INTRODUCTION
The Colombian creole horse is considered one of the country's most valuable genetic resources, their phenotypic qualities and sporting performance are characteristic, being one of the greatest reproductive and commercial importance species at national and international level (Jiménez and Ariza 2005).
In horses, cardiovascular exploration requires skills for processing, and abilities to perform and interpret complementary diagnostic tests that allow recognize sounds and/or physiological arrhythmias of pathological, as well as cardiac abnormalities during rest or triggered by exercise (Durando 2003). To do this, it is necessary to have knowledge of equine cardiac physiology, and cardiovascular parameters of equines, which may vary by breeds (Garrido-Lestache 2003).
In horses, cardiac abnormalities may occur in the absence of disease and are considered physiological due to the influence of vagal tone at resting condition. Among these are sinus arrhythmia, sinoatrial block, sinus bradycardia, sinus arrest, migratory pacemaker, atrioventricular blocks of first, and second degree. However, they should be differentiated from pathological cardiac disorders, such as atrial fibrillation, third- degree atrioventricular block, atrial and ventricular extrasystole (Fernandes et al. 2004).
Cardiac parameters, such as heart rate (HR), heart's electrical conduction, cardiac rhythm, AP, among others, may vary between breeds, sexes, and at different stages of animal's life (Diniz et al. 2008; Magdesian 2004; Melchert et al. 2012). Thus, there are gaps, and the literature is very scarce in regard to electrocardiography in Colombian creole horses, so it is necessary to standardize the main cardiovascular parameters in creole horses of the Bogotá Savannah, and thus expand the cardiovascular system clinical examination in Colombian creole horses in this region. The study pretended to standardize electrocardiographic parameters and AP of Colombian creole horses of the Bogotá Savannah, clinically normal, at different ages and in both sexes.
MATERIALS AND METHODS
This project was approved by the ethic and animal welfare committee of the Agrarian Sciences Faculty of the Agrarian University Foundation of Colombia (FCA-UNIAGRARIA), registered at the Research Committee of the Veterinary Medicine Program with CIPA-VET-003 protocol.
Location
The study was conducted in the region of the Bogotá Savannah, which is located in the department of Cundinamarca, on the Eastern Mountain Range, formed on a plateau with an average altitude of 2.600 masl. (Montoya and Reyes 2005). And facilities of ECOPARQUE research center in Tenjo Cundinamarca, of Universidad Agraria de Colombia (UNIAGRARIA).
Animals and experimental groups
100 clinically normal horses were evaluated between males and females aged 1 to 19 years old. The animals were divided into four groups of 25 horses (G1, G2, G3, G4), according to age. Group 1 was composed of horses between 6 months to 1 year old, group 2 was composed of horses between 1 year and 4 years old, group 3 composed of horses between 4 and 10 years old, and the group 4 composed of horses older than 10 years old.
Experimental Protocol
All animals were subjected to general physical examination with a prior medical history, followed by specific cardiovascular system physical examination. During the clinical examination were taken into account aspects, such as horse's attitude, and alertness, body condition, and were examined parameters such as mucous color, capillary refill time, cardiac auscultation, respiratory, and temperature. The age of the animals was determined by the records of the animals on farms.
Electrocardiographic examination
The ECG examination was performed using a computerized electrocardiograph (ECG PC TEB®). The evaluated animals were previously in stables, placed in a squeeze chute, and kept in station on a rubber mat to prevent interference on the ECG tracing. Electrodes were attached to the skin by metal clamps covered with plastic, using alcohol and gel as a contact in attachment points; then they were subjected to 10 minutes of adaptation period, followed by 5 minutes of ECG evaluation in all the groups of animals. The tracing was recorded in the I, II, Ill bipolar leads; aVR, aVL, and aVF unipolar leads, placing the electrodes at the olecranon region in the upper limbs, and the patella in the pelvic limbs, according to Fernandes et al. (2004). Precordial leads were also evaluated, fixing the electrodes in different positions of the thorax: V1, at the right low sixth intercostal space level under the costocohondral union; V2, at the left low sixth intercostal space level; V4, at the left high sixth intercostal space level, and V10 at the spinous process of the seventh thoracic vertebra, based in methods cited by Fernandes et al. (2004), Bello et al. (2012) and Costa et al. (2014). ECGs were analyzed in the II bipolar lead (LU), at the speed of 25 mm/second, calibrated for centimeter equals to 1mV.
