Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Colombiana de Química
Print version ISSN 0120-2804
Rev.Colomb.Quim. vol.41 no.2 Bogotá May/Aug. 2012
THEORETICAL MODEL OF CARBON NANOTUBES AS DELIVERY TO FLUOROURACIL (Anticancer)
MODELO TEÓRICO DE NANOTUBOS DE CARBONO COMO LIBERADORES DE FLUOROURACILO (Anticancerígeno)
MODELO TEÓRICO DE NANOTUBOS DE CARBONO COMO LIBERADORES DE FLUOROURACIL (Anticancerígeno)
Mohanned J. Ala-nber1* and Najim A. Al-Masoudi2
1 Department of Physics, College of Science, Basrah University, Basrah City, Iraq.
2 Department of Chemistry, College of Science, Basrah University, Basrah City, Iraq.
Recibido: 07/04/2012 – Aceptado: 16/07/2012
ABSTRACT
The quantum modeling interaction properties of fluorouracil radicals on the single walled carbon nanotubes surface is researched via MNDO/d calculations. We have studied the effect of diameter, length, position and rotational characteristics of CNT on binding fluorouracil. Our results suggest that the binding energy is lower as the CNT diameter increases, while as the CNT length increases the binding energy initially increases and then slightly increases.
Key words: anticancer, drug delivery, CNT, fluorouracil, semi-empirical, binding energy.
RESUMEN
Las propiedades de interacción, según el modelo cuántico, de los radicales de flourouracil ubicados en la superficie de los nanotubos de carbono de pared simple se investigan a través de cálculos MNDO/d. Hemos estudiado el efecto del diámetro, la longitud, la posición y las características rotacionales de CNT en la síntesis de fruorouracil. Nuestros resultados sugieren que la energía de síntesis baja en la medida en que aumenta el diámetro, mientras que a medida que aumenta la longitud, la energía de síntesis inicialmente disminuye y luego aumenta levemente
Palabras clave: anticáncer, administración de fármacos, fluorouracilo, semiempírico, energía de enlace.
RESUMO
As propriedades da interação, segundo o modelo quântico, do radicais de flourouracil localizados na superfície de nanotubos de carbono com paredes simples são pesquiçadas a traverso de cálculos MNDO/d. Estudamos o efeito do diâmetro, comprimento, posição e características rotacionais do CNT na síntese de fruorouracil. Nossos resultados sugerem que a energia de síntese baixa à medida que aumenta o diâmetro, enquanto que à medida que o comprimento aumenta, a energia de síntese inicialmente diminui e depois aumenta um pouco.
Palavras-chave: adminsitração de drogas, anticancerigeno, fluorouracil, semiempírico, energia de ligação.
INTRODUCTION
The development of some nanomaterials has had a promising effect on the treatment of several cancers. They have been used as delivery for drugs. Clinical research has approved the use of a number of conjugated materials, such as nanoparticles, liposomes, and polymers. The requirements for new drug delivery systems, improving the pharmacological profiles while decreasing the toxicological effects of the delivered drugs, have also envisaged carbon nanotubes (CNT) as one of the potential carriers for the cancer therapy. The physicochemical properties and behavior of nanomaterial, which were discovered by Iijima (1), have been given a new field in science. The quantum nature comes back due to their atomic and molecular sizes. How can the experiments approach to the atomic dimensions to do nanomeasurements? Carbon nanotubes are huge cylindrical molecules consisting of a hexagonal arrangement of sp2 hybridized carbon atoms. CNT can be synthesized by the techniques of electric arc discharge, laser ablation and catalytic decomposition of hydrocarbons [2-8]. Several applications are drug delivery, biosensing, antigen recognition and DNA hybridization, due to their unique properties. The penetration ability of CNT into cells offers the potential of using CNT as vehicles for the delivery of drug and antibiotic molecules without toxic effects [9-20]. Bianco et al. (2003) developed a new strategy for the multiple functionalization of CNT with amphotericin B (AmB) and fluorescein, where the CNT has the ability to form electrostatic interactions with nucleic acid as a complex [21]. This CNT-DNA complex acts as a non-viral vector to deliver plasmid DNA into the cell [22]. On the other side, Azamian et al (2002) used a simple non-covalent route to attach reactive molecules to sidewalls of CNT[23]. This work is of interest to the development of biosensors based on nanotubes. Wong et al (1998). Have shown that CNTs are ideal probe tips for AFM due to their small diameter [24]. The CNTs will present potential technological advances in bioengineering [25]. Up to now, there have been a lot of researches on the functionalization of CNTs with various molecules [20, 26-27].
