (2013).
Statistical analysis
Sex frequencies in the masculinizing test were analyzed by the Fisher exact test at 5.0% significance (Zar, 2010), testing the ratio between males and genotypic females (female with normal gonads + intersex females + females with masculinized gonads) for each masculinizing dose and the control. This test detects whether there is a difference from the balanced 1:1 ratio and the ratio of normal females and neomales (females with masculinized gonads) in each masculinizing treatment.
To assess the effect of the hormone, a one-tailed test was used to check whether the proportion of masculinized females, the intersex fish, and females with female gonads was higher in the masculinizing treatments than in the control. Fish with undifferentiated gonads were excluded from all statistical analyses.
Results
At 150 DPH, fish presented similar growth in all treatments (p>0.05), with mean (± SD) body weight of 10.09 (± 1.70) g and total length of 8.47 (± 4.39) cm.
After histological analysis, gonads were classified as undifferentiated (the presence of the primordial cells did not allow sex identification), female gonads, male gonads, intersex gonads (female and male gonadal tissues were present simultaneously) and masculinized gonads (ovarian lamellae containing exclusively male germ cells characterized the female masculinized gonads of R. quelen neomales).
Histological analysis of gonads
In the undifferentiated gonads, primordial germ cells (PGC) were observed, distributed separately between cells of connective tissue and somatic cells. The primordial germ cells (PGC) presented an elliptical-rounded shape, with less basophils and a bulky nucleus, with some blood vessels (BV) observed nearby (Figure 1).
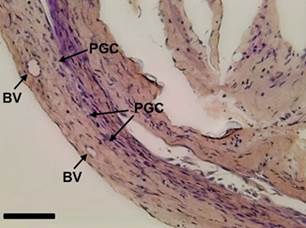
Figure 1 Longitudinal section of an undifferentiated gonad of Rhamdia quelen showing the blood vessels (BV) and the primordial germ cells (PGC) in fish at 150 days post-hatching. Staining: Schiff’s periodic acid + iron hematoxylin + Metanil Yellow (MY). Bar: 20 µm.
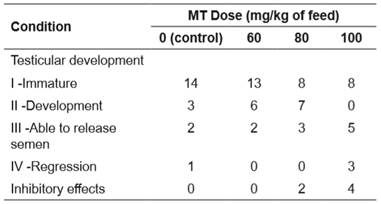
The gonads of males presented four stages of testicular development: I - immature, II - development, III - able to release semen, and IV - regression (Table 1). Males that showed inhibitory effects on gonadal development were not classified.
Table 1 Number of male Rhamdia quelen (fed diets supplemented with 0, 60, 80, or 100 mg of 17α-methyltestosterone (MT) per kg of feed) and testicular development.
In immature testes, only spermatogonia cords (SC) were observed. However, in some testes the cellular organization of the spermatic duct (COSD) was noted (Figure 2a).
In the development phase, groups of spermatogonia (SG), spermatocytes (SC), and spermatids (ST) were observed in the spermatocysts, which indicated the beginning of spermatogenesis. Some males presented spermatozoa in the lumen of the seminiferous tubules and in the spermatic ducts. The maturation of germ cells occurred from the finger-like projections into the spermatic duct, with the presence of a continuous epithelium along the testicles (Figure 2b).
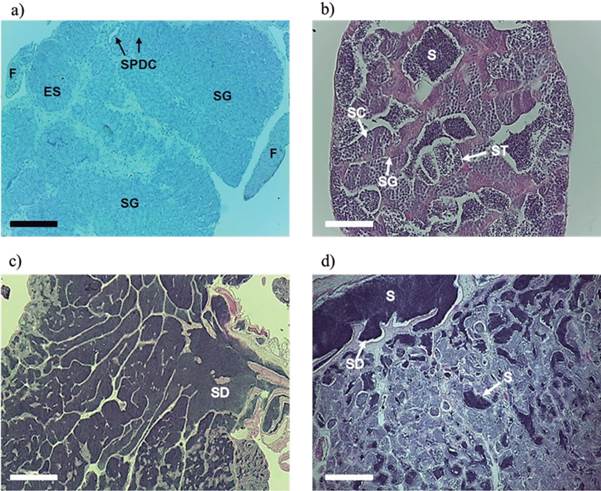
Figure 2 Sections of Rhamdia quelen testes at 150 days post-hatching. (a) Longitudinal section of the immature stage: basophilic areas (purple color) with the cords of spermatogonia (SG), beginning of cellular organization as a precursor of the spermatic duct (SPDC), and the finger-like (F) projections. (b) Cross-section of the developmental phase: spermatogonia (SG), spermatocytes (SC), and spermatids (ST) grouped in spermatocysts, and the presence of sperm (S). (c) Longitudinal section of testis able to release semen: continuous germinal epithelium in the periphery and discontinuous near the spermatic duct (SD), with a large number of sperm present in the lumen of the tubules and sperm ducts. (d) Longitudinal section of testicular regression: sperm (S) residual in the spermatic duct (SD) and the lumen of the seminiferous tubules. Staining: hematoxylin/eosin (HE). Bars: 200 (a), 50 (b), 500 (c), and 200 µm (d).
