Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Colombian Journal of Anestesiology
Print version ISSN 0120-3347
Rev. colomb. anestesiol. vol.44 no.3 Bogotá July/Sep. 2016
Scientific and Technological Research
Transcultural and linguistic adaptation of the Richmond Agitation-Sedation Scale to Spanish*
Validación transcultural y lingüística de la escala de sedación y agitación Richmond al español
José Antonio Rojas-Gambasicaa, Albert Valencia-Morenoa, Víctor Hugo Nieto-Estradaa,**, Pablo Méndez-Osorioa, Daniel Molano-Francoa, Álvaro Tito Jiménez-Quimbayaa, Raúl Escobar-Modestob, Nayibe Cortés-Rodrígueza, Liliana Paola Correac
a Critical Medicine and Intensive Care, Clínica Universitaria Colombia, Bogotá D.C., Colombia
b Second Year Fellow in Intensive Care, Clínica Universitaria Colombia, Bogotá D.C., Colombia
c Critical Medicine and Intensive Care, Clínica Reina Sofía, Bogotá D.C., Colombia
* Please cite this article as: Rojas-Gambasica JA, Valencia-Moreno A, Nieto-Estrada VH, Méndez-Osorio P, Molano-Franco D, Jiménez-Quimbaya ÁT, et al. Validación transcultural y lingüística de la escala de sedación y agitación Richmond al español. Rev Colomb Anestesiol. 2016;44:216-221.
** Corresponding author at: Clínica Universitaria Colombia, UCI Cardiovascular, Tercer piso. Calle 23 # 66 - 46, Bogotá D.C., Colombia.
E-mail address: victor.nietoestrada@gmail.com (V.H. Nieto-Estrada).
Article history: Received 23 July 2015 Accepted 6 April 2016 Available online 1 June 2016
Abstract
Introduction: Goal oriented sedation is standard in the management of critically ill patients, but its systematic evaluation is not frequent. The Richmond agitation sedation scale's efficient operative features make it a validated instrument for sedation assessment.
Objectives: To translate and validate the Richmond agitation sedation scale into Spanish.
Method: A cultural and linguistic adaptation study was designed. Translation into Spanish included back-translation and pilot testing. The inter-rater reliability testing was conducted in Clínica Colombia's cardiovascular and general intensive care unit, including 100 patients mechanically ventilated and sedated. Inter-rater reliability was tested using Kappa statistics and Intra-class correlation coefficient. This study was approved by Fundación Universitaria Sanitas Research and Ethics Institute and Clínica Sanitas Research Committee.
Results: 300 assessments using the Spanish version of the Richmond agitation sedation scale were performed by three independent evaluators. The intra-class correlation coefficient was 0.977 (CI 95% 0.968-0.984). The kappa was 0.84 between the first and second evaluators 0.85 between the first and third evaluators and 0.86 between the second and third evaluators.
Conclusion: The product of this study, the Spanish version of the Richmond agitation sedation scale, is conceptually equivalent to the original scale, being reproducible and understandable to physicians whose native language is Spanish.
Keywords: Deep sedation, Conscious sedation, Critical care, Translating, Validation studies.
Resumen
Introducción: La sedación por metas es un estándar en el manejo del paciente crítico pero su evaluación sistemática no es frecuente, la escala de sedación y agitación Richmond es un instrumento con características operativas eficientes para evaluar sedación.
Objetivo: Traducir y validar la escala de sedación y agitación Richmond al idioma español.
Método: Se diseñó un estudio de adaptación transcultural y lingüística y validación de instrumento. La traducción al idioma español incluyó una traducción reversa y una prueba piloto. Las evaluaciones para la validación se realizaron con 100 pacientes bajo sedación, ventilados mecánicamente en dos unidades de cuidados intensivos, una polivalente y otra cardiovascular de la Clínica Universitaria Colombia. La fiabilidad entre los observadores fue probada utilizando el estadístico kappa y el coeficiente de correlación intraclase. El estudio contó con la aprobación del instituto de investigaciones y comité de ética de la Fundación Universitaria Sanitas y comité de investigaciones de Clínica Sanitas.
Resultados: Se realizaron evaluaciones secuenciales e independientes por tres entrevistadores, completando 300 valoraciones con la traducción de la escala en español. El coeficiente de correlación intraclase fue de 0,977 (IC 95% 0,968 - 0,984). La concordancia cualitativa entre los evaluadores también fue alta con un kappa de 0,84 entre el primer y segundo evaluador, 0,85 entre el primer y tercer evaluador y 0,86 entre el segundo y tercero.
