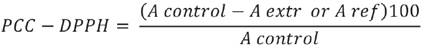Introduction
Many studies have reported that cacao beans contain abundant amount of flavan-3-ols as major compounds, these are identified as catechin and epicatechin (Donovan et al., 2012). In 2012, a scientific book stated five important cacao effects on: (1) vascular endothelial function and platelet activity, (2) antioxidant and anti-inflammatory, (3) lipid and lipoprotein metabolism, (4) insulin sensitivity, (5) cerebral blood flow and neurocognitive functioning (Donovan et al., 2012).
Dark chocolates contain more cacao and show a higher anti-oxidant capacity, a bigger phenolic content and contain more monomeric procyanidins than milk chocolate (Da Silva et al., 2015). Vinson y Motisi (2015) discovered a significant linear relationship between the percentage of cacao solids and the anti-oxidants. Even though there were individual differences in the polyphenol content in samples of the same chocolate category, a good correlation between the anti-oxidant potency and the declared cacao content was observed. Therefore, the information about cacao content can be a reliable indicator of the anti-oxidant potency of chocolates produced in Serbia (Todorovic et al., 2015).
Currently there is little information about the anti-oxidant properties of the dark chocolates that are sold in Lima (Peru). That is why the goal of this paper was the evaluation of the total polyphenol content (TPC), the total flavonoid content (TFC), the anti-oxidant capacity (AC) and the reducing power (RP) of eight dark chocolates that are sold in Lima, as well as to relate the cacao content to the anti-oxidant properties.
Materials and methods
Population and sample size. Random dark chocolate samples supplied by different chocolate companies were purchased from local market in Lima (Peru) in 3 months period from March to May 2015. The percentage and origin of cacao, either domestic or imported were printed on the chocolate's labels. Eight random samples were selected, from different large-scale chocolate companies, two of domestic origin and six of imported origin. The samples had different cacao contents (Chire, Valdivia, Orihuela and Ureña, 2017).
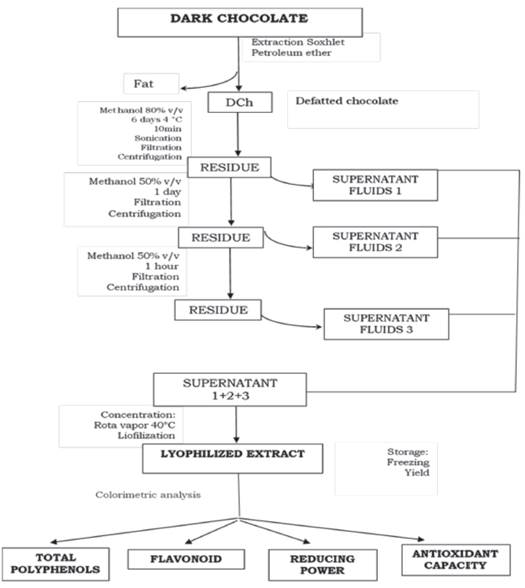
Sample treatment. The chocolate samples were dried using the 931.04 method (Horwitz and Latimer, 2016) and then they were defatted with petroleum ether using the Soxhlet 963.15 method (Horwitz and Latimer, 2016). The fat was removed and defatted chocolate (DCh) were obtained (Hu et al., 2016). The solvent excess was removed at room temperature. DCh showed light brown color, they were packaged in polyethylene bags and stored in refrigeration until the extraction process was conducted.
