Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Colombian Journal of Anestesiology
Print version ISSN 0120-3347
Rev. colomb. anestesiol. vol.36 no.1 Bogotá Jan./Mar. 2008
ARTÍCULO DE INVESTIGACIÓN BÁSICA
Molecular Diagnostics of Porcine Stress Syndrome Susceptibility Associated with the Arg615Cys Mutation Using Real-Time PCR with Fluorescent Hybridization Probes
Jesús E. Rojas1*, Miriam A. Wilches1*, Libia A. Cepeda1, María F. Garóes1, Miguel A. Suarez1, Rita M. Baldrich2, Cesar A. Vélez3, Mario F. Guerrero4, Martha R. García5 Iván H. Moreno5, Susana B. Bravo6, Radoslav Omelka7 and Jorge E. Caminos1,6
1 Department of Physiology
2 IGUN, Faculty of Medicine
3 Faculty of Veterinary Medicine
4 Department of Pharmacy, Faculty of Sciences
5 CEIF, National University of Colombia, Bogotá, Colombia.
6 Department of Physiology, School of Medicine, University of Santiago de Compostela, Santiago de Compostela, Spain.jecaminosp@unal.edu.co
7 Department of Botany and Genetics, Constantine the Philosopher University, Slovak Republic.
*These authors contributed equally to this work
Recibido para publicación marzo 31 de 2008, Aceptado para publicación abril 9 de 2008
ABSTRACT
Objective: The purpose of the presera study was to devélop a molecular genotyping method test by using a real time PCR hybridization probé and applying it to the analysis of C1843T mutations of the Sus scrofa RYR1 gene.
Animáis population Three PSS-susceptible and PSS non-susceptible crossbred swine races were used for the experiments: Pietrain X Landrace Belga, Pietrain X Large White and Pietrain X Duroc.
Methods: We have devéloped a genotyping method by using a hybridization probé and applied it to the analysis of C1843T mutations of the RYR1 gene, associated with PSS susceptibility. Genotyping results obtained by hybridization probé strategies were confirmed by restriction analysis and sequencing. In addi-tion, phenotype/genotype correlation analyses were devéloped by using the in vitro contracture test and confirmed the in vivo hálothane-succinylcholine challenge.
Results: The real-time PCR with fluorescent hybridization probé methodology was designed to identify ho-mozygous PSS-resistant, PSS-susceptible animáis as well as heterozygous carriers. All cases genotyped by fluorescent hybridization probes were in agreement with PCR restriction enzyme digestión and sequencing and showed a 100% concordance between the in vivo and in vitro porcine stress syndrome (PSS) susceptibility results.
Conclusions and clinical relevance: The real-time PCR with fluorescent hybridization probé method described here provides a rapid, easily interpretable and réliáble tool for genotyping the C1843T (Arg615-Cys) polymorphism of the RYR1 gene. This new methodology may be useful in the wide-scale genotyping of PSS-susceptibility and genetic selection.
Keywords: Málignant Hyperthermia, Caffeine-Hálothane Contracture Test, Málignant Hyperthermia Diagnosis, Halothane Test, Succinilcholine Test, Porcine stress syndrome susceptibility, Arg615Cys, C1843T SNP, molecular diagnostic, real-time PCR, fluorescent hybridization probes
INTRODUCTION
The Porcine Stress Syndrome (PSS) susceptibility is a hypermetabolic syndrome involving skeletal muscle disorder associated with a failure of the calcium homeostasis in muscle fbers, characterized by hyperthermia, muscle rigidity, and autonomic dysfunction(1,2). Ryanodine receptor 1 (RYR1) and the dihydropyridine receptor (DHPR) are involved in the release of the calcium (Ca2+) stored in sarcoplas-mic reticulum(3,4). PSS-susceptibility is inherited as an autosomal recessive dysfunction that has been clearly demonstrated and associated with a single point mutation C1843T (Arg615Cys) in the Ca2+ re-lease channel at the sarcoplasmic reticulum RYR1, where the susceptible mutant allele is denominated T and the wild-type - nonsusceptible allele C(5,6).