Conclusión: La versión en español de la escala de sedación y agitación Richmond producto de este estudio, resulta conceptualmente equivalente a la original, es reproducible y comprensible para médicos de habla hispana.
Palabras clave: Sedación profunda, Sedación consciente, Cuidado crítico, Traducción, Estudios de validación.
Introduction
Goal oriented sedation has become standard in the management of critical patients, with notable benefits in clinical outcomes.1 It allows for an adequate level of patient-ventilator interaction, alleviates patient anxiety about medical care, favors sleep architecture by conserving the sleep-wake cycle, increases tolerance to procedures like tracheal aspiration, and reduces the frequency of unexpected events like self-extubation and the removal of intravascular devices.2,3 Another benefit described and associated with an appropriate sedation plane in critical patients is the lower quantity of circulating systemic catecholamines with a decrease in oxygen consumption.4 It has also been credited with lower barotrauma in patients with reduced pulmonary compliance.5
Deep sedation, on the other hand, leads to a series of risks that are potentially avoidable for the patient: increase in the incidence of ventilation-associated pneumonia,6 more days on mechanical ventilation,7 prolonged hospital stay, difficulty in neurological evaluations, and neuromuscular weakness in the critical patient.8
Although sedation is universally used in intensive care services, its systematic evaluation is infrequent.9 Objective and subjective methods exist for evaluating sedation.10 Overall, the subjective evaluation of the level of sedation through scales is preferred to more elaborate techniques.11 Any evaluation instrument in medicine must be validated and submitted to processes of cultural and linguistic adaptation in order to avoid barriers in the application and the variability in the results.12
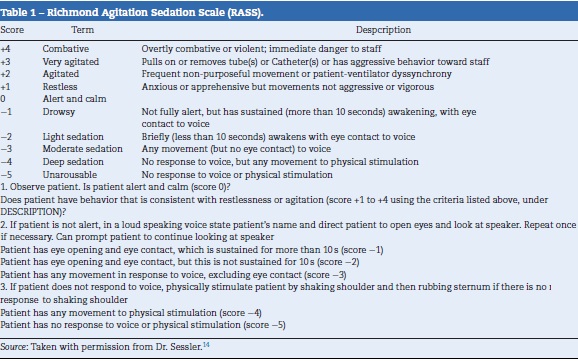
One of the scales with efficient operative characteristics and reproducibility in the systematic evaluation of sedation is the Richmond Agitation Sedation Scale (RASS).13 The RASS was developed in 2012 by a multidisciplinary group in the University of Richmond, USA. It consists of a 10-point scale that can quickly evaluate a patient and place them in a level of sedation or agitation through three clearly defined steps14 (Table 1). The RASS is widely used, even in countries like Colombia. It has been validated in other languages such as French,15 Swedish16 and Portuguese,17 but no reports exist of official translations or validations into Spanish.
The creation of a scale is a complex process. The process of adaptation and validation is more quickly achieved since it originates from a tested instrument. The difference in languages or cultures may affect the way in which it is applied or in which one responds to an instrument of measurement. As such, linguistic equivalence is an obligatory step in the validation of an instrument to another language.
The objective of this endeavor was to create a linguistic equivalent of the RASS and validate the version translated into Spanish in order to have a tool for Spanish-speaking physicians that would allow them to monitor the level of sedation in adult critical patients.
Methodology
The protocol was approved by the Research Committee of the Sanitas University Foundation of Colombia. According to Resolution No. 008430 of 1993 of the Colombian Ministry of Health, which regulates research on human beings in Colombia, this study is classified in the "no risk" category. There was no requirement of informed consent. The study was carried out in two phases: first, the translation of the RASS from English to Spanish, followed by the measurement of the reliability of the translated scale. This later phase was performed in both intensive care units of the Colombia University Clinic, a university health center with fourth level complexity and 28 intensive carebeds: 13 for polyvalent care and 15 for cardiovascular care.
Phase 1: translation and linguistic equivalency of the scale
A translation and cultural adaptation of the RASS from its original language to Spanish was performed based on ISPOR norms.18 The linguistic equivalency was achieved through a series of stages, with recorded proceedings of each result and individual conclusion:
Preparation: Permission was requested from the original author of the RASS, Dr. Curtis Sessler. He conceded this permission.
