Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
Biomédica
versão impressa ISSN 0120-4157versão On-line ISSN 2590-7379
Biomédica v.27 n.1 Bogotá jan./mar. 2007
Genetic diversity of Giardia intestinalis populations in Colombia
Zaava Ravid 1, Sofía Duque 2, Adriana Arévalo 2, Rubén Santiago Nicholls 2, Moisés Wasserman 1, 3
1 Laboratorio de Investigaciones Básicas en Bioquímica LIBBIQ, Departamento de Química, Facultad de Ciencias, Universidad Nacional de Colombia, Bogotá D.C., Colombia.
2
Laboratorio de Parasitología, Instituto Nacional de Salud, Bogotá D.C., Colombia.3
Laboratorio de Bioquímica, Instituto Nacional de Salud, Bogotá D.C., Colombia.Recibido: 31/07/06; aceptado: 01/11/06
Introduction.
Giardia intestinalis is a protozoan parasite that causes a gastrointestinal infection known as giardiosis, which is transmitted primarily through fecal-oral contamination. Genetic studies of axenically cultivated Giardia isolates have identified two major genetic groups distributed throughout the world. In the present study 24 native strains of the parasite were analyzed by the RAPD technique (Random Amplified Polymorphic DNA).Objective. To determine the level of polymorphism and the complexity of Giardia intestinalis circulating strains in specific areas of Colombia.
Materials and methods. The RAPD method was used, as it allows for a quick, simple and reliable analysis that requires no prior knowledge of the genetics of the parasite. A RAPD analysis was conducted on native isolates collected in Colombia between 1997 and 2001, established in continuous cultures. Several primers were tested separately, in order to enhance the capacity for discrimination of the method.
Results. Of the 24 strains that were included in the study, 22 were arranged in independent clusters. The strains that were from the same geographic area and collected at about the same time, generally displayed highly similar but distinguishable RAPD patterns. Clones isolated from a strain were analyzed as well, and it was possible to differentiate them molecularly.
Conclusion. The studied strains showed to belong to genotype A .The results suggest that the Colombian strains studied consist of a heterogeneous mixture of closely related populations.
Key words: Random amplified polymorphic DNA technique, Giardia lamblia, variation (Genetics), genetics, population.
Diversidad genética de poblaciones de Giardia intestinalis en Colombia
Introducción. Giardia intestinalis es un parásito protozoario que causa la infección gastrointestinal conocida como giardiosis, la cual se transmite principalmente a través de la contaminación fecal-oral. Estudios genéticos de aislamientos de Giardia cultivados axénicamente han identificado dos grupos genéticos principales distribuidos en el mundo. En el presente estudio se analizaron 24 cepas nativas del parásito por medio de la técnica RAPD (ADN polimórfico amplificado aleatoriamente).
Objetivo. Determinar el nivel de polimorfismo y la complejidad de las cepas circulantes en áreas específicas de Colombia.
Materiales y métodos. Se utilizó el método RAPD debido a que permite obtener un análisis rápido, sencillo y confiable, que no requiere conocimiento previo de la genética del parásito. El análisis RAPD se realizó en cultivos continuos de aislamientos nativos recolectados en Colombia entre 1997 y 2001. Se evaluaron separadamente diversos iniciadores con el objeto de incrementar la capacidad de discriminación del método.
Resultados. De las 24 cepas incluidas en el estudio, 22 se agruparon en clusters independientes. Las cepas que provenían de la misma zona geográfica, y que fueron recolectadas aproximadamente al mismo tiempo, generalmente presentaron patrones RAPD sumamente similares pero distinguibles entre sí. Se analizaron así mismo los clones aislados de una misma cepa y fue posible diferenciarlos molecularmente.
Conclusión. Los resultados de las cepas estudiadas indicaron que éstas corresponden al genotipo A, y sugieren que consisten en una mezcla heterogénea de poblaciones estrechamente relacionadas.
Palabras clave: técnica del ADN polimorfo amplificado aleatorio, Giardia lamblia, variación (Genética), genética de población.
Giardia intestinalis is a protozoan parasite that causes a gastrointestinal infection known as giardiosis, which is transmitted primarily through fecal-oral contamination. Disseminated in a variety of geographic areas, it is frequently present in places with inadequate sewage systems; thus it has a greater incidence in the developing countries (1).
