Introduction
Tropical dry forests cover more than 40% of the tropical vegetation which corresponds to 1,048,700 km2, mainly distributed between America, Africa and Eurasia, but also in a small proportion (3.8%) in Australasia and southeast Asia (Miles et al., 2006). Floristically, the Leguminosae represent the dominant family in these forests, which may lead to believe that these forests are presumed a hotspot of symbiotic N fixation.
This high diversity of species of legumes in Caatinga may be providing the maintenance of the diverse population of rhizobia in the soil. These soils represent natural repositories of new potential elite strains of rhizobia for inoculation of legumes (Boakye et al., 2016).
Rhizobia are classified into two major types according to Gyaneshwar et al. (2011), including a-Proteobacteria and p-proteobacteria. Liu et al. (2007) thoroughly studied rhizobia obtained from native leguminous trees through phenotypic characteristics and genotypic and molecular classification of isolates within the following genera: Rhzobium, Bradyrhizobium, Sinorhizobium, Mesorhizobium, Allorhizobium and Azorhizobium, and Liu et al. (2012) proposed a new reclassification of strains with new genera: Burkholderia, Azorhizobium, Methylobacterium, Devosia, Burkholderia, Cupriavidus and Shinella, respectively.
Lafay and Burdon (2007) studying the molecular diversity of legume-nodulating bacteria, observed that, in Australia tropical areas are centers of biodiversity and biodiversification to root-nodulating legumes. In other words, the biodiversification aims to spread the risk, minimize the ecological loss and maximize the overall performance of the ecosystem, while biodiversity refers to the variety and variability of microorganisms in the environment.
Polyphasic studies are being increasingly applied in microbial diversity, considering many morpho-physiological parameters of colonies to help group the native strains, e.g.: growth time, consistency, change in pH (acid and alkali production), mucus, edge of the colony, shape, color and diameter of the colonies; besides the use of fingerprint molecular markers such as BOX, ERIC (Enterobacterial Repetitive Intergenic Consensus) and REP (Repetitive Extragenic Palindromic Sequences) (Versalovic et al., 1994). It is also observed the use of specific genes such as 16S rRNA (Kim et al., 2014) and nifH and nodC (Fernandes Júnior et al., 2013; Languere et al., 2001) for studies on the diversity and phylogeny of microorganisms.
The most important factor for greater understanding of the diversity of soil microflora and the optimization of biological processes which increase the sustainability of agricultural systems is the concept of species diversity, assessed by two components: species richness, based on the total number of species and uniformity, which is based on the relative abundance of species and the degree of dominance, according to Simpson, Margalef, Wiener and Shannon index (2015). The application of these indices reveals the extent of diversity of an ecosystem, as well as its stability.
A study by Freitas et al. (2014), with native isolates from nodules of Mimosa spp. in the Caatinga, using molecular techniques, found that the isolates are more related to p-Rhizobia than to a-Rhizobia and all have fast growth habit. Mishra et al. (2012) demonstrated that Mimosa spp. forms symbioses with both types of rhizobia in their native environments in South and Central America, including Burkholderia, Cupriavidus and Rhizobium species. This study aimed to assess the diversity of microorganisms in isolates from nodules of three native tree legumes of the dry forest.
Materials and methods
Soil sampling and tree legume cultivation
Soil samples from the 0 - 20 cm superficial layer were collected in areas of preserved Caatinga vegetation in three municipalities, with different climate conditions (Table 1): (1) Santa Terezinha, in the Sertáo Zone of the state of Paraíba; (2) Remigio, in the Agreste zone of this same state; and (3) Serra Talhada, in the Sertáo Zone of the state of Pernambuco. The composition and structure of the vegetation in the three areas were described by Souza (2011). Number of species and tree heights and stem diameters are higher in the Agreste zone than in the Sertáo Zone and, regarding this last zone, the values were higher in Serra Talhada than in Santa Terezinha, probably reflecting higher water availability.
Table 1 General characteristics of preserved Caatinga areas in three municipalities, in the States of Paraíba (PB) and Pernambuco (PE), from the Brazilian Dry Forest.