Measurement of blood pressure
The measurement of arterial pressure (AP) was performed by non-invasive method by pulse oscillometry (Dixta® modelo DX2710 - Dixtal-Manaus-AM), used in horses (Canola et.al 2002) and with which it has been obtained blood pressure results closer to those given by doppler vascular method (Pereiraneto et al. 2014). The diastolic arterial pressure (DAP), mean (MAP), and systolic (SBP) were automatically determined. Seven measurements were performed for each animal, being excluded pressures with variations higher at 10%. Measurements were made with an occlusive cuffon the tail base, at the coccygeal artery. The inflatable cufFarea had a length corresponding to 80% of the tail circumference and a 20% width of the same.
Statistical method
The data were evaluated by descriptive statistics to obtain the mean, and standard deviation subsequently they underwent to variance analysis (One way ANOVA), the Kruskal-Wallis test was performed followed by Dunn's test for multiple comparisons; The minimum level of significance was set at 95% probability, with significant values of P < 0,05. All analyses and graphics were made by the "Graphpad Prism version 5.00 for Windows®", Graphpad Software, San Diego, California, USA program.
RESULTS
Cardiac Rhythm
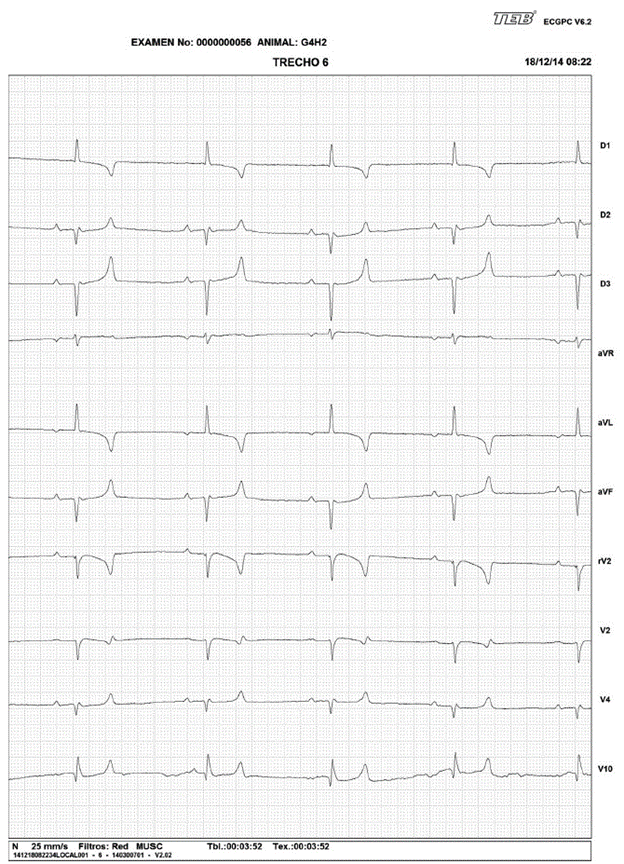
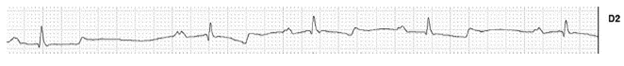
In the heart rate evaluation, the presence of sinus rhythm in 99 animals (99%) was observed; Meanwhile, two animals showed sinus rhythm with intraventricular conduction defect (ICD), showing characteristics in the ECG similar to the left fascicle anterior block (2%), being a female of G2 and another female of G4 (Figure 1). The presence of sinus arrhythmia was observed in 1 animal (1%), being a female of G3 (Figure 2). A female of G2 (1%) presented arrhythmia, characterized by the presence of 3 premature ventricular complexes.
