Introduction
Heart failure (HF) is a clinical syndrome characterized by typical signs and symptoms in addition to the objective evidence of a structural or functional abnormality of the heart 1. It is associated with significant morbidity and mortality, with one-year mortality rates of 7.2% and hospitalization rates of 31.9% in patients with chronic HF, and these outcomes increasing to 17.4% mortality and 43.9% hospitalization in patients with acute HF 2. Five-year survival is 56.7% (95% CI 54-59.4%) and 10-year survival is 34.9% (95% CI 24-46.8%) 3.
Traditionally, left ventricular ejection fraction (LVEF) has been the most commonly used classification method, due to its practical implications for determining an appropriate treatment strategy, although over the last few years the measurement of global longitudinal strain and myocardial contraction fraction have gained ground 4. The European Society of Cardiology classifies it in three categories according to LVEF: HF with preserved LVEF when it is equal to or greater than 50%, HR with mildly reduced LVEF if it is 40-49%, and HF with reduced LVEF (HF-rLVEF) when it is less than 40% 5.
Heart failure with preserved and mid-range LVEF has no specific evidenced-based treatment to date, and its treatment is aimed at managing risk factors and controlling comorbidities 1,2,5, while HF-rLVEF has clearly evidence-based treatments with an increasingly broad therapeutic arsenal, and currently even offers new treatment paradigms 6. Thus we, in this systematic review of the literature, deal with the current treatment of HF-rLVEF.
Data collection
The study design is a systematic review following the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines 7. The systematic search performed in drafting the "Clinical Practice Guidelines for the Prevention, Diagnosis, Treatment and Rehabilitation of Heart Failure in People Over the Age of 18, Classification B, C and D" 1 was updated.
Objective
The population, intervention, control, and outcome (PICO) questions were:
1. In patients over the age of 18 with HF-rLVEF, does pharmacological or interventional treatment or treatment with devices decrease the combined primary outcome - mortality and hospitalizations for any cause or for cardiovascular causes - compared with a placebo or active treatment or not using them?
Elegibility criteria
Studies were selected by two investigators, and differences were resolved by consensus. The included studies were randomized clinical trials which compared the use of pharmacological or interventional treatment or devices in HF-rLVEF (defined as LVEF less than 40%) with placebo, active treatment or, in the case of devices, with not using them, and which reported relevant combined outcomes: mortality from any cause or cardiovascular mortality, hospitalizations for any cause or for HF. The baseline characteristics and the outcomes evaluated were extracted from each study.
Search strategy
A systematic search was performed in the following bibliographic databases: PubMed, Embase, CENTRAL, DARE, Epistemonikos, SciELO, LILACS and OpenGrey, along with the web pages of clinical trial registries such as Clinical-Trials. The manual search included international cardiology congress pages, web sites and cardiology electronic media like Twitter from institutions such as the European Society of Cardiology, the American Heart Association, the Canadian Cardiovascular Society and the American College of Cardiology, as well as primary studies included in these societies' clinical practice guidelines and those of the National Institute for Health Care Excellence (NICE) and meta-analyses. The update included studies in English from January 2014 to December 2020.
A general and manual search was performed including the following terms: "heart failure" or "congestive heart failure" or "left ventricular dysfunction" or "reduced ejection fraction" or "systolic heart failure" and "treatment" "management" or "drug therapy" and randomized controlled trial or controlled clinical trial or randomized or randomly or trial".
Statistical analysis
In line with the individual characteristics of the included studies, the heterogeneity among them and the fact that they act as complementary therapies, the study group consensus was to perform a systematic review rather than a meta-analysis. The individual papers were analyzed according to their baseline characteristics and defined outcomes, which are presented in tables for comparison. The risk of bias was assessed using the strategy proposed by Cochrane, and the fragility index was calculated for new studies along with a comparison with the classic studies using the outcome of mortality from any cause. The fragility index is defined as the minimum number of events that would have to change to a "non-event" status in order to change from a significant to a nonsignificant result, as evidence of the robustness of the studies, since many of these depend on a difference of three or fewer events; its interpretation shows that the smaller the index, the more fragile the result 8.
Results
The systematic and manual search recovered 1,424 results, of which only original clinical trials were analyzed, excluding publications analyzing individual subgroups or secondary outcomes or subrogates. New studies of angiotensin converting enzyme (ACE) inhibitors or angiotensin II receptor blockers (ARBs) or beta blockers were not recovered, nor were new studies of mineralocorticoid receptor antagonists (MRAs) or ivabradine.
The risk of bias for the six new studies analyzed was considered to be low for all the evaluated aspects: generation of the allocation sequence, allocation concealment, blinding of participants and personnel, blinding of outcomes, incomplete outcomes and selective reporting (Table 1).
Table 1 Risk of bias in the clinical trials.
| Study | Random sequence generation | Allocation concealment | Blinding of participants and personnel | Blinding of outcome assessment | Incomplete outcome data | Selective reporting |
|---|---|---|---|---|---|---|
| PARADIGM HF | Low | Low | Low | Low | Low | Low |
| DAPA HF | Low | Low | Low | Low | Low | Low |
| VICTORIA | Low | Low | Low | Low | Low | Low |
| EMPEROR REDUCED | Low | Low | Low | Low | Low | Low |
| GALACTIC HF | Low | Low | Low | Low | Low | Low |
| SOLOIST WHF | Low | Low | Low | Low | Low | Low |
The fragility index shows robust results for sacubitril/ valsartan for both the primary combined outcome as well as for cardiovascular death, death from a cardiovascular source and hospitalization for HF. For dapagliflozin, the results are robust for the primary combined outcome and hospitalization for HF. The empagliflozin study showed robust results for the primary combined outcome and hospitalization for HF; for sotagliflozin it is robust for the primary outcome; and vericiguat and omecamtiv mecarbil had very low fragility indices, especially the latter one (Table 2).