In this work, we introduce a model for making CNT a delivery means of fluorouracil (anticancer drug) by examining the interaction of the fluorouracil radicals on the surface of single-walled carbon nanotubes SWCNT, which are defined as bond-alternation patterns of an armchair [28]. Then we examine this interaction as a function of CNT length and diameter. Also we investigate the effect of changing the position of the fluorouracil-CNT bond on the interaction energy. Lastly, we examine the effect of fluorouracil radical rotation about bond, which it makes on the CNT surface as a function of the interaction energy.
COMPUTATIONAL DETAILS
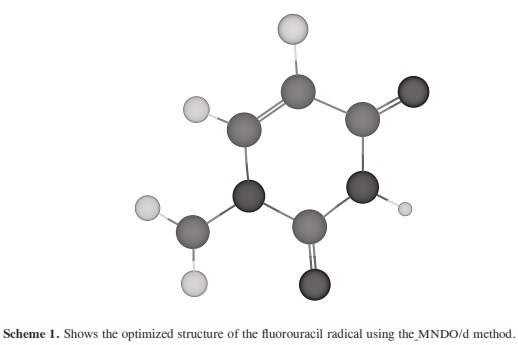
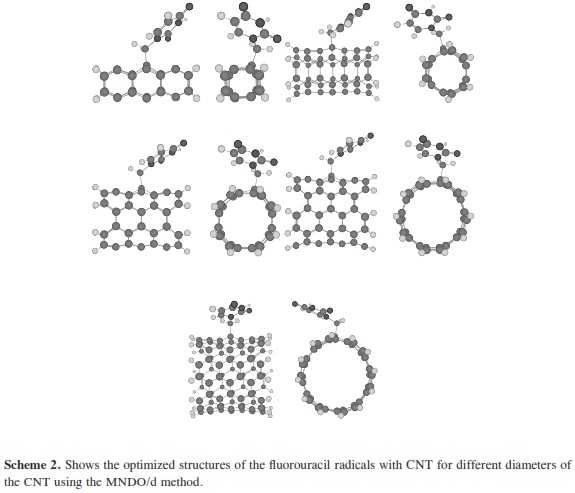
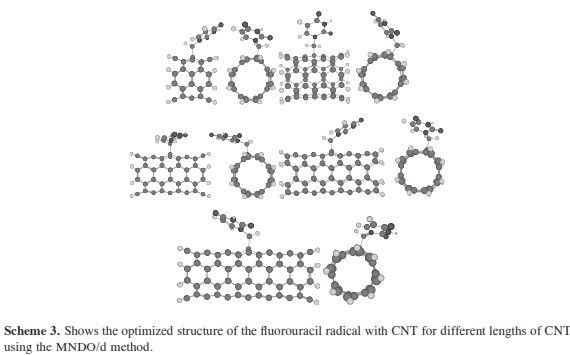
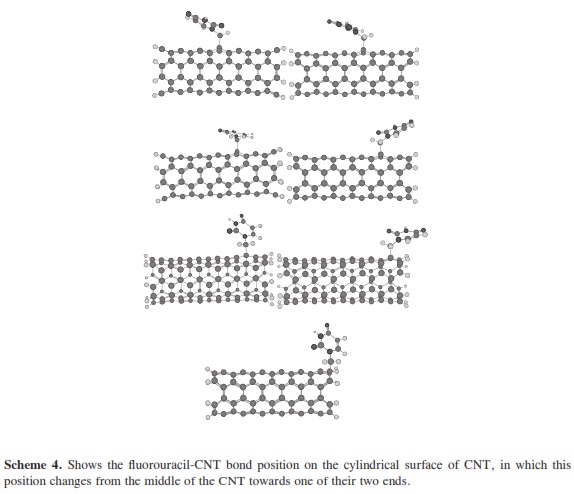
The theoretical calculations of molecular quantum mechanics can be used to research properties beyond the scope of current crystallographic methods and to bridge gaps in understanding experimental results. In many cases the results of the experimental methods are unable to accurately describe small complex systems or they can be used in further research and to predict the physical nature of hydrogen bonding interactions. Molecular quantum mechanics provides the interaction energies that are not provided by x-ray or NMR experiments. To research the structural and electronic properties of CNTs decorated with the fluorouracil radicals, we used the MNDO/d method. MNDO/d is a version of MNDO with d-orbital capability. Parameters for several new elements are included in the implementation of the MNDO/d method [29, 30]. MINDO/ 3 stands for Modified Intermediate Neglect of Differential Overlap, version 3; it is a modification of the INDO method. The method was proposed and developed by Dewar and coworkers [31-34]. Using the same nomenclature as for the INDO UHF Fock matrix approximation. All non-zero integrals over atomic orbitals on the two centers are set equal, as in CNDO/INDO. The two-center coulomb integrals were calculated from first principles using s orbitals as in CNDO/INDO [35]. The two-center one-electron integral, sometimes called the resonance integral, is approximated in MINDO/3 by using the overlap integral. The one-center twoelectron integrals used in MINDO/3 are derived from an analysis of atomic spectra [35]. The problem in quantum computational that arises is how to perform an accurate calculation for a nano-sized system without ending in a prohibitly large computation. The dangling bonds at the ends of the tubes were saturated by hydrogen atoms. The resolution of MNDO/d, as implemented in the HyperChemTM Release 7.52 for Windows Molecular Modeling System program package [36], was employed for the geometry optimizations with UHF, SCF controls (convergence limit 0.01, lteration limit 50), algorithm Polak-Ribiere (conjugate gradient), and RMS gradient