Genetic studies of axenically cultivated Giardia isolates have identified two major genetic groups distributed throughout the world (2). These two groups have been described in Europe as "Polish" and "Belgium", in North America as Groups 1/2 and 3 (3), and in Australia as Assemblages A and B (4). In 2001 Adam (5) proposed unifying the nomenclatures by calling them genotype A and genotype B. In their study, Homan and Mank (6) demonstrated a 100% correlation between the severity of the infection and the parasite genotype. Isolates of genotype A were detected in patients with intermittent diarrhea, while isolates of genotype B were found in patients with persistent diarrhea. However, Read et al. (7) did not find a strong correlation between a symptomatic infection and a specific genotype of Giardia in children under five years old.
The RAPD (Random Amplified Polymorphic DNA) technique has been widely used for the study of genetic diversity, allowing for the detection of polymorphisms in the DNA sequence, using primers of arbitrary sequence (8,9). One of its main advantages is that it provides complex electrophoretic patterns, which allow for a clear distinction, in one assay, without having any previous sequence information. It is a sensitive method and extremely useful when limited quantities of DNA are available.
The assessment of genetic variation in Giardia is of outstanding value in order to a) clarify the taxonomy of the parasite, b) identify the molecular markers associated with infection, virulence and drug resistance and c) understand the epidemiology, specifically concerning the transmission patterns in different geographical areas (10).
In the present study, an adequate RAPD was developed using various primers separately to enhance the capacity to distinguish between the samples. With such RAPD analysis, 24 native strains of Giardia intestinalis, collected in Colombia, were studied to determine the level of polymorphism and the complexity of circulating strains.
Materials and methods
Cells and DNA extraction
Giardia intestinalis trophozoites were established in continuous cultures from clinical samples from various places and years. The Parasitology Group at the Colombian Instituto Nacional de Salud (INS) was responsible for this process. The year of isolation of the 24 analyzed strains and their codes were the following: 1997: MHOM/Co/97/G1, MHOM/Co/97/G4, MHOM/Co/97/G5, MHOM/Co/97/G7, MHOM/Co/97/G8, MHOM/Co/97/G9; 1998: MHOM/Co/98/G14; 1999: MHOM/Co/99/G6, MHOM/Co/99/G15, MHOM/Co/99/G16, MHOM/Co/99/G17, MHOM/Co/99/G18, MHOM/Co/99/G19, MHOM/Co/99/G21, MHOM/Co/99/G22, MHOM/Co/99/G38; 2000: MHOM/Co/00/G27, MHOM/Co/00/G32, MHOM/Co/00/G33, MHOM/Co/00/G34; 2001: MHOM/Co/01/G35, MHOM/Co/01/G36, MHOM/Co/01/G37, MHOM/Co/01/G40.
The geographical origin of these strains was: Amazonas departament: 2; Boyacá department: 3; Bogotá D.C.: 19.
A G. intestinalis strain was established by inoculating gerbils (Meriones unguiculatus) with the clinical sample infected with cysts of the parasite, using a gastric catheter. The animals were sacrificed fourteen days later and the upper part of their small intestine was extracted (11). Parasites were cultured at 34°C, in a TYI-S-33 medium with antibiotics, as developed by Diamond in 1978 (12) and modified by Keister in 1983 (13). Genomic DNA was extracted and purified using the Easy-DNA Kit of Invitrogen (Catalog Number - K1800-01).
RAPD analysis
Eight random primers previously employed in our laboratory (14) were assessed (R1 5 GAC TAC TCG C 3; R2 5 GTC ACG TAG T 3; R3 5 GGC TAC ATC A 3; R4 5 GAG TAC TCA G 3; R5 5 AAT CGG GCT G 3; R6 5 GGG TAA CGC C 3; R7 5 GTG ACG TAG G 3 and R8 5 GTG ATC GCA G 3). The primers used in the study by Deng and Cliver (15) were assessed as well (GPD52 5 ATT GCG TCG A 3; GPD61 5 GCG TGT GAA C 3; GPD72 5 ACG AGC GTG G 3; GPD81 5 CCA GCG TCG C 3 and GPD82 5 GGC CGA CCG T 3).
