Introduction
Soybean (Glycine max (L.) Merr.) is one of the most important crops in the world economy. The soybean grains are largely used in the agroindustry, mainly in the production of plant oil, animal feed, chemical products and food. Currently, soybean is also an alternative for biofuels. In addition, the soybean crops are very valuable due to their high levels of produced proteins. In Brazil, this fabacea plant is becoming increasingly economically viable due to the inoculation with strains of Bradyrhizobium and co-inoculation with Azospirillum (Hungria, Nogueira & Araujo, 2013).
Nitrogen (N) is the most demanded nutrient by the soybean crop. It is estimated that it takes 80 kg of N to make 1000 kg of grains. Basically, the N sources available to grow soybeans are the nitrogen fertilizers and the biological nitrogen fixation (BNF) (Hungria, Campo & Mendes, 2007).
Today, the bacterial BNF made by the Bradyrhizobium genus has decreased production costs and promoted more sustainable soybean crops. Consequently, it has resulted in an increased competitiveness of this commodity in the world market. This is possible because the bacterial BNF contributes sufficiently to a high soybean production, in addition to replace the N mineral fertilization (Hungria, Franchini, Campo & Graham, 2005; Hungria & Mendes, 2015).
However, the bacterial BNF efficiency depends on various edaphoclimatic factors, especially in the tropical regions. Moreover, management practices such as treatment with fungicides prior to inoculation also interfere in the bacterial BNF efficiency (Hungria, Campo & Mendes, 2007; Campo, Araújo, Mostasso & Hungria, 2010; Zilli, Campo & Hungria, 2010; Zilli, Gianluppi, Campo, Rouws & Hungria, 2010; Hungria & Mendes, 2015).
The BNF is the principal source of N for soybean crops. Bacteria of the Bradyrhizobium genus enter the root via infection of the root hairs, forming nodules (Hungria & Mendes, 2015).
The use of Bradyrhizobium nitrogen fixing bacteria is currently indispensable to grow soybean in the Brazilian fields (Hungria & Mendes, 2015). The efficiency of these microbes has enabled high grain yields, dispensing mineral nitrogen (Alves, Boddey & Urquiaga, 2003). The soybean inoculation with Bradyrhizobium via sowing furrows is a strategy to match the agrochemical seed treatment with the inoculation technique (Hungria, Campo & Mendes, 2007; Hungria, Nogueira & Araujo, 2013; Vieira-Neto, Pires, Menezes, Menezes, Silva, Silva & Assis, 2008a,b; Silva, Carvalho, Schoninger, Monteiro, Caione & Santos, 2011). This management practice is technically recommended and commonly used. However, information on benefits of this practice is scarce, especially in the case of sandy and little organic soils.
The Bradyrhizobium nodulation begins in soybean during the V2 growth stage (2-Trifoliolate), reaches its peak at the R2 stage (complete flowering) and then starts decreasing, when occurs an intense process of mobilization of N from the vegetative parts to the reproductive organs.
The BNF occurs in a sequence of processes that begins with the bacteria adaptation to the plant and culminates with the atmospheric N2 fixation (Hungria & Mendes, 2015). Nodulation begins about 2h after contact between bacteria and roots. The primary nodules develop in the root stretching regions, mainly in the growth zone of small root hairs, which are the main site for infection of nitrogen-fixing bacteria (Hungria, Campo & Mendes, 2007).
According to Hungria, Campo & Mendes (2007)), the activity of nitrogenase decreases at beginning of the formation of beans due to the competition for photosynthetic products among nodules, pods and beans. This competition can restrict the N availability for filling the grains due to the inadequate supply of photosynthetic products to the nodules (Hungria et al., 2007). Hungria et al. (2007) also explain that occurs a decrease in the N concentration in the vegetative parts of the plant during the pod development stage, as the N is mobilized to form pods and beans. So, in the reproductive stage, the N is moved to supply a stronger plant demand, represented by beans and pods, which causes a decrease in the BNF.
Some studies have assessed the performance of inoculations made in the sowing and planting furrows and their results have encouraged the use of this technique in peas, beans and soybeans (Campos, Hungria & Tedesco, 2001; Vieira Neto et al., 2008a,b; Zilli et al., 2010a,b). Regardless the way that the inoculant is applied, the yields from inoculation in areas previously cultivated with soybeans are lower than in areas unprecedentedly cultivated with soybeans (Campos, 1999). However, an average increase of 4.5% in the grain yields was already observed in areas previously cultivated with soybeans (Hungria et al., 2007). The natural occurrence of B. japonicum in uncropped soils is unlikely as soybean is an exotic plant and one of the few species that associates with these bacteria. However, it is possible that some new-introduced strains survive and naturalize in the soil through artificial inoculation or via seeds, even in the Biological nitrogen fixation and agronomic features of soybean (Glycine max (L.) Merr.) crop under different doses of inoculant presence of native strains (Campos, Hungria & Tedesco, 2001).
