Introduction
One of the major challenges that humanity is at present facing is to reconcile its evolution and development with environmental preservation, especially when it comes to increasing agricultural production. Currently, main biofuels with the capacity to replace fuels derived from petroleum are obtained from plant biomass like biodiesel and ethanol. In this way, the study and improvement of raw materials is essential for the consolidation of biofuels in the world energy matrix (Silveira, 2008; Loyola, Urra & Acuña, 2016; Lareo, Ferrari, Guigou, Fajardo, Larnaudie, Ramírez & Martínez-Garreiro, 2013).
Alcohol production from amylaceous crops is still not widespread in Brazil however, in other countries this technique is widely adopted, e.g., United States that produces ethanol fuel from maize hydrolysis followed by fermentation. Despite having a higher use of maize (Zea mays L.) as raw material for fuel production, it shows adaptation problems to certain climatic conditions, low energy balance values, besides competing directly with food chain (Lareo et al., 2013).
In this sense, other raw materials with ethanol production potential are being studied, especially beet (Beta vulgaris L.), cassava (Manihot escalenta Crantz.) and sweet potato (Ipomoea batatas (L.) Lam.). Among these three crops, sweet potato has favorable characteristics that allows to classify it as a promising species for fuel production (Velásquez, Lucas & Quintero, 2017; Loyola, Urra & Acuña, 2016). This is mainly because sweet potato is a crop that adapts quickly to low and medium fertility soils. Moreover, is more resistant to pests and diseases compared to other crops and shows a short production cycle between four and six months. Furthermore, all parts of the crop can be harvested, i.e. roots for alcohol extraction, aerial parts, and residues from distillation for animal production (Gonçalves-Neto, Maluf, Gomes, Gonçalves, Silva & Lasmar, 2011). Additionally, it is important to note that in this case, these characteristics influence agrochemical use reduction, allowing an improvement in production costs and environmental impacts, in addition to using all parts of the plant.
Furthermore, Silveira (2008), and Manas, Mishra & Thatoi (2013), also highlights the environmental sustainability in species cultivation because it seems as a great alternative for family agriculture. This author also points out that through his researches, yields of 170 liter of ethanol per ton of root were obtained, having however a yield capacity of 40 to 60 tons per hectare, with yields of 6800 to 10200 liters per hectare.
However, the process to obtain alcohol from amylaceous plants occurs through several steps, which directly interferes with the productivity and quality of the alcohol obtained. Some studies have already suggested improvements on critical points of the acid and enzymatic hydrolysis of sweet potato and its fermentation (Pavlak, Abreu-Lima & Carreiro, 2011).
Consequently, the aim of this study was to evaluate the potential of alcohol production from sweet potato genotypes, as well as to assess the effect of fermentation time, use of Saccharomyces cerevisiae yeast in commercial forms (tablet and powder) and enrichment of fermentative medium with micronutrients. This research will open new agricultural frontiers and markets for alcohol production, mainly for regions as the southern region of Brazil that does not have favorable conditions for sugarcane cultivation, that is the species from where alcohol is currently produced.
Materials and methods
Study site
The study was carried out in the Plant Physiology and Horticulture Laboratory of Midwestern State University-UNICENTRO, located in the municipality of Guarapuava-Paraná, Brazil, at a latitude of 25° 41' 00'' S and a longitude of 51° 38' 00" W and at an altitude of1100 meters above sea level. The climate is type Cfb (subtropical mesothermic humid) and temperate according to the classification of Kõppen, without a defined dry season, but with a hot summer and a moderate winter.
Plant material
Eight sweet potato genotypes from the germplasm collection of UNICENTRO were selected based on productivity and adaptation to local climatic conditions as follows: UGA 92, UGA 77, UGA 49, UGA 11, UGA 08, UGA 07, UGA 06 and UGA 05 (Camargo, Morgon, Resedente & Da-Silva, 2013). Seedlings were cultivated during October 2012 to April 2013 in a plot with soil classified as Distroferric Bruno Latosol.
