Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista de la Facultad de Medicina Veterinaria y de Zootecnia
Print version ISSN 0120-2952
Rev. Med. Vet. Zoot. vol.59 no.1 Bogotá Jan./Apr. 2012
COMPOSITION AND BACTERICIDAL ACTIVITY AGAINST BENEFICIAL AND PATHOGENIC BACTERIA OF OREGANO ESSENTIAL OILS
FROM FOUR CHEMOTYPES OF Origanum AND Lippia GENUS
COMPOSICIÓN Y ACTIVIDAD BACTERICIDA CONTRA BACTERIAS BENÉFICAS Y PATÓGENAS DE ACEITES ESENCIALES DE CUATRO QUIMIOTIPOS
DE LOS GÉNEROS Origanum Y Lippia
L. Betancourt1,2*, V. Phandanauvong3, R. Patiño4, C. Ariza-Nieto4, G. Afanador-Téllez1.
1 Departamento de Producción Animal, Facultad de Medicina Veterinaria y de Zootecnia, Universidad Nacional de Colombia, sede Bogotá.
Cr. 45 nro 26-85, Bogotá (Colombia).
2 Facultad de Ciencias Agropecuarias, Universidad de La Salle.
Cr. 7 nro. 174-85, Bogotá (Colombia).
3 Laboratorio Interinstitucional de Detección y Monitoreo de Organismos Genéticamente Modificados, Instituto Nacional de Vigilancia de Medicamentos y Alimentos (Invima).
Av. Cll. 26 nro. 40-31, Bogotá (Colombia).
4 Corporación Colombiana de Investigación Agropecuaria (Corpoica).
Centro de investigación Tibaitatá, km 14 vía Mósquera, Cundinamarca (Colombia).
*Autor para correspondencia: lcbetancourt@unisalle.edu.co
Artículo recibido: 3 de febrero de 2012; aprobado:13 de marzo de 2012
ABSTRACT
Several studies have showed the antibacterial activity of oregano essential oils (OEO) chemotypes, carvarol and thymol against Gram positive and Gram negative pathogenic bacteria, but lack information on OEO antibacterial activity against beneficial bacteria. The object of this study was to compare the composition and minimum bactericidal concentration (MBC) of Origanum gender chemotypes: O. vulgare L. ssp. hirtum (OH), O. majorana (OM) y O. vulgare L. (OL) harvested in Colombia, against Lippia origanoides Kunth (LO) from Alto Patía in Colombia, and O. vulgare L. ssp. hirtum (OG) from Greece. The OEO composition was analyzed by gas chromatography coupled to mass spectrometer and its antibacterial activity by broth dilution and agar culture method. Three chemotypes were evaluated, OH and OG were carvacrol-type, LO and OL were thymol-tipe and OM was rich in sabinyl compound. The highest precursors content was showed to OEO from oregano varieties produced under greenhouse conditions at high altitude. The same bactericidal activity against Salmonella enteritidis (0.098 mg/mL) for OH and LO was founded. The lowest bactericidal activity against beneficial bacteria Lactobacillus acidophilus and Bifidobacterium breve was observed for OM (6.25 mg/mL) and LO (50 mg/mL), respectively. These results showed that LO native to Colombia had a comparable effect to OH and carvacrol against pathogenic enterobacteria and the lowest bactericidal activity against beneficial bacteria. These results showed a selective bactericidal effect against pathogenic beneficial bacteria of Origanum ssp. and L. origanoides chemotypes. A good potential to use L. origanoides as antimicrobial additive for animal and human health was found.
Key word: Bactericidal, Escherichia, Lactobacillus, Salmonella, oregano.