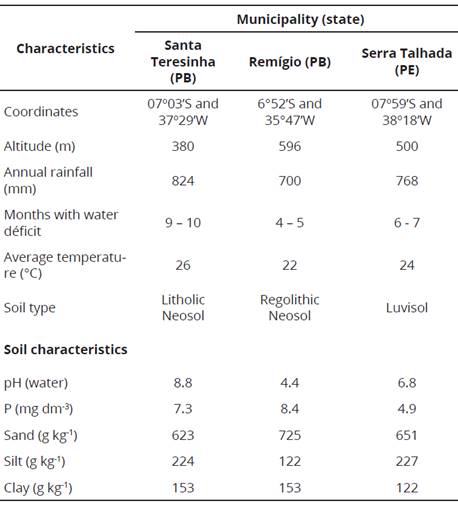
Soil subsamples were analyzed for some chemical and physical characteristics (Table 1) following the methodology described by Solos (1997). The samples were dried, passed through a 6 mm mesh sieve, and portions of 1 kg were placed in pots maintained under greenhouse conditions. Seeds of Mimosa tenuiflora (Willd.) Poir., Piptadenia stipulacea (Benth.) Ducke and Mimosa caesalpiniifolia Benth. were collected from single mother trees in Remígio and Serra Talhada. The seeds were surface-disinfected in ethanol (70% v/v - 3 min) and sodium hypochlorite (1 % v/v - 3 min), rinsed five times with sterile distilled water, rolled onto YMA plates to test for surface sterility and then sown in the pots. The pots received 100 ml of nutrient solution without nitrogen very week until harvest, 120 days after seed germination. At harvest, the root nodules were separated, dehydrated in silica gel and stored.
Rhizobia isolation and phenotypic characterization
Rhizobia were isolated from the nodules in yeast mannitol agar (YMA, pH 6.8) medium [18] with 25 mg kg-1 (w/v) of congo red. The typical rhizobial colonies were purified and stored at -20 °C, in microtubes with 1 ml YM medium (YMA without agar) plus 15% sterilized glycerol. The colonies isolated in YMA with 25 mg kg-1 (w/v) bromothymol blue as pH indicator were observed for the following characteristics: growth period, pH alteration of growth medium, colony morphology (shape, size, border, transparency, surface) and amount of extracellular polysaccharides (EPS). These characteristics were converted to binary data employed in a cluster analysis using the UPGMA (Unweighted Pair Group Method Using Arithmetic Averages) algorithm and the Jaccard similarity index.
The results of the cluster analysis were used to calculate richness (Taxa S and Margalef), diversity (Shannon H), dominance (Simpson 1-D) and uniformity (Evenness, J) indices for the soils and species, where each morphological group, at 60% of the similarity (de Concei^áo Jesus et al., 2005), was considered as one operational taxonomic unit. The PAST (Palaeontological Statistics) program was used to perform cluster analysis and diversity indices calculation (Hammer et al, 2001).
Genetic characterization
Confirmation of isolates: nifH- and nodC-PCR amplification.
The nifH amplification was performed using the nifHKD primer (Fernandes Jr. et al, 2013), whose sequences were PolF (TGC-GAYCCSAARGCBGACTC) and PolR (ATSGCCAT-CATYTCRCCGGA). For amplification of the nodC gene, it was used the nodCr (AYGTHGTYGAYGAC-GGTTC) and nodCr (I) (CGYGACAGCCANTCKC- TATTG) primers, according to Laguerre et al. (2001). The composition of the duplex PCR reaction mixture was as follows: reaction buffer 1X Taq Polymerase Invitrogen, MgCl2 (2.5 mM), dNTPs (1.2 mM), Taq Polymerase (0,3U), primers (1 ^M nifH + 0.6 ^M nodC, template DNA (40 ng). The amplification conditions in the thermal cycler Biometra® were an initial cycle of denaturation (94 °C for 5 min); 35 cycles of denaturation (1 min at 94 °C), annealing (45 sec at 55 oC) and extension (1 min at 72 °C); and a final extension cycle at 72 °C for 1 min.
PCR amplification with REP, ERIC and BOX- PCR.
