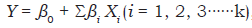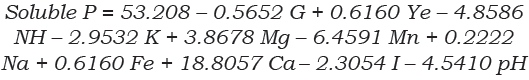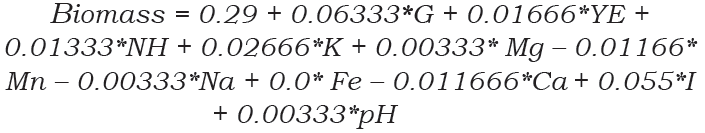Introduction
Phosphorus (P) is one of the most important macronutrients for plant growth and development and is a key nutrient for achieving sustainable agriculture. Phosphorus plays a fundamental role in all major metabolic processes in plants including photosynthesis, macromolecular biosynthesis, energy transfer, signal transduction and respiration. In nature, tricalcium phosphate (Ca3(PO4)2) is one of the main P reserves; however, cultivated plants are unable to use this chemical form (Sharma et al., 2013).
To release P from Ca3(PO4)2, the activity of soil microorganisms such as bacteria, soil borne micromycetes and mycorrhiza is required. In agricultural soils with low abundance and diversity of P-solubilizing microorganisms, bio-fertilizing microorganisms can be directly applied using a bioaugmentation strategy (Khan et al., 2014). In this sense, soil borne micromycetes have been described as suitable microorganisms for releasing P from inorganic sources such as Ca3(PO4)2, FePO4 and AlPO4 (Oliveira-Mendes et al., 2016; Zúñiga-Silva et al., 2016).
Solubilization of inorganic phosphates is gaining greater attention in organic and sustainable agriculture; several soil borne micromycetes have been isolated for this purpose (Hernández-Leal et al., 2011; Hernando-Posada et al., 2012, 2013). These isolates can be inoculated directly into the soil as biofertilizers or can be used in bioreactors to solubilize rock phosphates or other inorganic phosphates in the production of P fertilizers (Khan et al., 2014).
However, to produce P fertilizers from inorganic P sources using fungi, a suitable fungal strain and an optimized culture media are required (Oliveira-Mendes et al., 2016). Some studies have focused on the optimization of inorganic phosphate solubilization using liquid fermentation, considering one independent variable in addition to other variables fixed at a certain level. These single experimental designs are usually laborious and time consuming and screen few experimental factors (Scervino et al., 2011). For instance, Relwani et al. (2008) used Pikovskaya broth to study the influence of C and N source on the solubilization of Ca3(PO4)2 by a mutant strain of Asperigillus tubingensis (AT1). The study reported that glucose and sucrose promoted Ca3(PO4)2 solubilization compared with fructose, lactose and xylose. In addition, potassium nitrate increased Ca3(PO4)2 solubilization in comparison with ammonium sulfate, ammonium nitrate, asparagine and tryptophan. In other study, Chun-Qiao et al. (2008) optimized the NBRIP culture medium for rock phosphate solubilization using soil microfungi isolated from phosphate mines. The authors reported that the optimum culture conditions for rock phosphate solubilization were 32 °C, 160 r.p.m., 2.5 g/L of rock phosphate and 0.5 mg/mL of EDTA. In addition, Penicillium expansum resulted in the highest soluble P at a C:N ratio of 40:1. In the studies of both Relwani et al. (2008) and Chun-Qiao et al. (2008) factorial experimental designs were used. Thus, the best culture conditions could not be determined through this experimental design. In comparison, Palckett-Burman experimental design (PBED) represents one means of identifying the main effects and the efficiency of a large number of factors on a specific response (Plackett and Burman, 1946).
Accordingly, we considered this experimental design to be useful in the screening of a large number of factors during the optimization of the solubilization of Ca3(PO4)2 by soil-borne micromycetes. The objectives of this study were. (1) to evaluate the solubilization of Ca3(PO4)2 by native soil borne micromycetes isolated from the rhizosphere of Carica papaya plants, and (2) to determine the significant nutritional factors involved in the solubilization of Ca3(PO4)2 by the best P-solubilizing fungi using a Plackett-Burman experimental design.