Initial Translation: Two native authors with fluency in both languages translated the scale from English to Spanish and compared their results.
Reconciliation: Resolution of discrepancies between the original and the translations by third native translator.
Back-Translation: The RASS in Spanish was translated back into English by a bilingual physician without knowledge of the scale in the original language.
Review of the Quality of the Back Translation and Harmonization >of conceptual discrepancies in the items of the scale. Carried out by the group of researchers.
Cognitive Review: Evaluation of understanding through a survey of 20 specialists in Critical Medicine. The goal was to determine comprehensibility, understanding, writing, spelling, and difficulties that arise when the translated scale is applied.
Final correction of grammatical and typographic errors.
Final Report: The final, translated and corrected version of the RASS in Spanish is presented. With this product, a pilot trial on 30 patients was performed to familiarize the evalua-tors with the translated instrument.
Phase 2: inter-evaluator reliability of the RASS in Spanish
The reliability tests between evaluators were performed on 100 sedated adult patients that received invasive mechanical ventilation in the Intensive Care Units of the Colombia University Clinic between February, 2013 and July, 2014.
Each patient was submitted to grading by a multidisci-plinary group made up of three evaluators: two physicians with a first specialty either in Internal Medicine or Anesthesiology, but both with a second specialty in Critical Medicine, and a third specialist in Critical Medicine and Intensive Care.
The evaluations were performed consecutively and independently by the three evaluators at different times of the day or the night. The order of the graders was chosen randomly and they were blinded to the grades of the others. Pharmaceuticals administered for the sedation was part of the institutional protocol aimed at addressing the patient's clinical condition.
Statistical analysis
The numerical variables were submitted to normality testing with the Kolmogorov-Smirnov test. Depending on their distribution, they are summarized with averages and standard deviation or median and interquartile range. The categorical variables are expressed in relative frequencies and percentages. The RASS is, by definition, an ordinal variable, but it has 10 defined levels in numerical values, which permits its quantitative analysis. To measure the inter-observer reliability, the coefficient of intra-class correlation (CIC) for quantitative data and Cohen's kappa statistic were used for the categorical variables. With a CIC greater than 0.8, the evaluators were considered to be in almost perfect agreement, and a kappa value of 1 means complete agreement. The information was analyzed with the statistical program IBM®SPSS® version 22.
Results
Translation phase
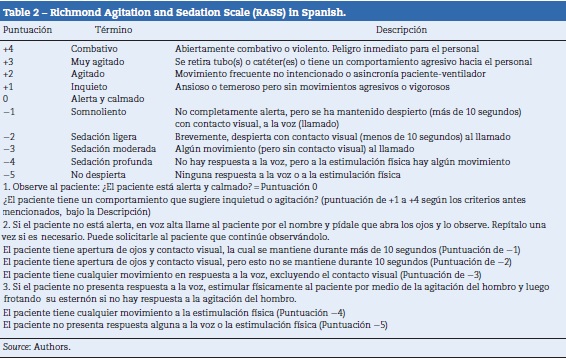
The product of this phase is the translated, corrected and unified version of the scale in Spanish. 30 patients were used in the pilot test. 90 measurements of the level of sedation were made, and the objective of familiarizing and accepting the new instrument was achieved (see Table 2).
Validation phase
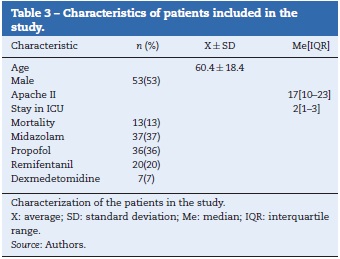
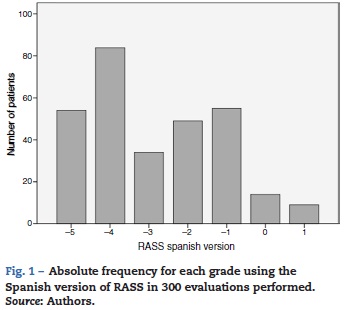
The analyzed series consisted of 100 adult patients with a median age of 63 years, 53% male; the median stay in the ICU was 2 days IQR [1-3] , while that of the Apache II score was 17 IQR [10-23] (see Table 3). The patients came from two intensive care units with different medical care profiles, some polyvalent and others cardiovascular. This allowed for the application of the scale in varying clinical scenarios, including: neuro-intensivism, surgery due to major trauma, post-operative care for heart surgery and sepsis. Each patient included in the study was submitted to three consecutive evaluations. A distribution of grades between the categories —5 and 1 was found. The mode was category —4, and 37% of the patients were cataloged under superficial sedation — —2 to 0 — (see Fig. 1). The sedation of the patients was performed with midazolam in 37% of patients, propofol in 36%, remifentanil in 20%, and, in 7% of patients, conscious sedation with dexmedetomidine. All options were combined with opioid analgesics in accordance with the patients' needs (see Table 3).