The isolates from the nodules of the three species and the three soils and six reference strains (ER1 - Rhizobium SP NGR234, ER2 - Mesorhizobium mediterraneanus BR 523, ER3 - R. etli CFN42, ER4 - Ensifer fredii USDA205, ER5 - Bradyrhizobium japonicum BR111 and ER6 - R. tropici CIAT899), provided by EMBRAPA Agrobiology, were placed to grow in liquid TY. After 48 hours of growth, 1.5 uL aliquots of the cultures were transferred to microcentrifuge tubes, being centrifuged for 3 min. at 13,000 rpm. Each Isolates received the following legend: we use the initial two letters of the common name of the plant and the last one of the place collected for example: (Jurema Branca - Serra Talhada - JBS), (JuremaPreta - Patos - JPP), (Jurema Branca - Remigio - JBR) and (Sabiá - Remigio -SBR).
The genomic DNA extraction was performed using the Wizard Genomic DNA kit (PROMEGA) as described by the manufacturer. The DNA was diluted in the ratio 1: 500, leaving a final concentration of about 20 to 40 ng.juL-1.
Isolates were amplified by repetitive element polymerase chain reaction (rep-PCR), using BOX-A1R primer (5’-CTACGGCAAGGCGAC-GCTGACG-3’), and by ERIC-PCR, using ERIC-1 (5’-ATGTAAGCTCCTGGGGATTCAC-3’) and ERIC-2 (5’-AAGTAAGTGACTGGGGTGAGCG-3’) primers, to assess genetic diversity. The BOX-PCR reaction containing 5 pL GoTaq Colorless polymerase (PROMEGA), 1.0 juM BOX-A1R primer and 1 uL template (60 ng), supplemented with water to a final volume of 10 juL. The cycling was performed as follows: initial denaturation at 95 °C for 7 min; 35 cycles of denaturation (1 min at 94 °C), annealing (1 min at 55 °C) and extension (8 min at 65 °C); and a final extension cycle at 72 °C for 16 min (Freitas et al. 2013).
Products obtained by using BOX-PCR were analyzed using horizontal gel electrophoresis in a 1.5% agarose gel containing SybrGold. A 10,000 bp molecular marker (INVITROGEN) was loaded alongside BOX-PCR products to estimate the DNA fragment size. The gel was run at 150 V for 1 h, with final visualization under UV radiation.
Regarding ERIC-PCR reaction, it contained 5.0 uL GoTaq Colorless Master mix (PROMEGA), 0.4 mM each primer, 2 juL free of ultrapure water and 100 ng of genomic DNA with a final volume of 10 uL. The amplification cycle was performed in a thermocycler Biometra® in the following conditions: 95 °C for 7 min, 30 cycles (1 min at 95 °C, 1 min at 55 °C; 2 min at 72 °C) and a final extension cycle of 72 °C for 16 min. The amplified fragments were separated by electrophoresis at 80 V, for three hours, in agarose gel at 1.5%, stained and photographed in a photodocumenter Loccus.
The profiles of BOX and ERIC bands were analyzed for the presence (1) or absence (0) of each amplicon gel using the Simple Matching coefficient. The program used to perform cluster analysis was nTsYS-PC 2.1. The dendrograms were constructed using UPGMA (Unweighted Pair Group Method Using Arithmetic Averages), which is a SAHN (sequential, agglomerative, hierarchical, non-overlapping) clustering technique, and graphics were made using tree plot (Sokal, 1986)
Results and discussion
The diversity indices of Shannon-Wiener, Margalef, Menhinick, Simpson and Pielou were applied to assess the richness, dominance and evenness of the isolates. It is observed for the Shannon-Wiener index, which expresses the richness and evenness of the species, that the isolates of M. tenuiflora have greater richness compared to M. caesalpiniifolia and P. stipulacea. Consequently, it has a lower dominance, when observing the Simpson index for the isolates of the three species. The evenness index of Pielou showed the lowest abundance for the studied isolates of M. caesalpiniifolia, while the isolates of P. stipulacea showed the greatest abundance. That is, the evenness is the inverse of dominance, as demonstrated in Figure 1.