In males able to release semen, all stages of spermatogenesis were present in spermatocysts (SG, SC, and ST), and the germinal epithelium was continuous on the periphery an discontinuous near the spermatic duct (SD). Sperm was present in the lumen of the seminiferous tubules and in the sperm ducts in large quantities (Figure 2c).
Males in regression were characterized by depleted testes with residual spermatozoa in the lumen of seminiferous tubules and/or spermatic ducts. A simultaneous regeneration of the germinal epithelium was noted, with proliferation of spermatogonia and spermatocysts without spermatozoa release (Figure 2d).
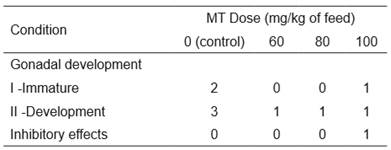
Histological analysis of females showed fish in only two stages of gonadal development: I - immature and II - development (Table 2).
Table 2 Number of Rhamdia quelen females (fed diets supplemented with 0, 60, 80, or 100 mg of 17α-methyltestosterone (MT) per kg of feed) at various stages of gonadal development and those that presented inhibitory effects.
The immature phase was characterized by the presence of oogonia (OO) and oocytes in primary growth (OPG) (pre-vitellogenic oocytes), and by the proximity to the ovarian lamellae (Figure 3a). In the development phase oocytes at several stages of primary growth were present, and also oocytes at the final stage of secondary growth (OSG) and some atretic oocytes (AO) were observed (Figure 3b). This phase was characterized by early vitellogenesis, in which oocytes at various stages (alveolar cortical) and oogonia of secondary growth were present, mainly on the lamellae base.
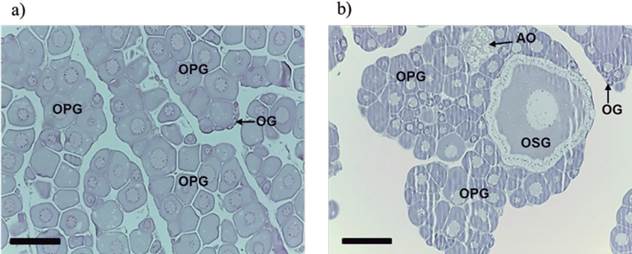
Figure 3 Longitudinal sections of ovaries of Rhamdia quelen females. (a) Longitudinal section of the immature stage: oocytes in primary growth (OPG), oogonia (OG). (b) Longitudinal section of the development phase: oogonia (OG), oocytes in primary growth (OPG), atretic oocytes (AO), and oocyte in secondary growth (OSG). Fish at 150 days post-hatching. Staining: hematoxylin/eosin (HE). Bars: 200 µm.
Histomorphological effects of 17α-methyltestosterone in gonads
The MT hormone produced important histomorphological effects in both sexes, resulting in genotypic females with masculinized gonads (neomales) or with intersex gonads. Higher doses inhibited development of both sexes. None of the fish in the control group presented such changes.
Intersex fish were observed in all groups treated with MT. Intersexual gonads presented ovigerous lamellae containing the male line germ cells together with primary growth oocytes and areas with cellular reabsorption and reorganization (Figures 4a and 4b).
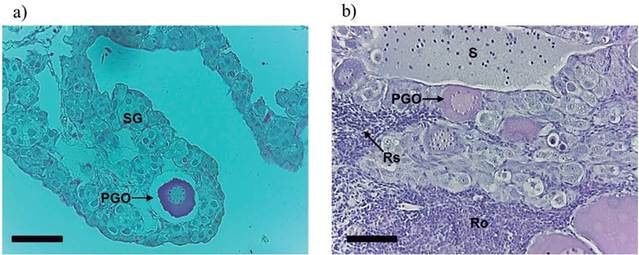
Figure 4 Longitudinal section of intersex gonads of Rhamdia quelen. (a) Interlamellar space with spermatogonia (SG) and primary growth oocyte (PGO). (b) Interlamellar space with spermatozoa (S), cellular reabsorption (Rs), and reorganization (Ro) areas and primary growth oocyte (PGO). Staining: hematoxylin/eosin (HE). Fish at 150 days post-hatching. Bars: 50 µm.
