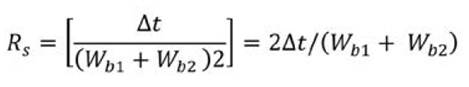Introduction
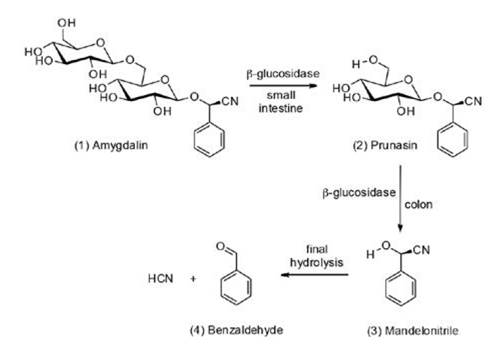
Cyanogenic glycosides are cytotoxic secondary metabolites widely diffused in the vegetal kingdom, with a presence in more than 2650 species, where they exert a defensive action against plant pathogens and herbivores by releasing poisonous hydrogen cyanide through enzymatic hydrolysis (Vetter, 2000). In particular, amygdalin [D-mandelonitrile-b-D-gentiobioside, (1) in Figure 1] is a popular and well-documented cyanogenic disaccharide highly abundant in the seeds of apricots, almond, cherries, plums, peaches, apples, and other rosaceous plants used in human nutrition (Viorica-Mirela, Socaciu, Jianu, Florica, and Florinela, 2006), as well as in several traditional Chinese medicines (TCM): loquat (Eriobotrya japónica Lindl.) flower (Chunhua, et al., 2007), Semen pruni armeniacae and Semen prunipersicae (Li, et al., 2016).
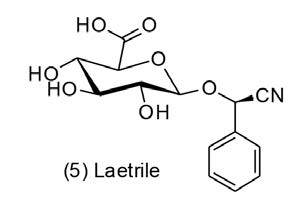
Amygdalin has been the focus of a controversial debate both within the scientific community and in civil society for more than 40 years (Blaheta, Nelson, Haferkamp, and Juengel, 2016). On the one hand, this compound is attributed highly therapeutic effects, such as anti-inflammatory and analgesic actions (Yang, et al., 2007), anti neurodegenerative diseases agency, and as an alternative treatment for asthma, bronchitis, emphysema, leprosy and diabetes (Zhou, et al., 2012). But more importantly, amygdalin is cataloged as an antineoplastic agent (Moon, et al., 2015), especially since the early 1970s, when its semisynthetic derivative Laetrile (Laevo- mandelonitrile - Figure 2) or ‘Vitamin B17’ (not a vitamin), sometimes named improperly as ‘amygdalin’, was commercially launched as an anticancer drug (Blaheta, et al., 2016).
On the other hand, amygdalin is cataloged as a dangerous substance capable of producing highly toxic effects in humans, even death (Sauer, et al., 2015); as matter of fact, the US Food and Drug Administration (FDA) has banned its use and commercialization since 1977 (Kennedy, 1977). Regardless of the position on the therapeutic benefits of amygdalin, the vast majority of research agrees that information on this compound is still scarce and many pharmacological and biochemical studies have yet to be performed (Blaheta, et al., 2016).
Enzymatic catabolism of amygdalin in mammals occurs in two stages (see Figure 1): a ‘first pass’ to produce (2) prunasin (D- mandelonitrile b-D- glucoside) and a second path, which gives rise to mandelonitrile (3), which then hydrolyzes to benzaldehyde (4), which induces an analgesic action, and hydrocyanic acid, which is an antineoplastic (Kuroki and Poulton, 1986). Based on this, some studies have concluded that oral administration of amygdalin in mammals should be more potentially toxic than intravenous injections (Moon et al.; 2015). Interestingly, however, the communities with the lowest rates of cancer worldwide, such as the Abkhazians, the Hopi and Navajo Indians, the Hunzas, the Eskimos and the Karakorum, have in common a diet rich in products with a high content of amygdalin (Enculescu, 2009).