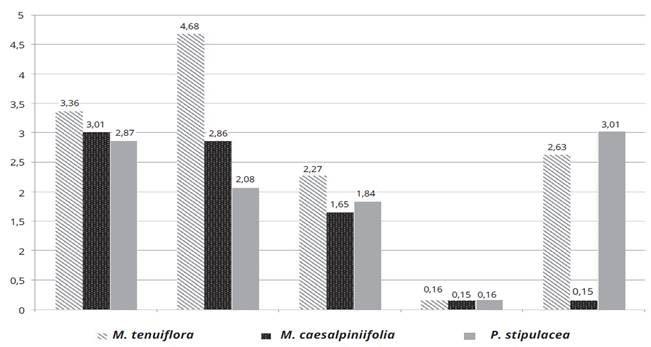
Figure 1: Diversity index of Shannon-Wiener, Margalef, Menhinick, Simpson and Evenness of isolated the Indigenous Tree Legumes: Mimosa tenuiflora (Willd.) Poir., Piptadenia stipulacea (Benth.) Ducke and Mimosa caesalpiniifolia Benth. from the Brazilian Dry Forest.
Margalef and Menhinick indices are also used to measure the richness of species and both of them showed similar results to those obtained by Shannon index, only differing in values. The studied isolates have a high diversity that includes a wide variety and relative abundance of species.
To molecularly authenticate the 27 studied isolates (Figure 2) amplification with the nifH and nodC genes was performed, being observed that three JBR (from P. stipulacea) isolates showed no amplification with any of the genes: JBR02, JBR09 and JBR18. The isolates JBR13, JBR06 and JBR12 showed identical profiles with an amplicon of 500 bp and 700 bp, except for the isolate JBR13, which did not show the band of 700 bp. The isolates JBR16, JBR05, JBR11, JBR14, JBR42 proved to be extremely polymorphic in duplex PCR, with fragments that could be considered for the two genes.
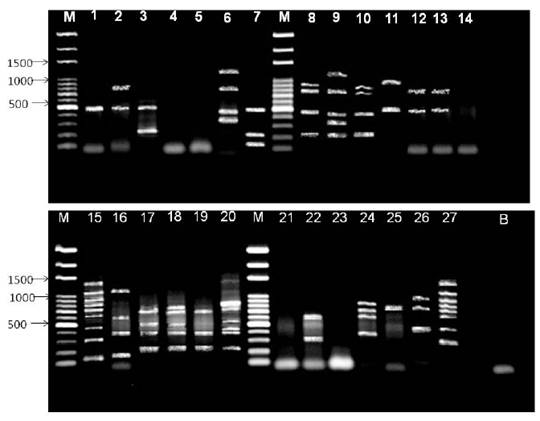
Figure 2: Duplex-PCR Simultaneous duplex amplification of the nifH and nodC genes in legume isolates: Mimosa tenuiflora (Willd.) Poir., Piptadenia stipulacea (Benth.) Ducke and Mimosa caesalpiniifolia Benth. from the Brazilian Dry Forest for molecular authentication. Legend: M: Amresco 100 pb, 1:JBR13, 2: SBR15, 3:JBS06, 4:JBR02, 5: SBR10, 6:JPP18, 7:SBR01, 8:SBR20, 9:JPP13, 10:SBR02, 11: SBR06, 12:JBR06, 13:JBR12, 14:JBR09, 15: SBR19, 16:JBR16, 17:JBR05, 18:JBR11, 19:JBR14, 20:JBR42, 21 :JBR18, 22:JPP08.2, 23:JPP14, 24:SBR04, 25:JBS02, 26:SBR14, 27:JBS05, B: Branco.
Only one of the SBR isolates from the bait plant M. caesalpiniifolia was not amplified (SBR10) with any of the two primers. The SBR15 isolates presented two amplicons of 500 and 700 bp (nodC); SBR01 (3 amplicons; 500 bp - nodC, 200 bp and 100 bp - nifH); SBR20 and SBR02 (amplicons of 200, 480 bp - nifH; 700, 900 bp - nodC). The isolate SBR06 showed two bands with sizes of 500 and 1000 bp, which may be related to the nodC gene. The most interesting fact was that the amplification of these genes in isolate SBR19 proved to be a real fingerprint, what is quite intriguing because it would be necessary to clone each fragment of it and make a sequence to know to which fragment it belongs. This same behavior was observed with the isolate JBS.
