Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Acta Agronómica
Print version ISSN 0120-2812
Acta Agron. vol.65 no.2 Palmira Apr./June 2016
https://doi.org/10.15446/acag.v65n2.46432
http://dx.doi.org/10.15446/acag.v65n2.46432
Effect of nitrogen fertilization on soil microbial biomass in an Oxisol cultivated with irrigated barley in the Brazilian Cerrado
Efecto de la fertilización nitrogenada en la biomasa microbiana del suelo en Oxisuelos cultivados con cebada irrigada en el Cerrado de Brasil
Lidia Tarchetti Diniz 1, Maria Lucrecia Gerosa Ramos 1, Walter Quadros Ribeiro Junior 2, Andre Freire Cruz 3*, Leonice Vieira de Franca1, Bruno Tarchetti Diniz 1 and Renato Fernando Amabile
1Universidade de Brasilia. Facultade de Agronomia e Medicina Veterinaria, Campus Universitário Darcy Ribeiro, Brasília, Brasil; 2 Embrapa, Empresa Brasileira de Pesquisa Agropecuária. Ministério da Agricultura, Pecuária e Abastecimento, Brasil; 3 Kyoto Prefectural University, Graduate School of Life and Environmental Sciences, 1-5 Shimogamohangi-cho, Sakyo-ku, Kyoto 606-8522, Japan. *Author for correspondence: andre@kpu.ac.jp
Rec.: 20.10.2014 Acep.: 25.05.2015
Abstract
The aim of this study was to evaluate the effect of different nitrogen doses and five period of sample collection, on soil microbial biomass - nitrogen (SMB-N), total nitrogen (total N) and percentual ratio of the microbial biomass and total N (SMB-N/total N) in a Oxisol cultivated with barley (Hordeum vulgare L.). The experiment was installed in June, 2005, in an area located at Embrapa Cerrados, Federal District. The experimental design was a randomized block, with three replicates. The plots received doses of nitrogen: 20 - 40 - 80 kg ha-1 N and a control without it and the subplots were period of soil sample. Three applications of N were realized: 10 kg ha-1 on the 5th day (06/14) after sowing; the rest of N was parceled in two applications with fertigation, on tillage, on the 27th (07/08) DAP, e no 43rd (07/22) DAP. Soil samples layer (0 - 10 cm deep) were collected for (SMB-N) determination and total N in six periods: 02 days before of the first fertigation; 02 days after of the first fertigation; 04 days before of the last fertigation and 04 days after of the last fertigation; on flowering stage and after harvesting. There was effect of the doses of N and the period of soil collection on the SMB-N, total N and in the ratio SMB-N/total N. The average values of total N revealed steadier in short-term (cycle of the culture) and this was not a good parameter to evaluate the behavior and N transformations in the soil-plant system.
Keywords: Cerrado, Hordeum vulgare L., Microbial biomass, Nitrogen, Soil quality.
Resumen
El objetivo de este estudio fue evaluar el efecto de diferentes dosis de nitrógeno y cinco período de muestreo en la biomasa microbiana del complejo suelo - nitrogeno (BMS-N), nitrógeno total (N total) y la relación porcentual de la biomasa microbiana y N total (BMS-N/N total) en un Oxisol cultivado con cebada (Hordeum vulgare L.). El estudio se inició en junio de 2005 en la estación experimental de la Empresa de Pesquisa Agropecuaria (Embrapa-Cerrados), Distrito Federal, Brazil. El experimento se dispuso en bloques al azar con tres repeticiones. Las parcelas recibieron dosis de nitrógeno: 20 - 40 - 80 kg/ha de N más un control sin N, y las subparcelas fueron el periodo de muestro. Las aplicaciones de N se realizaron de la forma siguiente: cinco días después de la siembra (dds) se aplicaron 10 kg/ha y el resto de la dosis se aplicó con fertirrigación en dos dosis 27 y 43 dds. Las muestras de suelo (0-10 cm de profundidad) para determinar BMS-N y N total fueron tomadas, 2 días antes e igual número de días después de la primera fertirrigación y 4 días antes y después de la última, en floración y después de la cosecha. No se encontró efecto de las dosis de N y el período de muestreo en el BMS-N, N total y en la relación BMS-N/N total. Los valores medios de N total fueron más estable en el corto plazo (ciclo de la cultivo) lo que indica que éste no es un buen parámetro para evaluar la dinámica del N y sus transformaciones en el sistema suelo-planta.