One-hundred and eighty (180) days after transplanting roots were harvested; these were identified, selected, washed, weighed, and fresh mass (g) was established. Subsequently, samples were grated and taken to a drying oven at 65 °C for 72 hours until constant weight was reached, establishing dry mass (g) values. Then, samples were ground to obtain sweet potato flour to establish levels of reducing sugars (%) and starch (%).
Moreover, starch content was determined by titration with Fehling reagents in a complementary manner. Reducing sugar content was determined by the Lane-Eynon analytical titration method (IAL, 1976), the initial mass results of sweet potato flour, volume of sample spent on titration and standardization of the Fehling reagents A and B were used to calculate starch content and reducing sugars in sweet potato samples.
Then, according to the reductive sugars (%) and starch (%) results, two genotypes were selected for fermentation process. Obtaining alcohol sequentially comprised the steps of enzymatic hydrolysis of amylaceous material with purified enzymes, α-amylase and amyloglucosidase (Sigma-Aldrich). Sterilization was carried out by autoclaving (1 atm and 120°C). Fermentation was carried out by addition of Saccharomyces cerevisiae in the form of dry and fresh biological yeast, followed by distillation of the alcohol, and finally it was characterized according to its alcoholic content in °GL.
Enzymatic hydrolysis or saccharification was performed according to a methodology adapted from Pavlak, Abreu-Lima & Carreiro (2011) , starting with a thermostatic bath by mixing three grams of sweet potato flour into 50 mL of McIlvaine buffer at pH 5.6; starch was gellified at 90 °C for 15 minutes. Subsequently, amylase (13 μL) was added maintaining the temperature at 90°C for 1 hour at 15-minute intervals, stirring and homogenizing the medium. Therefore, the medium was cooled and pH was adjusted to 4.5 with 0.1 mol.L-1 of HCl, reducing the temperature to 60°C, to improve the action of the amyloglucosidase enzyme (54 μg-1 of enzyme). It remained in a one hour bath and stirring was carried out in 15-minute intervals. These conditions have allowed an increasing in a-amyloglucosidase (57 μL) enzymatic activity as verified by Stroparo, Beitel, Resende and Knob (2012) .
For instance, was centrifuged with a Hettich Zentrifuger MIKRO 220R centrifuge, rotating at 2200 rpm for eight minutes. The supernatant generated had its volume completed to 50 mL and autoclaved for 10 minutes in a vertical autoclave model CS (Prismatec, Brazil). This process have allowed to evaluate fermentation, since the medium was sterilized and free from the action of microorganisms that could interfere with the activity of Saccharomyces cerevisiae, added at a concentration of 8 g.L-1. Thus, after yeast addition the system was agitated for homogenization for 2 minutes at a frequency of 10 rpm on a Labstore shaker table, Model 109. The entire system was then deaerated with the passage of nitrogen gas, N2 (g) for 10 minutes; samples were taken to oven at a temperature of 36°C during the two evaluated fermentation times: 72 and 96 hours.
At the end of fermentation times, volatile compounds present in the sample were removed at a temperature of 78°C, allowing the alcohol to be distilled at a temperature near its boiling point. Finally, alcohol content in °GL was established in all distilled alcohol samples according to standard NBR 13920, where each test was repeated in duplicate. The method comprised the construction of a standard absorbance curve as a function of known concentrations of ethyl alcohol.
Factors such as fermentation time, the addition of nutrients for yeast supplementation, the form of yeast used (dry or fresh), and the behavior of each sweet potato genotype used were assessed. The results for °GL were analyzed using a completely randomized design of two factorials (22 and 23).
In the factorial design 22, the variation between two levels was determined for each study factor. The first factor was (1) fermentation time: coded in (-) 72 hours and (+) 96 hours; and the second factor was (2) fermentation medium: encoded in (-) without addition and (+) with nutrients [0.30 g.L-1 of (NH4) 2HPO4; 0.25 g.L-1 MgSO4.7H2O; and 0.20 g.L-1 of ZnSO4], and the cultivar employed was UGA 05.