Preparation of lyophilized extracts from DCh. The was carried out using basically the method described by Belscak et al. (2009), only slight modifications were introduced (Table 1). For the first extraction, 80% methanol (v/v) was used for six days. The sample was protected from light at 4 °C and then it was placed in an ultrasound bath (BRANSON 1510) for 10 minutes at room temperature, only once. After this, it was filtered (Whatman paper No. 4) and centrifuged (SORWALL) at 5000 r.p.m. for 10 minutes. For the second extraction with 50% methanol was carried out for 24 hours and for the third extraction the same quantity of methanol was used for only one hour under the same conditions. The filtrates from the three extractions were combined, then centrifuged at 7500 r.p.m. and 5 °C. After this the extracts were concentrated under reduced pressure in a rotary evaporator (BOECO) at 540 r.p.m. and 40 °C, then the extracts were lyophilized (RIFICOR) (Figure 1). The lyophilized extracts (LE) showed a color that varied between brown and yellowish brown and had a soft touch consistency. The yield was obtained by weight difference and was stored in refrigeration. The chocolates’ characteristics and the extracts’ details are described in Table 1. The lyophilized extracts (LE) were expressed based on one gram of chocolate (Ch).
Source: Own elaboration
Figure 1. Experimental scheme for the assessment of the anti-oxidant properties of chocolate.
Total polyphenol content (TPC). The analysis was carried out according to the technique described by Singleton, Orthofer, Lamela-Raventos (1999), only slight modifications were introduced. LE were weighed and dissolved in bidistilled water to find a compatible dilution with the absorbances of the calibration curve. The aqueous solutions for each of the extracts were prepared from 0.35 to 0.91 mg/mL. A 100 μL of the extract solution was mixed with 150 μL of diluted Folin-Ciocalteu reagent, five minutes later, 150 μL of 20% sodium carbonate was added and it was filled with bidistilled water to complete an amount of 1 mL. After 30 minutes in the dark, spectrophotometer readings were recorded (Spectroquant, Pharo 300 Merck) at 760 nm. Each of the eight LE was analyzed by triplicate. A standard calibration curve was made (gallic acid of 1.0-7.5 mg/mL, y = 0.0848x + 0.0291 and R2 = 0.9928) and the concentration was expressed in mg Gallic Acid Equivalent in one gram of chocolate (mg GAE/g).
Total flavonoid content (TFC). The method of Lee, Kim, Lee and Lee (2003) was used to determine the TFC, only some modifications were introduced. The LE were dissolved in a mixture of ethanol and water at 50% v/v and from each sample 0.2 mL were measured, then 0.06 mL of 5% NaNO3 were added. Six minutes later 0.06 mL of 10% AlCl3.6H2O were added. Again, six minutes later 0.8 mL of 1M NaOH were added. The samples were stirred for 30 seconds each time a reagent was added. Finally, the mixture was diluted to 2 mL and two minutes later the absorbance was measured at 510 nm. A calibration curve was made with catechin (2.5 - 17.0 Hg/mL, y = 0.0301x + 0.0038 y R2 = 0.9907). The results were expressed as mg Catechin Equivalent in one gram of chocolate (mg CE/g).
Determination of anti-oxidant capacity
Capture capacity of the radical DPPH. The anti-oxidant capacity was determined by the method described by Brand-Williams, Cuvelier and Berset (1995). A methanolic solution of the 0.1 mM 1,1-diphenyl-2-picrylhydraz (DPPH) radical was prepared. Each of the eight LE were used to prepare five aqueous solutions in increasing concentrations. The reaction tubes contained 0.40 mL of the LE in aqueous solution with 0.800 mL of the methanolic DPPH solution and were stirred for 30 seconds. After 30 minutes in the dark at room temperature, the absorbance was recorded at 517 nm (A extr spectrophotometer). In parallel, a control tube was prepared; another blank sample was used to correct the absorbances of the extracts, the tests were run in triplicate. Simultaneously, a calibration curve was made using 6 -hy dr oxy- 2,5,7,8- tetr amethylchr oman- 2 - carboxylic acid (Trolox) (Aref), its concentrations were in the range of 1.8 - 5.4 mg/mL. The results were expressed as IC50, these values were obtained by designing the linear equation in the graph, for the percentage of the capture capacity of the DPPH radical (PCC-DPPH) versus the extract concentration of each sample (mg/mL extract). The same procedure was used for the trolox (y = 18.275x - 6.4 y R2 = 0.9906). El PCC-DPPH was obtained by the following relationship:
Capture capacity of the radical ABTS. The test was performed as described by Re et al. (1999). The first step was to generate the ABTS radical cation [2,2’-Azino-bis (3-ethylbenzthiazoline-6-sulfonic acid)]; 7 mM of the radical were prepared, this was kept in the dark at room temperature for 16 hours and then it reacted with potassium persulfate at 2.45 mM. As a next step, the solution was adjusted with water at 0.7 ± 0.05 absorbance at 734 nm. The reaction mixture consisted of 0.98 mL of the ABTS solution with 0.02 mL of the extracts dissolved in water with concentrations from 25 to 400 mg/mL. The mixture was stirred and incubated for seven minutes in the dark at room temperature and then the absorbance was measured at 734 nm. Trolox (1-4 Mg/mL) was used as reference standard. The results were expressed in terms (mol Trolox Equivalent Anti-Oxidant Capacity (TEAC) in one gram of chocolate ((mol TEAC/g).