Direct seed farming is the most commonly used method of planting in soybean fields. It promotes an ecological environment different from the conventional, especially in the first centimeters of soil depth, where decreased temperature fluctuations and increased moisture can positively influence the microbial activity (Campo and Gnatta, 2006). Benefits are also observed in the rhizobium-legume symbiosis, such as, higher number of cells and diversity of Bradyrhizobium, higher number and weight of nodules, greater depth of nodules in soil and higher rates of BNF (Hungria et al., 2007).
In direct seed farming, the inoculation made in the sowing furrow can favor nodulation because rhizobia are better positioned next to roots right after the seedling emergence (Voss, 2002). Little is known about the differences between inoculation made in the sowing furrow and mixed in the seed. Similarly, there is little information on the effects of inoculation comparing soybean planting areas with areas not planted with soybeans.
Therefore, this study aims to analyze the effect of inoculating nitrogen-fixing bacteria, comparing doses of the Bradyrhizobium japonicum liquid inoculant with and without Ca+B on the soybean ((Glycine max (L.) Merr.) crop development and growth.
Material and Methods
Plants were grown for 90 days in 80 plastic pots, each containing 3 kg of sand and vermiculite (v/v) autoclaved at 121oC for 1h. The experiment was conducted in a completely randomized design with five replications in a greenhouse. The 8 treatments were defined as follows: 1-) 0 inoc.+ 0 N + 0 Ca+B; 2-) 600 mL ha-1 inoc. + 0 N + 0 Ca+B; 3-) 1200 mL ha-1 inoc. + 0 N + 0 Ca+B; 4-) 0 inoc. + 100 kg N ha-1 + 0 Ca+B; 5-) 0 inoc. + 100 kg N ha-1 + 1 L Ca ha-1 (Ca 41%) + 0,4 L B ha-1 (B 14%); 6-) 0 inoc.+ 0 N + 1 L Ca ha-1 (Ca 41%) + 0,4 L B ha-1 (B 14%); 7-) 600 mL ha-1 inoc. + 0 N + 1 L Ca ha-1 (Ca 41%) + 0,4 L B ha-1 (B 14%); 8-) 1200 mL ha-1 inoc + 0 N + 1 L Ca ha-1 (Ca 41%) + 0,4 L B ha-1 (B 14%).
The experiment started by October 2013. Three soybean seeds were sowed in each pot uniformly. The COODETEC CD 202 cultivar was recommended for this region and therefore used in this study. The Stoller Masterfix liquid inoculant containing Bradyrhyzobium japonicum (SEMIA 5079 + SEMIA 5080) was applied to treatments with inoculation. The calcium and boron used on the soybeans seeds at the time of sowing in this experiment were obtained from the Prime Agro Company, in the proportions of 1 L Ca ha-1 (Ca 41%) + 0.4 L B ha-1 (B 14%); recommended by the Prime Agro Company. Seeds were covered with 1-2 cm of autoclaved substrate and wet with nutrient solution. The treatments were irrigated as required, with nutrient solution according to Hoagland & Arnon (1950); however with and without N, following each treatment definition and the N was partitioned during the experiment.
In the V8 vegetative stage, 5 pots per treatment (40 pots) were selected to determine the chlorophyll index of the plants. The chlorophyll index of 5 completely developed leaves was measured in each pot by a portable chlorophyll meter (ClorofiLOG® CFL 1030), operating in the middle of the leaves, according to manufacturer instructions (Falker, 2008).
The plant heights (cm) and the root lengths (cm) were obtained with a tape measure. The nodules and leaves were numbered manually. The shoot, nodules and root were separated from plants and dried in an oven at 65°C for 48h. Then, these parts were weighted in a semi-analytical balance to determine the dry weight of nodules, root and shoot.
Subsequently, the shoot was ground to determine the level of N in the shoot (NS) with sulfuric acid digestion, followed by distillation according to the Kjeldahl method, as described by Silva (2009).
In the R8 reproductive stage, 5 pots per treatment (40 pots) were selected to determine the number of pods per plant (NPPL); number of grains per pod (NGP); and number of grains per plant (NGPL).
Data were subjected to ANOVA and means were compared by the Tukey test (p<0.05), using the SPSS software version 16.0 for Windows (SPSS Inc., Chicago, IL, USA).
Results
The inoculation with Bradyrhizobium at sowing increased the number and dry weight of nodules (Table 1). The number and dry weight of nodules increased significantly in treatments with inoculation at sowing in comparison to treatments without inoculation (Table 1).
Table 1 Values of P in ANOVA1 of number of nodules (NN), dry matter of nodules (DMN), root dry matter (RDM), shoot dry matter (SDM) and total dry matter (TDM).