Palabras clave: Cerrado, Hordeum vulgare L., Biomasa microbiana, Nitrogeno, Calidad del suelo.
Introduction
The barley cropping in Brazilian Cerrado is a suitable alternative for crop rotation, due to the local climate and soil well adapted, high yield potential, and can be employed non-tillage system due to high dry matter production.
The soil fertilization practice is a fundamental practice for the farmers, when applied properly. Fertilization primary aims to maintain or increase the availability of soil nutrients and organic matter, because the incorporation of essential elements can replace those lost by soil leaching, erosion, complexion, immobilization, fixation, volatilization, denitrification, and plant uptake (Sanchez, 1981; Hargrove & Thomas, 1984).
In tropical zones, the soils in general have been weathered and leached consequently having low fertility. In this situation, the soil organic matter can be considered a major source of energy and nutrients to the system due to their ability to maintain soil productivity (Xavier et al., 2006). Thus, the soil natural fertility depends on organic matter dynamics and nutrient cycling, which are usually catalyzed by soil microbial biomass (SBM) (Melloni et al., 2001; Billings & Ballantyne, 2013).
The SBM is defined as the smallest fraction of soil organic C and constitutes a significant portion of the available N available to plants. The SBM represents a fast cycling, strongly responds to seasonal fluctuations in humidity and temperature, the cultivation and organic matter management. The variations on macroclimatic conditions affect the activity and soil microbial biomass and subsequently in global cycle of C and N (Gama-Rodrigues et al., 2005).
The soil microbiota functions are decomposition of soil organic matter, nutrient cycling and energy flow, It also changes carbon storage and mineral nutrients availability (Matsuoko et al., 2003; Singh et al., 2011). In addition, biochemical transformers of organic compounds, SMB compose a reservoir of N, P and S (Wardle, 1992; Melloni, 2001). It contains between 2 to 5% of carbon, 1 to 5% of nitrogen and 2 to 20% of organic phosphorus in tropical soils (D'Andrea et al., 2002). The amount of nutrients immobilized in biomass can reach values above 100 kg ha-1 of N. The N retained in biomass is released with the death of microorganisms and are mineralized by the remaining, in environmental stress soils, most of the mineralized N might be of originated from microbial source. The MB acts as a buffer for soil nitrogen, because it controls the availability of nutrients through mineralization processes and immobilization (Paul, 2007). The microbial activity and biomass are influenced, among other factors, by temperature, moisture, aeration, fertilizer and availability of substrate in soil (Coser et al., 2007; Harrison-Kirk et al., 2014).
The monitoring of soil biota allows to evaluate not only the soil quality but also the production system, since it is related to decomposition and nutrient cycling in the soil-plant interface.
The application of fertilizers through irrigation, the fertigation, is a routinely adopted practice, with some advantages such as: savings in laboring, the application of the products at any stage of the cropping, divide the doses of nutrients, and finally to efficiently control the using of nutrients (Costa et al., 1994; Ebrahimian, 2014).
The microbial biomass nitrogen (SMB-N) is easily available fraction, but little is known about the biotic and abiotic factors that affect their dynamics on growth of malting barley (Hordeum vulgare L.) in the Brazilian cerrado with fertigation. Thus, studies on microbial biomass could allow associating the amount of immobilized N and microbial activity with the potential of soils to supply this nutrient to barley.
This study aimed to evaluate the effect of nitrogen applied through fertigation, and harvest dates on soil microbial biomass nitrogen (SMB-N), total soil nitrogen (total N) and the percentage ratio of nitrogen from SMB and soil total nitrogen (SMB-N / total N) in barley.