The grades with the Spanish version of the RASS in our study showed excellent reliability among the evaluators. The coefficient of intra-class correlation was "almost perfect": 0.977 (CI 95% 0.968-0.984).
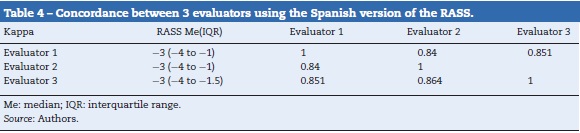
The qualitative concordance between the evaluators was also high with a kappa of 0.84 between the first and second evaluators, 0.85 between the first and third evaluators, and 0.86 between the second and third evaluators. Table 4 summarizes the grading of the three evaluators — Evaluator 1 (physician), Evaluator 2 (physician), Evaluator 3 (nurse) — and shows the respective concordance.
Discussion
When we tend to a patient in the Critical Care Unit, physicians concentrate on protecting primary organs such as the heart and the brain as functional units, forgetting "the biological cost of the depression of consciousness" and the deleterious consequences of the alteration of the state of consciousness.19 Therefore, a rational model for the management of sedation in critical patients is vital, recognizing that it is not only a question of putting a patient to sleep to spare them suffering but of understanding all of the physiological and physio-pathological processes that are compromised when the functional state of the brain is altered with a sedative.20 In any scenario, be it in the operating room, in the postanesthesia care room, or in the ICU, this model should be initiated without fail with a sensitive, objective, and validated evaluation of the patient's level of sedation.
In our country, one of the scales for objectively evaluating the level of sedation-agitation of critical patients is the RASS. Until now, this scale has not been translated to and validated in Spanish. The RASS has already been translated to other languages like French and Portuguese with satisfactory results.15,17
With these antecedents, a process in phases was carried out to translate and validate the scale in Spanish. This has resulted in a version that is conceptually equivalent to the original, is reproducible, and comprehensible to Spanish-speaking physicians. This new instrument features appropriate theoretical and psychometric support for its use, has adequate internal consistency and construct validity, like the original scale. In total consensus, the group of evalua-tors were well satisfied with and accepted the instrument. This study has notable advantages, namely the heterogeneous population — patients of medical, surgical, coronary and traumatic problems — to which the product was applied. It is also a project with a greater sample size and number of evaluations compared to similar studies with translation to other languages.
Upon analyzing the results, the kappa statistic showed a significant qualitative concordance with a result of 0.87 for an expected 0.80. The quantitative concordance among the evaluators, measures by CIC, was also excellent (0.97), showing that this new instrument, the RASS translated to Spanish, is valid, useful and precise for measuring the level of sedation by Spanish-speaking physicians in critically ill adult patients. As limitations of the study, we recognize the internal socio-cultural variability between the different regions of our country and external variability with other countries that also speak Spanish. A high adherence to sedation goals in critical patients in the participating institution meant an additional limitation in the validation process due to the absence of categories higher than 1 on the RASS in the results obtained.
Having access to a valid instrument specifically designed for measuring the level of sedation-agitation in Spanish will allow physicians to obtain reliable data, achieve real sedation goals, and avoid adverse consequences derived from not achieving these goals. This finished product is a proven tool for use not only in the clinical field but also in research contexts.
The Spanish version of the RASS shows an appropriate concordance with the original version in terms of validity, reliability, and applicability. This scale should be used systematically with all critical patients hospitalized in the ICU with the goal of reducing the negative impacts of overdosing and/or agitation.
Ethical disclosures
Protection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of data. The authors declare that no patient data appear in this article.
Right to privacy and informed consent. The authors declare that no patient data appear in this article.
Conflicts of interest
The authors have no conflicts of interest to declare.
Financing
The authors did not receive sponsorship to carry out this article.