Electrocardiogram (ECG)
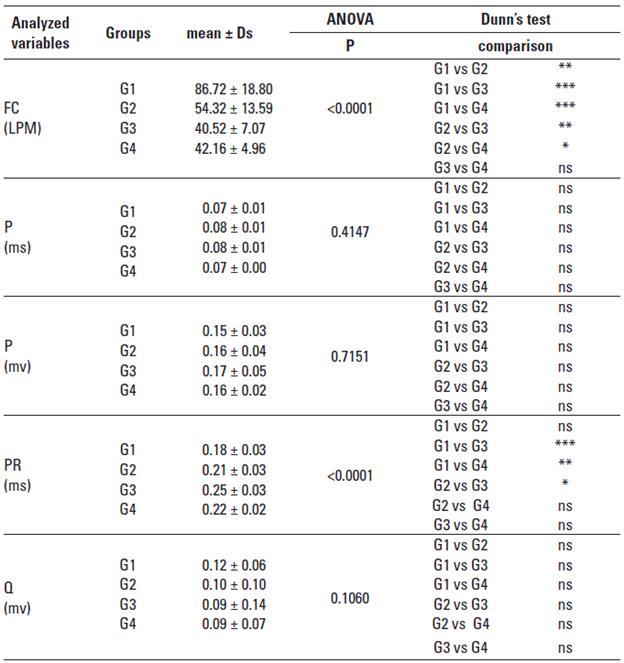
Heart Rate: The younger horses group, the G1, showed the highest mean HR of the four groups, obtaining significant differences compared to groups G2, G3, and G4 (p < 0.0001). The G4, and G3 groups were the groups showed lower heart rates, the G1 group had a double value of beats per minute that these two groups (Table 1). There were no significant differences between males and females of the same group.
TABLE 1 Means, standard deviation, and statistical data relating to heart rate/heartbeat per minute (HR/HPM), P wave in milliseconds (Pms), P wave in millivolts (Pmv), and PR interval in milliseconds (PRms).
ns = non significant (p > 0.05), * significant (p between 0.01 and 0.05), ** very significant (p between 0.001 and 0.01), *** extremely significant (p < 0.001), according to Martinez and Carvalho (2010).
P Wave: Regarding the P wave, both in duration and amplitude, there were no significant differences between the four groups of animals. There were no significant differences between males and females of the same group. 3 types of P wave were identified: Bifid, unique, and biphasic (Figure 3). The unique, and positive P wave was the one that presented at most, primarily in the G1, G2, and G4 groups; for the G3 group was frequent the Bifid P wave, being the second morphology in frequency of occurrence in the G1, G2, and G4 groups. The P wave biphasic showed the lowest frequency in all groups.
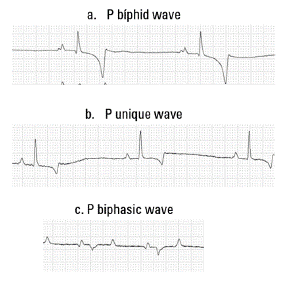
FIGURE 3 Morphologies of the P wave identified in this study. (a) P Biphid, (b) P unique and (c) P biphasic.
The PR interval showed statistically difference significantly less between the G1 group compared to G3 (p < 0.001), and G4 (p < 0.01); and between G2, and G3 (p < 0.05). Being the PR interval of G1 group the lower value between the groups (Table 1). There were no significant differences between males and females of the same group.
QRS Complex: The QRS complex duration showed statistically difference significantly less for G1 animals respectively compared with the G3, and G4 animals (p < 0.0002) (Table 2). There were no significant differences between G1 and G2, neither between males and females of the same group.
QRS types found correspond to QR, QRS, R, and RS type (Figure 4) being the QR type of the highest frequency in all four groups evaluated, followed by the R type in groups G2, G3, and G4, the QRS type and finally the RS type.
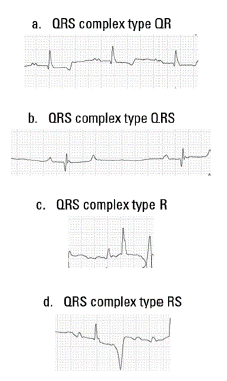
FIGURE 4 Morphologies of the QRS complex founded in this study. (a) QR type, (b) QRS type, (c) R type and (d) RS type.
R Wave: The G3 and G4 groups were the ones who obtained a higher value for the R wave amplitude and found highly significant differences between G1 and G3 groups, G1 and G4, and between G2 and G4 (p < 0.001) (Table 2). There were no significant differences between males and females of the same group.
T Wave: The T waves morphologies found were unique positive, unique negative and biphasic (Figure 5). The biphasic T wave was the most frequent wave type present in the G2, G3, and G4 groups, and the second in the G1 group. In the G1 predominated the T wave unique negative, being this the second most commonly found in the study, especially in the G1, G3, and G4 groups.