Materials and methods
Experimental area characterization
The field experiment was conducted between June and September of 2005, at the Experimental Station of Embrapa Cerrados (CPAC) in Planaltina city-DF-Brazil, (15°35’30”S and 47°42’30”W, with 1,007 m of altitude).
The soil of the experimental area was classified as a typical red clayey Oxisol (LVd) (Embrapa, 1999), cultivated during 15 years. Historically, the brachiaria grass (Brachiaria decumbens) was cultivated in the last three years before barley cultivation. At the end of each cycle, the grass was cut and removed from the area in order to exhaust the soil nitrogen. Before cultivate the plant drought was performed for the direct seeding, but the machine damaging prevented the planned system, so the conventional tillage was applied for that.
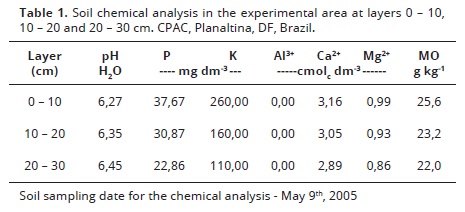
The area preparation consisted of plowing down, followed by disking. The fertilizers were applied before planting as follows: 165.2 kg ha-1 of potassium chloride, and 138.8 kg ha-1 of triple superphosphate, distributed manually and then the fertilizers were incorporated into the soil by disking. The soil chemical properties were determined according to the methods proposed by Embrapa (2006), which are shown in Table 1.
The experiment was conducted under conventional tillage system in randomized blocks with three replications; plots received nitrogen rates: 20 - 40 - 80 kg N ha-1 and a control, and the subplots represented soil collected stages. The urea [(NH2)2CO2] was applied as a nitrogen source through fertigation. Nitrogen fertilization was applied three times (Table 2).
The barley genotype used was the AF9585 due to its response to nitrogen fertilizer and might be launched as an alternative to irrigated farming in the Brazilian cerrado. The crop seeding was done in June 9, 2005, where each plot (2 × 4 m), with spacing of 0.20 m between lines, composing 10 rows of plants per plot. The sampled area of each the plot was established by five lines to evaluate chemical and biological soil properties.
Irrigation was performed when the tensiometers reached the predetermined values of 100 kPa for barley, and 400 mm of water were applied during the whole crop cycle.
Soil samples were taken using the Dutch auger at 0-10 cm, inside the plots to avoid the borders effect, in a zigzag system between the lines, with attention to not collect samples from different periods in same location. The soil sampling was done in six periods for the SMB-N and Total N (Table 3).
Results and discussion
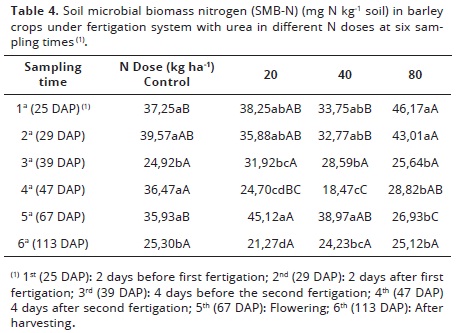
There was a significant interaction between nitrogen and sampling times in the SMB-N (Table 4). The overall data showed a downward trend in SMB-N at the end of the crop cycle in all treatments. In the first and second sampling times, there was no significant difference between treatments in SMB-N (within each dosage), but in the control treatments, these values were similar in the 4th and 5th sampling time and higher than other sampling times (3rd and 6th). Probably the N immobilization by microorganisms could occur in the first two collections, whereas at 25 and 29 days after seeding the plant was still in the early stages of development so the demand for nutrient was low.
In the 3rd sampling, the control had reduced their values significantly from 33.10% compared to the first time. With this reduction the SMB-N, perhaps the soil N was not immobilized by microorganisms may promote N availability to plants which at this stage (39 days after seeding) the tillage was done. In 4th sampling of the same treatment there was a significant increase of 46.35% in SMB-N, compared to the 3rd one. It suggests that a favorable condition to stimulate microorganisms could occur, such as water content and substrate availability. Maybe because the plant was under a stress condition caused by lack of N necessary to meet their demands, which could stimulate the growth of root system to make easier the N immobilization at this stage (before flowering). The higher root density can release more carbon and other compounds to the soil especially through the decomposition of dead roots.