For factorial planning 23 the factors of greatest interest chosen were: 1 fermentation times coded in (-) 72 hours and (+) 96 hours; 2 cultivars coded as (-) UGA 05 and (+) UGA 77; and 3 is the action of tyeast type encoded in dry (-) biological or (+) fresh biological strains. Each assay was carried out in duplicate and in a randomized performance, and statistical data analysis both for factorial planning 22 and 23, was performed using the Design-Expert® software version 8 (Stat-Ease).
Results
Biomass
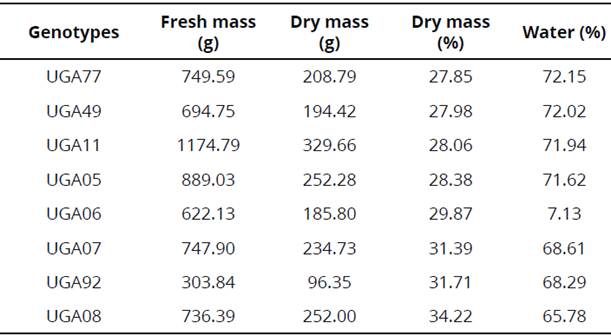
Fresh mass quantities were very promising for sweet potato genotypes UGA11 (1174.79 g), UGA05 (889.03 g), and UGA 77 (749.59 g). Moreover, results related to water loss after the drying process of sweet potato genotypes, indicated high water contents ranging from 65.78 % (UGA 08) to 72.15 % (UGA 77), as shown in Table 1.
Genotype UGA 11 (329.66 g) showed higher dry mass, followed by UGA 05 (252.28 g), UGA 08 (252 g), UGA 07 (234.73 g), UGA 77 (208 g), UGA 49 (1954.42 g), UGA 06 (185.80), and UGA 92 (96.35 g). These data are essential, but must be compared to the amount of fresh mass produced to have a higher percentage of dry mass for ethanol production. The highest dry weight percentages was found in genotype UGA 08 (34.22 %), followed by UGA 92 (31.71 %).
Solely taking into account our data and considering only a small amount of properties, it is possible to state that UGA 08 and UGA 92 have more interesting characteristics than UGA 11, UGA 05, and UGA 07. However, regarding dry mass and its percentage these are only indicative, since reducing sugar and starch must be considered primarily.
Starch and reducing sugar levels
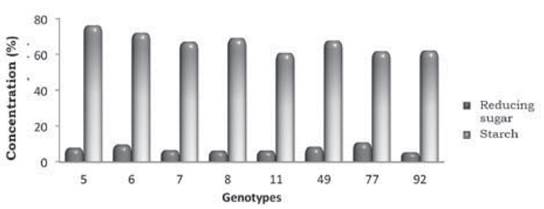
Starch content of the genotypes UGA 05 (76.24 %) and UGA 06 (72.05 %) was the highest compared to the other genotype, and it is expected that with an effective hydrolysis, these cultivars should show great potential for alcohol conversion (Figure 1).
Moreover, for reductive sugar contents, genotypes UGA 77 and UGA 06 obtained the highest results (11.00 %, and 9.70 %, respectively), although levels of 5.48 % were also observed.
Experiment in a factorial scheme 2 2
Percentage of distilled alcohol was verified in relation to total must volume during seven days, where alcohol fermentation showed a production peak after 72 hours of fermentation, with a decreasing in the following hours.
Establishment of alcohol content in °GL for UGA 05 genotype, have allowed the verification of a statistically significant difference for fermentation times; the period of 72 hours of fermentation stood out compared to the one of 96 hours (Table 2).