Each primer was assessed individually by amplifying DNA from the MHOM/Co/97/G1 strain of Giardia intestinalis. The conditions for DNA amplification with the RAPD method were adjusted on the basis of the optimization by Deng and Cliver (15) and included the following: MgCl2 (4.0 mM), dNTPs: dATP, dGTP, dCTP,dTTP (200 mM); primer (1.0 mM); Taq DNA Polymerase (2.5 U) and PCR buffer comprised of Tris-HCl (10 mM) pH 9.0, KCl (50 mM) Triton X-100 (0.1%). The amplification reaction volume was 25 µL. The thermal cycle program consisted of denaturation at 94°C for three minutes, 45 cycles of: denaturation at 94°C for one minute, annealing at 36°C for one minute, and extension at 72°C for two minutes; finally an additional extension at 72°C for seven minutes. The amplification products were detected through horizontal electrophoresis in 1.5% agarose gels, visualized, under UV illumination, after ethidium bromide staining. Each sample was analyzed at least three times to confirm reproducibility.
Phylogenetic analysis
Trees portraying the phylogenetic relationship between the isolates were constructed using the RAPDistance software package (16).
A binary matrix of normalized data was constructed, registering the presence or absence of the principal bands, i.e., those not shared by all the strains. The patterns generated with each of the primers were combined and the pairwise distances for the combined band patterns were calculated using the Dice algorithm described by Nei and Li (17). An unrooted tree was constructed on the basis of the neighbor-joining method (NJ) by Saitu and Nei (18), using the NJTREE and TDRAW software included in the RAPDistance package.
Cloning in semi-solid agarose
This was done according to the method described by Gillin and Diamond (19). It consists of suspending a small number of parasites in borosilicate tubes with TYI-S-33 medium and 0.22% agarose. The MHOM/Co/97/G1, MHOM/Co/01/G37 and MHOM/Co/01/G40 strains were cloned by seeding 5, 10, 20 and 50 parasites in separate tubes. The culture tubes were incubated at 34°C for approximately 20 days, during which time colonies suspended in the semi-solid medium were produced. The genomic DNA was isolated after further growth of the clones in TYI-S-33.
Results
Definition of polymorphic patterns with different primers
Eight primers (14) were initially used, and a good amplification pattern was obtained for primers R5, R6, R7 and R8 (Figure 1A). Primers R5 and R8 produced the most complex amplification patterns. Five primers that were previously used on DNA from G. intestinalis (15) were assessed as well (Figure 1B). Only primers GPD52, GPD72 and GPD82 generated a complex electrophoretic pattern. Primers R5 and GPD82 generated the largest number of amplification products. Nevertheless, they produced some amplification in the negative control (reaction with no DNA template) through internal self-annealing. This rendered them useless for our application. Consequently, primers R8, GPD52 and GPD72 were selected for simultaneous but separate analysis on the G. intestinalis strains. The usefulness of a primer in subsequently characterizing the strains was determined on the basis of two criteria: i) if it displayed a band pattern that was complex enough to allow for distinguishing between strains; and ii) if the polymorphisms were reproducible among samples of the same strain.
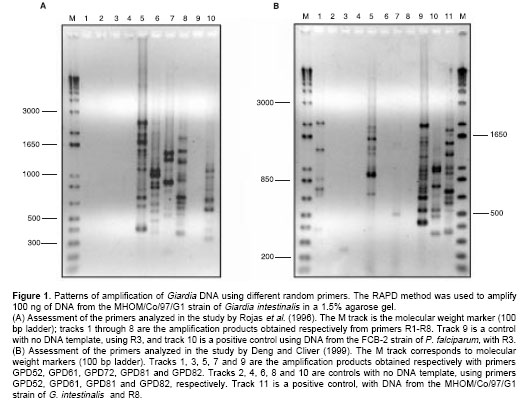
Our analysis of the RAPD patterns was based on a comparison of the RAPD profiles of each strain, using the previously selected primers. By way of example,
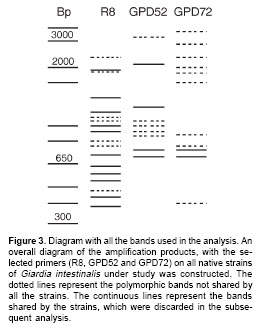
figure 2 shows the result of amplification for some of the strains, using primer R8. Figure 3 is a diagram summarizing the amplification products for all the strains, it indicates whether or not the amplified DNA fragment was a product shared by all the strains, or was present only in some of them.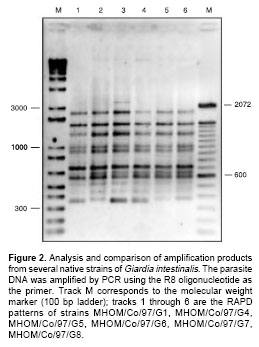
