What do we know about this problem?
Hepatic chemoembolization (HC) is an interventional radiology procedure used worldwide for the treatment of patients with hepatocellular carcinoma. To perform this procedure, different clinical professionals, including anaesthetists, must be pre-sent inside the X-ray room. Due to its complexity, chemoembolization procedures demand high fluoroscopic times and digital subtraction angiography images, exposing patients and medical staff to high radiation doses. For HC procedures we have previously reported on the ocular doses received by the main operator but, as of this date, no dosimetric data are available regarding anesthetist exposure levels.
What does this study contribute?
In this study, occupational dosimetry was performed in order to estimate the doses received by anaesthetists in hepatic chemoembolization procedures.
Having a record of occupational exposure in this type of procedure helps to optimize radiation protection practices of each professional in the interventional radiology room and also understand the biological response mechanisms to low doses of X-radiation.
INTRODUCTION
Hepatic chemoembolization (HC) is an interventional radiology procedure used worldwide for the treatment of patients with hepatocellular carcinoma. To perform this procedure, different clinical professionals, including anesthetists, must be present inside the X-ray room. Due to its complexity, chemoembolization procedures demand high fluoroscopic times and digital subtraction angiography images, exposing patients and medical staff to high radiation doses 1,2.
Annual effective doses for radiologists performing chemoembolization procedures (3.16 mSv) are comparable or higher than those received in other interventional procedures, including radiofrequency ablation (4.26 mSv), cardiac angiography (1.41 mSv) and cerebrovascular interventions (0.83 mSv), respectively 3.
Khoury et al. 2 showed that radiologists performing HC procedures can receive total body and eye lens doses of up to 41 μSv and 894 μSv per procedure, respectively. Based on these results, and considering the current occupational dose limits recommended by the International Atomic Energy Agency 4, the authors conclude that a radiologist who performs chemoembolization procedures without radiation protection measures would reach the annual limit of equivalent dose in the eyes of 20 mSv with just one procedure per week.
Most studies have reported data on the occupational dose received by the main operator in HC; however, up to now there are no dosimetric data about the exposure levels received by anesthetists in this type of procedure. The objective of this study was to assess the radiation exposure received by one anesthetist and the main operator over the course of seven consecutive HC procedures conducted in a private hospital in Recife, Brazil.
METHODS
This study was conducted in a private hospital in the city of Recife, Brazil between January 21 and 27, 2023, and was approved by the Brazilian National Research Ethics System (SISNEP) under certified number 53083016.2.0000.5198 (CAAE). A 35-year old anesthetist was monitored during seven consecutive hepatic chemoembolization procedures (one procedure per day) performed in patients with hepatocellular carcinoma (HCC). The procedures were carried out by one experienced interventional radiologist under local anesthesia and mild sedation through a unilateral right femoral artery access using an Artis zeego interventional angiography system (Siemens-Healthineers). The hepatic arterial vasculature was delineated by means of digital subtraction angiography (DSA) images using a posteroanterior (PA) beam projection.