RESUMEN
Diversos estudios demuestran la actividad antibacterial de distintos quimiotipos de aceites esenciales de orégano (OEO), carvacrol y timol, contra bacterias patógenas Gram positivas y Gram negativas, pero muy poca información está disponible sobre el efecto de los OEO en bacterias benéficas. El objetivo de este estudio fue comparar la composición y concentración mínima bactericida (CMB) de OEO de quimiotipos del género Origanum: O. vulgare L. ssp. hirtum (OH), O. majorana (OM) y O. vulgare L. (OL) cultivados en Colombia, frente Lippia origanoides Kunth (LO) nativo del Alto Patía en Colombia y O. vulgare L. ssp. hirtum cultivado en Grecia (OG). Los OEO se obtuvieron por arrastre de vapor, la composición se determinó por cromatografía de gases acoplada a espectrometría de masas y la actividad antibacteriana mediante el método de dilución en caldo y cultivo en agar. Se evaluaron tres quimiotipos de OEO: OH y OG tipo-carvacrol, LO y OL tipo-timol y OM rico en compuestos sabinilo. Las variedades de orégano producidas bajo condiciones de invernadero a alta altitud mostraron un alto contenido de precursores. Un valor similar de concentración mínima bactericida contra Salmonella enteritidis (0.098 mg/mL) fue observada para carvacrol, OH y LO. La más baja actividad bactericida contra las bacterias benéficas Lactobacillus acidophilus y Bifidobacterium breve fue observada para OM (6.25 mg/mL) y LO (50 mg/mL), respectivamente. Estos resultados mostraron que LO, nativo de Colombia presentó un efecto antibacteriano comparable a OH y carvacrol contra enterobacterias patógenas y una baja actividad bactericida contra las bacterias benéficas. Estos resultados mostraron un efecto bactericida selectivo contra bacterias benéficas y patógenas de los quimiotipos Origanum ssp. y L. origanoides. Se comprobó un buen potencial de L. origanoides para uso como aditivo antimicrobiano para la salud humana y animal.
Palabras clave: Bactericida, Escherichia, Lactobacillus, Salmonella, orégano.
INTRODUCTION
Origanum genus covers a wide range of more than 60 plant species used as spice. The majority of them belong to Lamiaceae and Verbenaceae families; among these species, Origanum vulgare L. and Lippia graveolens Kunth, are considered species of economic importance with a large distinction between them (Padulosi et al. 2002).
Antimicrobial activity and mechanism of action for OEO as well as two of its main constituents, thymol and carvacrol, has been documented (Hammer et al. 1999; Ultee 1999; Nostro 2004; Trombetta 2005; Lambert et al. 2001). This property has been attributed to the presence of hydroxyl groups in their phenolic components (Ben et al. 2006); Ultee et al. (2002) showed that the presence of a free hydroxyl group and a delocalized electron system are essential for antimicrobial activity. The OEO has showed activity against Gram-negatives and Gram-positives bacteria (Dorman y Deans 2000).
The antibacterial action mechanism has been explained by its effect on altering bacterial membrane integrity. Lambert et al. (2001) found that the OEO damaged in-membrane integrity, which affects pH homeostasis and equilibrium of inorganic ions. Ultee et al, (1999) showed that carvacrol decreased intracellular potassium, the membrane potential and increased the extracellular potassium. But the effect of OEO is not only limited to the bacterial membrane, a transfer of monoterpene through the lipid bilayer to cell and the interaction with their components is suggested (Trombetta et al. 2005). On the other hand, Vattem et al. (2007) found that OEO inhibited the quorum sensing (QS) of Escherichia coli and Pseudomonas aeruginosa; QS is a mechanism that modulates the expression of genes involved in processes related to survival and pathogenesis. Based on these broad spectra of antibacterial activity, oregano seems to be one of the most inhibitory species ever tested; however, there are few references that compare the antimicrobial activity of Origanum gender species versus Lippia origanoides as well as the effect against pathogenic respect to benefic bacteria strains for animals and humans health.
It is well known that the regulation of essential oils production and synthesis of metabolites is integrated into the plant physiology and depends on the metabolic state and its adaptation to the ecosystem. Latitude and elevation determine the climate which, in turn, affects soil types and vegetation. In particular, Colombia shows that its altitudinal gradient along its mountains is associated with alteration in a number of environmental factors, such as air temperature, rainfall, wind exposure, light intensity, UV-B radiation, soil fertility, ozone density and partial CO2 pressure. The combination of all these factors exerts stress on plants, which triggers, not only in morphological and anatomical adaptations, but also biochemical changes and productivity patterns (Kofidis et al. 2003).