Table 2 Fragility index for all the outcomes in the new trials included.
| Fragility index | PARADIGM HF Active treatment/ superiority LCZ696/ enalapril | DAPA HF Placebo/superiority Dapagliflozin/placebo | VICTORIA Placebo/superiority Vericiguat/placebo | EMPEROR R Placebo/superiority Empagliflozin/placebo | GALACTIC HF Placebo/superiority Omecamtiv/placebo | SOLOIST WHF Placebo/superiority Sotagliflozin/placebo |
|---|---|---|---|---|---|---|
| Primary outcome | 118 | 62 | 8 | 50 | 1 | 72 |
| Cardiovascular death | 66 | 5 | 0 | 0 | 0 | 0 |
| Total death | 49 | 8 | 0 | 0 | 0 | 0 |
| Hospitalization for heart failure | 54 | 43 | 0 | 50 | 0 | NA |
A comparative analysis of the classic and new studies was performed for the outcome of mortality (Table 3). The results showed estimates below 1 in almost all cases, except for valsartan, losartan and omecamtiv mecarbil. Those with a 95% CI above 1 were valsartan and candesartan in the CHARM Added trial, losartan in ELITE II and HEAAL, carvedilol in ANZ, nebivolol, digoxin, ivabradine vericiguat, empagliflozin, omecamtiv mecarbil and sotagliflozin. The fragility indices were robust for beta blockers, spironolactone and sacubitril/valsartan.
Table 3 Comparison of the fragility index of classic HF trials.
| Study | Total mortality | Estimated 95% CI | Fragility index |
|---|---|---|---|
| CONSENSUS | 1 year • 36 vs. 52% | RR 0.69 (0.52-0.91) | 7 |
| SOLVD T | 2 years • 35.1 vs. 39.7% | RR 0.84 (0.74-0.95) | 10 |
| Val-HeFT | 2 years • 19.7 vs. 19.4% | RR 1.02 (0.88-1.18) | 0 |
| CHARM Alternative | 3 years • 26.1 vs. 29.1% | aHR 0.83 (0.7-0.99) | 0 |
| CHARM Added | 3.5 years • 29.5 vs. 32.4% | HR 0.89 (0.77-1.02) | 0 |
| ELITE II | 1.5 years • 17.7 vs. 15.9 % | HR 1.13 (0.95-1.35) | 0 |
| HEAAL | 4.7 years • 33 vs. 34.8 % | HR 0.94 (0.84-1.04) | 0 |
| CIBIS II | 1.3 years • 12 vs. 17% | HR 0.66 (0.54-0.81) | 37 |
| MERIT HF | 1 year • 7.2 vs. 11% | RR 0.66 (0.53-0.81) | 34 |
| COPERNICUS | 10.4 m • 11.4 vs. 18.5% | RR 0.66 (0.53-0.81) | 30 |
| ANZ CARVEDILOL | 1.6 years • 9.7 vs. 12.5% | RR 0.76 (0.44-1.32) | 0 |
| COMET | 5 years • 34 vs. 40% | HR 0.83 (0.74-0.93) | 33 |
| SENIORS | 1.75 years • 15.8 vs. 18.1% | HR 0.88 (0.71-1.08) | 0 |
| RALES | 2 years • 35 vs. 46% | RR 0.70 (0.6-0.82) | 54 |
| EMPHASIS HF | 1.75 years • 12.5 vs. 15.5% | HR 0.76 (0.62-0.93) | 5 |
| DIG | 3 years • 34.8 vs. 35.1% | RR 0.99 (0.91-1.07) | 0 |
| A-HeFT | 10 m • 6.2 vs. 10.2% | HR 0.57 (no CI) RR 0.63 (0.41-0.96) | 3 |
| SHIFT | 2 years • 16 vs. 17% | HR 0.9 (0.8-1.02) | 0 |
| PARADIGM | 2.25 years • 17 vs. 19.8% | HR 0.84 (0.76-0.93) | 49 |
| DAPA HF | 18.2 m • 11.6 vs. 13.9% | HR 0.83 (0.71 to 0.97) | 8 |
| VICTORIA | 10.8 m • 20.3 vs. 21.2% | HR 0.95 (0.84-1.07) | 0 |
| EMPEROR R | 16 m • 13.4 vs. 14.2% | HR 0.92 (0.77 to 1.10) | 0 |
| GALACTIC HF | 21.8 m • 25.9 vs. 25.9% | HR 1.00 (0.92 to 1.09) | 0 |
| SOLOIST WHF | 9 m • 13.5 vs. 16.3% | HR 0.82 (0.59-1.14) | 0 |
Pharmacological treatment
Angiotensin converting enzyme (ACE) inhibitors
The ACE inhibitors decrease the concentration of angiotensin II and aldosterone, increase renin release and the concentration of angiotensin 9-12, decrease the plasma concentration of epinephrine, norepinephrine and vasopressin, and increase the production of bradykinin 10,13,14.
The CONSENSUS Trial 15 with enalapril was the first drug (1986) to show reduced mortality in HF, in patients in New York Heart Association (NYHA) class IV. Six-month mortality significantly reduced in the group receiving enalapril (44% vs. 26%). Since then, many studies have shown that RAAS inhibitors reduce morbidity and mortality in HF-rLVEF, with reductions in mortality from all causes ranging from 20-30% 16-20.