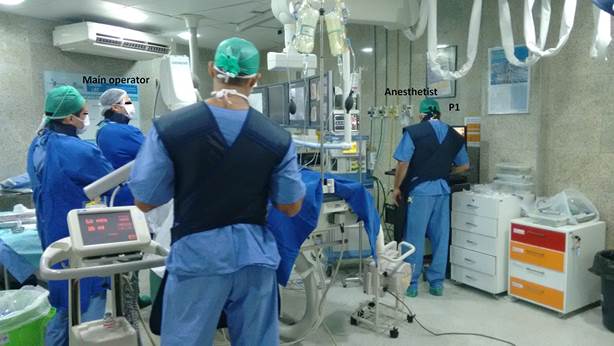
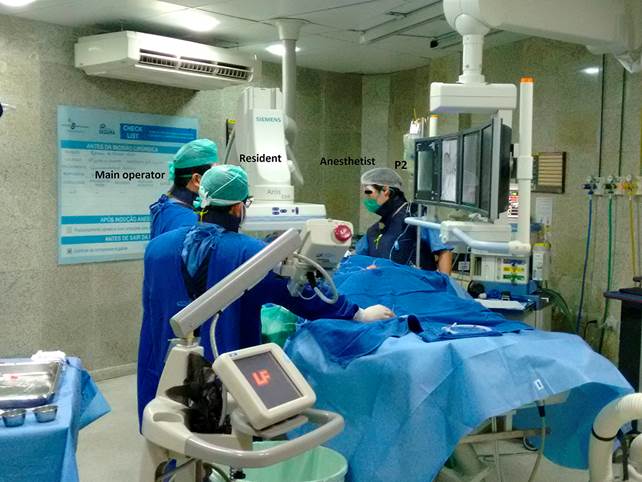
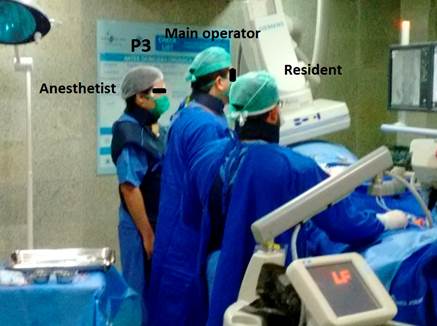
In most procedures, the anesthetist remained in a relative safe position in the X-ray room (P1; approximately two and half meters from the isocenter of the angiography equipment), as depicted in Figure 1. During two of the procedures, the anesthetist remained close to the patient in order to monitor hemodynamic function as illustrated in Figure 2 (position P2) and Figure 3 (position P3). For all procedures, both the main operator and the anesthetist wore protective aprons and thyroid collars both with an equivalent lead thickness of 0.5 mm, but they didn't wear leaded glasses.
For each procedure, dosimetric parameters such as air kerma - area product (PKA) and fluoroscopic time were extracted from the dose report generated by the angiographic equipment, through the DICoM (Digital Imaging and Communications in Medicine) system.
Anesthetist and main operator absorbed doses were recorded using TLD-100 thermoluminiscent dosimeters (Thermo Scientific) placed next to the eyes, close to the thyroid (over the shielding) and on the chest (under the apron). Absorbed doses to the eyes were expressed in terms of the operational quantity, personal dose equivalent, Hp (3), which is the suitable metric for eye lens dose monitoring 4. The effective dose was estimated by means of the following double-dosimetry algorithm proposed by von Boetticher et al 5: E = 0.84H p,u (10) + 0.051H p,o (10) where H p,u (10) and H p,o (10) are the personal dose equivalent Hp (10) readings of dosimeters placed on the chest under the apron and on the neck over the thyroid protector, respectively.
Average ratios of eye lens dose to personal dose equivalent at the neck Hp,o (10) were assessed to evaluate possible eye dose correlations with whole body dose. The best correlations are given by the lowest spread on the average eye lens dose/ Hp,o (10) ratio.
For data analysis, quantitative variables were expressed as means and standard deviations. The Originpro 9.1 software was used for the statistical analysis.
RESULTS
Table 1 shows means and standard deviations for eye doses, as well as effective dose and personal dose equivalent Hp (10) values at the chest (under the apron) and the neck (above the apron) per procedure for the monitored anesthetist in this study. The mean PKA and fluoroscopy time values in this study were (338.2 ± 198) Gy*cm2 and (18.5 ± 7.6 min), and are in agreement with those reported in the literature for hepatic chemoembolization 1,2. For the seven procedures evaluated in this study, the anesthetist received, on average, absorbed radiation doses to the glabella, the left eyebrow, the right eyebrow, and effective dose of 142.4 ± 72 μSv, 117.3 ± 66 μSv, 137.8 ± 71 μSv and 12.4 ± 8.4 μSv, respectively. As for the main operator, the mean Hp (3) values per procedure for the glabella, the left eyebrow and the right eyebrow were 257.2 ± 119 μSv, 345.7 ± 169 μSv and 88 ± 62 μSv, respectively, as shown in Table 2. The mean effective dose was 15 ± 10 μSv.