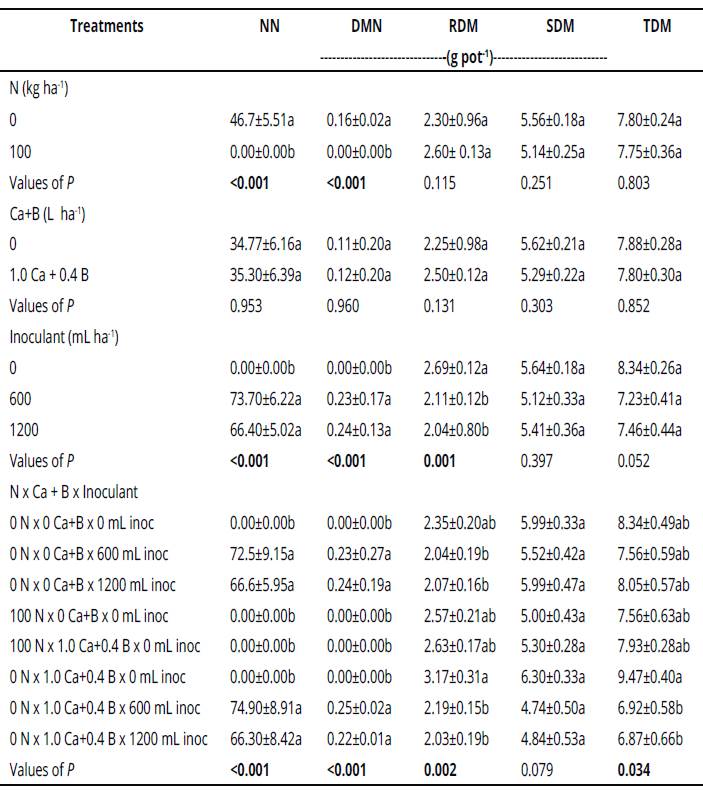
*Mean values (± standard error); different letters in the same column differ significantly by the Tukey test (p < 0.05). Values of P in bold are significant (p < 0.05).
The plants become taller in the treatments with Bradyrhizobium inoculation at sowing and addition of Ca+B (Table 2). The increment in height was 67% compared with the control, with inoculation (600 mL ha-1) and addition of Ca+B.
Nitrogen content in the shots was increased by Bradyrhizobium inoculation at sowing (Table 3).
The soybean yield increased significantly in the treatment with 600 mL ha-1 inoculant and addition of Ca+B (Table 3). This result is demonstrated by the values of NPPL, NGP and NGPL found for this treatment, which are higher than those of other treatments (Table 3). In this case, the soybean yield increased at 10% in terms of grain production.
Table 2 Values of P in ANOVA2 of plant height, root length (RL), total plant length (PL), number of leaves (NL) and ratio shoot/root (S/R).
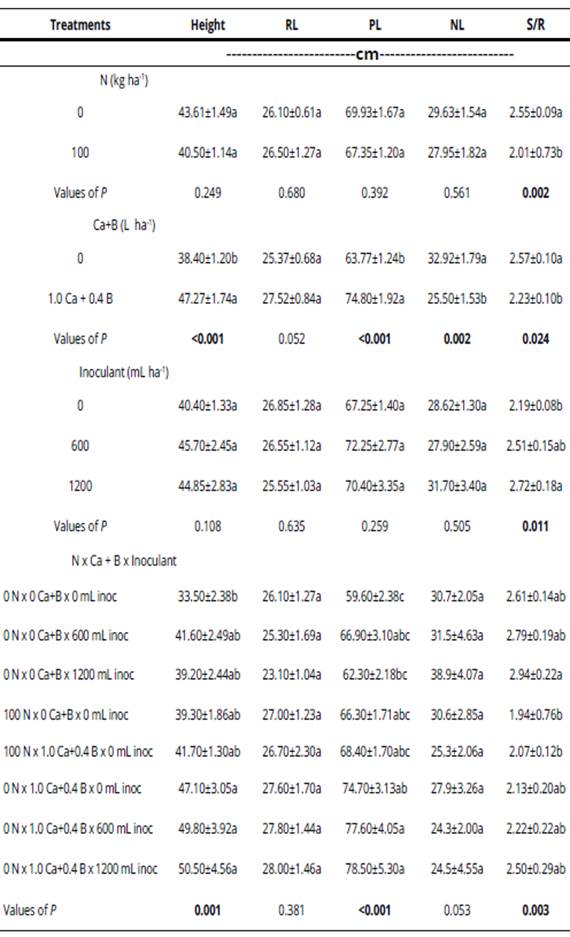
*Mean values (± standard error); different letters in the same column differ significantly by the Tukey test (p < 0.05). Values of P in bold are significant (p < 0.05).
Table 3 Values of P in ANOVA* of N content in the shoot (NS), index of chlorophyll (IC), number of pods per plant (NPPL), number of grains per plant (NGPL) and number of grains per pod (NGP).