Angiotensin II receptor blockers (ARBs)
The clinical trial results indicate that ARBs are not superior to ACE inhibitors, but are an alternative for patients with intolerance. Clinical trials have evaluated the efficacy not only of losartan (OPTIME-ELITE II), but also of valsartan (Val-HeFT) and candesartan (CHARM) in patients with HF, in terms of cardiovascular morbidity and mortality 21-24. However, they have not been able to surpass the ACE inhibitors in consistently reducing morbidity and mortality; their main advantage lies in a better tolerance. Their use as an alternative treatment is based on the results of the CHARM-Alternative study in patients with symptomatic HF and LVEF <40% who were not taking ACE inhibitors due to intolerance. In these patients, the use of candesartan achieved a significant reduction in both mortality and the number of hospitalizations for HF 25.
Beta blockers
Beta blockers should be part of the treatment of all chronic, stable patients, and they should be started early after compensating patients with acute, newly diagnosed HF 1,5. They have proven benefits, regardless of the etiology of the HF. These include symptom relief and improved quality of life, reduced risk of hospitalization and incidence of sudden death, as well as improved survival. In addition, they have significant effects on reverse ventricular remodeling, with LVEF recovery 26-30. Beta blocker treatment should be used with caution in patients with borderline BP, recent inotrope treatment or NYHA functional class IV.
A recent meta-analysis raises questions about the effect of beta blockers on patients with HF and atrial fibrillation (AF), as they have a different impact in these patients than in patients with a sinus rhythm 31. The recent RATE AF clinical trial shows that digoxin, compared with bisoprolol, has similar effectivity, but with fewer adverse events 32.
Mineralocorticoid receptor antagonists (MRAs)
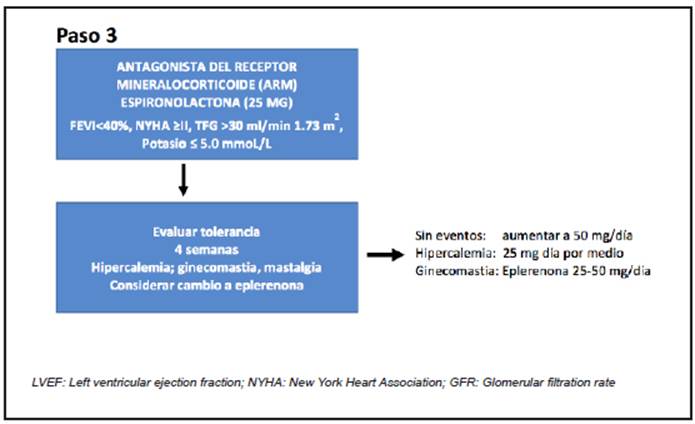
The MRAs (spironolactone and eplerenone) reduce the aldosterone escape phenomenon, contribute to the RAAS block 33-35 and reduce mortality by 15 to 30%. Furthermore, they decreased hospitalizations for HF by 15 to 40% in three randomized clinical trials (RALES, EMPHASIS, EPHESUS) 36-38, in patients with chronic HF-rLVEF, including patients who had had a myocardial infarction, excluding patients with a baseline serum creatinine greater than 2.5 mg/dL (or an estimated glomerular filtration rate [GFR] <30 mL/min/1.73 m2) or a serum potassium level >5.0 mEq/L.
Recent evidence, using indirect comparison, suggests greater survival benefits with a starting strategy using the three agents compared with conventional therapy 39. New drugs such as finerenone have shown benefits in a Phase 2 trial (ARTS-HF) and in type 2 diabetic patients with chronic kidney disease 40-41.
Angiotensin receptor-neprilysin inhibitors (ARNIs)
Initially, as a complement to the neurohormonal block, an attempt was made to raise natriuretic peptide levels with nesiritide and carperitide, with discouraging results 42-45. Subsequently, isolated neprilysin inhibition was attempted with racecadotril, candoxatrilat and ecadotril, which was unsuccessful, as their effect was rapidly lost 46. The next step was to use a new group known as vasopeptidase inhibitors which have a dual mechanism, inhibiting neutral endopeptidases (such as neprilysin) and ACE (omapatrilat). The OVERTURE trial showed noninferiority compared with enalapril, but a greater number of patients with angioedema [24 (0.8%) vs. 14 (0.5%)] 47, due to neprilysin degrading aminopeptidase P which, like ACE, degrades bradykinin.
Finally, sacubitril (a neprilysin inhibitor) was combined with an ARB-II (valsartan), which was tested in the PARADIGM HF trial 48 in patients with LVEF <35%, with proven tolerance to ACE inhibitors or ARBs, NYHA ≥II, elevated natriuretic peptides, systolic arterial pressure (SAP) > 100 mmHg, GFR > 30 mL/min/1.73m2 and potassium ≤5.2 mEq/L. Sacubitril/valsartan showed superiority in doses of 200 mg every 12 hours, compared with enalapril at 10 mg every 12 hours, in cardiovascular mortality, including sudden death and worsening HF (HR 0.8, CI 0.73-0.87) and hospitalizations for HF (HR 0.79, CI 0.71-0.89).
The PIONEER HF trial 49 proved the safety of beginning this drug in uncompensated HF in patients with LVEF <40%, BNP ≥400 ng/L or NT-proBNP ≥1,600 ng/L, no inotrope requirement in the last 24 hours, nor use of vasodilators, SAP <100 mmHg or increased diuretics in the last six hours. Sacubitril/valsartan showed superiority at a dose of 200 mg every 12 hours compared with enalapril at doses of 10 mg every 12 hours, with regard to decreased NT-ProBNP levels and a reduction in 30-day readmissions as a secondary outcome.