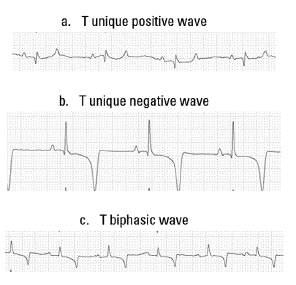
FIGURE 5 Morphologies of the T wave founded in this study. (a) T unique positive, (b) unique negative and (c) T biphasic.
QT interval: The QT interval was statistically higher in G3 and G4 groups. Significant differences between G1 and G3, G1 and G4, and between G2 and G4 groups (p < 0.001) (Table 2) were observed. There were no significant differences between males and females of the same group.
TABLE 2 Means, standard deviation, and statistical data relating to QRS complex in milliseconds (QRSms), R wave amplitude in millivolts (Rmv), QT interval in milliseconds (QTms), and T wave amplitude in millivolts (Tmv).
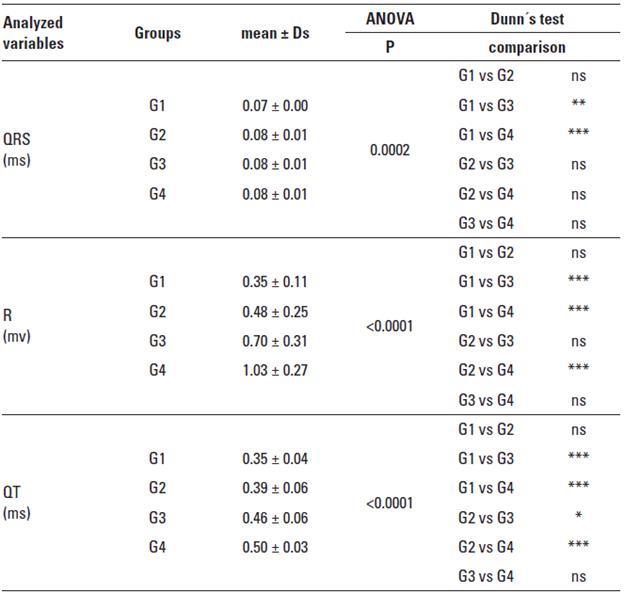
ns = non significant (p > 0.05), * significant (p between 0.01 and 0.05), ** very significant (p between 0.001 and 0.01), *** extremely significant (p < 0.001), according to Martinez and Carvalho (2010).
Cardiac axis: The cardiac axis showed statistically significant differences for animals of G1 compared to the other groups (p < 0.001), being predominantly negative in this group with deviation to the left, and cranial (Table 3).
TABLE 3 Means, standard deviation, and statistical data relating to the cardiac axis (Axis), Systolic Arterial Pressure in mmHg (SAPmmHg), Mean Arterial Pressure in mmHg (MAPmmHg) and Diastolic Arterial Pressure in mmHg (DAP mmHg).
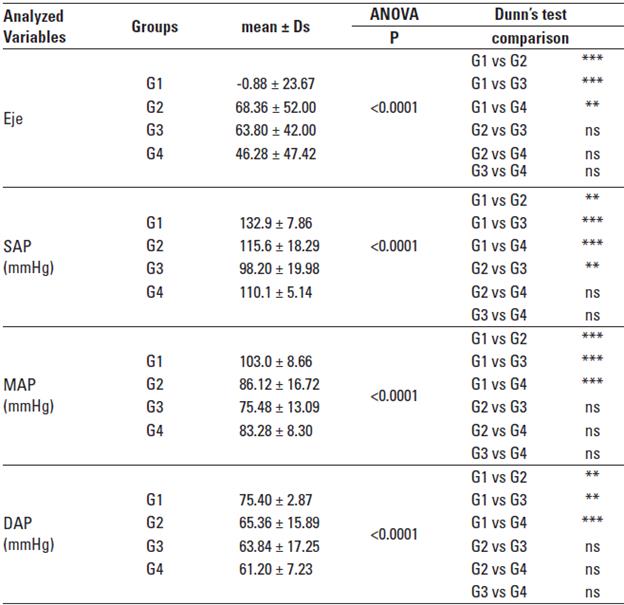
ns = non significant (p > 0.05), * significant (p between 0.01 and 0.05), ** very significant (p between 0.001 and 0.01), *** extremely significant (p < 0.001), according to Martinez and Carvalho (2010).