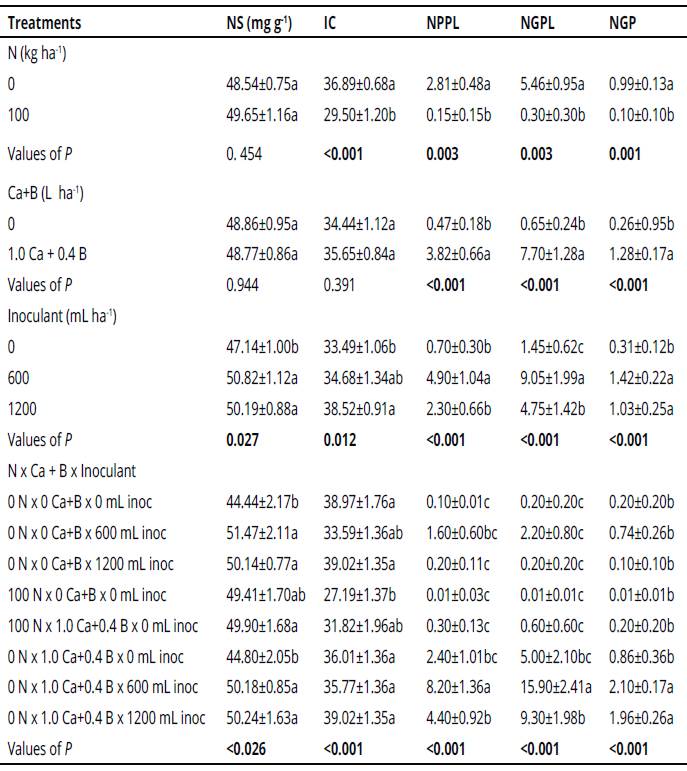
*Mean values (± standard error); different letters in the same column differ significantly by the Tukey test (p < 0.05). Values of P in bold are significant (p < 0.05).
Discussion
The number and dry weight of nodules were increased by BNF inoculation. It can be explained by the highest efficiency of the inoculant when applied at sowing, verified by the highest number of nodules. Similar results were also observed in other studies (Viera-Neto et al., 2008a; Campos & Gnatta, 2006). Benefits are also observed in the rhizobia-legume symbiosis; in the case of soybean, it is found a higher number of cells and greater diversity of the Bradyrhizobium genus, higher number and weight of formed nodules, deeper distribution of nodules across the soil profile and higher BNF rates by the soybean plants (Hungria, Nogueira & Araujo, 2013).
The amount of nodules per plant found in the treatment with 600 mL ha-1 inoculant demonstrate that this dose is appropriate for supplying the N required by the plant for a normal development. Moreover, the application cost of 600 mL ha-1 inoculant is lower than that for 1200 mL ha-1.
Plants were higher when inoculated with BNF and addition of Ca+B. Moretto & Viecelli (2012), also observed a significant increasing in the plant height with application of higher doses of Ca (1.65 L ha-1 and 2.50 L ha-1). In this study, the increased plant height from the Ca application can be understood by the increased soil pH, which favored the availability of nutrients in the soil solution and consequently a better use of these by the plant. However, this is an untested hypothesis.
The soybean crop yield increased significantly in the treatment with 600 mL ha-1 inoculant and addition of Ca+B. In this case, the soybean yield increased at 10% in terms of grain production. Campos & Gnatta (2006), observed that the inoculation made in the sowing furrow could minimize some deleterious effects of the seed chemical treatments. They also verified a better crop performance when high amounts of Bradyrhizobium cells were inoculated together with the soybean seeds (up to 2.5 million cells per seed). In a different way, Silva et al. (2011), did not find significant differences between the two doses of B. japonicum inoculant (3 and 6 mL kg-1 seeds) tested with mineral N; both in the first year of using the soil for soybean crop and in the following year.
The productivity of four soybean cultivars inoculated with BNF was evaluated as a function of foliar mineral fertilizer application containing 8% Ca and 2% B in R1 stage (early flowering 50% of flowering plants) and R3 (final flowering, pod up to 1.5 cm in length) (Souza, Sá, Carvalho & Simidu, 2008). The productivity was significantly higher when the solution based on Ca and B was applied. In the present study, soybean yield increased at 10% in terms of grain production when with 600 mL ha-1 inoculant and addition of Ca+B being a viable technology to the soybeans producers.
Conclusion
The dose of600 mL ha-1 of the inoculant combined with Ca+B significantly increaded number of pods per plant, number of grains per pod and number of grains per plant. The inoculation of soybean plants with Bradyrhizobium at 600 mL ha-1 along with applications of Ca+B is a viable practice due to the practicality of operations and the increased soybean yield up to 10%.