of 0.1 kcal/(Ao mol), the molecular systems which have been considered in the present work. Scheme 1 shows the optimized structure of the fluorouracil radicals; scheme 2 shows the optimized structure of the fluorouracil radicals with CNT for different diameters of the CNT using MNDO/d method, while scheme 3 shows the optimized structure of fluorouracil radicals with CNT for different lengths of the CNT. Scheme 4 shows the fluorouracil-CNT bond position on the cylindrical surface of CNT, where this position changes from the middle of the CNT towards one of their two ends.
RESULTS AND DISCUSSION
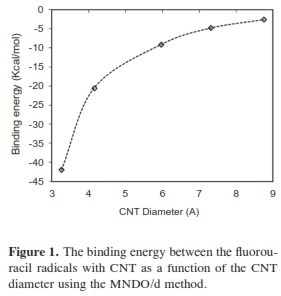
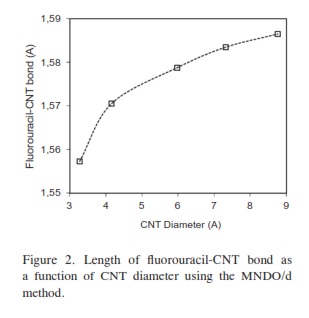
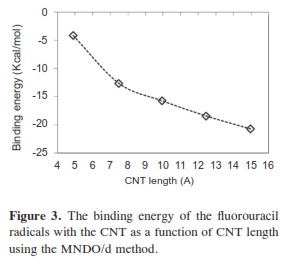
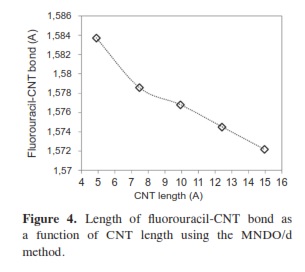
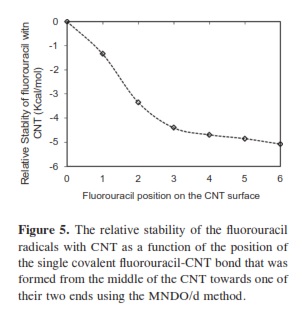
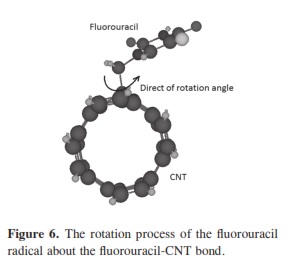
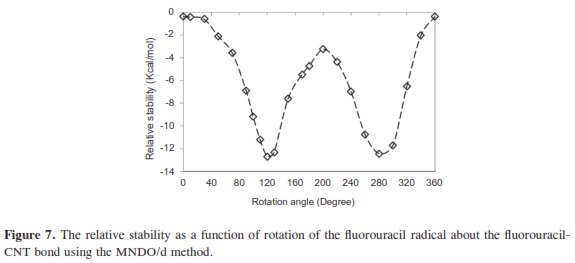
In our research, it was important to determine the most stable form of the fluorouracil radical as a complex with CNT. We calculated the interaction, binding energy BE of the fluorouracil radical with CNT, BE = Efluorouracil+CNT - (Efluorouracil +ECNT); where Efluorouracil+CNT is the energy of the complex of fluorouracil and CNT. The first important factor is the interaction of fluorouracil with CNT as a function of the CNT diameter. The effect of increases in the CNT diameter on the BE of the fluorouracil radicals with CNT (for constant length equal to 7.35 Ao) are shown in Figure 1. Note that in each issue the fluorouracil-CNT bond got fixed in the middle of the CNT surface. An increase in diameter of CNT leads to a decrease in the binding energy of the fluorouracil radicals with CNT. This case shows the mechanism of lower (or higher) binding between the fluorouracil with CNT as the CNT diameter increases. From Figure 1 we note that the energy may be required to finish the binding energy between the fluorouracil radicals with CNT, 2.67~41.98 Kcal/ mol, in visible regions of the spectrum. In other words, sunlight may be able to split the drug from the delivery. So, the CNT with narrow diameter must be chosen. We may be able to deduce the binding energy between the fluorouracil and the CNT diameter according to the formula BE=nln|d| where d is the CNT diameter. In the fluorouracil binding, a single covalent fluorouracil-CNT bond is formed with CNT. The fluorouracil- CNT bond becomes longer as the diameter of the CNT increases (Figure 2). The increases of the CNT diameter showed exponential increase in the bond length and decrease in the binding energy (Figure 1). This suggests there is a relationship between the bond length increase and the decrease in the binding. The second important factor is the interaction of fluorouracil with different lengths of CNT (for constant diameter equal to 5.45Ao). The CNT length during their synthesis is a very important property. The binding