Arterial pressure: SAP values were higher in G1 and G2 groups. G1 showed significant differences compared to the G2 (p < 0.01), G3, and G4 (p < 0.001) groups. The G2 group presented only a significant difference compared with G3 animals (p < 0.01). MAP was higher in animals of the G1 group, showing highly significant differences compared to the other groups (p < 0.001). The G1 group had also higher values for DAP, being highly significant when compared to groups G2, G3 (p < 0.01), and G4 (p < 0.001) (Table 3).
DISCUSSION
Orientation, amplitude, and duration of ECG waves depend on various factors, including the horse's age, breeds, sex, size of the heart chambers, the degree of training, ventilation phase, among others (Bonagura and Reef 2004).
The HR in Colombian creole horses of this study was 86.72 ± 18.80 for foals between 6 months to one year old, 54.32 ± 13.59 for horses between 1 year to 4 years old, 40.52 ± 7.07 for animals between 4 and 10 years old, and finally 42.16 ± 4.96 for horses older than 10 years old. Similar results were described in adult horses, considering the HR normally between 28 to 44 beats/minute, diminishing in trained racing horses, and increasing in young horses, foals, and healthy but nervous animals (Bonagura and Reef 2004). However, different HR benchmarks according to the animal's breed have been described in the literature (Melchert et al. 2012).
The HR results of this study obtained in foals between 6 months to 1 year old are consistent with the HR values of 65 to 135 beats per minute previously described in foals (Lombard 1990), meanwhile, differ with the HR values of 62.290 ± 16.56 beats per minute described in English Thoroughbred breed horses of 2 to 11 months old (Fernandes et al. 2004).
The literature is scarce regarding the HR of animals in Group 2 and 3 of this study, therefore the discussion of this animals group started from the analysis of various items. In the study of Fernandes et al. (2004) were evaluated horses over one year old of English Thoroughbred breed with HR values of 49.960 ± 8.590. In adult horses for hard testing at rest, were described HR of 35.3 to 38.8 beats per minute for the Arab breed, for Mangalarga and Mestiza breed, HR values of 49 to 51 beats per minute (Dumont et al. 2010).
In Lusitano Thoroughbred adult horses, the evaluation of normal HR was considered between 25 to 46 beats per minute (Melchert et al. 2012).
References of HR values were described for adult equines between 25 to 40 beats per minute (Feitosa 2004). While similar these values, the results indicate minimal differences in HR values for Colombian creole horses of the G2 and G3 groups.
HR values of G2 group showed higher when compared with HR described of Mangalarga Marchador breed horses from 1 to 3 years old (Diniz et al. 2008), however, HR parameters of animals of the G3 group were lower than those described in the results of this author for horses between 4 and 10 years old of the same breed.
In a study performed in Colombian creole horses in the Piedemonte llanero region, average heart rates found in animals aged between 1 and 5 years old were slightly lower than those found in horses of group G2 of this study. Also, G1 animals also showed higher HR values, than those reported by the authors described (Corredor-Matus et al. 2005). The differences in environmental and climatic conditions, feeding, management, and purpose of these animals can be causes of this difference (Corredor-Matus et al. 2005). Although the studies agree that higher heart rates are found in younger animals, also slight differences between breeds are found.
HR Parameters described in Mangalarga Marchador breed equines showed mean values of38.8000 ± 4.696 beats per minute in horses older than 10 years old (Diniz et al. 2008); these values were lower to those shown in animals of G4. The mean heart rates of G3 and G4 groups obtained in this study were higher than those reported in Lusitano Thoroughbred horses at rest between 4 to 12 years old (Melchert et al. 2012). Possibly this increase is due to the geographical, environmental and climatic conditions. To verify this fact, other investigations under different conditions would have to be carried out.
No significant differences for HR between males and females of the same group were found. Similarly, other authors did not find gender differences for HR in their study performed in creole horses from Piedemonte llanero (Corredor-Matus et al. 2005).
Furthermore, 99% of the animals showed sinus rhythm, being predominant in the animals of this study, similar results were reported (Diniz et al. 2008; Fernandes et al. 2004), who observed a sinus rhythm in foals, Thoroughbred and Mangalarga Marchador breeds horses respectively evaluated. The presence of sinus arrhythmia has been slightly evidenced in animals older than one year old, as in this study (1%) (Fernandes et al. 2004; Melchert et al. 2012; Vicenzi et al. 2000). Meanwhile, the presence of sinus arrhythmia in 13.3% of adult Mangalarga horses at rest was described (Diniz et al. 2008).