In the 5th sampling there was a reduction on values, without any significance. Interestingly, the most expected rhizosphere effect during flowering period was not detected here. The rhizosphere of grasses have strong effect due to its extensive root system, which usually has high rate of nutrient cycling (Santos et al., 2004) to make available organic compounds of easy degradation. Such compounds are used as an energy source on microbial growth (Freixo et al., 2002). Also, root exudates have strong effect on soil microbial communities (Chaparro et al., 2012)
In the 6th one there was a reduction of 29.58% in the control treatments compared to the previous sampling and storage of SMB-N at end of crop cycle corresponding to 32.08% of the first sampling. These data suggest that part of SMB-N have been used by the culture.
In the treatments with 20 kg ha-1 N and 40 kg N ha-1 until the 3rd sampling, the SMB-N was similar, with a reduction up to 39 days. In 4th collection there was a significant reduction of 22.62% and 35.39% compared to the 3rd one in SMB-N. This N removing from the system may be due to uptake by the plants, volatilization and leaching that could reduce the availability of N to soil microorganisms and these immobilized lower amounts of N compared to previous season.
In the 5th sampling (flowering period), there was a significant difference between the sampling times, with an increasing of 15.22% and 13.40%, respectively for doses 20 and 40 kg ha-1 N comparing to the first collection; and an increase from the previous season, respectively, for the doses 20 and 40 kg ha-1 N of 45.25% and 52.60%. The higher amount of SMB-N obtained was probably due to the rhizosphere effect. The root system itself can provide a higher amount of organic matter easily degradable in the soil surface (Moore et al., 2000; Chaparro et al., 2012) and probably induce more microbial activity, which can immobilize nutrients by rhizodeposition (Martens, 1995). The largest amount of SMB-N in cultivated areas is fundamental for N cycling through the microbial biomass. The immobilized N by microorganisms would be available to plants in short and medium term, according to cycling this environment (Souza & Melo, 2000) and avoid N losses by leaching, providing N according to plant demand (Kuzyakov and Xu, 2013).
In 6th sampling with the dose of 20 kg N ha-1 there was a reduction by 52.86% compared to the previous sampling and a storage at the end of crop cycle of 16.98 mg N kg-1 soil corresponding to the first sampling time. For the dose 40 kg N ha-1 a reduction of 37.83% was observed compared to the previous sampling and storage of SMB-N of 9.52 mg N kg-1 soil, taking out the SMB-N from the 1st and 6th soil samples (28.20%). This N removal from the system could occur mostly due to exportation of these nutrients through grains.
In the 80 kg N ha-1 treatment there was no difference in SMB-N in the first and second sampling. In the third one there was a significant reduction of 44.46% in SMB-N compared to the first and perhaps a portion of N added by fertigation was not immobilized by the soil microorganisms. These N may have been removed from the system by plants uptake, nitrate leaching or volatilization. In 4th, 5th and 6th samples the SMB-N were similar with a reduction of 7.2% and SMB-N storage was 21.05 mg N kg-1 soil (45.59% compared to the first sampling).
Within the sampling times, the first one (25 days after seeding), except for the control, the other N rates (20 - 40 and 80 kg ha-1) contained only the basic fertilizer (10 kg ha-1 N), it was observed that the dose of 80 kg ha-1 N was significantly higher than that of 40 kg ha-1 and the control. At the 2nd time, 29 days after seeding (after fertigation, where treatments received half the recommended dosage: 5-15 to 35 kg ha-1 N), the results followed the same trend. Also the dose of 80 kg ha-1 N was significantly similar to 20 kg N ha-1 and the control, and the dose 40 kg N ha-1 was significantly lower. These results could be explained by lack of management uniformity for several years which results in ranges in SMB influenced by soil temperature and addition of plant residues (Patra et al. 1990). In 3rd sampling, 39 days after planting (DAP), no significant difference was detected between treatments. In 4th sampling at 47 DAP, after the second fertigation, the control plots and treatments of 80 kg ha-1 N were similar, but the control had the largest absolute value (36.47 mg N kg-1 soil).