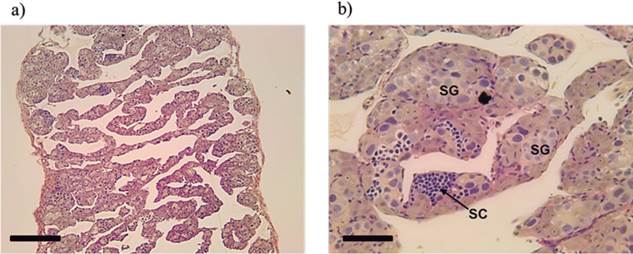
Figure 5 Longitudinal section of masculinized gonads Rhamdia quelen (neomales) (a) Lamellae exclusively containing ovarian male germline cells. (b) Details of spermatogonia (SG) and spermatocytes (SC) present in the lamellae. Staining: Schiff’s periodic acid + iron hematoxylin + Metanil Yellow (MY). Fish at 150 days post-hatching. Bars: 200 (a) and 20 µm (b).
In neomales, gonads with ovarian lamellae containing only the male lineage cells were observed (Figures 5a and 5b).
One female that received 100 mg MT/kg feed treatment showed areas with greater presence of atretic oocytes, areas of reabsorption and cell reorganization similar to ovarian regression, and cytoplasmic breakdown. This suggested an inhibitory effect on gonadal development of this dose (Figure 6a). Two males that received 80 mg MT/kg feed and four that received 100 mg MT/kg feed showed anomalous development of gonads, presenting areas with cells at the very early stages of development and areas where the lumen of the seminiferous tubules and sperm ducts were filled with sperm (Figure 6b).
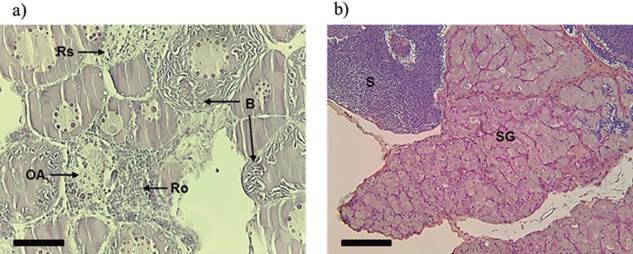
Figure 6 Longitudinal sections showing inhibitory effects on gonadal development in Rhamdia quelen caused by masculinizing hormone 17α-methyltestosterone. (a) Ovary showing reabsorption areas (Rs) and reorganization (Ro) cells, the presence of oocyte atresia (OA), and breakdown (B) of cytoplasm oocyte. (b) Testis showing areas at different stages of development: spermatogonia (SG) and spermatozoa (S). Staining: hematoxylin/eosin (HE) (a) and Schiff’s periodic acid + iron hematoxylin + Metanil Yellow (MY) (b). Fish at 150 days post-hatching. Bars: 50 µm.
Masculinizing test
Significant deviation (p<0.05) was observed in the sex ratio in favor of males, both in the control and in groups treated with MT (Table 3). Irrespective of the masculinizing treatment, genotypic females (gonads female + intersex gonads + gonads masculinized) occurred at low frequency (≤ 20%), limiting the number of fish susceptible to sex reversal. However, MT significantly influenced (p<0.05) gonadal differentiation of genotypic females at 60 and 80 mg MT/kg diet (Table 3) compared to the control, with sex reversal occurring at all tested doses.
The percentage of neomales produced from genotypic females (gonads female + intersex gonads + gonads masculinized) that could be masculinized was 50, 40, and 20% from the lowest to the highest MT doses, respectively. However, no significant differences were observed between doses (Table 3).
Discussion
Wild populations of R. quelen present a balanced (1:1) sex ratio (Baldisserotto et al., 2010), however, a higher frequency of males (72% of the analyzed gonads) were found, a condition also recorded by Sulis-Costa et al. (2013) with the same lineage. As the offspring used in this masculinizing trial presented a lower frequency of females, fewer animals were available for the sex reversal process.
Table 3 Classification of gonads of Rhamdia quelen fed diets supplemented with 0, 60, 80, or 100 mg of 17α-methyltestosterone (MT) per kg for 21 days. Fish at 150 days post-hatching.
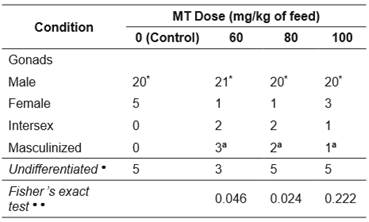
* Significant deviation (p<0.05) in favor of males (male gonads), from an expected ratio of 1:1 between males (male gonads) and genotypic females (gonads female + intersex gonads + gonads masculinized).
Superscript letters that differ indicate that the proportion of masculinized females was significantly different (p>0.05; Fisher’s exact test) between doses.
● Fish with undifferentiated gonads were excluded from all statistical analyses.