Also, 100% of the foals of this study showed sinus rhythm, coinciding with reports (Fernandes et al. 2004; Lombard 1990) that described this rhythm as normal in foals. It is described that foals undergoing to ECG evaluation do not present respiratory sinus arrhythmia but may present sinus tachycardia and premature supraventricular complexes considered benign in most foals, being indicative of nonspecific stress reactions (Lombard 1990). Such alterations were no evidenced in foals in this study, possibly attributed to the contact they have had from birth with those people responsible for their care, as well as the good holding of this animals group studied which considerably reduces their stress.
Premature ventricular complexes are considered as pathological arrhythmias (Dörner and Godoy 2009). These results demonstrate the importance of ECG evaluation, as an important component of the clinical examination in creole horses of the Bogotá Savannah, as well, the female of G2 presented this arrhythmia, showed no clinical signs or symptoms of disease, becoming the ECG test in a monitoring tool and supporting, in the arrhythmia evaluation in this animal. Moreover, in cases like these, it is recommended monitoring the frequency and heart rate through the Holter test within 24 hours of evaluation.
The presence of intraventricular conduction disturbances has been rarely reported in horses; branches blocks are rare, and difficult to identify in this species (Bonagura and Reef 2004). The morphological findings described for an evidenced blocking in a female of G2 and another of G4, were based on human literature and veterinary, identifying this for its characteristics in the ECG, as an anterior block of the left fascicle (Kulbertus and Demoulin 1976; Tilley et al. 1992; Watt and Pruitt 1965). Experimental studies in dogs have shown that alterations consistent with the anterior blocking of the fascicle may occur either by lesions in the septal branch or in the anterior branch (Myerburg et al. 1972). In humans, the anterior fascicle is usually more susceptible to injury by their anatomical, and physiological characteristics including, its little irrigation, it is also long and thin and is located in the outflow tract of the left ventricle (MacAlpin 2003). In cats, this type of blocks has been reported in feline hypertrophic cardiomyopathy, as well as left ventricular hypertrophy, hyperkalemia, and inadequate heart position within the thorax (Tilley et al. 1983). Animals that had this type of block showed no signs or symptoms of disease, this is another alteration that also needs to be monitored and accompanied, as well as correlated with ecodopplercardiographic tests and radiography, which were not allowed to perform in these animals, meanwhile periodical clinical follow-up, and ECG assessment for these animals is suggested.
The equine heart has anatomical features, which make their electrocardiogram differs in some aspects from other species. The surface location of the sinoatrial node, for example, leading to the electrical transmission through the atria, is made in a way that the P wave generated can be bimodal, thus, the electrical activity expands radially through the right atrium, causing the first component of the bimodal P wave in the ECG. Depolarization of the left atrium seems to occur spontaneously without contributing to the P wave formation of ECG in the body surface, being the depolarization of the atrial septum the second component of the bimodal P wave (Dörner and Godoy 2009).
The P wave is generated by atrial depolarization, being normally on a bifid P wave, but can also be found biphasic, unique, and polyphasic P wave in healthy horses. The P wave morphology may vary under the influence of vagal tone. In the presence of tachycardia, the P wave can be shortened and increase its amplitude and is followed by a wave of atrial repolarization deflecting downward the PQ segment (Bonagura and Reef 2004; Garrido-Lestache 2003; Melchert et al. 2012). This is consistent with the different morphologies of P wave found in the groups of animals studied, where three types of P wave morphology were found, the most common being the unique positive P wave and bifid P wave, and less frequent biphasic P.
The occurrence of different P wave morphologies, including P bifid, P unique and positive, and P biphasic was described in Lusitanos and Andalusians horses (Ayala et al. 1994; Melchert et al. 2012). Similar results to these authors were shown in this study.
The P wave amplitude showed no statistically significant differences, however there was a tendency to increase in the age of the animals in each group studied, observations also described by Diniz et al. (2008) and Fernandes et al. (2004); Meanwhile, the results obtained for the P wave amplitude show higher values than those reported by these authors.