Table 2 Alcohol content (°GL) in factorial design 22 trials as a function of fermentation time and nutrient insertion in genotype UGA 05
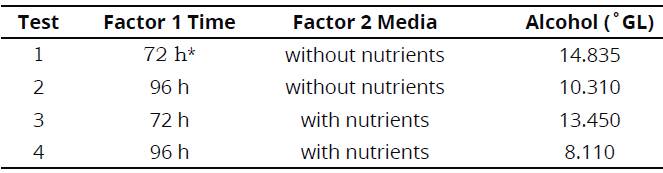
Similarly, the medium without nutrient insertion also showed a statistical difference at the 5 % probability level with the i-test, compared to the medium that received nutrients as nitrogen, magnesium and zinc. However, the interaction between factors 1 (fermentation time) and 2 (fermentation medium) was not significant.
Experiment in Factorial Scheme 2 3
Factorial design 23 was composed of the following variables: 1 (fermentation time of 72 and 96 hrs), 2 (UGA 05 and UGA77 genotypes) and 3 (yeast in dry and fresh biological presentations). Results of alcoholic content expressed in °GL are shown in Table 3.
Table 3 Alcohol content (°GL) in factorial design 23 trials as a function of fermentation time, genotype, and yeast
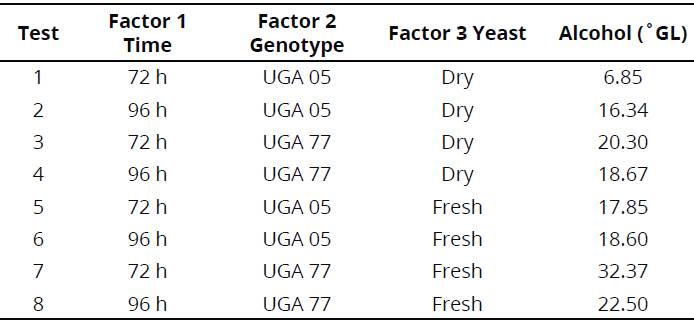
For the eight trials carried out, only the difference among genotypes (effect 2) and yeast form (effect 3) used in the process were significant, with values of 17.10 and 14.59, respectively, for the interval of 95% confidence. Additionally, values obtained in the calculation of the effects show a positive modulus by inferring that the alcohol obtained was better when the assay varied from the coefficients (-) to (+). Subsequently, the effect 2 (genotype: UGA 77) and effect 3 (yeast: fresh biological) showed the best results for alcohol content in °GL (Table 4).
Table 4 Effects calculated for main effects and for factorial design 22 and 23, plus their standard errors
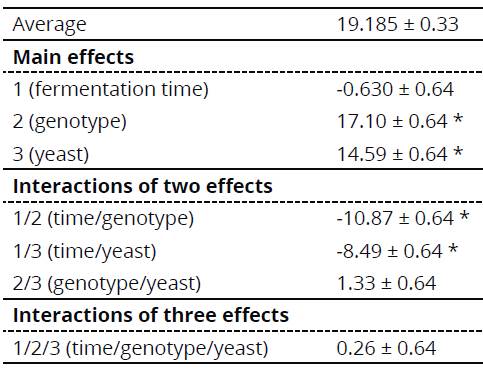
* Significant > .05
Coding of variables for each factor - Factor 1: Time: 72 hours (-), 96 hours (+); Factor 2: Genotype: UGA 05 (-), UGA 77 (+), and Factor 3: Yeast: Biological Dry (-), Fresh Biological (+).
Only time/genotype and time/yeast interactions were significant with values of -10.87 and -8.49. The effects of significant double interactions can be best visualized in Figure 2.