Colombian oregano has two different geographic origins: cultivated oregano which is European oregano used as a spice in foods. The wild oregano that bears a resemblance to Mediterranean oregano in terms of flavor, aroma, but its genus is Lippia, with an outstanding feature characterized by its differences in the essential oil composition reported for the same species from different local ecosystems. Cultivated oregano belongs to O. vulgare L., native to Greece and O. majorana, cultivated in Europe (Skoula and Harborne 2002). While wild oregano, classified as L. origanoides, is a species found in countries like Colombia, Mexico, Guatemala, Cuba, Venezuela and Brazil (Oliveira et al. 2007; Stashenko et al. 2010). L. origanoides grows at low altitudes with high temperatures (Albesiano and Rangel-Ch 2006) such as Patía region (23 °C). In contrast, O. vulgare L., is cultivated at high altitude and low temperature (13 ºC) in Sabana de Bogotá.
The OEO is composed of more than 50 phytochemically different molecules, terpens, phenols, alcohols, organic acids, aldehydes and ketones. Many Origanum plants are characterized by a wide range of volatile secondary metabolites and by the existence of chemical differences with respect to essential oil content, composition and antibacterial activity. Previous studies have agreed that it is necessary to separate and identify the active components with an antimicrobial effect in the essential oil. Chemo typing (biochemical composition) and accurate classification of the plant will allow us to evaluate the pharmacological action of the oil (Skoula and Harborne 2002) in order to find convenient practical applications.
Most studies have assessed the antimicrobial activity and the essential oil composition of Origanum genus against pathogenic microorganisms, while a few studies have been conducted to evaluate the antimicrobial activity of OEO against benefic strains bacteria for animals and humans health. Within pathogenic bacterial groups, Salmonella spp. is a major concern of public health and represents one of the most important causes of gastroenteritis in humans (Uribe and Suarez 2006). Because of that, the development of new antimicrobials that act on novel bacterial targets and that are less susceptible to resistance is of primary importance for the animal industry.
The aim of this study was to relate the chemical composition of the hydro-distilled essential oil by GC/MS with the antimicrobial activity of the essential oil of Origanum majorana, Origanum vulgare L., Lippia origanoides and Origanum vulgare L. ssp. hirtum by broth microdilution method against the following benefic microorganisms: Lactobacillus acidophilus, Bifidobacterium breve and the pathogen microorganisms: Salmonella enteritidis, Salmonella typhimurium and Escherichia coli.
MATERIALS AND METHODS
Plant material
Aerial parts of O. vulgare L. (OL) and O. majorana (OM) were collected from greenhouses of Sabana de Bogotá ecosystem and L. origanoides (LO) obtained from wild-growing populations located in the Alto Patía ecosystem. The plants were submitted and identified in the Herbarium of Universidad Nacional de Colombia, Faculty of Science.
Essential oils extraction and analysis
The essential oils were extracted using hydro distillation, with a Clevenger-type similar apparatus for 3 h and the oil was collected and stored at 4 ºC. An Agilent Technologies 6890 Plus gas chromatograph coupled with an ion trap detector mass spectrometer, equipped with a FID detector was used. A DB-5 capillary column, 60 m x 0.25 mm, film thickness 0.25um, (J & W Scientific, Folsom, CA, USA). Injector and detector temperatures, 240 oC; split ratio, 50:1 mL/min. The essential oils constituents were identified by comparison with their relative retention times and mass spectra, using data base NIST and Wiley Registry of Mass Spectral Data. A standard solution of n-alkanes was used to obtain the retention indices (Laboratorio de Cromatografía, Universidad Industrial de Santander).
Test microorganisms and preparation of inocula
The reference strains of American Type Culture Collection (ATCC) were obtained from the National Laboratory of Veterinary Diagnostic of Colombia Agricultural Institute (Bogotá, Colombia). Antimicrobial assay was against Lactobacillus acidophilus ATCC 4356, Bifidobacterium breve ATCC 15700, Salmonella enteritidis ATCC 13076, Salmonella typhimurium ATCC 14028 and Escherichia coli ATCC 25922 by broth dilution method. The concentration of S. enteritidis and S. typhimurium strains were confirmed on Mueller-Hinton broth plates (Oxoid CM337) in an aerobic atmosphere at 37 ºC for 24 h. B. breve was tested on MRS broth (MRSB, Oxoid, Basingstoke, Hampshire, UK) in an anaerobical condition at 37 ºC for 96 h. Lactobacillus acidophilus was confirmed on MRS broth in an anaerobic condition at 37 ºC for 24-48 h. After this period of time the culture was diluted in Peptone Buffer Solution (Oxoid CM225) and a concentration of 106 CFU/mL was obtained, and the final concentration of the microorganism in each cell of the assay was approximately 5 x 105 CFU/mL. Cell and inoculum were carried out for the recount on the plates in order to confirm the CFU concentration (NCCLS 1993).