The frequency of the PSS gene in swine popula-tions varies according to genetic breeds and country of origin. Porcine stress syndrome has been reported in different swine breeds and the incidence is higher in lean, heavily muscled breeds such as Duroc, Lar-ge White, Pietrain, and Landrace. This susceptibility trait was related to the incidence of acute stress and sudden death, and an inferior quality meat generally referred to as pale, soft, exudative, dark, frm and dry, with back muscle necrosis(7).
Despite the number of laboratory tests developed with the aim to identify PSS-susceptible animals or carriers, none enables rapid diagnosis of PSS in an acute situation and most of these lack the sensitivity and specifcity to identify PSS-susceptible animals. The in vitro contracture test remains the standard test for the diagnosis of malignant hyperthermia (MH) susceptibility in which live muscle specimens are exposed to halothane and caffeine in the labo-ratory and the contracture response correlates with elevated myoplasmic Ca2+ concentrations(8,9). In spi-te of the fact that the sensitivity of the in vitro con-tracture test is 97%, and the specifcity is approxi-mately 85%, when this was used for subjects at high risk(10), this methodology had limited application in animals. This was because special laboratory faci-lities are required and the test must be run within minutes after the specimen is obtained. Genotyping must meet several key criteria, including specifcity, sensitivity, fast turnaround, and cost-effectiveness. Real -time PCR using fuorescein-labeled hybridiza-tion probes fulfls these criteria and furthermore is a rapid, versatile, and cost-effective method for this type of analysis(11).
The purpose of the present study was to deve-lop a molecular genotyping method test by using a hybridization probe and applying it to the analysis of C1843T mutations of the RYR1 gene, which is associated with the PSS susceptibility. Hybridization probe methodology was designed to identify homo-zygous PSS-resistant, PSS-susceptible animals as well as heterozygous carriers. Genotyping results obtained by hybridization probe strategies were confrmed by restriction analysis and sequencing. In addition, phenotype/genotype correlation analyses were developed by using the in vitro contracture test and confrmed the in vivo halothane-succinylcholine challenge. Our results showed that the simplicity of real-time Polymerase Chain Reaction (PCR) te-chnologies using fuorescence quenching schemes, allowed the diagnosis of the three genotypes of the PSS. This new methodology is a tool that may contribute to genetic selection in order to obtain resistant pigs to Porcine Stress Syndrome. The real-time PCR with fuorescent hybridization probes method des-cribed here provides a rapid, easily interpretable and reliable tool for genotyping the C1843T (Arg615Cys) polymorphism of the RYR1 gene.
EXPERIMENTAL PROCEDURES
Three PSS-susceptible and PSS non-susceptible crossbred swine races were used for the experi-ments: Pietrain X Landrace Belga, Pietrain X Large White and Pietrain X Duroc. Swine were housed in standard lighting and were given water and food ad libitum until the beginning of the experiment, when they were moved to a surgery room. All procedures were in accordance to the Guide for the Care and Use of Laboratory Animals (National Institute of Health), and were approved by the Ethics Commission of the Veterinary School at the National University of Colombia.
Muscle samples biopsies, subjected to halotha-ne-caffeine in vitro contracture tests were tested according to the protocol of the North American Malignant Hyperthermia Group protocol(8,12). Geno-typing was done using a real-time PCR-based system (LightCycler, Roche, and Mannheim, Germany) with hybridization probes specifc for the RYR1 C1843T SNP. Additionally, individual samples representing the C/C, C/T or T/T genotypes analyzed by real-time PCR were also genotyped by DNA sequencing and restriction fragment length polymorphism techniques, proving the accuracy of the real-time PCR method.