Subsequently, the TITRATION trial was performed to determine the safety of conservative (standard) titration or condensed (aggressive) titration of the dose, with good results in both arms 50. Similarly, the TRANSITION trial 51 showed that initiating sacubitril/valsartan before or after discharge did not affect the achievement of the target dose. Finally, the PROVE HF study 52 found a weak, but statistically significant, relationship between NT-ProBNP levels and the change in LVEF, since the bioavailability of valsartan in the ARNI is 50% greater than that of valsartan alone, which means that 400 mg of the combination contains ~203 mg of valsartan, equivalent to 320 of valsartan sold alone; in addition, it has proven cost-effectiveness (Tables 4-6) 19.
Table 4 Comparison of the new HF treatments. Baseline characteristics.
| Variable | PARADIGM HF Active treatment/superiority LCZ696/enalapril | DAPA HF Placebo/superiority DAPA/placebo | VICTORIA Placebo/superiority Vericiguat/placebo | EMPEROR R Placebo/superiority Empagliflozin/placebo | GALACTIC HF Placebo/superiority Omecamtiv/placebo | SOLOIST WHF Placebo/superiority Sotagliflozin/placebo |
|---|---|---|---|---|---|---|
| Group | ARNI | ISGLT2 | SGCE | ISGLT2 | CMA | ISGLT2 |
| Year | 2014 | 2019 | 2020 | 2020 | 2020 | 2020 |
| Median follow up | 27 months | 18 months | 11 months | 16 months | 21.8 months | 9 months |
| Sample size | 4,187/4,212 | 2,373/2,371 | 2,526/2,524 | 1,863/1,867 | 4,120/4,112 | 608/614 |
| Design | RCT | RCT | RCT | RCT | RCT | RCT |
| Age (years) | 63.8 ± 11.5/63.8±11.3 | 66.2±11/66.5±10.8 | 67.5±12.2/67.2±12.2 | 67.2±10.8/66.5±11.2 | 64.5±11.3/64.5±11.4 | 69 (63-76)/70 (64-76) |
| Average GFR | 68±20/68±20 | 66±19.6/65.5±19.3 | 61.3±27/61.7±27.3 | 61.8±21.7/62.2±21.5 | 58.7 (43.8-73.7)/58.8 (44.3-74.3) | 49.2 (39.5-61.2)/50.5 (40.5-64.6) |
| Ejection fraction (%) | 29.6±6.1/29.4±6.3 | 31.2±6.7/30.9±6.9 | 29±8.3/28.8±8.3 | 27.7±6.0/27.2±6.1 | 26.6±6.3/26.5±6.3 | 35 (28-47)/35 (28-45) |
| Ischemic (%) | 59.9/60.1 | 55.5/57.3 | 59.8/56.8 | 52.8/50.7 | 53.2/54 | |
| NYHA I/II/III/ | 4.3/71.6/23.1/0.8; 5/69.3/24.9/0.6 | 0/67.7/31.5/0.8; 0/67.4/31.7/1.0 | 0/58.6/40/1.4; 0.1/59.3/39.4/1.2 | 0/75.1/24.4/0.5; 0/75/24.4/0.6 | 0/53.3/43.7/3; 0/52.8/44.1/3 | Hospitalized + IV diuretic |
| Hypertension (%) | 70.9/70.5 | 79.3/79 | 72.4/72.3 | |||
| Diabetes (%) | 34.7/34.6 | 41.8/41.8 | 48.6/45.3 | 49.8/49.8 | 40.1/40.3 | Type 2 100% |
| Atrial fibrillation | 36.2/37.4 | 38.6/38 | 43.5/46.4 | 35.6/37.8 | 27.8/26.7 | |
| Beta blocker (%) | 93.1/92.9 | 96/96.2 | 93.2/93 | 94.7/94.7 | 94.2/94.4 | 92.8/91.4 |
| Aldosterone antagonist (%) | 54.2/57 | 71.5/70.6 | 69.3/71.4 | 70.1/72.6 | 77.6/77.8 | 66.3/62.7 |
| ACE inhibitor/ ARB (%) | 84.5/82.8 | 73.3/73.6 | 70.5/68.9 | 87/87 | 82/83 | |
| Sacubitril/valsartan (%) | 10.5/10.9 | 14.3/14.7 | 18.3/20.7 | 19.9/19 | 15.3/18.2 | |
| ICD | 14.9/14.7 | 26.2/26.1 | 27.6/27.9 | 31.0/31.8 | 32.2/31.3 | |
| CRT | 7/6.7 | 8/6.9 | 14.7/14.6 | 11.8/11.9 | 14.4/13.8 | |
| NT-proBNP pg/mL | 1,631/1,594 | 1,428/1,446 | 2,803.5/2,821 | 1,887/1,926 | 1,977/2,025 | 1,816/1,741 |
Table 5 Comparison of the new HF treatments. Outcomes.