Minimum bactericidal concentration
Determination of the minimum bactericidal concentration (MBC) of the OEO was carried out by using the broth microdilution method, described by the National Committee for Clinical Laboratory Standards (NCCLS 1993). The MBC is the lowest concentration of one product that is capable of inhibiting visible growth of a microorganism on the agar plate. Initial emulsions of oils were prepared at 100 mg/mL in sterile distilled water with 10% Tween 80 (Sigma-aldrich, St Louis, MO, USA). Serial dilutions of the stock solutions in broth medium, 50µL of Mueller-Hinton broth (MHB; Becton Dickinson, Sparks, MD, USA) and 50µL of stock solution, were prepared in a microtiter plate (96 wells), final volume of 100 µL by well (50 µL of serial dilutions and 50 µL of inoculum) ranging from 50 mg/mL - 0.0976 mg/mL. The final concentration of the microorganism in each well after inoculation was approximately 5 x 105 CFU/mL.
In all assays a positive growth control was included (well with bacterial inoculum in distilled water with 10% Tween 80, but without any oil) and a negative growth control (well with 100 µL of stock solution of oils emulsion at 50 mg/mL without the inoculum) (Peñalver et al. 2005). Bacterial growth was indicated by the presence of turbidity and a 'pellet' on the well bottom.
After incubation, 10 μl of broth was removed from each well and inoculated on Mac Conkey agar (E. coli), Xylose-Lysine-Desoxycholate Agar (Salmonella) and MRS agar (Lactobacillus and Bifidobacteria) and then incubated again at 37 °C for 24 h. After this, each well was read and total absence of bacterial colonies in the agar plate was considered a positive result. Every experiment was repeated three times for each oil or compound at each test concentration and the modal and MBC values were selected (Cosentino et al. 1999).
RESULTS
Composition of oregano essential oils
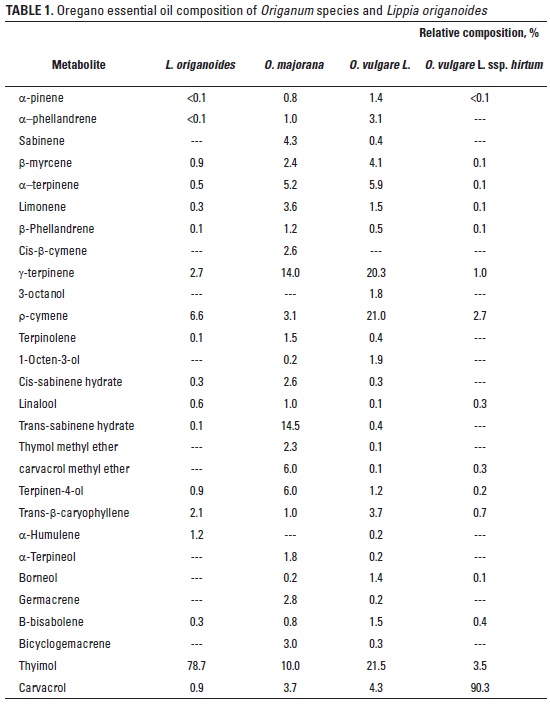
Fifty four compounds were identified in the volatile fraction of Origanum and Lippia genus essential oils (table 1). Major biochemically related groups of compounds were observed in O. vulgare ssp. phenolic monoterpenoids, mainly represented in oregano by carvacrol and thymol, their precursors, p-cymene and g-terpinene. Carvacrol showed the highest value in O. vulgare L. ssp. hirtum (90.3%) while thymol (78.7%) was highest in L. origanoides. L. origanoides is an endemic species Patía region in south Colombia. O. majorana was mainly represented by bicyclic monoterpenoids sabinene (4.3%) and cis- and trans-sabinene hydrates (17.1%), but also included g-terpinene (14.0%), terpinen 4-ol (6%), very low p-cymene (3.1%), thymol (10.0%), carvacrol (3.7%), carvacrol metyl eter (6%) and sesquiterpenoids componds germacrene and bicyclogermacrene. In contrast, the major volatile compounds found in O. vulgare L. was thymol (21.51%) and carvacrol showed the lowest value (4.3%), acyclic monoterpenoid b-myrcene (4.1%) and sesquiterpenoid trans-b-caryophyllene (3,7%).