energy of the fluorouracil radicals with CNT depends on the length of CNT (Figure 3). As the length of the CNTs increase, the binding energy between the fluorouracil radicals and CNT also increases. Note that in each case we put the fluorouracil-CNT bond in the middle of the CNT. Beyond a few Angstroms of length, ~8Ao, the binding energy increased less than the initial state. Thus we conclude that the binding between fluorouracil and CNT depends on the diameters of CNT more than on their lengths. The fluorouracil-CNT bond length as a function of CNT length is shown in Figure 4. Initially, there are repaid decreases for a few Angstroms of CNT length, and then its decrease becomes linear as the CNT length increases. However, the fluorouracil-CNT bond increases as the length of CNT increases. This interaction behavior of the fluorouracil radical with CNT was opposite to the interaction of the glycine radical with CNT [32]. The relative stability of the fluorouracil radicals with CNT decreases as a function of the fluorouracil-CNT bond, which positions on the cylindrical surface of CNT, as this position changes from the middle of the CNT towards one of their two ends (Figure 5). The complexes formed by the fluorouracil radical and the single-tube wall are more stable when the reaction occurs in the middle of the CNT. This behavior may be similar to the state of stability of the glycine, when its reaction happened in the middle of the CNT surface, too [37]. Beyond short distance from the middle of the CNT surface, the decreases in binding energy start lowering. The change in binding energy of the fluorouracil-CNT bond positions on the cylindrical surface of CNT, as this position changes from the middle of the CNT towards one of their two ends, in range 1.34~5.06 Kcal/mol which got located in the initial region of the visible spectrum. Figure 6 shows the direction of the rotation process of the fluorouracil radical about the fluorouracil-CNT bond on the surface of CNT (for constant distance equal to 5.71 Ao and length equal to 9.82 Ao). Figure 7 shows the relative stability as a function of rotation of the fluorouracil radical about the fluorouracil-CNT bond. Optimized case is considered at 0o angle, and then we rotated the fluorouracil from angle 0° to 360°. The relative stability decreases until 120°, and then it increases until 200°; then it drops again. Figure 7 shows frequent behavior with the rotation angle; it also shows two potential walls, but with different depths. The nearest distance for the fluorouracil radical from the surface of CNT occurs at a rotation angle equal to 200°, which may decrease the related stability. There is another minimum at rotation angle equal to 280°. The energy that is required for these rotations is still in the visible spectrum energy. However, the thermal energy is not enough to do this rotation about the bond.