| Outcome | PARADIGM HF Active treatment/superiority LCZ696/enalapril | DAPA HF Placebo/superiority DAPA/placebo | VICTORIA Placebo/superiority Vericiguat/placebo | EMPEROR R Placebo/superiority Empagliflozin/placebo | GALACTIC HF Placebo/superiority Omecamtiv/placebo | SOLOIST WHF Placebo/superiority Sotagliflozin/placebo |
|---|---|---|---|---|---|---|
| Primary combined (%, HR, events per 100 patient years) | 21.8/26.5 HR 0.80 (0.73-0.87) | 16.3/21.2 HR 0.74 (0.65-0.85) 11.6/15.6 | 35.5/38.5 HR 0.90 (0.82-0.98) 33.6/37.8 | 19.4/24.7 HR 0.75 ( 0.65-0.86) 15.8/21.0 | 37/39.1 HR 0.92 (0.86-0.99) 24.2/26.3 | 40.3/ 57.8 HR 0.67 (0.52-0.85) 51/76.3 |
| CV death | 13.3/16.5 HR 0.80 (0.71-0.89) | 9.6/11.5 HR 0.82 (0.69-0.98) 6.5/11.5 | 16.4/17.5 HR 0.93 (0.81-1.06) 12.9/13.9 | 10/10.8 HR 0.92 (0.75-1.12) 7.6/8.1 | 19.1/19.4 HR 1.01 (0.92-1.11) 10.9/10.8 | 8.4/ 9.5 HR 0-84 (0.58-1.22) 10.6/12.5 |
| First hospitalization for heart failure | 12.8/15.6 HR 0.79 (0.71-0.89) | 9.7/13.4 HR 0.70 (0.59-0.83) 6.9/9.8 | 27.4/29.6 HR 0.90 (0.81-1.0) 25.9/29.1 | 13.2/18.3 HR 0.69 (0.59-0.81) 10.7/15.5 | 27.7/28.7 HR 0.95 (0.87-1.03) 18/19.1 | |
| Total hospitalization | 39.7/43.5 HR 0.88 (0.82-0.94) | HR 0.85 (0.75-0.95) | 28.6/30.1 HR 0.93 (0.86-1) 18.7/20.3 | |||
| Total death | 17/19.8 HR 0.84 (0.76-0.93) | 11.6/13.9 HR 0.83 (0.71-0.97) 7.9/9.5 | 20.3/21.2 HR 0.95 (0.84-1.07) 16/16.9 | 13.4/14.2 HR 0.92 (0.77-1.10) 10.1/ 10.7 | 25.9/25.9 HR 1 (0.92-1.09) 14.4/14.4 | 10.7/ 12.4 HR 0.82 (0.59-1.14) 13.5/16.3 |
| KCCQ change | "-2.9±0.36/-4.63±0.36" HR 1.64 (0.63-2.65) | 6.1±18.6/3.3±19.2 HR 1.18 (1.11-1.26) | 5.8±0.4/4.1±0.4 HR 1.7 (0.5-3.0) | 5.8±0.3/6.3±0.3 -0.5 (-1.4 to 0.5) | 17.7/13.6 | |
| Kidney worsening | 2.2/2.6 HR 0.86 (0.65-1.13) | 1.2/1.6 HR 0.71 (0.44-1.16) 0.8/1.2 | 1.6/3.1 HR 0.50 (0.32-0.77) |
Table 6 Comparison of the new HF treatments. Adverse events.
| Adverse event | PARADIGM HF Active treatment/superiority LCZ696/enalapril | DAPA HF Placebo/superiority DAPA/placebo | VICTORIA Placebo/superiority Vericiguat/placebo | EMPEROR R Placebo/superiority Empagliflozin/placebo | GALACTIC HF Placebo/superiority Omecamtiv/placebo | SOLOIST WHF Placebo/superiority Sotagliflozin/placebo |
|---|---|---|---|---|---|---|
| Symptomatic hypotension (%, p) | 14/9.2 p<0.001 | 0.3/0.5 | 9.1/7.9 p=0.12 | 6/4.9 | ||
| Hypoglycemia | 1.5/0.3 | |||||
| Renal adverse event | 3.3/4.5 p<0.007 | 6.5/7.2 p=0.36 | 2.1/2.2 | |||
| Hypercalemia | 16.1/17.3 p=0.15 | 0.1/0.2 | 4.4/5.6 | |||
| Cough | 11.3/14.3 p<0.001 | 4.4/4.2 | ||||
| Angioedema | 0.4/0.2 | |||||
| Volume depletion | 7.5/6.8 P=0.4 | |||||
| Syncope | 4/3.5 p=0.30 | |||||
| Urinary tract infection | 4.8/5.1 | |||||
| Diarrhea | 6.1/3.4 | |||||
| Anemia | 7.6/5.7 | |||||
| Ischemic cardiac event | 4.9/4.6 | |||||
| Severe ventricular arrhythmia | 2.9/3.1 |
Sodium-glucose cotransporter-2 (SGLT-2) inhibitors
Beginning in 2016, with the publication of the EMPAREG-OUTCOME 53 study which found a reduction in hospitalization for HF in patients with diabetes mellitus (DM) receiving empagliflozin treatment, there has been a growing interest in the effect of this group of medications on HF treatment. The proposed mechanism of action of SGLT-2 inhibitors is, first of all, natriuresis and glucosuria which, in addition to decreasing preload and postload, decreases interstitial edema which is related to ventricular hypertrophy, although this effect is neutralized around week 12 of treatment 54. The second proposed effect is decreased BP, secondary to decreased sympathetic tone through direct inhibition of noradrenaline synthesis by blocking renal tyrosine hydroxylase 55. The third mechanism is decreased cardiomyocyte glucose consumption in favor of the use of fatty acids and ketones which, besides being more energy efficient, have the ability to decrease oxidative stress 56. Finally, although there are several ideas to explain improved contractility through calcium metabolism, direct blocking of the NHE1 receptor by SGLT-2 inhibitors and the subsequent reduction in intracellular sodium and calcium, as well as increased mitochondrial calcium, has the greatest evidence 56.
Their clinical effect has been proven in recent years in patients without DM; thus, the DEFINE HF 57 study found that patients with LVEF <40%, NYHA II-III, elevated natriuretic peptides and GFR ≥ 30mL/min/1.73m2 dapagliflozin at 10 mg/day was superior to placebo in decreasing the dual co-primary outcome which included reducing NT-proBNP levels by at least 20% and increasing the Kansas scale by ≥ 5 points (OR 1.8 CI 1.03-3.06), although there were no differences in the other co-primary outcome of the average NT-proBNP levels at 6-12 weeks.