Reducing power (RP). The reducing power was evaluated as described by Beyhan, Elmastas and Gedikli (2010), some modifications were introduced. The “A” mixture was prepared, it contained the aqueous dilution of the LE in different concentrations that varied between 0.039 and 0.515 Mg/mL. From each one 0.5 mL were measured, then 0.5 mL of phosphate buffer (0.2 M, pH 6.6) and 0.5 mL of potassium ferricyanide 1% were added, the mixture was stirred and incubated at 50 °C for 20 minutes. Then it was allowed to cool and 0.5 mL of 10% trichloroacetic acid were added. After that, 0.5 mL were measured and taken from each “A” mixture and placed in another tube with 0.5 mL of bidistilled water plus 0.1 mL of 0.1% ferric chloride; 30 minutes later the results were read at 700 nm. The absorbances of each of the eight LE were compared with a calibration curve (Padilla, Rincon and Bou-Rached, 2008) of L-ascorbic acid (0.2 - 10.0 Mg/mL, y = 0.082x + 0.0688, R2 = 0.998), the results were expressed in mg Ascorbic Acid Equivalent in one gram of chocolate (mg AAE/g).
Statistical analysis. Eight different samples of LE corresponding from each type of dark chocolate were used, each trial was carried out in triplicate. A Completely Randomized Design (CRD) was applied (p < 0.05), the results were expressed as the mean ± standard deviation. Tukey reports and Pearson correlation coefficients were done between treatments. The SAS® program 9.1 version, was used to perform the respective statistical analysis.
Results and discussion
As it is stated on the packages’ labels, the cacao content of the eight samples of dark chocolate varied between 50 and 74% of cacao with different fat content (Table 1). The weight of DCh used to carry out the aqueous methanol extraction was between 2.88 and 10.95 g. DCh were used to make three successive extractions. The yield of lyophilized extracts ranged from 0.90 to 3.29 g.
Table 1 Characteristics and extraction yields of chocolate traded in Peru.
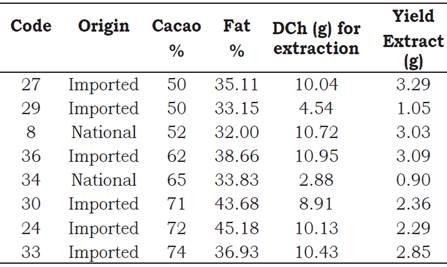
DCh. = Defatted chocolate.
Total polyphenol content (TPC). The TPC varied from 1.69 ± 0.02 to 5.39 ± 0.17 mg GAE/g Ch for chocolate containing 50%, 52%, 62%, 65%, 71%, 72% and 74% of cacao (Table 2), in this regard, Todorovic et al. (2015) found between 7.21 ± 0.49 and 12.65 ± 0.45 mg GAE/g in five chocolate samples whose cacao content was between 65% and 75%. These results were obtained in a study by Grassi et al. (2005), where 5 mg of polyphenols correspond to 1 g of chocolate, whereas Vinson et al. (2006) found that 23.9 mg of polyphenols correspond to 1 g of chocolate. In both studies the polyphenol standard used is not mentioned. However, in our study the gallic-acid standard was used.