Minimum bactericidal concentration
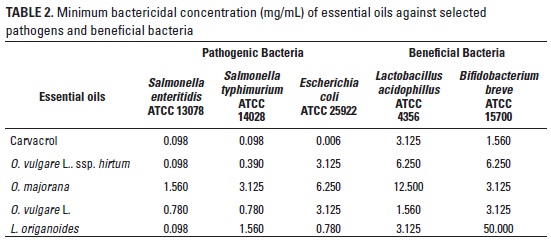
The MBC against S. enteritidis, S. typhimurium, E. coli, L. acidophilus and B. breve showed by O. vulgare L. (OL), O. vulgare L. ssp. hirtum (OH), O. majorana (OM), L. origanoides (LO) and carvacrol, are presented in table 2. Different bacteria demonstrated a wide range of sensitivity to different compounds. Carvacrol presented the lowest MBC against E. coli (0.006 mg/mL) and S. typhimurium (0.098 mg/mL) followed by L. origanoides against E. coli, and Origanum vulgare L. ssp. hirtum and O. vulgare L. against S. typhimurium. The lowest MBC against S. enteritidis was presented with carvacrol, Origanum vulgare L. ssp. hirtum and L. origanoides (0.098 mg/mL). The higher OEO concentrations were needed to act against E. coli respect to S. enteritidis and S. typhimurium. However, beneficial bacteria presented the higher MBC when they were compared with pathogenic bacteria. The greatest difference was for B. breve, while the MBC obtained for carvacrol was 1.56 mg/mL, to L. origanoides was 50 mg/mL. The lowest MBC against beneficial bacteria was obtained for O. majorana (6.25 mg/mL) against L. acidophilus and L. origanoides against B. breve. The O. majorana essential oil showed the lowest MBC against pathogenic and beneficial bacteria.
These results clearly showed that L. origanoides essential oil, rich in thymol from Patía ecosystem could have a desirable antibacterial effect.
DISCUSSION
This study found that L. origanoides is a thymol type species with a selective antibacterial activity against beneficial and pathogens microorganisms. Although, the subspecies O. vulgare L. ssp. hirtum is widely used as a spice under the name 'Greek oregano', it is considered to have the highest quality in the market. The essential oil composition of this subspecies, however, was not homogenous. Origanum genus can be divided into three chemotypes: carvacrol-type (O. vulgare L. ssp. hirtum), thymol-type (O. vulgare L.) and Sabinene-type (O. majorana) (Skoula et al. 1999). Franz (1990) observed that the most important factors for crude drug quality of oregano are: high carvacrol content in the essential oil, the quantity of the oil, and its sensorial characteristics. From this point of view, the Origanum genus has a large variability and thus provides an excellent basis for selection activities.
Lippia origanoides has been shown to tolerate dry soils and long droughts (Camargo-Parra and Rodríguez 2008) which is a typical condition of Patía region. Previous studies conducted in Brazil by Do Santos et al. (2004) showed that analyses of the oils from L. origanoides leafs demonstrated that carvacrol was the major component together with g-terpinene, thymol, and p-cymene. In contrast, in this study, carvacrol was present only as a trace constituent (0.9%). Ramirez et al. (2009) reported timol contents from 1.4 to 74.4 %. New chemotypes have been proposed for this specie (Lee et al. 2003). It is also known that crop, weather and seasonal conditions can affect secondary metabolite production (Skoula and Harborne 2002).
The O. majorana composition was similar to the thujanes group which are mainly represented by sabinene and by cis- and trans-sabinene hydrates, but also include their derivatives cis- and trans-sabinene hydrate acetates, trans-sabinol and sabina ketone, together with a-thujene. These are referred to as 'sabinyl' compounds. The sabinyl and the cymyl compounds are mutually exclusive. This profile corresponds to sweet marjoram described by Tabanca et al. (2004). Most Origanum species are rich either in sabinyl compounds or cymyl compounds. It was suggested that one or more components of the cymyl pathway act throughout the Origanum gender, to suppress the sabinyl pathway (Skoula et al. 1999).