Initially, random samples of peripheral blood were obtained from ninety-fve swine in a farm located outside the metropolitan area of Bogotá, Colombia. DNA samples plus controls (CC, CT and TT RYR1 genotyping, donated by Dr. R. Omelka, from the Department of Botany and Genetics, Constantine the Philosopher University, Slovak Republic) were pro-cessed for screening the RYR1 C/T SNP (C1843T), using polymerase chain reaction and restriction fragment length polymorphism analysis with the enzyme HhaI (Invitrogen), as described elsewhere(13). For this purpose 5 mL of peripheral venous blood was gathered from each swine by standard vene-puncture and each blood sample was collected in sterile tubes containing K3EDTA solution. The ani-mals were genetically tested by standard methods using genomic DNA isolated from blood leucocytes (ChargeSwitch® gDNA Blood Kit, Invitrogen, USA), according to the manufacturer's recommendations. The purity and concentration of the extracted DNA were determined by a UV–Vis spectrophotometer. Twenty-one swine genotyping with the restriction enzyme HhaI for the RYR1 C1843T SNP (CC n=9, TC n=7, and TT n=5), were randomly selected for the later development of the study.
Sequences for PCR primers and hybridization probes used for SNP detection can be found in Table 1. The DNA sequencing templates were PCR amplifed from the Sus scrofa strain Yorkshire RYR1 gene (Accession M91456.1 [GenBank]) and primers were designed using Primer3 software http:// frodo.wi.mit.edu/cgi-bin/primer3/primer3_www.cgi) The primers amplifed a 329 bp fragment encompassing the polymorphism C1843T.
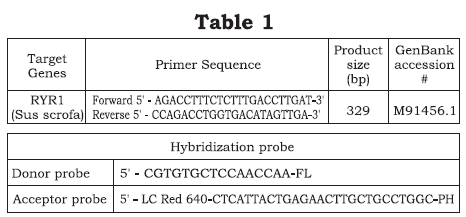
The detection probe attached to the polymorphic site and the nucleotides fanking it were specifc for the T-allele (figure 1). Hybridization probes for genotyping RYR1 gene whose mutation C1843T (Arg615Cys) is associated with PSS -susceptibility, were designed and obtained from Roche Molecular The primer and Hhybridization probé sequences are shown. Donor probé is labéled with fuorescein at the 3', whilst the adjacent acceptor probé is labéled with the specifc fuorophore Red 640 (LC Red 640) at the 5' end región.
Biochemical®. Acceptor probé were labéled at the 5'-end with LC Red 640 and phosphorylated on the 3'-end by block extensión. The donor probé was labéled on the 3'-end with fuorescein and was designed to bind at a distance of three bases from the acceptor probé.
The PCR reaction was carried out using the LightCycler real-time PCR machine 2.0 (Roche Diagnostics, Germany) and the software versión 4.0. The reaction was performed in a volume of 20 ul with 2.5 mM MgC12, 0.25 uM of both primers, 0.25 uM of both probes and 2 ul of 10 x LightCycler ® FastStart DNA Master HybProbe buffer (Roche Diagnostics), and about 50 ng of témplate DNA. The PCR protocol consisted of initial denaturation at 95 °C for 10 min, followed by 37 cycles of denaturation (95 °C for 5 s, 20 °C/s), annealing (60 °C for 15 s, 20 °C/s) and elongation (72 °C for 20 s, 20 °C/s). This was followed by melting curve analysis consisting of 1 cycle at 95 °C for 30 s (20 °C/s), 40 °C for 20 s (20 °C/s) and a temperature rise to 85 °C at a slope of 0.2 °C/s with continuous measurement of fuo-rescence. The genotyping results were interpreted according to derivative melting curves [- d (F2 / F1) / dt] plotted against temperature.