Table 2 Total polyphenols and flavonoid content of chocolates traded in Peru.
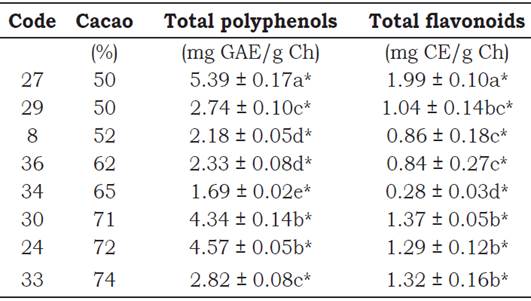
*The values are expressed as mean ± standard deviation (n = 3).
GAE. - Gallic acid equivalent. CE.- Catechin equivalent.
There is an increase in TPC in chocolates with 52%, 62%, 71% and 72% cacao, according to Belscak et al. (2009), who state that the TPC de- pends on the content of cacao solids in the product.
A very specific case is the chocolate with the lowest percentage of cacao (50%) and with different TPC values, one with 5.39 ± 0.17 mg GAE/g Ch (code 27, 35.11% fat, imported) and the other with 2.74 ± 0.10 mg GAE/g Ch (code 29, 33.15% fat, imported). In the first case its increase is significant, because on the packages' label said “chocolate with at least 50% of cacao content”. There are several reasons that explain these differences: (1) If the cacao beans were roasted at low temperatures, (2) If the produced chocolate contains exogenous polyphenol as an additive, as it is the case with the vanilla (Sun, Da Silva and Spranger, 1998) which is usually used in chocolate production for mass consumption, (3) It may also be due to the presence of polyphenols in the cacao butter. Although the chocolates were defatted before determining the level of polyphe nols, there may also be vestiges of cacao butter (or tocopherols, natural antioxidant) in the product, which would impact the results, since cacao butter has been found to be a source of anti-oxidants that causes an increase in polyphenols (Hu et al., 2016). Regarding chocolate containing 72% (45.18% fat, imported) and 74% (36.93% fat, imported) cacao with 4.57 ± 0.05 and 2,82 ± 0.08 mg GAE/g Ch, respectively, there is no direct relationship between them. This can be explained by the reports of Belscak et al. (2009) and Todorovic et al. (2015) who indicated that variations in polyphenol levels depend on the variety of cacao bean, geographic origin, degree of ripeness and post-harvest conditions (fermentation and drying), which as far as we understand also affect manufacturing conditions such as temperatures and the amount of time used in the roasting and conching processes. In addition, Perea-Villamil et al. (2009) attribute variations in polyphenol content to roasting cacao beans in the manufacturing processes. Likewise, Cooper et al. (2008) report that the percentage of cacao that appears on the labels of chocolates cannot be used to estimate the concentration of polyphenols.
Chocolates with a percentage of 65%, 52% and 62% of cacao (two of them domestic products) had the lowest TPC values of the entire group, whereas chocolates with a lower percentage of cacao (50% code 27 and 29) have a higher TPC value. This is explained by Todorovic et al. (2015) who indicate that the quality of the raw material and the production processes can have a significant influence on the quality of the final product.
All samples assessed were different in TPC (p value < 0.05). Tukey results report first chocolate containing 50% cacao (code 27, 35.11% fat, imported) had high level of TPC and last level was chocolate contained 65% cacao (domestic product, 33.83% fat).