The isolates SBR04 and SBR14 presented bands in the following sizes: 380, 600, 700 and 800 bp; and 480, 700 and 800 bp, respectively, we consider that both amplified the nif and nod genes. JBS isolates showed two fragments: JBS06 - 500 bp and 250 bp; and JBS02 - 480 and 600 bp, we consider that both genes were amplified. Of the three isolates of JPP, only JPP08.2 amplified three band sizes: 500, 400 and 300 bp. Interestingly, these same primers were used in type strains of R. tropici, Ensifer sp., Bradyrhizobium sp., B. japonicum, Azospirillum brasilense, Herbaspirillum seropedicae in duplex PCR, and generate fragments of 360 bp for the nifH gene and 980 bp for the nodC gene (Fernandes Jr. et al., 2013). The phylogeny of the nodC and nifH genes are similar, however, cases of incongruity are detected, suggesting that a genetic rearrangement is occurring in the course of evolution.
Given the variation found for the nifH and nodC genes in this study, we observed that these isolates have distinct characteristics for these two genes compared to the type strains mentioned above, since it was found a pattern of bands with much variation forming a fingerprint profile. Many features of the nifH gene, based on the phylogenetic tree, are entirely consistent with the phylogeny of the 16S rRNA of nitrogen-fixing bacteria. Notwithstanding, the diversity of the nifH genes allows a wider representation of the taxonomic diversity of fixing bacteria, showing that they can be used for studies on the diversity of the nitrogen-fixing bacterial community. Studies comparing the phylogeny of the 16S rRNA, nifand nod genes support the idea of lateral transfer of symbiotic genes, and that genetic rearrangements may be involved in the acquisition and development of rhizobial symbiotic functions.
Multiplex PCR has been widely used in soil and environmental microbiology, especially in the rhizobiology, according to Fernandes Jr. et al. (2013). In the work reported by these authors, it was found a 360 bp amplicon relative to the nifH gene and a 980 bp amplicon relative to the nodC gene when amplified alone. But when amplified in duplex PCR, the amplicon of the nodC gene was shown to be much greater than 980 bp. Languere et al. (2001), studying the nodC and nifH genes of six bacterial genera, observed that the amplicon of the nodC gene can range between 930-1300 bp, and the amplicon of the nifH gene ranges between 780-890 bp. The same authors studying combined data of each gene through RFLP analysis observed that combination of analyses of the nif and nod genes revealed 50 symbiotic genotypes (nod-nif).
It can be concluded that the authentication with the nifH and nodC genes can result in false-positive data, since both genes are highly polymorphic between species and biovars. Since the isolates are originating from Caatinga and the lack of knowledge studied plants and kind of isolate, this technique is not advisable.
The results of the morpho-physiological characteristics of the isolates originated a dendro- gram where ten clusters were formed (Figure 3). 70% similarity was stipulated as a basis to compare the clusters. Cluster 1 was comprised of 10 isolates, namely: SBR01, SBR14, SBR04.2, SBR10 and JPP18 (with similarity of approximately 80%), and SBR04, JBR05, JBR12, JBR16, JBR02 (with similarity of 75%). It is noteworthy that in this Cluster are three of the four isolates that did not amplify with the nod and nif genes. Cluster 2 was formed by the isolates SBR02 and JPP14, with 86% similarity between them, and the isolates SBR15 and SBR19, identical with similarity of 82%.
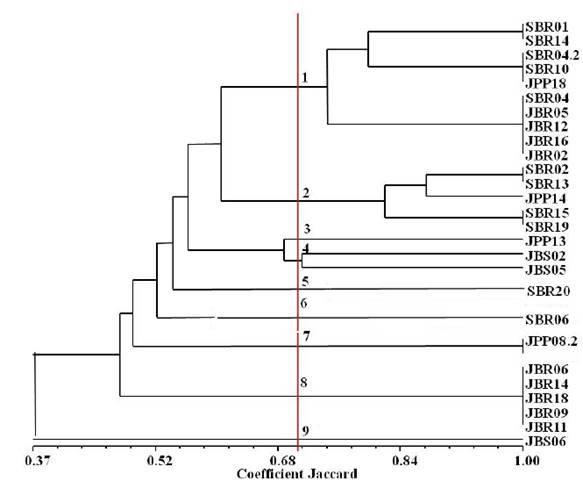
Figure 3: Dendrogram of similarity resulting from the morphological and physiological characteristics of the nodule isolates of Mimosa tenuiflora (Willd.) Poir., Piptadenia stipulacea (Benth.) Ducke and Mimosa caesalpiniifolia Benth. from the Brazilian Dry Forest.