Later, the DAPA-HF 58 trial showed that in patients with LVEF <40% and NYHA ≥II with elevated natriuretic peptides, despite being on optimal therapy, with GFR ≥ 30 mL/min/1.73m2, SAP > 95 mmHg, and without type 1 DM, dapagliflozin was superior at 10 mg/day compared with placebo in the combined outcome of worsening HF (unplanned hospitalization or an urgent visit to the emergency room for intravenous therapy) or cardiovascular death (HR 0.74 CI 0.65-0.85).
The EMPEROR REDUCED 59 study randomized patients with LVEF <30%, NYHA ≥II, and elevated natriuretic peptides (in cases with LVEF between 30 and 40%, hospitalizations for HF in the previous year or very high peptide levels were also required), despite being on optimal therapy, with a GFR ≥20 mL/min/1.73m2 and SAP >100 mmHg. In this study, empagliflozin proved to be superior at 10 mg/day compared with placebo in decreasing hospitalizations for HF or cardiovascular death (HR 0.75 95% CI 0.65-0.86).
Finally, the SOLOIST WHF study in type 2 diabetic patients with decompensated HF randomly assigned stabilized patients, before or within three days of discharge, to 200 mg of sotaglifozin (increasing to 400 mg, depending on side effects) versus placebo. After a median nine-month follow up, it significantly decreased the combined primary outcome (HR 0.67 95% CI 0.52-0.85). This study offers an interesting perspective by suggesting that early initiation of treatment has important effects and, furthermore, the effect is maintained in patients with an LVEF ≥50%, which suggests a promising effect for patients with HF with a slightly reduced and preserved fraction 60, as suggested by the results of the recent EMPEROR PRESERVED (N Engl J Med 2021 DOI: 10.1056/NEJMoa2107038/10.1056) study.
Although, given the consistency of the described evidence, it could be deduced that this is a group effect, the results of the VERTIS CV 61 trial raised doubts in this regard, since the use of ertugliflozin in patients with type 2 diabetes was unable to show superiority to the placebo, but did show non-inferiority, although the population of HF patients included did not exceed 25%. A subsequent report with the prespecified analysis of HF-related events suggested a reduction in hospitalization for HF (RR 0.70 95% CI 0.56-0.87) and the combined outcome of hospitalization for HF/cardiovascular death (RR 0.83 95% CI 0.72-0.96) 62.
Ivabradine
Ivabradine inhibits the sinus node's pacemaker activity by selectively blocking the hyperpolarization channels known as the "funny" (If) channels, decreasing the heart rate of patients with a sinus rhythm without affecting arterial pressure, cardiac contractility or intracardiac conduction 30. Among the studies found on this treatment is the initial background in the BEAUTIFUL 63 study on patients with coronary disease and ventricular dysfunction (LVEF <40%), which found no difference between ivabradine and placebo in the primary outcome (HR 1 95% CI 0.91-1.1) nor in the secondary outcomes which made up the combined primary outcome. However, in the subgroup analysis, in patients with a baseline heart rate ≥70 bpm, it reduced the incidence of hospitalization for myocardial infarction and coronary revascularization, although it did not decrease the primary objective nor overall mortality 64.
The SHIFT 65 study was conducted in patients with chronic HF and LVEF ≤35%, sinus rhythm and a resting heart rate ≥70 bpm. Ivabradine had an impact on the primary outcome of cardiovascular death or hospitalization for HF (HR 0.82 95% CI 0.75-0.90), as well as the outcomes related to HF (hospitalization for HF HR 0.74 95% CI 0.66-0.83; and death from HF HR 0.74 95% CI 0.58-0.94).
Patients treated with ivabradine have an average heart rate reduction of 8 bpm, while a meta-analysis of patients with HF-rLVEF with beta blockers showed a reduction of 12 bpm 66. Cullington et al. 67 showed that the proportion of patients for whom the addition of ivabradine would be indicated according to the SHIFT trial criteria dropped from 19.4% of the total at the initial visit to only 9% at the 12-month follow up, after adequately adjusting the beta blocker dose.
Hydralazine and isosorbide dinitrate
The evidence for the clinical usefulness of hydralazine and isosorbide dinitrate comes initially from the V-HeFT I trial, which compared three treatments: placebo, prazosin, or isosorbide dinitrate and hydralazine. After two years, there was a 34% reduction in the primary outcome of cumulative mortality in the hydralazine and isosorbide dinitrate group compared with placebo (p=0.028, 95% CI 4-54%) 68. Subsequently, the V-HeFT II study compared hydralazine combined with isosorbide dinitrate to enalapril; at two years, there was a significantly higher reduction in mortality from all causes in the enalapril group (ARR 6%, p= 0.016) and the differences in mortality between the two groups were not sustained at 2.5 years (ARR 5.4%, p=0.08), improved peak oxygen consumption and LVEF in the hydralazine-isosorbide dinitrate group compared with enalapril early on, as part of the secondary results 69.
The AHeFT study was carried out based on observations which suggested a greater benefit in African Americans. This study randomized 1,050 black patients with HF in functional class III/IV and ventricular dilation to receive a fixed dose of hydralazine and isosorbide dinitrate (n=518) compared to placebo (n=532) in addition to standard medical care. The study was ended early when it showed that hydralazine combined with isosorbide dinitrate resulted in significantly lower mortality compared with placebo (6.2% vs. 10.2%, p= 0.02). The outcome of side effects was also better in the intervention group, with a 43% reduction in death from any cause (HR 0.57; p=0.01), 33% reduction in the rate of first hospitalizations for HF (16.4% vs. 22.4%, p=0.01), and improved quality of life 70.