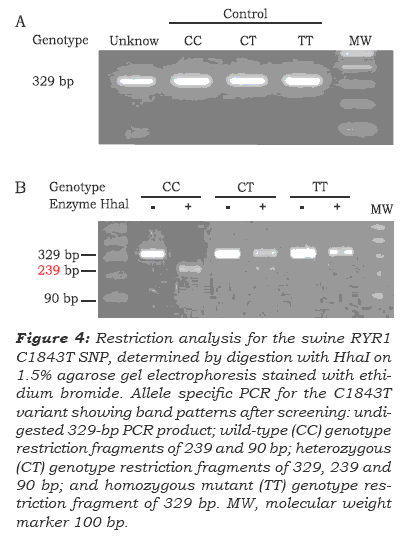
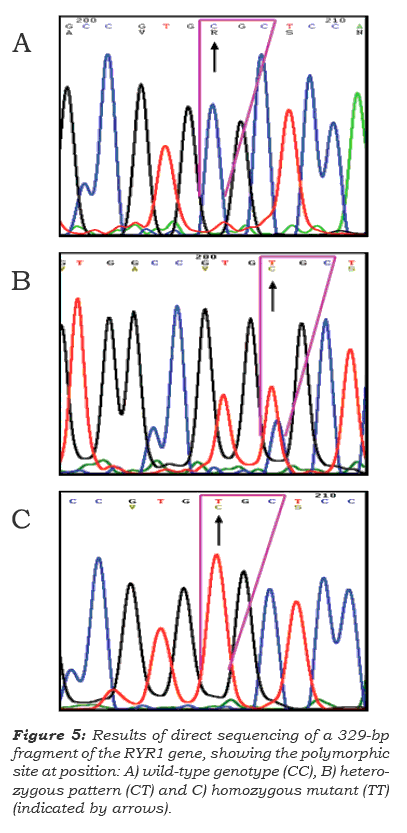
To confrm the genotyping results obtained by real-time PCR using hybridization probes strate-gies, restriction analysis and sequencing were used (figure 2, 3). All PCR products were purifed using Marligen's Gel Extraction Systems Kit (Marligen Biosciences Inc.), according to the manufacturer's recommendations. Sequencing was performed in both directions by DYEnamic™ ET Dye Terminator Kit (MegaBACE™) on a MegaBace DNA Analysis Systems (Amersham Biosciences). Sequencing results were analyzed using nucleotide-nucleotide BLAST 2 SEQUENCES software ([GenBank]) to locate and identify base changes. In addition, analyses of DNA RYR1 amplicons were subjected to restriction enzyme digestion during 4 hours, with the enzyme HhaI (Invitrogen), according to the manufacturer's recommendations. The digested DNA fragments were visualized on a 1.5% agarose gel, with a 100-bp DNA ladder (Invitrogen) as a molecular weight marker, and stained with ethidium bromide (10 mg/ mL). Images of the gels were taken using Molecular imaging Systems (Bio-Rad). In all assays, negative and positive sample controls with known SNP C/T genotypes were used.
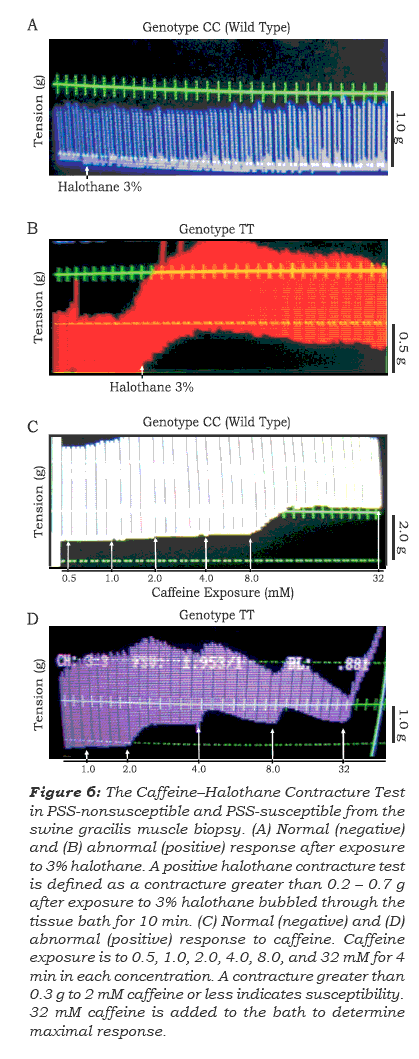
To determine the associations between genetica-lly tested swine for the RYR1 C1843T SNP and the stress susceptibility phenotype, six PSS-susceptible and PSS-nonsusceptible swine (C/C, n=2; C/T, n=2; T/T, n=2; weight, 30 +/- 10 kg) were investigated. In vivo studies with 3% halothane and in vitro con-tracture test from the gracilis muscle biopsy exposed to halothane-caffeine were undertaken(12). Parallel to the in vivo study, viable muscle specimens were taken and exposed to in vitro contracture tests following the protocol described by Marilyn Green Larach and the North American Malignant Hyper-thermia Group(8).