References
1. McGrane S, Pandharipande PP. Sedation in the intensive care unit. Minerva Anestesiol. 2012;78:369-80. [ Links ]
2. Hughes CG, McGrane S, Pandharipande PP. Sedation in the intensive care setting. Clin Pharmacol. 2012;4:53-63. [ Links ]
3. Tung A, Rosenthal M. Patients requiring sedation. Crit Care Clin. 1995;11:791-802. [ Links ]
4. Godet G, Gossens S, Prayssac P, Daghfous M, Delbrouck D, Aigret D, et al. Infusion of propofol, sufentanil, or midazolam for sedation after aortic surgery: comparison of oxygen consumption and hemodynamic stability. Anesth Analg. 1998;87:272-6. [ Links ]
5. Sánchez-Izquierdo JA, Alted E, Borges M, Sandumiege A. Sedoanalgesia-relajación en la Unidad de Cuidados Intensivos. Situación en España. Med Intensiva. 1998;22:1-6. [ Links ]
6. Cai Y, Li Y, Ji M, Yang H, Zhang Q, Jin Z. The effect of mild sedation on the prognosis and inflammatory markers in critical patients with mechanical ventilation. Zhonghua Jie He He Hu Xi Za Zhi. 2014;37:820-3. [ Links ]
7. Kollef MH, Levy NT, Ahrens TS, Schaiff R, Prentice D, Sherman G. The use of continuous i.v. sedation is associated with prolongation of mechanical ventilation. Chest. 1998;114:541-8. [ Links ]
8. Hodgson C, Bellomo R, Berney S, Bailey M, Buhr H, Denehy L, et al. Early mobilization and recovery in mechanically ventilated patients in the ICU: a bi-national, multi-centre, prospective cohort study. Crit Care. 2015;19:81. [ Links ]
9. Botha J, Le Blanc V. The state of sedation in the nation: results of an Australian survey. Crit Care Resusc. 2005;7:92-6. [ Links ]
10. Thuong M. Sedation and analgesia assessment tools in ICU patients. Ann Fr Anesth Reanim. 2008;27:581-95. [ Links ]
11. Sneyers B, Laterre PF, Perreault MM, Wouters D, Spinewine A. Current practices and barriers impairing physicians' and nurses' adherence to analgo-sedation recommendations in the intensive care unit - a national survey. Crit Care. 2014;18:655-67. [ Links ]
12. Gjersing L, Caplehorn JR, Clausen T. Cross-cultural adaptation of research instruments: language, setting, time and statistical considerations. BMC Med Res Methodol. 2010; 10:13. [ Links ]
13. Ely EW, Truman B, Shintani A, Thomason JW, Wheeler AP, Gordon S, et al Monitoring sedation status over time in ICU patients: reliability and validity of the Richmond Agitation-Sedation Scale (RASS). JAMA. 2003;289:2983-91. [ Links ]
14. Sessler CN, Gosnell MS, Grap MJ, Brophy GM, O'Neal PV, Keane KA, et al. The Richmond Agitation-Sedation Scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166:1338-44. [ Links ]
15. Chanques G, Jaber S, Barbotte E, Verdier R, Henriette K, Lefrant JY, et al. Validation of the french translated Richmond vigilance-agitation scale. Ann Fr Anesth Reanim. 2006;25:696-701. [ Links ]
16. Almgren M, Lundmark M, Samuelson K. The Richmond Agitation-Sedation Scale: translation and reliability testing in a Swedish intensive care unit. Acta Anaesthesio Scand. 2010;54:729-35. [ Links ]
17. Nassar Junior AP, Pires Neto RC, de Figueiredo WB, Park M. Validity, reliability and applicability of Portuguese versions of sedation-agitation scales among critically ill patients Sao Paulo Med J. 2008;126:215-9. [ Links ]
18. Wild D, Grove A, Martin M, Eremenco S, McElroy S, Verjee-Lorenz A, et al. Principles of Good Practice for the Translation and Cultural Adaptation Process for Patient-Reported Outcomes (PRO) Measures: report of the ISPOR Task Force for Translation and Cultural Adaptation. Value Health. 2005;8:94-104. [ Links ]
19. Ramsay M. El costo biológico de la depresión de la conciencia. Rev Colomb Anestesiol. 2015;43:119-21. [ Links ]
20. Moreno H. No es tan sencillo como «Te pondré a dormir»: una perspectiva neurobiológica sobre la sedación. Rev Colomb Anestesiol. 2015;43:173-5. [ Links ]











 text in
text in