Clusters 3 to 10, with the exception of Cluster 9, were basically constituted of1 or 2 isolates, yet Cluster 9 a larger cluster of isolates was formed P. stipulacea from Remigio. However, the similarity was very low, less than 52%. And among these isolates, one (JBR09) was not amplified with the nod and nif genes. It can be concluded that the morpho-physiological characteristics are incipient data to determine the diversity of an isolate, given that these isolates were shown to be very similar but with a similarity rather lower than expected. Chagas Jr. et al. (2010) verified that the morpho-physiological characteristics show a high diversity among populations and that this diversity may be related to molecular studies.
Figure 4 shows the dendrogram formed by the BOX-PCR marker. In this analysis, the type strains were inserted for comparison. Interestingly, the type strains of rhizobia gathered almost in the middle of the dendrogram, with a few isolates of this study grouped to the same. Only the isolate JBS02 showed a similarity of 72% with Ensifer fredii USDA205, a rhizobium of rapid growth. Cluster 7, formed by the isolates SBR14, SBR15, SBR19 and JBR13, showed a similarity of 71% with Rhizobium sp NGR234, also a rhizobium of rapid growth and fairly promiscuous. The remaining isolates studied with BOX marker formed distinct groups proving to be new isolates, many of them being identical among themselves.
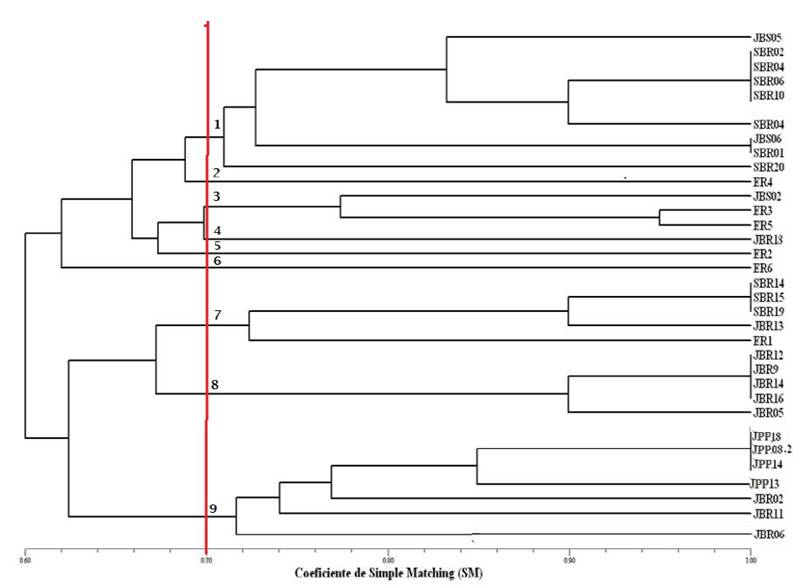
Figure 4: Similarity dendrogram resulting from the BOX-PCR fingerprinting marker of species nodule isolates of Mimosa tenuiflora (Willd.) Poir., Piptadenia stipulacea (Benth.) Ducke and Mimosa caesalpiniifolia Benth. from the Brazilian Dry Forest.
In Figure 5 it can be seen that the dendrogram formed by the ERIC-PCR marker showed a great diversity among the isolates. With 11 clusters formed, the type strains grouped in the lower part of the dendrogram, with a similarity ranging from 65%, with Rhizobium sp. NGR234 and M. mediterraneanus BR523, to 89%, with R. etli CFN42 and B. japonicum BR111. The most interesting in was that eighteen of the twenty-seven isolates were grouped in the upper part of the dendrogram without any type strain studied. In subcluster 7, the Ensifer fredii USDA205 ensifer - ER4 strain was grouped with three isolates: JBS6, SBR01, JBR11.