Materials and methods
Fungal sources
Soil-borne micromycetes were isolated from the rhizosphere of C. papaya plants, located in an organic orchard in Tecoman, Colima, Mexico (location: 18°50'16.8" N, 103°50'06.4" W). Isolation was carried out by washing the soil particles, which consisted in washing one gram of soil with three liters of sterile distilled water using three sieves (Alcon®, Mexico) of decreasing mesh size, one on top of the other, of 500, 250 and 150 ^m, respectively. Washed particles (150 μm) were deposited on sterile filter paper discs for 24 h to remove excess moisture. Ten particles were seeded on potato dextrose agar (PDA) + chloramphenicol (150 ppm, Sigma- Aldrich®, USA). Five Petri dishes were incubated at 25 °C and 75% relative humidity (RH). The fungal colonies were purified by re-isolation in PDA. Five fungal isolates were used to evaluate the solubilization of Ca3(PO4)2 in a Pikovskaya culture medium.
Molecular identification
Only Penicillium isolate SP16 was identified at molecular level using the methodology described by Zúñiga-Silva et al. (2016). After genomic DNA extraction, PCR amplification of the internal transcribed spacers (ITS regions: 26S and 16S regions) were carried out using the following primers ITS1 (5'-TCCGTAGGTGAACCTGCGG-3') and ITS4 (5'-TCCTCCGCTTATTGATATGC-3'). Purified PCR products were sequenced by Genomic Biotechnology Center-IPN (Reynosa, Tamaulipas, Mexico) using standard protocols. DNA sequence analyses were performed using the BLAST program, run against the NCBI database (https://www.ncbi.nlm.nih.gov/). The Index Fungorum (http://www.indexfungorum.org/) was used as a species authority.
Solubilization of Ca3(PO4)2 by fungal isolates
Pikovskaya broth was prepared according to Hernández-Leal et al. (2011). In Erlenmeyer flasks (250 mL), 120 mL of Pikovskaya broth was added and then autoclaved at 120 °C and 125 lb of pressure during 15 min. Soil-borne micromycetes were inoculated into the flasks using 50 μL of a conidia solution (1*106 conidia/ mL). Inoculated flasks were incubated for six days at 25 °C, 75% relative humidity (RH) and 12/12 h light/darkness. Five fungal isolates were tested; four independent replicates of each isolate were settled. Every 48 h, aliquots of 10 mL of supernatant were obtained aseptically in a laminar flow cabinet. The supernatant was filtered in nylon membrane disks (0.5 μm), avoiding fungal biomass. Free cell extracts were used to measure soluble P, total protein content and pH.
Studied variables
Soluble P was measured using a spectrophotometric method during the reduction ofphosphomolybdenum complexes to ascorbic acid (Watanabe and Olsen, 1965). Readings were made at 880 nm in a UV-Vis spectrophotometer (Luzeren®, model L6S, St. Louis, MO, USA). The standard curve for soluble phosphorus was: P-soluble=[absorbance-0.1000]/0.009074, r2=0.9976. As the P standard, KH2PO4 was used.
Total protein was measured according to the Bradford method. The standard curve to obtain total protein from the free cell extracts was: protein content=[absorbance-0.4306]/0.1188, r2=0.9915. As the protein standard, egg albumin was used.
To measure the pH of fungal extracts, a potentiometer OHAUS® (model Starter, Parsippany NJ, USA) was used. Finally, for fungal biomass quantification, mycelium from the five fungal screening cultures and the PBED were harvested by filtering the culture medium with a pre-weighed Whatman No. 1 filter paper and determining the difference in mass after drying over three days at 40 °C.