Our results were similar of those reported by Skoula and Harborne (2002) who have shown that the essential oil of O. vulgare L. is characterized principally by thymol and carvacrol (24.7 and 14.0% of the total oil, respectively) and by their two monoterpene precursors, g-terpinene and p-cymene, 11.7 and 14.6% of the total oil, respectively. In this study, the highest content of precursors was observed in oregano species that grows in green houses; it is known that composition depends on the climatic and environmental conditions under which they were grown (Sangwan et al. 2001).
Despite of great variability in the content of carvacrol and thymol, the European oreganos have been considered the best ones, and in general they have a higher content of carvacrol (Do Santos et al. 2004). However, in tropical America and Africa ecosystems, chemotypes with a high thymol contents might be important. Additionally, carvacrol has shown to have more negative effects than thymol (Lee et al. 2003), raising the question about if a high carvacrol content in the essential oil is a good indicative of its quality.
Previous data and our results confirm that most Origanum species are rich either in sabinyl or cymyl compounds but never together. Skoula and Harborne (2002) found that the hybrid between sabinyl-rich O. microphyllum and cymyl-rich O. vulgare L. ssp. hirtum contains mainly cimyl compounds while the sabinyl constituents have been suppressed.
This study found an bactericidal selective effect against pathogenic beneficial bacteria, Lactobacillus and Bifidobacterium were less sensitive than the pathogens Salmonella and E. coli to both carvacrol and OEO; in contrast, other studies have shown that OEO have a non-selective antibacterial activity against Gram-negatives and Gram-positives bacteria. Moreover, most reports on the antimicrobial activity of oregano involved pathogenic bacteria, only a limited number of authors studied the inhibitory effects of Origanum species on Lactobacillus, additionally, this study found that the lowest bactericidal activity was presented by L. oritanoides against Bifidobacterium breve. Similar to this study, Si et al. (2005) concluded that most of the oil/compounds demonstrated high efficacy against S. typhimurium DT104, E. coli O157:H7 and E. coli K88 with little inhibition towards Lactobacilli and Bifidobacteria. The results on OEO antibacterial activity show a good potential, including efficacy and selectivity towards bacterial pathogens, in reducing human and animal bacterial pathogens in intestinal tract. Additionally, Zaika and Kissinger (1981) have found that lactic acid bacteria became resistant toward the toxic effects of OEO when sublethal concentrations were applied and stimulated the growth and acid production.
The higher bactericidal activity was associated with chemotypes oils that have a high percentage of phenolic components (carvacrol and thymol) in comparison with oils containing high sabinyl compounds such as O. majorana. Similar results have been obtained for OEO from Origanum species against Salmonella (Si et al. 2005; Peñalver et al. 2005; Ben et al. 2006) and O. majorana (Hammer et al 1999). Unlikely, in our study the high thymol specie was Lippia origanoides, an established species in American tropical countries such as Colombia.
Compounds such as g-terpinene and p-cymene did not have any activity against the bacterial strains tested, whereas carvacrol and thymol exhibited high levels of antimicrobial activity (Dorman and Deans 2000).
Michiels et al. (2009) showed synergism of carvacrol and thymol, carvacrol inhibiting the growth of S. typhimurium at 0.4 mg/mL, but carvacrol + thymol inhibited at 0.1 mg/mL. Synergism between carvacrol and its biological precursor p-cymene had been noted when acting on Bacillus cereus (Oliveira et al. 2007), but no antimicrobial activity has been reported for p-cymene or g-terpinene (Dorman and Deans 2000). In this present study those effects were not tested.