On average, the P wave amplitude of the groups studied was slightly higher than reported by Corredor-Matus et al. (2005), in creole horses from the Piedemonte llanero region.
No significant differences between males and females of the same group were presented. Also, Corredor-Matus et al. (2005), found no difference in the P wave amplitude, when compared females with males in groups of creole horses between 1 and 10 years old from the Piedemonte llanero.
Conduction velocity of the electrical impulse that arises from the sinoatrial node and arrives at the atrioventricular node in normal horses is given by the vagal tone influence (diminishing it), and a sympathetic tone (increasing it). This isoelectric interval constitutes the PR interval, whose conduction velocity was higher in the G1 group (Mcintosh and Marr 2010).
The PR interval represents the time required for conduction through the atrioventricular node, and the His-Purkinje system. it is difficult to establish the normal maximum value in this range because they are common atrioventricular blocks (AVB) of first and second degree in normal horses (Robertson 1990). importantly, the four groups studied showed no AVB of the first or second degree. They are considered pathological PR intervals with values greater than 0,5 seconds (Bonagura and Reef 2004).
The results for the PR interval of this study were less than 0.5 seconds. The presence of AVB of first and second degree cannot be considered as pathological in creole horses of the Bogotá Savannah, a much larger number of animals study would have to carry, to ratify this feature of the PR interval on the ECG. Meanwhile, this parameter must be taken into account in ECG assessing for this breed of animals. Also, an important feature of creole equine from Bogotá Savannah is its small size in relation to other breeds, and its alert temperament characteristic of its manner.
in each group studied, the PR interval tended to increase with age, as reported similarly by Fernandes et al. (2004), and Garrido-Lestache (2003). The foals have a shorter PR interval, to maintain an accelerated HR, therefore adult horses tend to increase the PR interval, usually exacerbated by the influence of vagal tone (Garrido-Lestache 2003).
Females from G3 and G4 showed a longer mean duration of the PR interval. in the study of (Corredor-Matus et al. 2005), although the PQ segment was measured, it was found that empty creole females from the Piedemonte llanero region between 5 and 10 years old, showed a longer mean duration of the PQ segment than males within the same age range.
4 types of QRS were found: the QRS type, QR type, R type, and RS types. Similar results were found in one study of Lusitano Thoroughbred, also agreeing that the most found QRS type was the QR type (Melchert et al. 2012).
The duration of the QRS complex may vary from 0.08 to 1.4 seconds on normal horses (Mcintosh and Marr 2010). interval within are all duration means of QRS complex in the four groups of this study. in addition, were found significantly lower differences for the G1 group compared to the G3, and G4. This is consistent with that reported by Schade et al. (2014) and Fernandes et al. (2004), where the QRS complex duration tended to be higher in older horses.
G4 females showed the highest QRS, although only significant differences in the QRS complex duration between males and females of the G1 compared to G4 females were found. However, in the study by Diniz et al. (2008), there were significant differences in horses between males and females of one to 28 years old of Mangalarga Marchador breed, being largest the QRS complex duration in males, justifying the bodily differences between sexes as the main cause. QRS complex duration was lower in entire creole horses under 5 years old and empty females between 5 and 10 years old in the study of Corredor-Matus et al. (2005), consistent with the results of this study.
The dense and large branches of Purkinje fibers in equine heart makes at the time of the depolarization of the ventricles the electrical transmissions cancel each other, leaving only a small part without canceling and it is manifested in the electrocardiogram as a small vector corresponding to the R wave (Dörner and Godoy 2009).
The mean R wave amplitude in racehorses in lead il is from 0.8 to 1.1 mV (Bonagura and Reef 2004). This is consistent with the means R wave amplitude found in this study in all groups, which ranged from 0.35 to 1.03 mV. in the study by Corredor-Matus et al. (2005), they were reported very similar R wave amplitudes to those in this study, ranging between 0.35 and 1.15 mV wave, and as in this study, tended to show an increase as the age of the animal increases. R waves with amplitudes greater to 2.2 mV in lead il and 1.7 mV in lead I, are often abnormal (Bonagura and Reef 2004).
In one study in creole horses was found that empty females under 5 years old were the group showed lower amplitudes for the R wave (Corredor-Matus et al. 2005). Coinciding this result with this study.