CONCLUSIONS
We have performed MNDO/d calculations on the structural properties of CNT upon adsorption of fluorouracil radicals. The results of the diameter and length of the CNT on the binding energies with fluorouracil shows a decrease as the CNT diameter increases, while the binding energies increase with CNT length increase. The fluorouracil radicals are shown to prefer to bond in sit that at the middle of the CNT cylindrical surface. The stereo geometry (curvature of CNT surface) between the fluorouracil radicals and CNT may limit the binding energies behavior. The rotation of fluorouracil about the fluorouracil-CNT bond did not show a new optimized point. The nature of the fluorouracil-CNT bond will influence with some energy of the visible spectrum region. At least, the effect of temperature increasing will not cause the break in the bond that connect the fluorouracil with the CNT.
REFERENCES
1. Iijima, S. and Ichihashi, T. Single-shell carbon nanotubes of 1-nm dia meter. Nature 1993. 363: 603-604. [ Links ]
2. Bethune, D. S.; Klang, C. H.; De Vries, M. S.; Gorman, G. ; Savoy, R. ; Vazquez, J. and Beyers, R. Cobalt-catalyzed growth of carbon nanotubes with single-atomic-layer walls. Nature 1993. 363: 605-609. [ Links ]
3. Tomita, M.; Saito, Y. and Hayashi, T. LaC2 encapsulated in graphite nanoparticle. Jpn. J. Appl. Phys. 1993. 32: 280-291. [ Links ]
4. Ajayan, P. M.; Lambert, J. M.; Bernier, P.; Barbedette, L.; Colliex, C. and Planeix, J. M. Growth morphologies during cobalt-catalyzed single-shell carbon nanotube synthesis. Chem. Phys. Lett. 1993. 215: 509. [ Links ]
5. Lambert, J. M.; Ajayan, P. M.; Bernier, P.; Planeix, J. M.; Brotons, V.; Coq, B. and Castaing, J. Improving conditions towards isolating single- shell carbon nanotubes. Chem. Phys. Lett. 1994. 226: 364. [ Links ]
6. Thess, A.; Lee, R.; Nikolaev, P.;Dai, H.; Petit, P.; Robert, J.; Xu,Lee, Y. H.; Kim, S. G.; Rinzler, A. G.; Colbert, D. T.; Scuseria, G. E.; Tomnek, D.; Fischer, J. E. and Smalley, R.E. Crystalline Ropes of Metallic Carbon Nanotubes. Scien ce. 1996. 273: 483. [ Links ]
7. Guo, T.; Nikolaev, P.; Thess, A.; Colbert, D. T. and Smalley, R. E. Catalytic growth of single-walled manotubes by laser vaporization. Chem. Phys. Lett. 1995. 243: 49. [ Links ]
8. Witanachchi, S.; Mahawela, P. and Mukherjee, P. Laser triggered hollow cathode plasma process for film growth. J. Vac. Sci. Technol. 2004 22: 2061. [ Links ]
9. Iijima, S. Helical microtubules of graphitic carbon. Nature. 1991. 354:56. [ Links ]
10. Terrones, M.; Hsu, W. K.; Kroto,H. W. and Walton, D. R. M. Nanotubes: a revolution in materials science and electronics. Top. Curr. Chem. 1999.199:189. [ Links ]
11. Dresselhaus, M. S.; Dresselhaus,G. and Eklund, P. C. Science of fullerenes and carbon nanotubes. New York, Academic Press. 1996. [ Links ]
12. Tsang, S. C.; Chen, Y. K.; Harris,P. J. F. and Green, M. L. H. Simple chemical method of opening and filling carbon nanotubes. Nature.1994. 372:159. [ Links ]
13. Davis, J. J.; Green, M. L. H.; Hill,A. H.; Leung, Y. C.; Sadler, J.; Sloan, J.; Xavier, A. V. and Tsang, S. C. The immobilization of proteins in carbon nanotubes. Inorg. Chim.1998. 272:261. [ Links ]