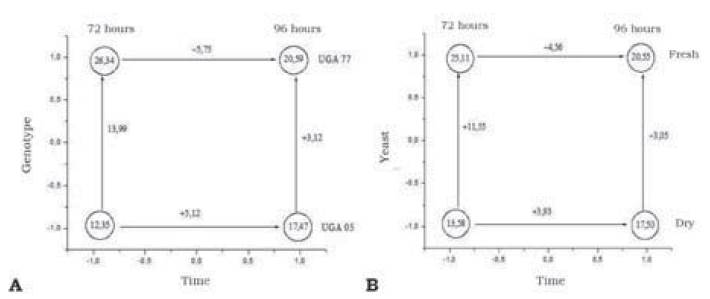
Figure 2 Combination of factors and levels of variation as a function of significant experimental design 23. A. Time (72 and 96 hours)/genotype (UGA 77 and UGA 05); B. Time (72 and 96 hours)/yeast (fresh and dry)
Figure 2A, shows that genotype UGA 77 had achieved a better alcohol production efficiency in 72 hours, while genotype UGA 05 has a better alcohol production efficiency in 96 hours. Additionally, in Figure 2B in time/yeast interaction Saccharomyces cerevisiae in dry biological presentation needs a more extended period for its higher efficiency, whereas in the fresh presentation, the period of 72 hours is sufficient for its performance.
Fresh mass quantities were very promising for genotypes UGA11 (1174.79 g), UGA05 (889.03 g), and UGA 77 (749.59 g), but water percentages in sweet potatoes are not attractive for alcohol production because sugar and starch contents in dry mass are what is basically sought. Hence, there must be a large amount of power generation contained in nutrients, agricultural practices, among others, for the production of biomass (Vitoria & Rodriges, 2016; Kumar, Singh, Surekha & Kumar, 2014).
On the other hand, Oliveira, Blank, Alves, Arrigon-Blank, Maluf & Fernandes (2017), reported an experiment in Sergipe, Brazil with cultivars showing a smaller amounts of dry mass than those found in the first cycle of three months for all cultivars of the experiment. Alternatively, cultivars of Sergipe, IPB 007, IPB 038, IPB 099, IPB 079 were compared with two of the cultivars used in our study, i.e. UGA 08 and UGA 92. Considering the second cycle of the experiment, Oliveira et al. (2017), found that cultivars IPB (34.44 %) and IPB 149 (38 %) had achieved a higher dry weight percentages than UGA 08 and UGA 92 (both showed 32%). In the third cycle with 210 days, cultivars Brazilândia Rosada and IPB 149 remained showing higher values than the genotypes of the highest value in the screening experiment. These higher values found in the Sergipe experimente, suggests a greater adaptation of these genotypes to hot climate and could be a good option for new experiments using this cultivar during hot seasons in Paraná, Brazil. However, the quantity produced by the cultivars UGA 08 and UGA 92 are quite satisfactory taking into account the percentage found, as well as the ones produced by cultivars UGA 11, UGA 05 and UGA 07, considering their dry mass amount; this, because sweet potato has higher ethanol productivity than sugarcane per hectare, depending on the fermentation method used and the dry mass produced.
It is possible to balance these dry mass amounts and percentages, since in a wide production scale there may be a wider difference since it should be mass produced to overcome the sugarcane monoculture production. This must be aimed at mainly in the Brazilian northeastern region due to competition with sugarcane plantations and considering regions with non-favorable conditions for sugarcane cultivation.
Starch and reducing sugar levels
Starch content of the genotypes UGA 05 (76.24 %) and UGA 06 (72.05 %) showed the highest values compared to the other genotype. Nonetheless, these levels were lower than those found by Oliveira et al. (2017), who found values above 18 % for all the genotypes studied; however, in this experiment different genotypes were used, as previously explained.
Low starch values found may be related to management, nutritional deficiency or due to the genetic material itself. However, climate may have also been a decisive factor for the low quantity found in the experiment of Oliveira et al. (2017).
According to this same author, concentration of starch in sweet potatoes influences directly alcohol production, and consequently, higher profits will occur when there is a high starch concentration. However, in an effective hydrolysis process, there may be a better use of the starch produced and better conditions for the cost of alcohol that can counterbalance the results obtained from the national and international average.
Sugar content indicates that molecules have the ideal form to start the fermentation process. Starch content indicates the amount of starch that can be converted into simple sugars after hydrolysis and hence alcohol. Results obtained for sugar content and starch in the eight evaluated sweet potato genotypes were based on the selection of genotypes UGA 77 and UGA 05, which were chosen for the fermentation process, i.e., for alcohol production.