In the 5th season at 67 DAP (flowering) the lowest value of SMB-N was obtained in the highest N dose; therefore, it is inferred that the addition of 80 kg N ha-1 led a decreasing of SMB-N at high activity in the root system during the cropping cycle. At this sampling time, the doses of 20 and 40 kg ha-1 N were significantly higher compared to 80 kg ha-1 N, and the dose 20 kg N ha-1 immobilized larger amount of N; and the control, was similar to the dose 40 kg ha-1 N.
At the flowering season, the highest values of SMB-N suggest greater immobilization of N in the SBM, probably due to the higher amount of N derived from root exudates and plant residues on soil surface that are immobilized by the microflora (Perez et al., 2005). In the 6th sampling to 113 DAP, after harvest, no difference was observed between doses with immobilization decreasing maybe due to N exportation through the grain.
Coser et al. (2007) evaluated the effect of different dosages (0 to 80 kg N ha-1) of N fertilization applied in the rows on microbial nitrogen biomass in the soil after barley harvest and obtained that these attribute varied between 20,03 to 38.39 mg N kg-1 soil at layer 0-10 cm. The authors concluded that there was no effect of different dosages on NBM at harvest. These results are similar to the preset paper.
The maintenance of long-term productivity in agricultural and forest agro-ecosystems depends on transformation of soil organic matter. Thus, the larger SMB-N value suggests a management and addition of fertilizers could stimulate the transformation of N in soil and microbiological processes involved in this nutrient cycling. These facts promote the N availability to plants according to their demand in the crop cycle. The largest amount of SMB-N may indicate greater potential for N mineralization, as observed by Santos et al. (2004).
There was a significant interaction between nitrogen levels and harvest dates, in the total nitrogen (Table 5). The total N for all samples within the doses showed a similarity, except the control treatment, which was lower at 1st, 2nd, 5th and 6th, compared to the 4th sampling.
The sampling time did not influence the other treatments (doses 20 - 40 and 80 kg ha-1 N). The treatment which did not receive urea and for doses of 20 and 40 kg ha-1 N, the highest value was observed in 4th sampling (47 DAP), after fertigation, suggesting an increasing of total N proportionally to the fertilizer dosage.
The sampling time probably influenced the SMB-N over the crop cycle by the fertilization at 80 kg ha-1 N, the rhizosphere effect; and other processes of N removal from the system: leaching (NO3-) and volatilization (NH3); plant uptake, export to the grain, unlike soil total N (Tables 4 and 5). These results indicate that SMB-N was most influenced by changes in biotic and abiotic factors during the crop cycle than the values of total N, apparently more stable in short-term changes. This suggests that this parameter by itself may not be a good indicator of transformations in organic matter and management systems on annual crops.
The magnitude of changes in total N depends on management intensity, the type and frequency of implements, the quality and quantity of fertilizers and organic residues replaced to soil (Beare et al., 1994; Gillespie, et al., 2014). According to Bayer & Mielniczuk (1999), the conventional soil tillage (plowing and harrowing) promotes faster decomposition rate of organic matter and lower addition of residues. These results in differences in the organic C amount and total soil N, but there was almost no fluctuation in total N, thus this parameter alone does not explain the N dynamics in the system. Santos et al. (2004) in studies on flat soils under different management systems found differences in soil total N in the 0 - 5 cm and 5-10 cm layers indicating that the effects of management systems with high or low plant residual addition could affect the total N in the soil. However, Moreira and Malavolta (2004) observed an increase SMB-N at 0 -10 cm layer, regardless the management used in cupuaçu (Theobroma grandiflorum) orchards, while the total N decreased in this layer.