Different mechanisms have been evaluated to explain the antibacterial activity of essential oils. Thymol and carvacrol are able to disintegrate the outer membrane of Gram-negative bacteria, release lipopolysaccharides (LPS), increase the permeability of the cytoplasmic membrane to adenosine triphosphate (ATP) and allow ions to leave the cytoplasm. The mechanism of inhibition appeared to be a combination of interference with acylated homeserine lactone (AHL) activity and modulation of the AHL synthesis. Vattem et al. (2007) results also indicated that various phytochemical extracts which inhibited quorum sensing (QS) also inhibited swarming of pathogenic bacteria, known to be modulated by QS. The observation that phytochemicals from foods can inhibit QS related processes opens up an exciting new strategy for antimicrobial chemotherapy and lead to the discovery of new category of antibiotics which can overcome the issues related to antimicrobial resistance. Due to these effects, essential oils as antimicrobial agents are considered to be low risk for resistance development by pathogenic microorganisms (Vattem et al. 2007; Aslim and Nihal 2008).
CONCLUSIONS
According to our results, a bactericidal selective effect against pathogenic beneficial bacteria of Origanum ssp. and Lippia origanoides chemotypes was found. This study showed that Lippia origanoides, a native species from Patía, Colombia, can be considered thymol type and presented a bactericidal activity similar to Greek oregano species (carvacrol type) against pathogen bacteria, but less activity against beneficial bacteria. This results show a good potential of this species to be used as an antimicrobial additive for animal and human health.
Acknowledgments
Laboratorio de Toxicología, Laboratorio de Cromatografía de la Universidad Industrial de Santander por el análisis de AEO, Laboratorio de Microbiología de Corpoica-Ceisa, Vicerrectoría de Investigaciones, Universidad de La Salle y Ministerio de Agricultura y Desarrollo Rural.
REFERENCES
1. Albesiano S, Rangel-Ch JO. 2006. Estructura de la vegetación del cañón del río Chicamocha, 500-1200 m; Santander Colombia: una herramienta para la conservación. Caldasia. 28(2): 307-325. [ Links ]
2. Aslim B, Nihal Y. 2008. In vitro antimicrobial activity of essential oil from endemic Origanum minutiflorum on ciprofloxacin-resistant Campylobacter spp. Food Chem. 107: 602-606. [ Links ]
3. Ben A, Combes S, Preziosi-Belloy L, Gontard N, Chalier P. 2006. Antimicrobial activity of carvacrol related to its chemical structure. Lett Appl Microbiol. 43(2): 149-154. [ Links ]
4. Camargo-Parra AA, Rodríguez NF. 2008. Respuestas fenotípicas de Lippia alba y Lippia origanoides (Verbenaceae) a la disponibilidad de agua en el suelo. Acta Biol Colomb. 13(3): 133-148. [ Links ]
5. Cosentino S, CIG Tuberoso, B Pisano, M Satta, V Mascia, E Arzedi, and F Palmas. 1999. In vitro antimicrobial activity and chemical composition of sardinian thymus essential oils. Lett Appl Microbiol. 29:130-135. [ Links ]
6. Dorman HJ, Deans SD. 2000. Antimicrobial agents from plants: antibacterial activity of plant volatile oils. J Appl Microbiol. 88(2): 308-316. [ Links ]
7. Do Santos FJ, Lopes JA, Cito GL, et al. 2004. Composition and biological activity of essential oils from Lippia origanoides H.B.K. J Essen Oil Res. 16: 504-506. [ Links ]
8. Franz C. 1990. Sensorial versus analytical quality of marjoram. Herb Hung. 29: 79-86. [ Links ]
9. Hammer KA, Carson CF and Riley TV. 1999. Antimicrobial activity of essential oils and other plant extracts. J Appl Microbiol. 86(6): 985-990. [ Links ]
10. Kofidis G, Bosabalidis AM, Moustakas M. 2003. Contemporary seasonal and altitudinal variations of leaf structural features in oregano (Origanum vulgare L.). Ann Bot. 92(5): 635-645. [ Links ]
11. Lambert RJ, Skandamis PN, Coote P, Nychas GJ. 2001. A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. J Appl Microbiol. 91(3): 453-462. [ Links ]