Plackett-Burman experimental design
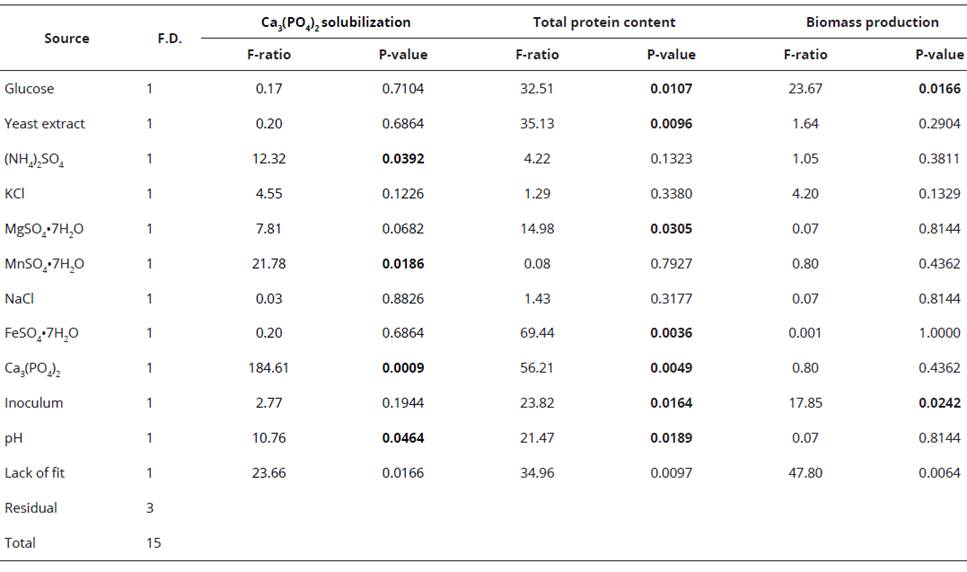
To determine the significant nutrimental factors that influence Ca3(PO4)2 solubilization and fungal growth of P. hispanicum (isolate SP16), a PBED was implemented. The statistical screening was based on the main effects of the experimental factors, although their interaction effects were not considered. Pikovskaya broth was modified to evaluate the influence of different nutrimental factors on Ca3(PO4)2 solubilization, total protein and biomass production. Eleven factors were assessed: the components of Pikovskaya broth (9 nutrients), inoculum amount (conidia/mL) and pH (Table 1). Each screened factor was set at two levels, coded as high (+1) and low (-1). Four central points (0) were screened by running 16 experiments, as shown in Table 2. The PBED was executed three times, and the average of each experiment was used for the statistical analysis. Factors significant at the 5% level (P < 0.05) in the regression analysis were considered to have a significant impact on the response variables. Equation 1 was used to fit the experimental data, which includes the individual effects of each studied factor. The PBED, the response variables were P-solubilization, total protein content and biomass production, which were measured as previously described.
Table 1 Original and coded levels of the independent variables used in the Plackett-Burman experimental design.
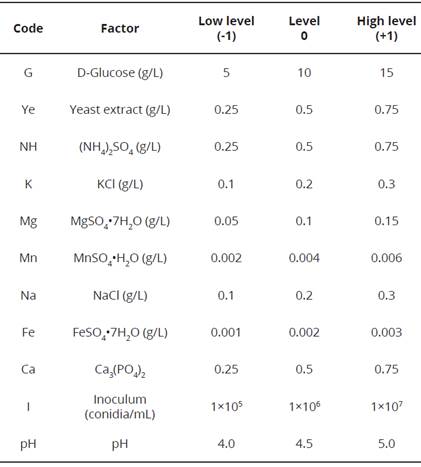
Table 2 Plackett-Burman experimental design matrix for evaluating the influence of nutrimental factors for Ca3(PO4)2 solubilization, total protein content and biomass.
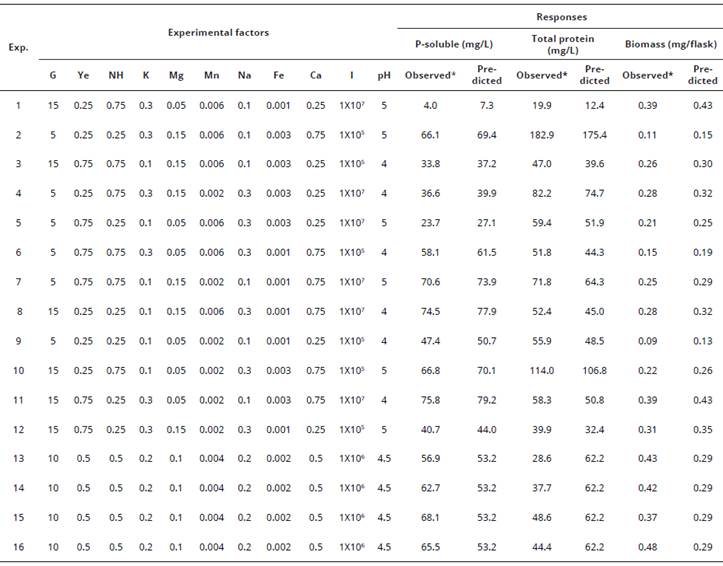
Prroduction of Penicillium hispanicum. *Observed values were the mean of three replicates. Exp.=Experiments.
where:
Y = Response (soluble P, total protein, biomass)
X = Independent variable
β 0 = Model intercept
βi. = Estimated variable
Data analysis
An analysis of variance (ANOVA) and a múltiple range comparison test (least significant difference, P = 0.05) were applied to the solubilization of Ca3(PO4)2, total protein content and biomass production in the screening of the five soil-borne micromycetes. The PBED was designed and analyzed using the StatGraphics Plus® software.