There was an interaction between nitrogen levels and sampling time in terms of ratio of N percentage from SMB-N and soil total nitrogen (SMB-N / Total N) (Table 6). It suggests that the SMB-N / N ratio expresses the overall efficiency of the microbial biomass to immobilize N according to the quantity and quality of available substrate, and lower ratio indicates less efficiency of microbial biomass to immobilize the C and N (Monteiro & Gama-Rodrigues, 2004).
Comparing the sampling times within N rates, the SMB-N / total N ratio varied between seasons (Table 6), suggesting that this variation could occur due to factors that change the balance and influenced the microbial activity, such as root development during the crop cycle.
Concerning on the sampling time in control plots, the ratio SMB-N / total N were higher and similar at the 1st, 2nd, 4th and 5th sampling due to higher values of SMB-N shown in this treatment (Table 4). The lowest ratio was observed in 3rd sampling and after harvest; it is important to emphasize that ratio SMB-N / total N confirmed the little ranging between sampling times compared to treatment without urea.
In the treatment of 20 kg ha-1 N there was a large fluctuation on SMB-N, decreasing within the cycle with the highest value during the flowering (3.2%). The one with 40 kg N ha-1, there was less fluctuation between the values, where the highest ratio SMB-N / Total N was obtained in 5th sampling (flowering) and the lowest value in 4th sampling (after 2nd fertigation). Nevertheless, in the dose of 80 kg N ha-1 there were few variations between the results; in the 1st and 2nd samplings it was observed the highest values, whereas in the other samples the values were similar.
At the flowering stage, even availability of substrate increased by rhizodeposition, the ratio SMB-N / N total was not affected as occurred at 40 kg. Among the N concentrations, within each season, in the first sampling, the highest ratio was obtained at the highest dosage (3.2%) and the lowest at dose 40 kg N ha-1 (1.2%). In the 2nd and 6th samples no difference was found between N dosages in 3rd sampling, in particular the control value was statistically similar to 20 kg ha-1 N, demonstrating that at 39 DAP, the addition of N resulted in a similar ratio SMB-N / N total.
In 4th sampling (after 2nd fertigation), the SMBN / total N was higher in the treatment without N and 80 kg ha-1 N and the lowest at doses of 20 and 40 kg ha-1 N. Whereas in the 5th sampling (flowering), the lowest values were found at the highest dose of N and at 20 kg N ha-1, the highest values of SMB-N / N total. At the dose of 40 kg ha-1 N it was similar to those without N. The soil amendment with N induced higher conversion of available N in the microbial tissue, which is desirable, because a higher N recycling system could occur through the microbial activity. In 6th sampling, there was no difference on ratio SMB-N / total N between the N rates.
The ratio SMB-N / total N may be an indirect estimation of the organic matter quality, i.e. the lower the value indicates a lower substrate quality, thus less efficiency in immobilization of microbial biomass C and N (Monteiro & Gama-Rodrigues, 2004).
Supposing an average of 1.5 g kg-1 for total of N experimental area in the layer (0 - 10 cm), would correspond to 1500 kg ha-1 of total N. Whereas SMB-N / Total N ratio, averaged 2%, has the ratio SMB-N 30 kg / 1.5 g total N / ha, then an average of 20 kg of N / g total N, originating from microbial source may be cycled and would become available for plants and microorganisms throughout the crop cycle.
Conclusions
- There was an effect of N rates and sampling time in SMB-N, total N and SMB-N / total N.
- The average N values were more stable in the short term (crop cycle), which is not a good parameter for evaluating the dynamics and transformations of N in the soil-plant system.
- In terms of soil microbial biomass preservation and fertility the N dosage of 40 kg ha-1 could be recommendable.