Table 1 Anesthetist's mean Hp(3) values per eye region, effective dose and Hp(10) per procedure for the seven HC procedures in this study.
| Procedure | Hp(3) μSv | Effective dose (μSv) | Hp,u(10) (μSv) | Hp,o(10) (μSv) | ||
|---|---|---|---|---|---|---|
| Glabella | Left eyebrow | Right eyebrow | ||||
| 1 | 111.6 | 78.5 | 130.2 | 7.0 | 1.3 | 115.9 |
| 2 | 204.0 | 229.8 | 196.5 | 19.8 | 5.7 | 294.6 |
| 3 | 257.7 | 1_148.1 | 257.9 | 25 | 12.2 | 288.1 |
| 4 | 46.6 | 44.3 | 48.6 | 4.7 | 3.0 | 41.7 |
| 5 | 79.0 | 1_81.1 | 71.4 | 12.2 | 9.6 | 80.3 |
| 6 | 144.9 | 74.7 | 136.8 | 2.2 | 1.7 | 15.9 |
| 7 | 152.9 | 164.7 | 123.3 | 16 | 8.2 | 179 |
| Mean | 142.4 | 1_117.3 | 137.8 | 12.4 | 6.0 | 145.1 |
| Standard deviation | 72.3 | 65.6 | 71.3 | 8.4 | 4.2 | 113 |
Hp,u(10): Reading of dosimeter placed on the chest under the apron.
Hp,o(10): Reading of dosimeter placed on the neck over the collar.
Source: Authors.
Table 2 Anesthetist and main operator mean eye doses and effective dose values per procedure for the seven HC procedures in this study.
| Physician | Hp (3) ± SD (μSv) | Effective dose ± SD (μSv) | ||
|---|---|---|---|---|
| Glabella | Left eyebrow | Right eyebrow | ||
| Anesthetist | 142.4 ± 72.3 | 117.3 ± 65.6 | 137.8 ± 71.3 | 12.4 ± 8.4 |
| Main operator | 257.2 ± 119 | 345.7 ± 169 | 88 ± 62 | 15 ± 10 |
SD = Standard deviation.
Source: Authors.
Comparison of our results with data from other studies (Table 3) shows that anesthetist doses reported in this work for hepatic chemoembolization are comparable with doses reported from other complex interventional procedures such as prostatic artery embolization and neurological procedures 6,7.
Table 3 Anesthetist dose comparisons (μSv Gy-1cm-2) between this and other studies.
| Study | Cases | Procedure | Hp(3) eye lens /PKA Mean (range) | (E)/P KA Mean (range) |
|---|---|---|---|---|
| Our study (2023) | 7 | Hepatic chemoembolization | 0.392 (0.137-0.734) | 0.047 (0.003-0.094) |
| Garzón et al. (2019) | 10 | Prostatic artery embolization | 0.373 (0.109-0.833) | 0.045 (0.004-0.213) |
| Kong et al. (2015) | 12 | Neurology | 0.384 (0.093-1.353) | 0.371 (0.004-1.287) |
| 9 | Vertebroplasty (thorax) | 0.592 (0.144-1.337) | 0.050 (0.013-0.156) |
Source: Authors.
The average eye lens dose / Hp,o (10) ratios for the glabella, left eyebrow and right eyebrow were 2.1 ± 3.1, 1.1 ± 0.5 and 2.0 ± 2.9. These results show that the best correlation between Hp(3) and Hp,o (10) was observed for the left eye dose measurement.
DISCUSSION
Although several studies have reported on occupational radiation levels to which anesthetists are exposed in various interventional radiology procedures 8-11, little is known about radiation doses during hepatic chemoembolization procedures. In this study, both the eye lens dose and effective dose were estimated for one anesthetist assisting seven consecutive HC procedures. In addition, the occupational dose of the radiologist who performed the procedures is reported.
Results showed that the mean doses per procedure to the main operator were higher than those received by the anesthetist, except to the right eyebrow region. This was expected given that the main operator remains on the right side of the patient and the X-ray tube during the procedure. In fact, in almost all endovascular radiological procedures, the interventional radiologist performs the intravascular access through the right femoral artery at the groin 12. Compared with the main operator, anesthetists normally stand to the left of the patient and their position varies 7.