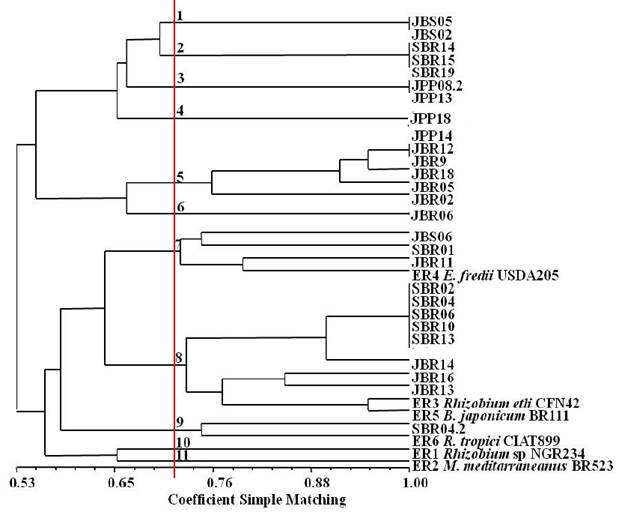
Figure 5: Similarity dendrogram resulting from the ERIC-PCR fingerprinting marker of species nodule isolates of Mimosa tenuiflora (Willd.) Poir., Piptadenia stipulacea (Benth.) Ducke and Mimosa caesalpiniifolia Benth. from the Brazilian Dry Forest.
Moreover, the isolates of this study were shown to be different from the type strains studied here, and may be new species not yet known. When compiling the data of BOX-PCR and ERIC-PCR markers (Figure 6) little difference was observed. Only a few changes in the position of isolates were verified.
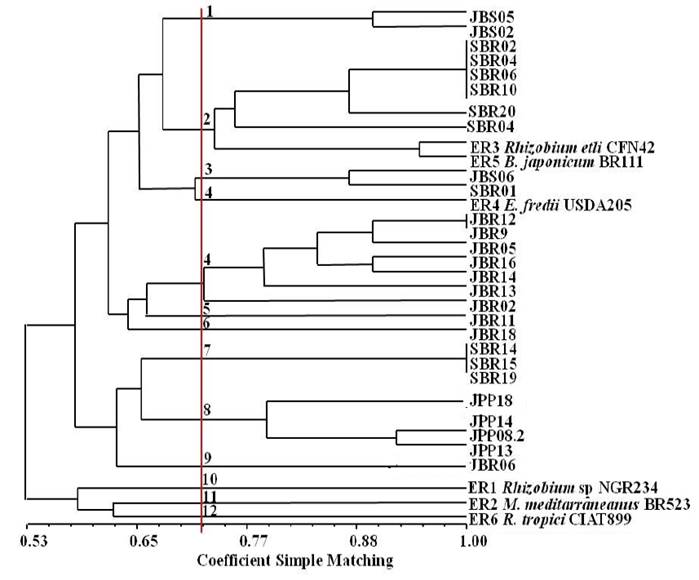
Figure 6: Dendrogram of similarity resulting from the compilation of BOX-PCR and ERIC-PCR fingerprinting markers of species nodule isolates of Mimosa tenuiflora (Willd.) Poir., Piptadenia stipulacea (Benth.) Ducke and Mimosa caesalpiniifolia Benth. from the Brazilian Dry Forest
But the real conclusion is that these isolates are distinct and need more studies to be characterized as new species originating from leguminous plants of Caatinga. Several authors, such as Versalovic et al. (1994) believe that these markers, for being associated with high levels of polymorphism, showing the gene region amplified by the REP, ERIC and BOX primers, may have a role in the adaptive evolution process, helping in the interaction of microorganisms with hostile environments such as high temperature, high aluminum content, salinity, among others. Freitas et al. (2014), working with native isolates of Mimosa spp. in soils of the Brazilian semiarid region, found that these isolates have different cultural characteristics from those obtained in the same soil but using other species of legumes as bait plants. Based on these results, it can be seen a great diversity among the isolates, leading us to conclude that there is a great variability in the studied isolates and that a polyphasic study considering other known markers to obtain a more accurate taxonomy is necessary.
Conclusión
The morpho-physiological and molecular characterization of the isolates originating from three tree legumes, M. tenuiflora, P. stipulacea and M. caesalpiniifolia, revealed a wide variety of bacteria colonizing this plant.
The authentication method of the nodC and nifH genes with rhizobia isolates still needs to be better adapted, once the primers used produce a very high polymorphism.
For this study, we accepted that the isolates showing amplicons greater than 500 bp would have amplified the nodC gene, and those which amplified fragments smaller than 500 bp would have amplified the nifH.
The analyses with molecular markers proved to be quite efficient, especially when these data were compiled. Thus showing that the larger the number of molecular markers used, the greater the capacity of distinction between species.