● ● Probability of Fisher’s exact test of whether the proportion of intersex + masculinized gonads was higher than female gonads in the control (α = 0.05).
Nevertheless, masculinization occurred at all MT doses; with 60 and 80 mg MT/kg feed causing greater influence on gonadal differentiation and producing a larger number of intersex and neomale gonads.
In Oreochromis niloticus, oral administration of 17α-methyltestosterone stimulates sexual maturation from 45 days after hatching (Zanardi et al., 2011). The different stages of gonadal development for R. quelen males (immature to regression) and females (immature and developing) during the same growth period showed that sexual maturation occurred earlier in males, a characteristic condition of species in captivity (Fracalossi et al., 2004; Ghiraldelli et al., 2007).
In undifferentiated R. quelen gonads, which represented 15% of total analyzed gonads, presence of primordial germ cells characterized the germinal epithelium that gives rise to ovarian lamellae or to the seminiferous tubules, delimited ovarian and testicular lumen (Grier and Lo Nostro, 2000; Mazzoni et al., 2010). The presence of fish with undifferentiated gonads at 150 DPH suggests that a longer period would be necessary to express the phenotypic sex of all fish. As fish with undifferentiated gonads also resulted in the control group; no relationship with MT doses was observed.
Genetic sex in fish is defined at the time of fertilization by the combination of the oocyte and sperm genes involved in the sexual determination, but sex phenotypical differentiation occurs later (Piferrer, 2001). At a particular developmental time, specific to each species, a chemical signal originating from a gene or set of genes is sent to the primordial germ cells, the embryological precursors of the ovary and testis, informing the direction in which they must develop (Martínez et al., 2014). In the timeframe prior to the signal, physiological sex can be altered if the animal ingest or absorb anabolic steroids, such as the 17α-methyltestosterone, that directs the expression of primordial germ cells towards a male or female phenotypic expression (Pandian, 2013).
Davis et al. (1990) showed that the effective time for sex reversal was 21 days after the first feeding for channel catfish Ictalurus punctatus. For Tachysurus fulvidraco, the period of sexual differentiation, knownas the labile period in which undifferentiated gonads respond to steroid action (Piferrer, 2001), occurs between 8 and 30 DPH, and is longer in females (Park et al., 2004). In Salvelinus fontinalis, the labile period occurs earlier and is longer compared to most salmonids, which typically exhibit this period between 18 and 28 DPH (Haffray et al., 2009).
As the labile period for R. quelen is unknown, a lower frequency of sex reversed fish could be expected, if among other factors, the period of diet supplemented with MT did not cover the crucial period. However, Amaral Junior et al. (2008) applied the direct sex reversal test at the same time as in this study and reported a 79% ratio of females.
The presence of fish with intersex gonads at all masculinizing doses may be an indication that administration of the hormone did not completely cover the labile period for R. quelen. Conversely, gonads could have been in the process of reversal, so a period longer than 150 DPH would be necessary to complete sex reversal. Intersex gonads can also result from suboptimal or very high MT doses. However, the presence of sterile individuals is usually observed at very high doses (Pandian and Sheela, 1995). Although gonadal intersex has been also observed in males exposed to MT (Kang et al., 2008), in this trial, R. quelen gonads showed that the intersexual tissue derived from female gonads was due to the presence of ovarian lamellae, a condition similar to that reported for Morone saxatilis (Schutz and Harrell, 1999).
Oral administration of MT induced masculinization in R. quelen with reversed fish presenting sizes and condition similar to normal fish at the time of gonad removal. Masculinized gonads showed ovarian lamellae filled with spermatogonia and spermatocytes, or presented exclusively male germline cells. The effectiveness of the tested doses, however, was lower than that found for other species of commercial interest, such as Oreochromis niloticus (Shelton et al., 1981). As fish were 150 DPH at the time of gonad removal, the final morphology of masculinized gonads could be different at the maturation stage, resembling gonads of true males able to release semen.Inhibitory effects on gonadal development may indicate that the MT doses used were very high for R. quelen; however, such changes were also recorded for other species (Ankley et al., 2001; Kang et al., 2008). Rivero-Wendt et al. (2013), using feed supplemented with 60 mg MT/kg feed for 28 days, recorded a marked decrease in spermatogenesis of Astyanax bimaculatus and an abnormal ovarian degeneration of Oreochromis niloticus.
Considering the effectiveness of the MT doses tested and the inhibitory effects on gonadal development caused by higher doses, 60 mg MT/ kg feed is recommended as optimal to masculinize female R. quelen. However, the feeding period of MT should be adjusted to increase the rates of sex reversal. Fish should also be analyzed over a period longer than 150 DPH to ensure that all offspring have completed sexual differentiation and can be properly identified.