Alcohol production from sweet potato shows a similar processing produce compared to sugarcane. Among its priorities, preparation of the raw material to be used for the manufacturing and its own fermentation system must be highlighted. However, sweet potato in addition to sugar, also has starch; therefore, is necessary to convert the starch to sugars and ferment these, having to add cooking and saccharification procedures from root samples (Martins, Romanzini, Baldin, Trierweiler & Trierweiler, 2016; Oliveira et al., 2017).
Furthermore, another essential factor besides starch and reducing sugar contents is the experimentation phase that will be mentioned below, treated differently by factorial scheme 22 and factorial scheme 23. In 22 time and culture media will be dealt with, and in 23 genotype and yeast to obtain alcohol percentage (°GL) response will be considered.
Experiment in a factorial scheme 2 2
Percentage of distilled alcohol was verified in relation to total must volume during seven days; in this period alcohol fermentation showed a production peak after 72 hours of fermentation, with a decreasing in the following hours. This is due to the fermentation medium becoming unsuitable for the survival of Saccharomyces cerevisiae yeast, causing a decreasing in fermentative activity until new adaptation to the environment is achieved.
Alternatively, fermentation times of 72 and 96 hours were established to verify the joint action of the time in relation to the enrichment of fermentative medium with micronutrients aiming at obtaining a better yeast action. Other authors have evaluated the fermentation time of 24, 72 and 120 hours, and observed higher efficiency in alcohol production in 120 hours, which corroborates the chosen fermentation times. Conversely, Maia, Antunes, Weirich, Borba, Corpo & Borsato (2014), studied fermentation medium supplementation and obtained with a concentration of 0.3 g.L-1 (NH4) 2HPO4 the best alcohol using the jerivá extract media compared to higher concentrations used, i.e. 0.35 and 0.40 g.L-1 (NH4) 2HPO4, although the absence of micronutrients has yet not been evaluated.
Genotype UGA 77 has a better alcohol production efficiency in 72 hours, while genotype UGA 05 needed at least 96 hours. This is because this last genotype has a higher proportion of starch; in this sense, polysaccharides need to break their a and 6 bonds to become a reducing sugar and subsequently be reduced to an alcohol. On the other hand, genotype UGA 77 has a higher content of reducing sugar, which is readily available for alcohol reduction, requiring a shorter processing time.
Time/yeast (Saccharomyces cerevisiae) interaction in dry biological presentation needs a more extended period of time for its higher efficiency, whereas, in its fresh presentation, the period of 72 hours is sufficient for its full performance (Manas, Mishra & Thatoi, 2013). This is because these organisms in their fresh biological form are immediately active and interfere with the speed of reactions. According to the nutritional information provided by the manufacturer, this is due to the conservation, before the fermentative process, of the fresh biological yeast occurring in a higher amount in water (70 %), oxygen and starch.
According to studies carried out by Camargo et al. (2013), evaluating data from 67 polymorphic loci obtained by 10 ISSR primers for sweet potato genotypes, including those evaluated in this study, they observed through the principal components analysis that UGA 77 and UGA 05 genotypes fit in the same group; this indicates their genetic similarity. Although the productivity of UGA 05 was 55.32 % lower than UGA 77, which can be attributed to its origin place, which for UGA 05 is Palmas (TO) and UGA 77 is Turvo (PR), the latter with similar soil and climatic conditions as our study site.
Conclusion
Considering the particularities of the study, it can be concluded that the ideal fermentation time for obtaining sweet potato alcohol is 72 hours, and the addition of micronutrients to the fermentative medium is not necessary; however, the use of fresh yeast excels its use in dry presentation. Given these concerns, sweet potato genotypes UGA 05 and UGA 77, had achieved highest potential to produce alcohol. Although it is emphasized that cultivation conditions, such as crop cycle, climate, productive region, and management, triggers significant changes in the nutritional composition balance, this subsequently interferes a great deal with the ability of these and other genotypes to produce alcohol.