The anesthetist's mean right eyebrow dose per procedure was 36% higher than that of the main operator (see Table 2). Analysis of the data showed that the absorbed radiation dose to the anesthetist's right eye was 3.1 (196.5 μSv vs 62.7 μSv) and 4 (257.9 μSv vs 64.9 μSv) times higher than that of the main operator for procedures 2 and 3, respectively. This fact can be explained because, during those procedures, the anesthetist remained with the right side of his body close to the X-ray tube (primary radiation source) and the patient (main scatter radiation source) as he checked hemodynamic and respiratory functions while the X-ray equipment was emitting radiation (Figures 2 and 3). Similar findings were reported by Garzón et al. 6 and Kong et al. 7 in neuroradiology and prostatic artery embolization procedures. We also noted that the anesthetist's effective dose can be similar or higher than that of the main operator for some procedures. For example, in procedure 2, the effective dose to the anesthetist and the main operator was the same (25 μSv), while in procedure 3 the anesthetist's effective dose was 4.7 times higher than the effective dose received by the main operator (19.8 μSv vs 4.2 μSv). Anastasian et al. found that, at a 1-m distance, the scattered dose to anesthetists in neuroradiology procedures can be four times greater on the side of the patient with the X-ray tube than on the side with the image intensifier 9. Distances of 1 m or less between the anesthetist and the X-ray tube and the patient were observed in this study.
Maximum anesthetist eye dose in this study was obtained for the right eyebrow in procedure 3 (257.9 μSv). Based on this result, it is possible to infer that the annual eye lens dose limit of 20 mSv (416.6 μSv/week) could be exceeded in this professional with only two hepatic chemoembolization procedures per week if radiation protection measures are not adopted during each procedure. Samara et al. assessed the effectiveness of different eye lens protection devices during fluoroscopy-guided interventional procedures. The authors found that doses to the eye lens can be reduced by 4.5 and 2 times with the use of full visor (lead equivalent protection of 0.1 mm) and lead glasses with 0.75 mm Pb equivalent frontal protection and 0.5 mm Pb equivalent lateral protection 13, respectively.
Other actions such as setting the anesthesia device as far away from the table as possible and not standing near the patient unless required when the X-ray beam is on, can further reduce radiation exposure to anesthetists.
In addition to the application of the basic principles for radiation safety (time, distance and shielding), anesthetists should participate in training programs that are relevant to the implementation of safe practices within the interventional suite.
Finally, from retrospective eye lens dose assessment based on whole-body dose in HC procedures, the correction coefficient found in this study should be used for cases where the anesthetist uses the personal dosimeter in the middle of the thyroid collar (equivalent thickness of 0.5 mm of lead) and the procedure involves PA projection. Our result is in agreement with those reported in the literature 6.
CONCLUSIONS
This study found that anesthetists assisting chemoembolization procedures may be subject to radiation exposure levels that are comparable with doses from other complex interventional procedures such prostatic artery embolization and neuroradiology. In some cases, doses to the anesthetist can be comparable or higher than those received by the main operator. The current occupational annual eye lens dose limit of 20 mSv can be exceeded with only two procedures per week if relevant radiation protection actions are not implemented. Measures such as not remaining close to the X-ray tube and the patient while the beam is on and using lead glasses or visors can reduce radiation doses to these professionals.
The results from this study showed that the personal dose equivalent Hp (10) at the neck (over the collar) can be used as complementary measure in estimating the anesthetist's eye dose if ocular protection is not used during procedures.
ETHICAL DISCLOSURES
Ethics committee approval
This study was approved by the Brazilian National Research Ethics System (SISNEP) under the certified number 53083016.2.0000.5198 (CAAE).
Protection of human and animal subjects
The authors declare that no experiments were performed on humans or animals for this study. The authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics commit-tee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).











 texto en
texto en