References
Bayer, C. and Mielniczuk, J. 1999. Dinâmica e função da matéria orgânica. In: Santos, G.A.; Camargo, F.A.O. (Ed.). Fundamentos da matéria orgânica do solo: ecossistemas tropicais e subtropicais. Porto Alegre: Genesis. 9-26. [ Links ]
Beare, M.H.; Cabrera, M.L.; Hendrix, P.F. and Coleman, D.C. 1994. Agregate-protected and unprotected organic matter polls in conventional-tillage and no-tillage soils. Soil Sci. Soc. Am. J. 58: 787-795. [ Links ]
Billings, S.A. and Ballantyne, F. 2013. How interactions between microbial resource demands, soil organic matter stoichiometry, and substrate reactivity determine the direction and magnitude of soil respiratory responses to warming. Global Change Biol. 19: 90-102. [ Links ]
Chaparro, J.M.; Sheflin, A.M.; Manter, D.K. and Vivanco, J.M. 2012. Manipulating the soil microbiome to increase soil health and plant fertility. Biol. Fertil. Soil 48: 489-499. [ Links ]
Costa, E.F.; Vieira, R.F. and Viana, P.A. 1994. Quimigação. Aplicação de produtos químicos e biológicos via irrigação. Brasília, EMBRAPA-SPI, 315p. [ Links ]
Coser, T. R.; Ramos, M. L. G.; Amabile, R. F.; Ribeiro Júnior, W. Q. 2007. Nitrogênio da biomassa microbiana em solo de Cerrado com aplicação de fertilizante nitrogenado. Pesq. Agropec. Bras., 42: 399-406. [ Links ]
D'Andréa, A.F.; Silva, M.L.N.; Curi, N.; Siqueira, J.O. and Carneiro, M.A.C. 2002. Atributos biológicos indicadores da qualidade do solo em sistemas de manejo na região do cerrado no sul do estado de Goiás. R. Bras. Ci. Solo, 26:913-923. [ Links ]
Ebrahimian, H.; Keshavarz, M. R.; Playán, E. 2014. Surface fertigation: a review, gaps and needs. Span. J. Agric. Res., 12: 820-837. [ Links ]
EMBRAPA. 1997. Manual de métodos de análises de solo. 2ª ed. Rio de Janeiro - Brazil. 212p. [ Links ]
EMBRAPA, 2006. Sistema Brasileiro de Classificação dos Solos. Rio de Janeiro: Embrapa Solos, 306 p. [ Links ] Ferreira, D.F. Sisvar versão 4.3. Lavras: DEX-UFLA, 2003. [ Links ]
Freixo, A.A.; Machado, P.L.O.A.; Guimarães, C.M.; Silva, C.A. and Fadigas, F.S. 2002. Estoque de carbono e nitrogênio e distribuição de frações orgânicas de Latossolo do cerrado sob diferentes sistemas de cultivo. R. Bras. Ci. Solo. 26: 425-434. [ Links ]
Gama-Rodrigues, E.F.; Barros, N.F.; Gama-Rodrigues, A.C. and Santos, G.A. 2005. Nitrogênio, carbono e atividade da biomassa microbiana do solo em plantações de eucalipto. R. Bras. Ci. Solo. 29: 893-901. [ Links ]
Gillespie, A.W., Diochon, A.; Ma, B.L.; Morrison, M.J.; Kellman, L.; Walley, F.L.; Regier, T.Z.; Chevrier, D.; Dynes, J.J. and Gregorich, E.G. Nitrogen input quality changes the biochemical composition of soil organic matter stabilized in the fine fraction: a long-term study. Biogeoch. 117: 337-350. [ Links ]
Harrison-Kirk, T., Beare, M.H., Meenken, E.D. and Condron, L.M. 2014. Soil organic matter and texture affect responses to dry/wet cycles: Changes in soil organic matter fractions and relationships with C and N mineralisation. Soil Biol. Bioch. 74: 50-60. [ Links ]
Kuzyakov, Y. and Xu, X. 2013. Competition between roots and microorganisms for nitrogen: mechanisms and ecological relevance. New Phytol. 198: 656-669. [ Links ]