Results
Fungal identification
According to sequence analysis, the isolate Penicillium SP16 was identified as P. hispanicum (Ascomycota: Trichocomaceae) C. Ramírez, A. T. Martínez and Ferrer, with 99% of similarity to the accessions: NR_138307.1, JX841247.7 and KJ775637.1 reported in the gen bank.
Solubilization of CaTPOby soil-borne micromycetes
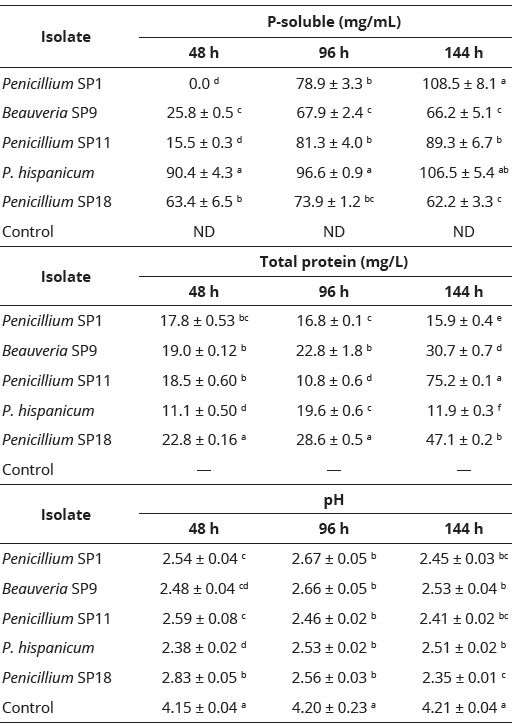
Soil-borne micromycetes isolated from the C. papaya rhizosphere showed statistical differences in the solubilization of Ca3(PO4)2. At 48 h of culture, P. hispanicum achieved the highest (F = 36.56, P = 0.00001) soluble P at 90.4 mg/mL; in contrast, Penicillium SP11 (15.5 mg/L) solubilized the lowest amount of Ca3(PO4)2 (Table 3). During the second evaluation period (96 h), P. hispanicum maintained the highest Ca3(PO4)2 solubilization at 96.6 mg/L compared with that of the other studied fungal isolates (F = 37.52, P = 0.00001). At the end of experiment, Penicillium SP1 (108.8 mg/L) and P. hispanicum(106.5 mg/L) solubilized all soluble P contained in Ca3(PO4)2, or 98.6 mg/L, according to the molecular weight of Ca3(PO4)2. In contrast, both Beauveria SP9 and Penicillium SP18 demonstrated the lowest solubilization of Ca3(PO4)2 (F = 3828.85, P = 0.00001), solubilizing 66.2 and 62.2 mg/L of P, respectively.
Total protein content, biomass production and pH in fungal extracts
At both 48 (F = 36.56, P = 0.00001) and 96 h (F = 37.52, P = 0.00001), Penicillium SP18 achieved the highest total protein content at 22.8 and 28.6 mg/L, respectively (Table 3). The lowest total protein content was produced by Penicillium SP1 at 17.8 and 16.8 mg/L at 48 and 96 h, respectively. At the end of evaluation, Penicillium SP11 (75.2 mg/L) achieved the highest total protein content (F = 3828.85, P = 0.00001) compared with that of the other isolates, while P. hispanicum showed the lowest total protein content (11.9 mg/L). Total protein content is an indirect parameter of the ability of a specific isolate to produce certain fungal enzymes that could be involved in the solubilization of Ca3(PO4)2.
Figure 1 shows the biomass production of the five studied isolates during the solubilization of Ca3(PO4)2. Biomass production ranged from 0.20 to 0.52 mg/flask (120 mL of Pikovskaya culture medium). The isolate with the highest (F = 30.06, P = 0.00001) biomass production was Penicillium SP18 (0.52 mg/flask). In contrast, Penicillium SP1 showed the lowest biomass production (0.20 mg/flask).