12. Lee KW, Everts H, Kappert HJ, Yeom KH, Beynen AC. 2003. Dietary carvacrol lowers body weight gain but improves feed conversion in female broiler chickens. J Appl Poult Res. 12(4): 394-399. [ Links ]
13. Michiels J, Missotten JA, Fremaut D, Smet S, Dierick NA. 2009. In vitro characterization of the antimicrobial activity of selected essential oil components and binary combinations against the pig gut flora. Anim Feed Sci Tech. 151(1): 111-127. [ Links ]
14. [NCCLS] National Committee for Clinical Laboratory Standards. 1993. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically-Third Edition. Approved Standard NCCLS Document M7-A3. 13(25): Villanova (PA): NCCLS. [ Links ]
15. Nostro A, Blanco A, Cannatelli MA, Enea V, Flamini G, Morelli I, Sudano Roccaro A, and V Alonzo. 2004. Susceptibility of methicillin-resistant staphylococci to oregano essential oil, carvacrol and thymol. Fems Microbiol Lett. 230(2): 191-195. [ Links ]
16. Oliveira DR, Gilda GL, Bizzo HR, Daíse L, Daniela S, Alviano C, Leitao SG. 2007. Chemical and antimicrobial analyses of essential oil of Lippia origanoides H.B.K. J Ethnopharm. 108: 103-108. [ Links ]
17. Padulosi S, Leanman D, Quek P. 2002. Challenges and opportunities in enhancing the conservation and use of medicinal and aromatic plants. J Herb Spices Med Plant. 9(10): 243-267. [ Links ]
18. Peñalver P, Belén H, Borge C, Astorga R, Romero R, Perea A. 2005. Antimicrobial activity of five essential oils against origin strains of the Enterobacteriaceae family. APMIS. 113(1): 1-6. [ Links ]
19. Sangwan NS, Farooqi AH, Shabih F, Sangwan RS. 2001. Regulation of essential oil production in plants. Plant Growth Reg. 34: 3-21. [ Links ]
20. Si W, Gong J, Tsao R, Zhou T, Yu H, Poppe C, Johnson R, Du Z. 2005. Antimicrobial activity of essential oils and structurally related synthetic food additives towards selected pathogenic and beneficial gut bacteria. J Appl Microbiol. 100(2): 296-305. [ Links ]
21. Skoula M, Gotsiou P, Naxakis G, Johnson CB. 1999. Chemosystematic investigation on the mono- and sesquiterpenoids in the genus Origanum (Labiatae). Phytochemistry. 52(4): 649-657. [ Links ]
22. Skoula M, Harborne J. 2002. The taxonomy and chemistry of Origanum genus. In: Kintzios SE, editor. Oregano, the genera Origanum and Lipia. London and New York: Taylor & Francis. p. 67-105. [ Links ]
23. Stashenko EE, Martínez JR, Ruíz CA, Arias G, Durán C, Salgar W, Cala M. 2010. Lippia origanoides chemotype differentiation based on essential oil GC-MS and principal component analysis. J Sep Sci. 33(1): 93-103. [ Links ]
24. Tabanca N, Özek T, Baser KHC. 2004. Comparison of the essential oils of Origanum majorana L. and Origanum x marjoricum Cambess. J Essent Oil Res. 16: 248-252. [ Links ]
25. Trombetta D, Castelli F, Sarpietro MG, Venuti V, Cristani M, Daniele C, Saija A, Mazzanti G, Bisignano G. 2005. Mechanisms of antibacterial action of three monoterpenes. Antimicrob. Agents and Chemother. 49(6): 2474-2478. [ Links ]
26. Ultee A, Kets EOW, Smid EJ. 1999. Mechanisms of action of carvacrol on the food-borne pathogen Bacillus cereus. Appl Environm Microbiol 65(10): 4606-4610. [ Links ]
27. Ultee A, Bennink MH, Moezelaar R. 2002. The phenolic hydroxyl group of carvacrol is essential for action against the food-borne pathogen Bacillus cereus. Appl Environ Microbiol. 68(4): 1561-1568. [ Links ]
28. Uribe C, Suárez MC. 2006. Salmonelosis no tifoidea y su transmisión a través de alimentos de origen aviar. Colomb Med. 37(2): 151-156. [ Links ]
29. Vattem DA, Mihalik K, Crixell SH, McLean RJ. 2007. Dietary phytochemicals as quorum sensing inhibitors. Fitoterapia. 78: 302-310. [ Links ]
30. Zaika LL, Kissinger JC. 1981. Inhibitory and stimulatory effects of oregano on Lactobacillus plantarum and Pediococcus cerevisiae. J Food Sci. 46(4): 1205-1210. [ Links ]