Total flavonoid content (TFC). The TFC varied from 0.28 ± 0.03 to 1.99 ± 0.10 mg CE/g Ch for chocolate between 50 and 74% cacao and Meng et al. (2009) found 0.28 mg CE/g defatted dark chocolate. Chocolate samples (Table 2) with a 50% (code 27), 71% and 74% cacao content show higher TFC values, whereas those that contain 52%, 62% and 65% cacao have lower ones. These results are quite different from those reported by Hu et al. (2016) who found values ranging from 0.02 to 1.16 mg catechin/g defatted chocolate and 0.02 to 6.01 mg epicatechin/g defatted chocolate. The differences could stem from the different evaluation techniques that were used. Another reason is the origin of the samples: the 50% (code 27), 71% and 74% cacao samples were imported, whereas the chocolate samples having 52% and 65% cacao were domestic products. It should be noted that for a long time domestic chocolate companies processed cacao paste (the raw material used to produce chocolate) under high temperatures, confirmed by results of Perea-Villamil et al. (2009). Todorovic et al., (2015) reported the TFC in dark chocolates. Thus, with samples containing 75% cacao they obtained 24.4 ± 0.6 gmol CE/g. In our study, using 74% cacao samples we obtained 1.32 ± 0.16 mg CE/g Ch. Several other studies suggest that epimerization reactions occur during the manufacturing of chocolate, where (-)-epicatechin is transformed into (-)-catechin (Lambert, 2017). This compound is not found naturally in cacao seeds (Gotti et al., 2006). A correlation (r = 0.85937) between TPC and TFC was observed (Meng et al. 2009).
All samples assessed were different in TFC (p value < 0.05). Tukey results report the first chocolate sample containing 50% cacao (Code 27, imported) had high level of TFC and last level was chocolate contained 65% cacao (domestic product).
Anti-oxidant capacity (AC). Table 3 show the results of the capturing capacity of the free radical DPPH and the radical cation ABTS. Todorovic et al. (2015) recommend that at least two methods be used to determine the anti-oxidant capacity in vitro since they offer us information about the total anti-oxidant capacity of food products. According to this study, the authors found between 63.0 ± 4.2 and 92.2 ± 1.8 gM TEAC/g in dark chocolates with 70% to 75% cacao produced in Serbia, while our study on ABTS obtained values of 27.87 ± 0.65 to 24.13 ± 0.08 gmolTEAC/g Ch in chocolate samples containing 71% to 74% cacao respectively, thereby indicating that our samples had a lower anti-oxidant capacity. Muñoz et al., (2002) grouped IC50 values and classified the degree of anti-oxidant capacity obtained by the DPPH radical technique. The values of IC50 obtained by the DPPH technique ranged from 52.97 ± 1.77 to 158.67 ± 2.04 gg/ mL extract (Table 3). Chocolates with 72% and 71% of cacao had a lower IC50 value with respect to the DPPH radical and this indicates a greater anti-oxidant capacity (Sánchez-Moreno, Larrauri and Saura-Calixto, 1998). There is a relationship between the TPC and TFC levels (r = 0.85937), such as five samples of chocolate with IC50 values have the following levels: 52.97 ± 1.77 (71% cacao); 53.19 ± 0.79 (72% cacao); 61.99 ± 1.60 (50% cacao, code 27); 73.39 ± 0.17 (50% cacao, code 29); and 75.83 ± 1.90 (74% cacao) gg/mL extract (Table 3). These values are considered to correspond to a moderate anti-oxidant capacity (IC50 > 50 gg/mL and IC50 <100gg/mL). And when the IC50 values are 129.06 ± 2.49 (62% cacao); 133.73 ± 1.17 (52% cacao) and 158.67 ± 2.04 (65% cacao) gg/mL extract, they show a low anti-oxidant capacity (IC50 >100 gg/mL and IC50 <200 gg/mL) (Muñoz et al., 2002). Therefore, chocolates with 52%, 62% and 65% of cacao (two of them domestic products) showed low anti-oxidant capacity due to the reduced polyphenols and flavonoid content unlike the other sample.
Table 3 Antioxidant capacity of chocolate traded in Peru with different cacao percentage.