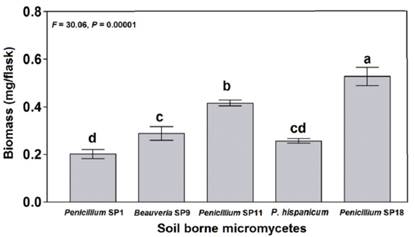
Figure. 1 Biomass production in Pikovskaya culture medium by five soil borne Micromycetes in the solubilization of Ca3(PO4^. Bars with the same letter are not significantly different from each other (n = 4, mean ± SEM, LDS test, a < 0.05).
All fungal isolates reduced the pH of the culture medium across time of fermentation (Table 3). The pH of the control was 4.15 at 48 h after incubation; this value increased slightly at 96 and 144 h to 4.20 and 4.21, respectively. However, in the culture of fungal isolates, the pH decreased significantly (F = 174.0, P = 0.00001) from 4.15 to 2.38 (in P. hispanicum) at 48 h of culture. At 96 h of evaluation, the pH of fungal cultures ranged from 2.46 to 2.67, statistically different from the control (pH = 4.20, F = 15.06, P = 0.00001). Meanwhile, at the end of evaluation (144 h), the pH ranged from 2.35 to 2.53 in the fungal cultures; these values were statistically lower (F = 444.59, P = 0.0001) than the control (4.21).
Placket-Burman experimental design
For P. hispanicum, Ca3(PO4)2 solubilization, total protein content and biomass production varied widely (Table 2). These variations evidence the importance of culture medium optimization to efficiently solubilize Ca3(PO4)2. In addition, parameters such as total protein production and biomass can reflect the efficiency of fungal growth.
For Ca3(PO4)2 solubilization, the highest soluble P was found in the 11th trial run (75.8 mg/L of soluble P), whereas the lowest activity was seen in the 1st trial run (4.0 mg/L of soluble P). Thus, 19.7% more soluble P was found in the 11th trial run compared with the central points (runs: 3, 16, 11 and 9). According to the Pareto chart (Figure 2A) and the ANOVA (Table 4), this level improved in presence of a high amount of Cag(PO4)2 (0.75 g/L) and low values of (NH4)2SO4 (0.25 g/L), MnSO4 (0.002 g/L) and pH (4.0).

Figure 2 Pareto chart of eleven-factor standard effects on: A) solubilization of Ca3(PO4)2, B) total protein content and C) biomass production by Penicillium hispanicum
The p-value was used to determine the main nutrimental factors at a confidence level of greater than 95%. According to the ANOVA, the main factors that influenced the solubilization of Ca3(PO4)2 by P. hispanicum were (NH4)2SO4 (P = 0.0392), MnSO4 (P = 0.0186) Ca3(PO4)2 (P = 0.0009) and pH (P = 0.0464) (Table 4). Polynomial Equation 2 represents the model for Ca3(PO4)2 solubilization by P. hispanicum. The significance of Equation 2 was checked using an F-test, and the value was highly significant [(P-value > F) = 0.0029]. The coefficient of determination (r2) of the model [Equation 2] was 0.90194, indicating that 90.19% of the variability in the experiment data could be explained by the estimated model. Also, the standard error was low (4.79).
Regarding total protein content, the highest value was found in the 2nd trial run at 182.0 mg/L. In the 1st trial run, we observed a protein content of 19.9 mg/L. Thus, protein content increased 3.6 fold in the 2nd trial run compared with that of the central points (runs: 13th to 16th). According to the Pareto chart (Figure 2B) and the ANOVA (Table 4), protein content improved significantly in conditions of low glucose (5 g/L, P = 0.0107), yeast extract (0.25 g/L, P =O.00396), inoculum amount (1*105 conidia/mL, P = 0.0164) and pH (4.0, P = 0.0189), and high MgSO4 (0.15 g/L, P = 0.0305), FeSO4 (0.003 g/L, P = 0.0036) and Ca3(PO4)2 (0.75 g/mL, P = 0.0049). Polynomial Equation 3 was obtained from the multiple regression analysis and describes the effect of the nutrimental factors on total protein production of P. hispanicum during the solubilization of Ca3(PO4)2. Statistical significance of the model was checked using an F-test [(P-value > F)=0.0538]. The r2 coefficient of the model [equation 3] was 0.8728, which indicated that 87.28% of the variability in the experimental data could be explained by the estimated model. The standard error of the model was low (8.72), indicating a low variation of the data.