RESULTS
A schematic representation of the adjacent fluorescent hybridization probes used for genotyping the RYR1 C1843T SNP is shown in figure 1. Table 1 lists the sequences for PCR primers and hybridization probes used for SNP detection. These probes were designed so that during hybridization the donor probé formed a mismatch with the wild-type alíele (C:T). The data collected during the melting phase were used to genotype each sample and melting peaks were obtained by plotting the fuorescence (F) versus temperature data as 2dF/dT.
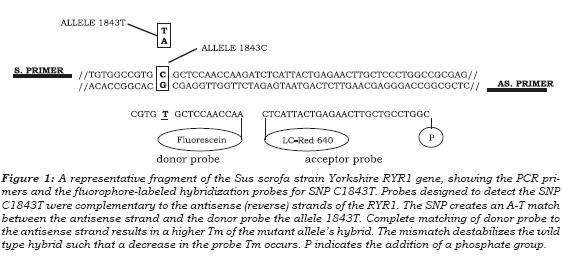
Genotype analysis for the C1843T SNP is shown in figure 2, where all genotypes are clearly distin-guishable. The peak assignment is as given in figure 3. The G:T mismatch formed between the wild-type alíele with the fuorescein probé, created a ATm of 4.62°C from the completely Watson-Crick paired dúplex (homozygous mutant alíele T). A distinct peak at 54.2 ± 0,10 °C is indicative of alíele C1843, whereas both the homozygous mutant alíele (TT) and the alíele with the 1843T mutation from the heterozygous samples, were annealed to the probé at the fuorescence acquisition temperature of 58,82 ± 0,35°C (see figure 3).
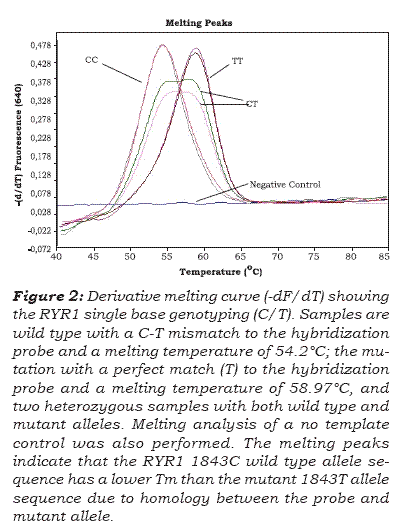
Swine single point mutation C1843T (Arg615-Cys) genotyping performed in the LightCycler instrument was compared with conventional PCR restriction fragment length analysis and sequen-ce. PCR restriction fragment length analysis was performed on all samples for the C1843T mutation with the enzyme Hhal (figure 4). Forward and reverse primers were designed to produce a 329-bp-long amplicon (see table 1 and figure 4A). The RYR1 C1843T mutation destroyed a Hhal restriction site that upon restriction digestión of the wild-type genomic DNA, yielded fragments of 239 and 90 bp (figure 4B). All cases genotyped by fuorescent hybridization probes agreed with PCR restriction fragment length analysis. In addition, results of genotyping with hybridization probes and restriction analysis were compared with PCR amplicon sequences and aligned to the Sus scrofa strain Yorkshire RYR1 gene to verify melting curve analyses (figure 5). Single base change at the targe-ted SNP site corresponded with results of melting curve analyses (Figure 2). Direct DNA sequencing analysis confrmed the different RYR1 genotypes (figure 5) and revealed a C-to-T substitution that results in an arginine-to-cysteine amino acid repla-cement at codon 615 (Arg615Cys). Variations from wild-type to mutant alíeles resulted in 4.62°C Tm change (Figure 3).