The T wave morphology was very variable in this study. According to some authors, the T wave is one of the most variable waves in the electrocardiogram, as it can be influenced by several factors such as exercise, training degree, excitement, medications, diseases of infectious type, vagal tone, electrolyte imbalance, hypoxia, myocarditis, influence of the equine thorax depth, and heart rate. For this reason, its diagnosis use is very difficult, must always correlate with clinical signs, and paraclinical tests, and ecodopplercardiographic. (Dörner and Godoy 2009; Muñoz et al. 2005). it can be considered as an abnormal T wave, when positive or biphasic in lead I, negative in aVR, and positive in the precordial lead V1R, V1L, V3R, and V3L. (Muñoz et al. 2005). Based on the analysis of T wave morphology of the author described, the morphologies of the T wave of this study were considered normal.
The QT interval indicates the ventricular systole duration, representing the time between the depolarization and repolarization of the ventricular muscle (Dörner and Godoy 2009). it increases as HR decreases. Therefore, it increases in older horses. This is consistent with the findings in this study, where the QT interval tended to increase as the age of the animals was higher (Diniz et al. 2008; Fernandes et al. 2004).
Regarding the cardiac axis, axis variation reflects a size alteration and position of the heart in the chest during growth and the result of physical maturation of the animal (Fernandes et al. 2004). in other species, dogs and cats, the clinical significance of the cardiac axis is that allow establishing criteria for right ventricular enlargement, and several atrioventricular conduction defects (Tilley et al. 1992).
In this study, the cardiac axis of foals (G1), showed significant differences with G2, G3, and G4 groups, presenting smaller and negative values, with a deviation to the left, and cranial. Similar axis behavior was found in English Thoroughbred foals in the study by Fernandes et al. (2004), where the cardiac axis in foals up to 11 months appeared with a negative trend. The axis in foals with 1 year old is often oriented cranially, while the axis of the most mature horses is oriented in the left and caudal direction (Bonagura and Reef 2004). Such observations were also evident in this study. However, other authors found predominantly negative values for Arab Thoroughbred breed horses between 6 and 12 years old, with at least 4 years of endurance training. Concluding that there are cardiac changes in the training mode impact on cardiac axis, due to cardiac hypertrophy (Silva et al. 2010).
For the MAP in foals is suggested as a guideline values from 65 to 120 mmHg. The mean MAP reported in the G1 found within this range. MAP reported for adult horses is 110-133 mmHg (Magdesian 2004). Differing from these results, mean MAP in G2, G3, and G4 of this study were lower.
For SAP, a 79-145 mmHg range, and 49 -106 mmHg for DAP is reported. Mean SAP and DAP for all groups agreed to these reference ranges (Bonagura and Reef 2004). Mean SAP, MAP, and DAP of G3, and G4 showed similar results to those reported by Walders and Gehlen (2014), in their study in horses between 4 and 31 years old, by a non-invasive method.
The AP can vary between breeds, also by the change in the horse's head position, by changes in the HR, and ventilation fluctuations (Bonagura and Reef 2004). According to these authors, the AP is lower in neonates, increases during the first month of life to adult value. in our study, SAP, MAP, and DAP were greater in G1 animals.
CONCLUSIONS
Creole horses of the Bogotá Savannah present higher HR compared to other breeds, even compared to creole horses from another region of Colombia. Variables such as geographical conditions, altitude of the study site, and daily exercise routine, could influence this parameter, however further studies are required to assess these variables. MAP tended to be lower in older animals, unlike other studies in other breeds. Although significant differences between some males and females of different groups for some parameters evaluated were evident, no significant differences were found between males and females in the same group for the same parameter evaluated, inferring that, perhaps sex is not a very strong variable, and influential, as is the age in the groups studied. Females who had left fascicles anterior block, and arrhythmia with premature ventricular complexes, did not manifest any sign of disease, emphasizing the importance of performing a complete cardiovascular examination with diagnostic tests, such as the electrocardiogram, which is very useful to identify these alterations. However, further studies, monitoring, and correlation with ecodopplercardiographic tests, radiography, and Holter are required, which were not allowed to perform, to learn more about the pathophysiology of these disorders in these horses. The groups of animals studied in the Bogotá Savannah did not show AVB of the first and second degree, a larger group of animals should be evaluated to determine if this parameter is characteristic of normal horses in this breed. Finally, these parameters can be used as a reference in ECG assessment of creole horses of Bogotá savanna.