Original extraction of amygdalin from bitter almond (Prunus amygdalus Batsch) was reported in 1830 by the French chemists Robiquet and Boutron (Robiquet and Boutron, 1830). Due to the high solubility of amygdalin in polar solvents, solid-to-liquid extractions in combination with High Performance Liquid Chromatography (HPLC) has become in a very convenient procedure for the isolation and determination of this compound from natural products. Reflux/HPLC methodology, for instance, has been employed for determination in seeds of Prunus armeniaca (Armeniacae semen) (Yan, Tong, Li, and Lou, 2006), bitter almond (Bolarinwa, Orfila, and Morgan, 2014) and apples (Bolarinwa, Orfila, and Morgan, 2015); meanwhile ultrasonic/HPLC has been employed for determinations in Loquat flowers (Chunhua et al.; 2007) and Soxhlet/HPLC treatments for apricot, plum and peach oils and kernels (Viorica- Mirela, et al., 2006). In this regard, Lv, Ding, and Zheng (2005) reported a comparative study for extraction of amygdalin from the TCMs Apricot- kernel and Prunus tomentosa thunb using three different methodologies: ultrasonic extraction by methanol, Soxhlet extraction by methanol, and reflux extraction by water (Lv, et al., 2005). In such study, refluxing by water was found to be the most effective extraction procedure.
In this work we report for the first time the isolation and HPLC determination of amygdalin in seeds of apples Malus pumila Mill. by using three different solid-to-liquid methodologies: reflux with ethanol, Soxhlet with methanol and microwave radiation in methanol. The aim of this study was not only to establish an optimal method for the extraction of amygdalin from the seeds of this species but also to differentiate the content of this cyanogenic glycoside contributed by both the dicotyledon and the episperm tissues.
Materials and methods
Collection and preparation of vegetal material
The seeds used were obtained from apples produced by small farmers in the Department of Boyacá (Colombia), in the village of San Antonio Norte (2620 m.a.s.l), in the jurisdiction of Duitama city. The main commercial destination of these fruits is the capital Bogota, where they are used for human consumption. Plant materials were taxonomically classified as belonging to the Family: Rosaceae, genus: Malus, species: Malus pumila Mill. by the Herbarium of the Universidad Pedagógica y Tecnológica de Colombia (collection code: UPTC 021203). In all, 240 fruits were used throughout this study, which were distributed in two stages of research: 190 specimens were required to define the optimum conditions of the three amygdalin extraction treatments and the HPLC method presented here, and 50 specimens for the final quantification of the compound. All fruit units were in healthy condition, and none presented evidence of thermal, mechanical or microbiological (fungal or bacterial) damage. Seeds were manually extracted from plant material using latex gloves and plastic knives and stored in sealed polypropylene plastic bags (Ziploc®) at 4 °C until use.
The seeds were dehydrated at 40 °C for 24 h and their tissue posteriorly separated into the episperm and dicotyledon parts, and pulverized using an electric mill (Wilmington, NC 28405). The study of efficiency of amygdalin extraction was performed by considering only the dicotyledonous tissue and then the best extraction procedure applied to the episperm. In total, 2.55 g of dried seed material was extracted from the 50 apples selected for determination of amygdalin, and distributed in three equal parts, i.e.; 0.85 g for each solid-liquid extraction treatment.
Solid-to-liquid extractions
Hereinafter, the reflux, Soxhlet and microwave extraction methods will be referred to as M1, M2 and M3, respectively.
All solvents used in solid-liquid extractions were HPLC grade (purity 2 99.6%, J. T. Baker). For the reflux extractions, 50 mL of ethanol was used per gram of sample and each reflux run for 100 min at a temperature of 70 - 90 °C, under stirring at 320 rpm. The extracts obtained were filtered under vacuum using nylon 0.45 mm pore (Acrodisc®, 13 mm). Soxhlet extractions were performed over 16 h at 65 - 70 °C using 200 mL of methanol for the 0.85 g of sample, with stirring at 180 r.p.m. These extracts were concentrated using a rotary evaporator until a concentration similar to that obtained in the reflux extracts was reached.
Microwave extractions were performed as follows: First, the 0.85 g of sample was immersed in 10 mL of chloroform at room temperature for 15 min under stirring at 280 rpm. Second, the mixture was gravity-filtered using paper (0.45 mm pore) and the solid material was dried at room temperature for 20 min. Third, 20 mL of methanol was added to this material and heated by microwave (600 W) to 50 - 70 °C for 90 s. The microwave extracts were finally gravity filtered on paper.
HPLC determination
The extracts obtained from the above-mentioned three solvent methods (M1 to M3) were quantitatively analyzed by HPLC. For this, a high performance liquid chromatograph Shimadzu Model LC-2030 with a solvent delivery, quaternary gradient, auto sampler injector and a dual UV detector settled at 218 nm was used. For the analytical separation a Macherey-Nagel C18 column (150 mm x 5 mm x 5 pm) was selected. Other chromatographic conditions included a column temperature of 30 °C, an injection volume of 100 pL, an eluent flow of 1.3 mL/min, a methanol/water 20:80 (v/v) eluent composition and a total elution time of 10 min.