In the current study, the correlation between RYR1 C1843T genotype determination above by different molecular approaches and the porcine stress syndrome susceptibility has been investiga-ted. Phenotype/genotype correlation analyses were developed by using the in vitro contracture test and confrmed by the in vivo halothane-succinylcholine challenge (Figure 6A andB), as was described elsewhere(12). All swine diagnosed as PSS-susceptible by in vitro contracture test, were T allele homozygous (TT) and heterozygous (CT) for the RYR1 mutation, and all PSS non-susceptible swine were homozygous for the C1843 allele (Figure 6). All results were in agreement with contracture test parameters seen with an increase in twitch height as Allen described elsewhere(12). Furthermore, all swine diagnosed as PSS-susceptible by the in vivo halothane-succinyl-choline challenge were homozygous for the 1843T allele whereas the wild-type (CC) and heterozygous swine were PSS non-susceptible (data not shown). These results confrm once again that autosomal recessive Porcine Stress Syndrome susceptibility disease is caused by the C1843T mutation in the gene encoding the skeletal muscle calcium release channel RYR1.
DISCUSSION
Porcine stress syndrome (PSS) susceptibility, has been associated with a single point mutation C1843T (Arg615Cys) in the Ca2+ release channel at the sarcoplasmic reticulum ryanodine receptor 1 (RYR1). The RYR1 gene is the best commercially available marker used for predisposition of stress susceptibility in several swine breeds. Changes in the intracellular free calcium concentration are refected in a variety of cellular responses, such as muscle contraction, neuronal excitability, gene expression and metabolism variation. The functional effect of the RYR1 Arg615Cys mutation (C1843T) linked to neuromuscular disorders lead to a dysregulation of intracellular calcium homeostasis. The mechanisms leading to the PSS-susceptibility crisis involve a high concentration of free calcium in the myoplasm released by a faulty membrane system in calcium storing organelles including sarcoplasmic reticulum(14). In the present study, we used molecular and physiological (invitro and in vivo) approaches to determinate the PSS susceptibility.
Current methods for genotyping the C1843T Porcine stress syndrome (PSS) susceptibility mutations include allele-specifc oligonucleotide hybridization and PCR restriction fragment length analysis, and these methods require multiple manual steps and are time consuming(5,15). An alternative is the use of fuorescent hybridization probes and rapid-cycle PCR, a technique that provides homogeneous amplifcation and genotyping in approximately 45 minutes. We have developed a genotyping method by using a hybridization probe and applied it to the analysis of C1843T mutations of the RYR1 gene, associated with PSS susceptibility.
Methods available for PSS- susceptibility genotyping are based on sequencing and restriction fragment analysis, which requires several technical steps. Although different PCR-based fuorescent techniques are being used for the diagnosis of PSS-susceptibility none of them genotype by allele / hybridization probe Tm(16). In addition, the tech-nology of fuorescence resonance energy transfer avoids additional steps, and the genotyping can be done directly on extracted genomic DNA from swine. Real time PCR using hybridization probe systems based on fuorescence resonance energy transfer (FRET) have been established as effective tools that allow the discrimination between closely related sequences on the basis of a melting curve analysis that confer very sensitive and highly specifc detection. This technology is useful for diagnostic purposes such as genotyping (SNP analysis) and determination of genetic predisposition to disease(11). In addition, Polymerase Chain Reaction amplifcation and genotyping by fuorescent probe melting temperature, which reduces the cost and invasiveness of sample collection, is a rapid, versatile, and cost-effective method for this type of analysis. Derivative melting curves for the three genotypes of the RYR1 C1843T (Arg615Cys) polymorphism are shown in Figure 2.