The highest value for biomass production of P. hispanicum was found in the central points (13th to 16th trial run) with a mean of 0.425 mg/ flask (120 mL of Pikovskaya culture medium). In contrast, the lowest biomass production was found in the 2nd trial run (0.11 mg/ flask). The PBED conditions did not lead to an improvement in biomass production. Therefore, the Pikovskaya culture medium was adequate for biomass production of P. hispanicum during the solubilization of Ca3(PO4)2. The regression model of PBED, the Pareto chart (Figure 2A) and the ANOVA (Table 4) showed that biomass production was significantly influenced by a high level of glucose (15 g/L, P=0.0166) and inoculum amount (1*107 conidia/mL, P = 0.0242), although biomass production did not improve. Equation 4 describes the biomass production of P. hispanicum during Ca3(PO4)2 solubilization. The model was significant at 95% [(P-value>F)=0.0285] and had a low determination coefficient (r2=0.4970), explaining only 49.70% of the variability in the data. Also, the standard error was low (0.045).
Discussion
In the soil the availability and efficiency of nutrients depend to a great extent on the biological activity of soil microorganisms. In this context, biofertilizers are an alternative means of providing plant nutrition (Khan et al., 2014). According to Fuentes-Ramírez and Cabello-Mellado (2005), a biofertilizer is “a product that contains living microorganisms, which exert direct or indirect beneficial effects on plant growth and crop yield through different mechanisms”.
Due to the benefits of biofertilizers, during last two decades, mexican agriculture has developed strategies for the biological fertilization of cultivated plants. Today, biofertilizers formulated from bacteria (i.e., Rhizobium spp., Bacillus spp., Pseudomonas spp., Azotobacter spp., and Azospirillum spp., among others) and mycorrihizae (i.e., Glomusintraradices, Gigaspora spp., and Acaulospora spp., among others) are commonly found (Armenta-Bojórquez et al., 2010; Malusá and Vassilev, 2014), although P-solubilizing micromycetes are not currently on the market. Therefore, the studies of P-solubilizing micromycetes adapted to specific environmental conditions are of particular interest.
In this sense, we found that Penicillium SP1 and P. hispanicum are able to solubilize at 100% the Ca3(PO4)2 in a Pikovskaya culture media; both strains achieved better solubilization than a Mexican native strain of Paecilomyces lilacinus, which was able to solubilize only 71.28 mg/L of P at 288 h of culture in Pikovskaya culture medium (Hernández-Leal et al., 2011). In another study on native SBM from a Mexican coffee plantation, Hernando-Posada et al. (2013) reported that Paecilomyces marquandii and Penicillium janthinelum released 1.9 and 7.0 mg/L of P from Ca3(PO4)2 in the culture medium at 24 days; these values were lower than those achieved by Penicillium SP1 and P. hispanicum. The ability of a fungal strain to release P from an inorganic source is known to be strain and species dependent and can also be influenced by multiple environmental factors (Zúñiga-Silva et al., 2016).
The PBED was able to show the main nutrimental factors in Ca3(PO4)2 solubilization by P. hispanicum. Also, a P increase of 19.7% was obtained with low N source ((NH4)2SO4), Mn+2 (MnSO4) and pH. Some authors have reported that the N source mediated the solubilization ability of fungi. Relwani et al. (2008) reported that KNO3 significantly increased the solubilization of Ca3(PO4)2 by a mutant strain of Aspergillus tubingensis in comparison with other N sources such as (NH4)2SO4, (NH4)NO3, asparagine and tryptophan. However, the analyzed amount of KNO3 was 0.5 g/L, a value higher than the best solubilization conditions for P. hispanicum, which only needed 0.25 g of (NH4)2SO4 as an N source.
In a related study, Chun-Qiao et al. (2008) reported the different effects of various C:N ratios on rock phosphate solubilization by fungal isolates. The content of soluble P released by Penicillium expansum increased with an increasing C:N ratio, up to 104.1 mg/L at a C:N ratio of 40:1 (10 g of glucose and 0.25 g of (NH4)2SO4). These data are in agreement with what we found in this study using a PBED: a low amount of (NH4)2SO4 (0.25 g/L) favored a high P solubilization of Ca3(PO4)2.