Vericiguat
Vericiguat is an oral, soluble guanylate cyclase stimulator which increases cyclic GMP activity, a second messenger involved in regulating cardio-renal and metabolic protective mechanisms. The SOCRATES-REDUCED study randomized 456 patients with HF with LVEF <45% and one recent decompensation episode (<4 weeks). A total of 377 patients finished treatment and there was no difference between the baseline and 12-week NT-proBNP levels between the group treated with vericiguat and the placebo group, with adequate tolerance and no differences in adverse events, with a linear relationship between the dose of vericiguat used and the degree of NT-proBNP reduction 71.
The VICTORIA study involved 5,050 patients with high-risk HF-rLVEF (40% in NYHA functional class III), severe impairment of systolic ventricular function (mean LVEF of 29%), with elevated natriuretic peptides and recent decompensation (80% within the last three months) to receive oral vericiguat with a target dose of 10 mg per day, compared with placebo plus guideline-directed treatment. The study showed a modest reduction in the primary outcome of cardiovascular death or first hospitalization for HF (35.5% vs. 38.5%, HR 0.9 [95% CI, 0.82-0.98, p=0.02]), derived mainly from the effect on hospitalizations for HF or any cause, with no impact on cardiovascular deaths or total deaths. Mean follow up was 10.8 months. Due to its vasodilating properties, there was symptomatic hypotension in 9.1% of the patients compared with 7.9% in the placebo group (p=0.12) and syncope in 4% compared with 3.5% in the placebo group (p=0.30) 72.
Omecamtiv mecarbil
Omecamtiv mecarbil is a selective cardiac myosin activator which increases contractility by binding to an allosteric site, for a faster transition from weak to strong binding, increasing the number of myosin heads bound to an actin filament, generating more strength. In addition, stabilization of the baseline status reduces ATP turnover in the absence of an interaction with actin, which improves energy efficiency.
The Phase 3 GALACTIC HF trial in 8,256 patients with chronic HF with LVEF ≤35% compared omecamtiv mecarbil with placebo and showed a reduction in the primary outcome of HF-related events or cardiovascular death (HR 0.92 95% CI 0.86-0.99), with no impact on cardiovascular mortality or quality of life assessed with the Kansas questionnaire. Despite its modest effect in the total group, the finding of the differential impact according to LVEF, with a median cut-off ≤ 28% generates an interesting hypothesis which should be tested in a subsequent clinical trial 73.
Digoxin
Digoxin, a sodium-potassium ATPase pump inhibitor 74 was evaluated in the DIG study in 6,800 patients with HF and LVEF <45%. The study showed no differences in mortality [34.8% vs. 35.1% (RR 0.99 95% CI 0.91-1.07, p=0.08)], but there was a reduction in total hospitalization and hospitalization for decompensated HF [26.8% vs. 34.7% (RR 0.72 95% CI 0.66-0.79, p=0.001)]. An analysis of secondary outcomes showed that digoxin had the tendency to reduce deaths attributable to worsening HF [RR 0.88 (95% CI, 0.77-1.01, p=0.06)] 75. The prespecified subgroup analysis of high-risk patients (NYHA III-IV, LVEF < 25%, cardiothoracic ratio > 55%) after two years of follow up showed a reduction in the outcome of hospitalization and death from any cause 76.
A Cochrane review showed that digoxin does not reduce mortality from all causes nor from HF, but it does reduce HF symptoms and readmissions by 32% [OR 0.68 (95% CI 0.61-0.75, p < 0.00001)]. The benefits were greater in patients with greater LVEF impairment (<25%) or NYHA functional class III/IV. A subgroup analysis by serum digoxin concentrations showed that patients with levels between 0.5 and 0.8 ng/mL had a 20% reduction in deaths from all causes (HR 0.8 [95% CI 0.68-0.94, p= 0.005]). There were more arrhythmia complications in patients with serum digoxin levels >1.2 ng/mL, especially in patients with kidney disease, who require close monitoring 77.
Diuretics
Their use is mainly based on small, controlled clinical trials in which they have proven to be well tolerated and to alleviate congestion, improving edemas and reducing body weight 78. Likewise, changes in neurohormonal parameters associated with congestion have been shown 63. Loop diuretics are preferred (furosemide, bumetanide, torsemide), although thiazides may be added in diuretic-resistant patients, as well as potassium-sparing diuretics 3,25. The main adverse events associated with their use are fluid and electrolyte disorders and renal dysfunction 79; the long-term effect on outcomes is unclear 80.
Studies of other treatment options for managing congestion, such as arginine vasopressin antagonists, have shown no impact on clinical outcomes, although they do increase urine volume and decrease weight 81,82.
Treatment of comorbidities and devices
Other types of therapies exist, including iron supplementation, management of atrial fibrillation, implantable automatic defibrillators, cardiac resynchronization, cardiac contractility modulation, mitral clips, CardioMEMS and ventricular assist devices which have proven beneficial in specific groups of patients, after optimizing what is considered to be standard treatment 83-129 (Annex 1).
Discussion
Given the current evidence, the papers published especially during the last year, the GPC recommended heart failure algorithm should be updated to introduce new therapies, considering the magnitude of the treatment benefit and that the effect could be considered to be separate from each of the medication groups, supported by the studies' subgroup analyses.