14. Balavoine, F.; Schultz, P.; Richard, C.; Mallouh, V.; Ebbesen, T. W. and Mioskowski, C. Helical crystallization of proteins on carbon nano- tubes: a first step towards the development of new biosensors. Angew. Chem. Int. 1999. 38:1912. [ Links ]
15. Guo, Z.; Sadler, P. J. and Tsang,S.C. Immobilization and visualization of DNA and proteins on carbon nanotubes. Adv. Mater.1998.10:701. [ Links ]
16. Bianco, A. and Prato, M. Can carbon nanotubes be considered useful tools for biological applications? Adv. Mater. 2003. 15:1765. [ Links ]
17. Pantarotto, D.; Partidos, C.; Graff, R.; Hoebeke, J.; Briand, J.; Pratt, M. and Bianco, A. Synthesis, structural characterization, and immunological properties of carbon nanotubes functionalized with peptide. J. Am. Chem. Soc. 2003. 125:6160. [ Links ]
18. Venkatesan, N.; Yoshimitsu, J.; Ito, Y.; Shibata, N.; Takada, K. Liquid filled nanoparticles as a drug delivery tool for protein therapeutics. Biomaterials. 2005, 26:7154. [ Links ]
19. Panthuis, M. Vaccine delivery by carbon nanotubes. Chem. Biol. 2003.10:897. [ Links ]
20. Basiuk, V. A. ONIOM studies of chemical reactions on carbon nanotube tips: effects of the lower theoretical level and mutual orientation of the reactants. J. Phys. Chem. B 2003.107:8890. [ Links ]
21. Bianco et al. Targeted delivery of amphotericin b to cells by using functionalized carbon nanotubes. Angew. Chem. Int. Ed. 2005.44:6358 [ Links ]
22. Singh et al. Binding and condensation of plasmid DNA onto functionalized carbon nanotubes: toward the construction of nanotube-based gene delivery vectors. J. Am. Chem. Soc. 2005.127:4388. [ Links ]
23. Azamian, R.; Davis, J.; Coleman, S.; Bagshaw, B.; Green, H. Bioelectrochemical single-walled carbon nanotubes. J. Am. Chem. Soc. 2002.124: 12664. [ Links ]
24. Wong, S.; Joselevich, E.; Woolley, A.; Cheung, C.; Lieber, C. Covalently functionalized nanotubes as nanometer probes for chemistry and biology. Nature. 1998.394:52. [ Links ]
25. Zanello, L.; Zhao, B.; Hu, H.; Haddon, R. Bone cell proliferation on carbon nanotubes. Nano Lett. 2006.6:566. [ Links ]
26. Gustavsson, S.: Rosen, A. and Bolton, K. Theoretical analysis of ethergroup derivatization at carbon nanotube ends. Nano. Lett. 2003.3:265. [ Links ]
27. Mavrandonakis, A.; Froudakis, G., E. and Farantos, S. C. Theoretical modeling of glycine radical addition to carbon nanotubes. Rev. Adv. Mater. Sci.2006.11:88. [ Links ]
28. Tanaka, K.; Yamabe, T. and Fukui,K. The science and technology of carbon nanotubes. Amsterdam, El sevier.1999. [ Links ]
29. Theil, W.; Voityuk, A. Theo. Chim. Acta. 1992.81:391-404. [ Links ]
30. Thiel, W.; Voityuk, A. Int. J. Quant. Chem. 1992.44:807-829. [ Links ]
31. Baird, N. C.; Dewar, M. J. S. J. Chem. Phys., 1969.50:1262. [ Links ] [ Links ]
33. Dewar, M. J. S. and D. H. Lo. J. Am. Chem. Soc. 1972.94:5296. [ Links ]
34. Bingham, R. C.; Dewar, M. J. S. and D. H. Lo. J. Am. Chem. Soc. 1975.97:1285. [ Links ]
35. Ohno, K. Theo. Chim. Acta. 1964.2:219. [ Links ]
36. www.hyper.com [ Links ]
37. Al-anber, M. Theoretical semi-empirical study of the biomolecules interaction with carbon nanotubes. J. Macrmol. Sci.B. 2011.50: 2481. [ Links ]