Martens, R. 1995. Current methods for measuring microbial biomass-C in soil: Potential and limitations. Biol. Fertil. Soils. 19: 87-99. [ Links ]
Matsuoko, M.; Mendes, I.C. and Loureiro, M.F. 2003. Biomassa microbiana e atividade enzimática em solos sob vegetação nativa e sistemas agrícolas anuais e perenes na região de Primavera do Leste (MT). R. Bras. Ci. Solo, 27: 425-433. [ Links ]
Melloni, R.; Pereira, E.G.; Trannin, I.C.B.; Santos D.R.; Moreira, F.M.S. and Siqueira, J.O. 2001. Características biológicas de solos sob mata ciliar e campo cerrado no sul de Minas Gerais. Ciênc. Agrotec. 25: 7-13. [ Links ]
Monteiro, M.T. and Gama-Rodrigues, E.F. Carbono, nitrogênio e atividade da biomassa microbiana em diferentes estruturas de serapilheira de uma floresta natural. R. Bras. Ci. Solo, 28:819-826, 2004. [ Links ]
Moore, J.M.; Klose, S. and Tabatabai, M.A. 2000. Soil microbial biomass carbon and nitrogen as affected by cropping systems. Biol. Fertil. Soils. 31: 200-210. [ Links ]
Moreira, A. and Malavolta, E. 2004. Dinâmica da matéria orgânica e da biomassa microbiana em solo submetido a diferentes sistemas de manejo na Amazônia Ocidental. Pesq. Agropec. Bras. 39: 1103-1110. [ Links ]
Patra, D.D.; Brookes, P.C.; Coleman, K. and Jenkinson, D.S. 1990. Season changes of soil microbial biomass in an arable and a grassland soil which have been under uniform management for many years. Soil Biol. Biochem. 22: 739-742. [ Links ]
Paul, E.A. 2007. Soil microbiology, ecology and biochemistry. 3rd Edition. Burlington: Elsevier, 531p. [ Links ]
Perez, K.S.S.; Ramos, M.L.G. and Mcmanus, C. 2005. Nitrogênio da biomassa microbiana em solo cultivado com soja sob diferentes sistemas de manejo nos cerrados. Pesq. Agropec. Bras. 40: 137-141. [ Links ]
Santos, V.B.; Castilhos, D.D.; Castilhos, R.M.V.; Pauletto, E.A.; Gomes, A.S.; Silva, D.G. 2004. Biomassa, atividade microbiana e teores de carbono e nitrogênio totais de um Planossolo sob diferentes sistemas de manejo. R. Bras. Agroc. 10: 333-338. [ Links ]
Singh, J.S.; Pandey, V.C. and Singh, D.P. 2011. Efficient soil microorganisms: A new dimension for sustainable agriculture and environmental development. Agric. Ecos. Env. 140: 339-353. [ Links ]
Souza, W.J.O and Melo, W.J. 2000. Teores de nitrogênio no solo e nas frações da matéria orgânica sob diferentes sistemas de produção de milho. R. Bras. Ci. Solo. 24: 885-896. [ Links ]
Thomas, G.W. and Hargrove, W.L. 1984. The chemistry of acidity. In: F. ADAMS (ed.) soil acidity and liming. 2ª ed. Madison. ASA, CSSA, SSSA. 3-56. [ Links ] Vance, E.D.; Brookes, P.C. and Jenkinson, D.S. 1987.
An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 19: 703-707.
Vargas, L.K. and Scholles, D. 1998. Nitrogênio da biomassa microbiana, em solo sob diferentes sistemas de manejo, estimados por métodos de fumigação. R. Bras. Ci. Solo. 22: 411-417. [ Links ]
Vargas, L.K. and Scholles, D. 2000. Biomassa microbiana e produção de C-CO2 e N mineral de um Podzólico vermelho-escuro submetido a diferentes sistemas de manejo. R. Bras. Ci. Solo. 24: 35-42. [ Links ]
Xavier, F.A.S.; Maia, S.M.F.; Oliveira, T.S. and Mendonça, E.S. 2006. Biomassa microbiana e matéria orgânica leve em solos sob sistemas agrícolas orgânico e convencional na chapada da Ibiapaba - CE. R. Bras. Ci. Solo, 30: 247-258. [ Links ]