The consistency of studies with ARNI place it as the treatment of choice, replacing ACE inhibitors/ARBs, and reserving these for a few cases, specifically patients who do not tolerate ARNI treatment due to hypotension. Likewise, the SGLT-2 inhibitors, together with the already positioned beta blockers and MRAs complement this first-line therapy. The results with other new medications such as vericiguat and omecamtiv mecarbil are modest and they should be reserved for only a few cases which are not stabilized using standard treatment. The risk of bias was considered to be low in all the items.
The fragility index analysis comparing the classical strategies with the new ones showed robust results for beta blockers, spironolactone and sacubitril/valsartan, and weak results for ACE inhibitors, ARBs, vericiguat and omecamtiv mecarbil, although it should be noted that this may be affected by factors such as the expected effect size, the number of events and the study's power 130.
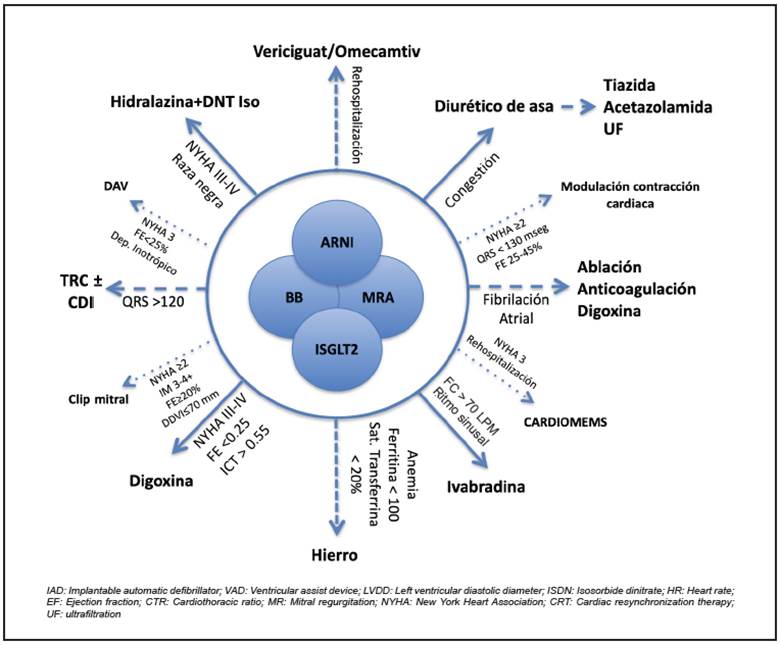
The proposal was designed as a decision circle with a central nucleus including what is considered to be the pillar of HF treatment (Figure 1), which is enlarged according to certain variables which may be considered or introduced into treatment, according to the clinical progress.
The central nucleus is organized hierarchically to highlight the fundamental pillars of treatment and provide a general recommendation for initiating and titrating doses according to monitoring and the patient's clinical progress, supported by the mentioned studies. The next orbit contains the interventions to be used after reaching the goals of the basic nucleus which contains four strategies, whose evidential strength is differentiated based on the support of designed clinical trials, the affected outcome and the variables to be considered for its use.
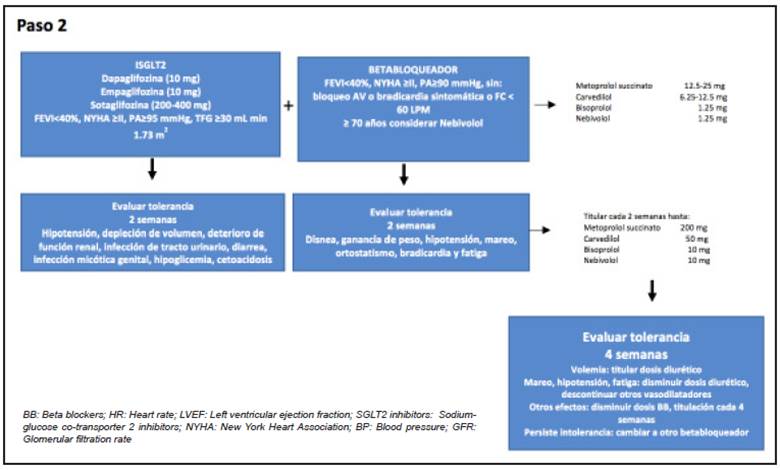
The suggestion is to begin in step one with ARNI (Figure 2); the only available one to date is the sacubitril/valsartan combination. In step 2, SGLT-2 inhibitors plus the beta blocker (Figure 3), and, finally, the MRA (Figure 4).
It is important to highlight that the algorithm is conceived in order to begin these treatments, considered to be pillars, early; and that the suggested titration may be modified according to the patient's clinical condition, although it is expected that the use of these recommendations will improve tolerance and adherence, in general, and that low doses of all the groups may be prescribed, to be subsequently increased according to the set sequence of steps 131.
Other therapies, such as cardiac rehabilitation, including an exercise program, vaccination and palliative care should be used concomitantly, ideally within a structured Heart Failure Clinic program. Patients who remain symptomatic despite the use of this strategy should be referred to a heart transplant group for assessment, especially if they are between 18 and 70 years old; have an NYHA functional status III or IV; a peak oxygen consumption (VO2 max) ≤ 14 mL/kg/minute or, with beta blockers, ≤ 12 mL/kg/minute, in patients < 50 years old add VO2 max ≤50% of the expected (predicted) value and in a submaximal test (RER <1.05) use the slope of the ventilatory equivalent of CO2 > 35 on the cardiopulmonary stress test; and are classified as high risk according to the Heart Failure Survival Score or have <80% expected one-year survival on the Seattle Heart Failure Model scale 132.











 text in
text in