Molecular characterization
The molecular characterization and verification of the purity of the extracted amygdalin was performed by FTIR, UV-vis and 1H and 13C NMR (mono and bi-dimensional) spectroscopies, using Shimadzu IR-Prestige-21, Shimadzu UV- 1800 and Bruker 400 MHz equipment models, respectively.
UV-vis spectra were measured on amygdalin extracts collected after chromatographic separation on the HPLC equipment at elution time. Dual UV- vis measurements were performed in the range of 190-400 nm using the mobile HPLC phase as background. FTIR and NMR characterizations used the crystalline-purified solid form of the extracted amygdalin. By doing this, we avoided its degradation by hydrolysis and confusing signals arising from the vegetal extracts.
The crystallization of the amygdalin was achieved by the following procedure: First, the solid material obtained after milling was washed three times with chloroform and allowed to dry at room temperature for 20 min. Next, this material was immersed in 30 mL of diethyl ether at 40 °C and cooled slowly for 24 h to about 2 °C. Finally, a very pure white sample of amygdalin was obtained by evaporation of the solvent. FTIR spectra were directly recorded on purified solid samples using ATR methodology, while for NMR measurements the solid samples were dissolved in DMSO-d6.
Results and discussion
Precision of the HPLC method
The precision of the HPLC method was determined by repeated analysis (n = 5) of amygdalin extracts obtained using treatment M1. As can be seen in Table 1, a maximum coefficient of variation (%CVmax) of 0.0949, as well as detection (LD) and quantification (LQ) limits of 0.0505 and 0.0548 mg/g, respectively, were determined; thus indicating a high reliability of the method.
Efficiency of the chromatographic separation
The efficiency of the chromatographic column to separate the amygdalin from the parent extracts was determined by calculating the value of the theoretical plates (N) in each of the three extraction methods (M1 to M3). Even though the extracts used to estimate the number of theoretical plates come from the same vegetal sample (dry seeds), different values of N were obtained among the three extraction methods. The highest number of theoretical plates (N = 175.75), thus the highest separation efficiency, was obtained for M1. In case of methods M2 and M3 values of N = 59,80 and 85,46 were found, respectively.
A very good chromatographic resolution (Rs) of 1.02 min was calculated for the column using bandwidths of W b1 = W b2 = 0.3 min and a delta time (At) of 0.306 min (obtained from M2), according to the equation 1:
Quantification of amygdalin in solid-liquid extracts
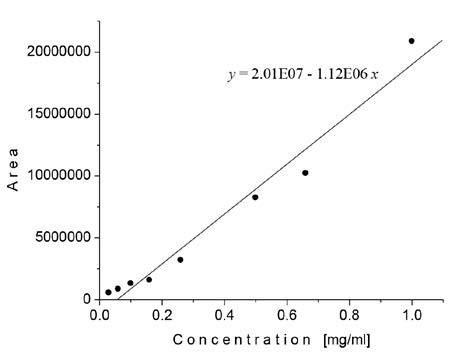
For the quantification of the amygdalin content in extracts, an external calibration curve was constructed using a Sunfood® certified seed pro duct containing 30 mg of amygdalin per 4.8 g. As presented in Figure 3 a good regression equation (y = 2.01E07 - 1.12E06x, R 2 = 0.98437) for HPLC determination of amygdalin was obtained, where y was the peak area of amygdalin and x was the amygdalin concentration (mg/mL).