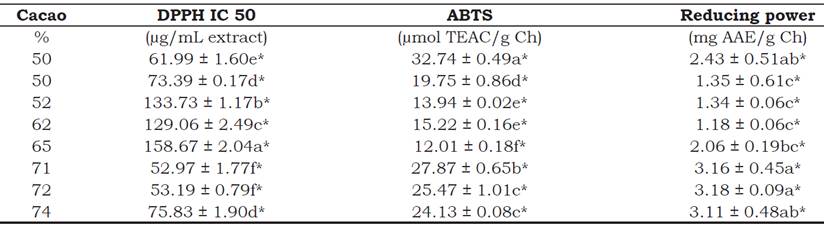
*The values are expressed as mean ± standard deviation (n = 3).
DPPH. - 1,1-diphenyl-2-picrylhydraz. ABTS. - 2,2’-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid). IC50.- The half maximal inhibitory concentration. TEAC.- Trolox equivalent anti-oxidant capacity.
Hu et al. (2016) reported that chocolate manufacturers use a variety of cacao cultivars, as well as processing and storage parameters, all of which impact the anti-oxidant capacity and phenolic profiles of the final products.
All assessed samples were different in AC (p value < 0.05). Tukey results report first chocolate sample containing 50% cacao (code 27, imported) had high level of AC in ABTS and the last chocolate sample contained 65% cacao (domestic product). Test for DPPH (IC50) was inverse than ABTS (r = -0.89835), and AbTs has good correlation with TPC (r = 0.93934).
Reducing power (RP). Finally, Table 3 shows the reducing power (RP) data in dark chocolate. Different studies have indicated that the ability to donate electrons reflects the reducing power (RP) of bioactive compounds (Beyhan et al., 2010).
Chocolates with 72%, 71% and 74% cacao contain 3.18 ± 0.09, 3.16 ± 0.45 and 3.11 ± 0.48 mg AAE/g Ch, respectively, and these show greater reducing power. In contrast, chocolates with 62%, 52% and 50% cacao (code 29) (one of them domestic product) contain 1.18 ± 0.06, 1.34 ± 0.06 and 1.35 ± 0.61 mg AAE/g Ch, and these exhibit a lower reducing power (Table 3). Padilla et al. (2008), studied the seeds of Theobroma cacao with a TPC of 6.66 ± 0.04 g GAE/100g and an RP of 5,80 ± 0.08 g AAE/100g, and found that the TPC level influences the RP value. A similar effect was found in this study, with the samples containing 71% and 72% cacao, for which there was a slight increase in RP for each increase of TPC. This may be due to the existence of different chemical structures in the TPC that react as electron donors, which will influence the RP (Padilla et al., 2008). The results showed that the methanol extracts of DCh are capable of donating electrons that can react with free radicals. The difference in the RP test is explained by Todorovic et al. (2015) who demonstrate that the quality of the raw material (liquor or cacao paste) and the production processes of chocolate can have a significant impact on the quality of the final product.
All samples assessed were different in RP (p value < 0.05). Tukey results report the first group chocolate sample containing 72 and 71% cacao had high level of RP and the last group chocolate samples contained 50% cacao (code 29), 52 and 62% cacao. RP has not a good relationship with others properties.
Conclusions
Dark chocolates marketed in Lima (Peru) had total polyphenol contents (TPC) of between 1.69 ± 0.02 and 5.39 ± 0.17 mg GAE/g Ch, and a total flavonoid content (TFC) of between 0.28 ± 0.03 and 1.99 ± 0.10 mg CE/g Ch. A relationship was found to exist between the TPC and anti-oxidant capacity (AC) (ABTS, r = 0.93934) of dark chocolate. For Ic50 values (DPPH) was inverse than TEAC (ABTS) (r = -0.89835). Chocolate containing 71 and 72% cacao had the highest values of reducing power among the group of eight dark chocolate samples due to polyphenol. Domestic dark chocolates had low antioxidant properties, suggesting studies with novel technologies.
The components investigated in chocolate are largely dependent on the percentage of cacao and it is worth noting that in some cases there was no dependence because the cacao bean goes through various technological processes, especially thermal treatments (roasting, sterilization and conching) that affect the TPC in chocolates. In addition, the biological nature and the variability of the cacao beans does not favor a direct relationship between the TPC and the percentage of cacao declared on the labels of the chocolates.