The thermal stability of a DNA duplex relies on GC content, duplex length, and Watson-Crick base pairing. Fluorescence from hybridization probes is the direct result of the hybridization of two indepen-dent probes and this result in very high specifcity. Changes from Watson-Crick pairing destabilize a duplex by varying degrees depending on the length of the mismatched duplex, the specifc mismatch, the position of the mismatch, and neighboring base pairs. The probe melting temperature is sequence dependent, providing a simple method to genotype mutations, including single base mutations(11). In this study, the single base mismatch G:T (C1843T) in the probe region decreases the melting temperature as little as 4.62 °C in comparison with the completely Watson-Crick paired duplex. The average melting temperature was 54.2 ± 0.10 °C for the Wild-type C allele and 58.82 ± 0,35°C for the mutant T allele. The donor fuorescein probe formed an G:T mis-match with the wild-type allele lowering the Tm of the probe by 4.62 °C from the Watson-Crick paired duplex, assuring a good separation of the C1843 and 1843T genotypes. The donor probe attached to the polymorphic site was specifc for the T-allele. All cases genotyped by fuorescent hybridization probes were in agreement with PCR restriction enzyme digestion and sequencing in a random set of samples representing all genotypes. When we validated the method with positive DNA control generously donated by Dr. R. Omelka (Department of Botany and Genetics, Cons-tantine the Philosopher University, Slovak Republic), the results matched completely.
Finally, we have demonstrated a 100% concor-dance between the in vivo and in vitro porcine stress syndrome (PSS) susceptibility results and the ones obtained by means of molecular genotyping such as real-time PCR using fuorescein-labeled hybridization probe, sequencing and restriction analysis. The simplicity of real-time PCR technologies using a fuorescence quenching schemes, allowed diagnosis of the three genotypes of the PSS, and this methodology is a tool that may contribute to the genetic selection and to obtain pigs resistant to PSS. In summary, this assay shows that real-time with fuorescent hybridization probes provide a rapid and sensitive analysis for genotyping the RYR1 C1843T (Arg615Cys) polymorphism and may be useful in the wide-scale genotyping of PSS-susceptibility. This new methodology is a tool that may contribute to the genetic selection and to obtain pigs resistant to PSS and may be useful in the wide-scale genotyping of PSS-susceptibility.
AGRADECIMIENTOS
Los autores agradecen al Dr. Henry Rosenberg, Dra Sheila Muldoon, Dr. Saiid Bina y Dr. Gregory Allen su decidida colaboración y apoyo en el desarrollo de este estudio.
Esta investigación se fnanció con aportes del Fondo de Investigaciones para Ciencias Básicas de la Sociedad Colombiana de Anestesiología y Reanimación soportado por la Revista Colombiana de Anestesiología: email: publicaciones@scare.org.co Fax: 6204619 Teléfono: 6196091 y la División de Investigación de la Universidad nacional de Colombia en Bogotá, D.C. jecaminosp@unal.edu.co, Teléfono: 3165000 ext. 15047
Dirección para correspondencia Address correspondence to:
Professor Jorge Eduardo Caminos
Department of Physiology, Faculty of Medicine,
National University of Colombia, Bogotá, Colombia.
(E-mail: jecaminosp@unal.edu.co).
Ciudad Universitaria - Bogotá D.C.- Colombia.