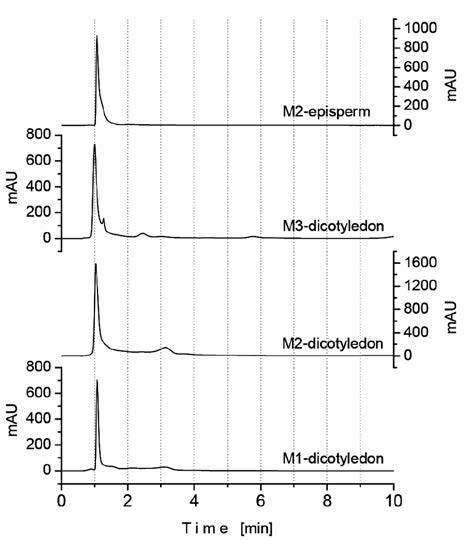
Chromatograms of amygdalin-containing extracts obtained from dicotyledon tissue using M1, M2 and M3 methods and from episperm tissue using M2 are presented in Figure 4. All the chromatograms are characterized by a single amygdalin peak around 1 min retention time: 1.16 (M1 and M2) and 1.04 (M3), indicating not only a high extraction specificity of the three treatments but also very fast separation of amygdalin by our HPLC method. Other HPLC methods, employing also methanol/ water mixtures as eluent in a similar ratio, have reported much longer retention times ranging from 4 to 25 min for the separation of amygdalin from natural products (Chunhua, et al., 2007; Yan, et al., 2006; Bolarinwa, 2014; Viorica-Mirela, 2006).
Concentration contents of amygdalin in extracts, expressed in mg/mL, as well as in the dry vegetal material (seeds), expressed in mg/g, are listed in Table 2. The largest amount of amygdalin extracted from seeds of apples Malus pumila Mill. was achieved by the M2 method. By employing this method, a total of 2.28 mg of amygdalin was extracted per gram of dry-seed material, i.e. 0.23% w/w, of which 61.4% (1.40 mg/g) was supplied by episperm tissue. This content of amygdalin (2.28 mg/g) is close to half the range of 0.95 to 3.91 mg/g recently reported for seeds of other apple varieties (Bolarinwa, 2015) (see Figure 5) and also similar to that reported in grains with an average content of glycosides, such as plum (0.26%) and apricot (0.30%) (Voldrich and Kyzlink, 1992).
Table 2 Amygdalin content in extracts and in dry vegetal material of seeds of apples Malus pumila Mill. determined in dicotyledon and episperm tissues using M1, M2 and M3 extraction methodologies.
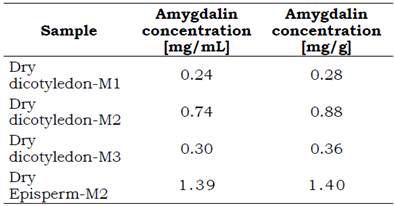
Concentrations of amygdalin in dicotyledon of 0.28 and 0.36 mg/g were determined using M1 and M3, corresponding to extraction efficiencies of 31 and 41%, respectively, of that of the M2 method.
M2, however, requires a much greater investment of energy, time and solvents compared to M1 and M3 (see Materials and methods section), making this method unsuitable for large-scale applications, and environmentally unfriendly.
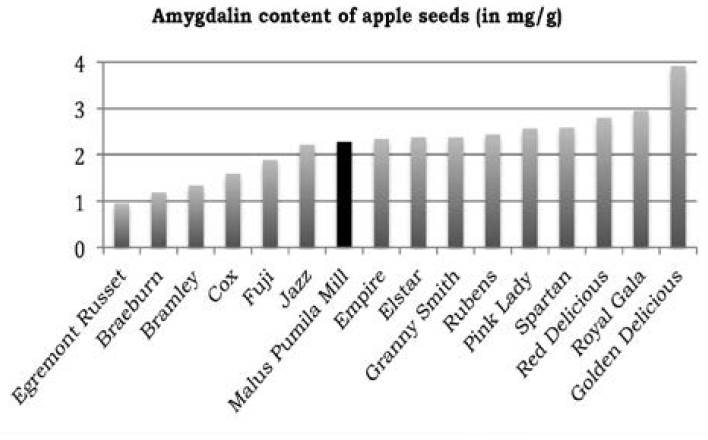
Figure 5 Concentration of amygdalin in seeds of Malus pumila Mill. compared to that reported in other apple varieties (Bolarinwa et al., 2015).
Spectroscopic characterization of amygdalin
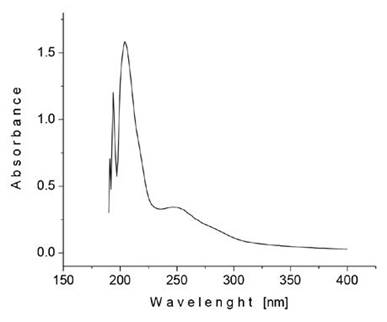
The UV-vis spectrum of a sample of dicotyledon extract collected after 1.1 to 1.2 min of column separation time, showing maxima around 190, 205 and 250 nm, is presented in Figure 6. These absorption maxima were found to be quite similar to those reported at 184, 203 and 256 nm, attributed to aromatic p ] p* transitions, for solutions of amygdalin in methanol (Savic, Nikolic, Savic-Gajic, Nikolic, Ibric and Gajic, 2015).