In another study, Scervino et al. (2011) reported that glucose- and (NH4)2SO4-based media showed the highest values (97.78 mg/L) of Ca3(PO4)2 solubilization by Penicillium purpurogenum in comparison with using fructose and L-asparagine as C and N sources. According to the molecular weight of added Ca3(PO4)2, P. purpurogenum solubilized 100% of P. In addition, P solubilization was optimized by Scervino et al. (2011) using a Box-Wilson design that varied the amount of C and N source. According to their results, the optimum amounts of glucose and (NH4)2SO4 were 13.66 and 0.34 g/L, respectively.
Recently, Padmavathi (2015) applied a PBED in the solubilization of Ca3(PO4)2 by Aspergillus niger in which P release was significantly influenced by glucose and (NH4)2SO4. Padmavathi (2015) suggested that the optimum (NH4)2SO4 value was 4.0 g/L. Our results with P. hispanicum suggest that a low value (0.25 g/L) of (NH4)2SO4 increased the P release from Ca3(PO4)2. This difference could be due to the utilized fungal species and also reveals the importance of fungal culture optimization.
Total protein production by P. hispanicum improved in presence of low amounts of glucose, yeast extract, inoculum and pH. This suggests that protein production occurs under slight stress conditions because protein production also improved in presence of high amounts of salts such as MgSO4, FeSO4 and Ca3(PO4)2. P-solubilizing fungi are well known to exhibit some enzyme activity, which contributes to P-solubilization; some enzymes include acid and alkaline phosphatase and phytase (Relwani et al., 2008; Gaind, 2016). Although enzyme activities were not evaluated in this study, total protein production suggests that there was a high amount of protein (according to total enzyme activity) in some of the culture conditions, for example, in the 2nd trial experiment. Future studies should evaluate and characterize these important enzymes. Recently, Gaind (2016) showed that depending on the fungal species, the mechanism for reducing pH and releasing P from inorganic sources may be different. For instance, in the solubilization of Ca3(PO4)2, Trichoderma harzianum increases its citric acid production to reduce the pH of the culture medium, while Aspergillus niger increases its phytase activity and decreases its acid phosphate activity.
Mycelial biomass is a significant parameter for evaluating the growth of fungi. According to the PBED, biomass production by P. hispanicum did not improve. We found the highest biomass in the central points, which suggest that the Pikovskaya culture medium is adequate for the biomass production of P. hispanicum. In the literature, several culture media for rock phosphate and Ca3(PO4)2 solubilization have been reported. For instance, Wakelin et al. (2004) used a basal medium (0.1 g NaCl, 0.4 g nH4C1, 0.78 g KNO3, 0.5 g MgSO4, 0.1 g CaCl2-2H2O and 10 g sucrose L-1) for rock phosphate solubilization and reported a high amount of biomass production by Penicillium bilaiae, Penicillium radicum and Penicillium sp.1, over 500, 300 and 250 mg/flask (100 mL of basal medium), respectively. More recently, Zhen et al. (2016) reported the biomass production of two P-solubilizing fungi: Penicillium oxalicum and Aspergillus niger. Under pH stress (1.5 to 6.5) in potato dextrose agar broth (PDAB), both solubilizing fungi showed high biomass production in comparison with P. hispanicum, studied in this work. Penicillium oxalicum and A. niger produced 497.28 and 389.13 mg of biomass/flask (100 mL of PDAB) at pH 4.5 and 2.5, respectively. In the case of P. oxalicum, biomass production was not possible at pH 2.5 and 1.5. The decrease in the pH of the culture medium is well associated with organic acid production (Oliveira-Mendes et al., 2016; Zhen et al., 2016); organic acids, in some cases, can reduce the biomass production of P-solubilizing fungi. This could be the reason why P. hispanicum had low biomass production despite showing high Ca3(PO4)2 solubilization. In addition, the pH values in Pikovskaya culture medium inoculated with P. hispanicum were low, between 2.8 to 2.83, during the evaluation period (2 to 6 days).
Conclusión
Penicillium hispanicum was able to solubilize 100% of Ca3(PO4)2 at six days of culture in Pikovskaya culture medium. The PBED revealed that P release by P. hispanicum was influenced by low values of MnSO4, (NH4)2SO4 and pH and a high amount of Ca3(PO4)2. Thus, P. hispanicum is a good candidate to use in field experiments of Ca3(PO4)2 solubilization in poor soils with low C and N sources, as glucose (as a C source) did not significantly affect Ca3(PO4)2 solubilization and only a low amount of (NH4)2SO4 (as an N source) was required.