Edifcio "Uriel Gutiérrez" Transversal 38 N° 40-01
Phone (57)(1) 3165000 – ext 15047
BIBLIOGRAFÍA
1. Brunson, D.B.; Hogan K.J. Malignant hyperthermia: a syn-drome not a disease. The Veterinary clinics of North America. Small. Anim. pract. 2004, 34, 1419-1433. [ Links ]
2. Balog, E.M.; Fruen, B.R.; Shomer, N.H.; Louis, C.F. Divergent effects of the malignant hyperthermia-susceptible Arg(615)-->Cys mutation on the Ca(2+) and Mg(2+) dependence of the RyR1. Biophys. J . 2001 , 81, 2050-2058. [ Links ]
3. Reggiani, C.; te Kronnie, T. RyR isoforms and fbre type-specifc expression of proteins controlling intracellular calcium concentration in skeletal muscles. J. Muscle. Res. Cell. M. 2006, 27, 327-335. [ Links ]
4. Dietze, B.; Henke, J.; Eichinger, H.M.; Lehmann-Horn, F.; Melzer, W. Malignant hyperthermia mutation Arg615Cys in the porcine ryanodine receptor alters voltage dependence of Ca2+ release. J. Physiol. 2000, 526, 507-514. [ Links ]
5. Fujii, J.; Otsu, K .; Zorzato, F.; de Leon ,S .; Khanna, V.K . ; Weiler, J.E .; O'Brien , P.J . ; MacLennan , D.H. Identification of a mutation in porcine ryanodine receptor associated with malignant hyperthermia. Science. 1991 , 253 , 448-451. [ Links ]
6. Fletcher, J.E.; Calvo, P.A.; Rosenberg, H. Phenotypes asso-ciated with malignant hyperthermia susceptibility in swine genotyped as homozygous or heterozygous for the ryanodine receptor mutation. Brit. J. Anaesth. 1993, 71, 410-417. [ Links ]
7. Kuchenmeister, U.; Kuhn, G.; Wegner, J.; Nurnberg, G.; Ender, K. Post mortem changes in Ca2+ transporting pro-teins of sarcoplasmic reticulum in dependence on malignant hyperthermia status in pigs. Mol. Cell. Biochem. 1999, 195, 37-46. [ Links ]
8. Larach, M.G. Standardization of the caffeine halothane muscle contracture test. North American Malignant Hyper-thermia Group. Anesth. Analg. 1989, 69, 511-515. [ Links ]
9. Allen, G.C.; Larach, M.G.; Kunselman, A.R. The sensitivity and specifcity of the caffeine-halothane contracture test: a report from the North American Malignant Hyperthermia Registry. The North American Malignant Hyperthermia Re-gistry of MHAUS. Anesthesiology. 1998, 88, 579-588. [ Links ]
10. Rosenberg, H.; Antognini, J.F.; Muldoon, S. Testing for malignant hyperthermia. Anesthesiology. 2002, 96, 232-237. [ Links ]
11. Pryor, R.J.; Wittwer, C.T. Real-time polymerase chain reaction and melting curve analysis. Method. Mol. Biol. 2006, 336, 19-32. [ Links ]
12. Allen, G.C.; Fletcher, J.E.; Huggins, F.J.; Conti, P.A.; Rosenberg H. Caffeine and halothane contracture testing in swine using the recommendations of the North American Malignant Hyperthermia Group. Anesthesiology. 1990, 72, 71-76. [ Links ]
13. Omelka, R.; Vasicek, D.; Martiniakova, M.; Bulla, J.; Bauerova, M. Simultaneous detection of malignant hyperthermia and ge-netic predisposition for improved litter size in pigs by multiplex PCR-RFLP. Folia Biol. (Krakow). 2004, 52, 113-115. [ Links ]
14. Lopez, J.R.; Contreras, J.; Linares, N.; Allen, P.D. Hypersen-sitivity of malignant hyperthermia-susceptible swine skeletal muscle to caffeine is mediated by high resting myoplasmic [Ca2+]. Anesthesiology. 2000, 92, 1799-1806. [ Links ]
15. de Oliveira, G.; Facioni, S. E.; Lopes, P. et al. Relationship between the Porcine Stress Syndrome gene and carcass and performance traits in F2 pigs resulting from divergent crosses. Genet. Mol. Biol. 2005, 28, 92-96. [ Links ]
16. China, B.; Leroy, B.; Dams, L.; Clinquart, A.; Daube, G. Genotypic frequencies of the ryanodin receptor gene mutation associated to stress sensibility in 5 pig production systems in Belgium. Les annales de Médecine Vétérinaire. 2005, 149, 161-165. [ Links ]