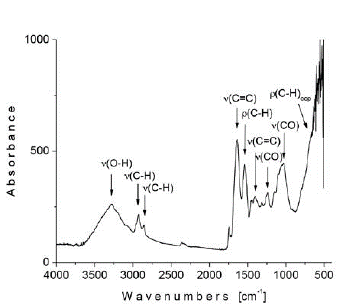
The FTIR spectrum of isolated amygdalin, crystallized from seed material by following the procedure described in section 2.4, is presented in Figure 7. In this spectrum, a broad band of medium intensity centered at 3288 cm-1 was assigned to the OH vibrations of the glucose moiety; meanwhile, the absorptions of lower intensity at 2926 and 2855 cm-1 were attributed to aromatic and aliphatic C-H stretching modes respectively. The bands at 1636 and 1400 cm-1 are the result of C=C stretching vibrations from the benzene ring, while the absorption at 1540 cm-1 was assigned to the deformation mode of the C-H groups. The stretching vibrations for C-O bonds, from ether and hydroxyl groups, are observed at 1240 and 1030 cm-1 respectively. Also, the intense absorption band at 690 cm-1 was assigned to out-of-plane C-H bending vibrations. No band was observed around 2200 cm-1 for CN group given its very low expected intensity.
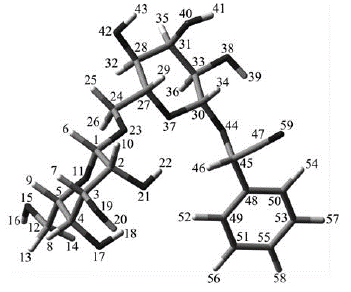
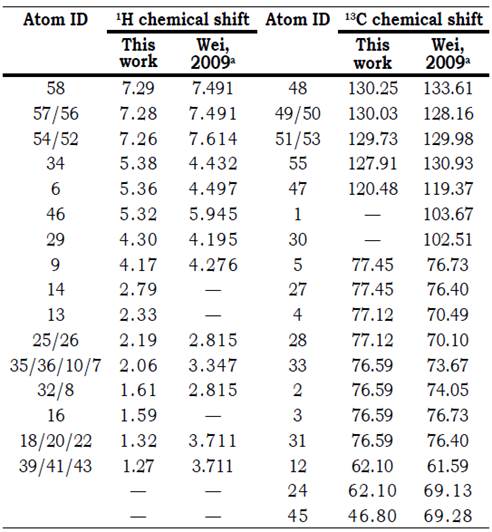
Additional structural characterization of isolated amygdalin was performed using NMR spectroscopy. 1H-NMR and 13C-NMR nuclear resonances of crystalline amygdalin diluted in DMSO-d6, are listed in Table 3. All the chemical shifts, proton integrations and multiplicities were found to be very similar to those reported previously for the compound (Wei, Xie, and Ito, 2009), thus confirming its identity as cyanogen glycoside amygdalin. For an easy structural location of the 1H and 13C nuclear resonances shown in Table 3, a molecular model of amygdalin including atomic labels is presented in Figure 8.
Table 3 1H and 13C NMR chemical shift assignments of amygdalin.
a Chemical shifts recorded in CD3COCD3 at 600 MHz.
Conclusion
In this work the content of the cyanogen disaccharide amygdalin in seeds of the Colombian apples Malus pumila Mill, differentiating the contribution given by the dicotyledonous tissue from that of the episperm, was determined for first time. Extraction of amygdalin was performed using three different solid-to-liquid methodologies: (1) reflux extraction in ethanol at 70 to 90 °C for 100 min, (2) Soxhlet extraction with methanol at 65 to 70 °C for 16 h, and (3) an extraction in methanol at 50 to 70 °C for 90 s using microwave radiation. The highest content of amygdalin was attained using Soxhlet treatment, which is attributed to its high effectiveness given the extended time of cycle extractions. However, microwave radiation is best recommended for the extraction of amygdalin given its very short time and small amount of solvent required. For the determination of amygdalin content, an optimized HPLC method, requiring around only 1 min elution time, was developed. In addition, the structure of the isolated amygdalin was confirmed by NMR (1H and 13C), FTIR and UV-vis spectroscopies.