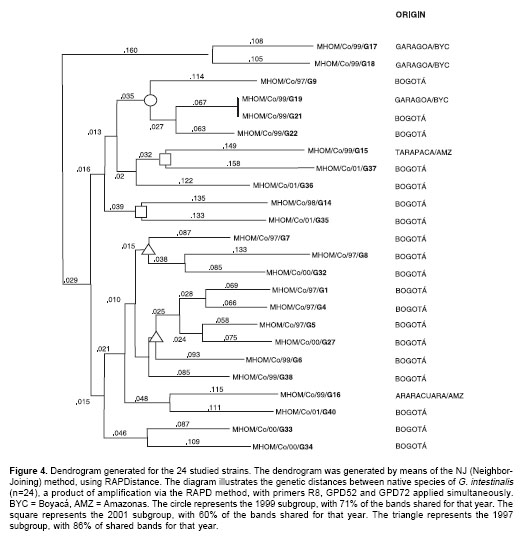
Phylogenetic analysis
A dendrogram was constructed from a combination of all the bands generated through amplification with the three primers. Two large groups were obtained (Figure 4): the first is comprised of the MHOM/Co/99/17 and MHOM/Co/99/18 strains; the second contains all the other strains.
DNA from standard cells Portland-1, ISR, AB and H7 (generously donated by Theodore E. Nash of NIAD-NIH, Bethesda, MD, USA) was assessed to determine if our samples belonged to genotype A or genotype B. Based on this analysis (data not shown), it was determined that all the strains under study probably belonged to genotype A, based on the similarity of their amplification profiles with those of standard culture lines. However, it should be noted that genotype A is often selected for in-vitro growth (20). This fact may affect the distribution results.
RAPD patterns in DNA from clones
The RAPD method allowed us to differentiate between isolates that are genetically very similar. Therefore, we decided to analyze clones from the same isolate. Clones were obtained for the MHOM/Co/97/G1, MHOM/Co/01/G37 and MHOM/Co/01/G40 strains (3, 5 and 4 clones, respectively). The amplification products between clones and with the strain of origin showed differences: two different bands were detected for the MHOM/Co/97/G1 strain and its clones, three different bands for the MHOM/Co/01/G37 strain and its clones, and two different bands for the MHOM/Co/01/G40 strain and its clones (data not shown).
Discussion
The RAPD method has been used widely to characterize different organisms because of its simplicity and potential in taxonomic studies. It enabled us to discover variations at the molecular level, without the need for precise knowledge of the specific sequences of each sample and with a rather high degree of resolution. The final result was a tree of genetic distances constructed with the information of the amplified fragments. The RAPD method was reproducible (in our hands) and very practical. It required small quantities of material, and the profiles it generated were simple to analyze and interpret. However, the method has its limitations. First and foremost, caution must be taken to avoid contamination with external DNA, since the method involves non specific primers, which could amplify any DNA present in the sample. Therefore, it should not be used directly on clinical samples, but only on axenic isolates.
The application of three different primers made it possible to detect differences between almost all the strains. Various fragments did not vary between samples. This reflects the close proximity of the strains subject to study. Seventeen out of 38 (45%) bands were shared by all the samples.
Using the data from the three amplification systems, a single set of data was developed in which each amplified DNA band had an equal value within the set and was used to construct a phylogenetic tree. The overall result shows native strains effectively have a high degree of proximity (
Figure 4), higher than that calculated with the same methods for two standard strains within the same genotype (not shown). All the Colombian strains were, in this way, defined to belong to genotype A.In the dendrogram, the 24 strains were arranged in 23 clusters. In other words, only two samples belonged to the same subgroup. We found a high degree of genetic proximity in strains from locations in the same vicinity. Nevertheless, the two strains from the Amazon region appeared far apart in the dendrogram. This might be explained by the distance between Araracuara and Tarapaca (310 km), where the strains were isolated, and the extraordinary difficulty of physical communication between these two locations, which in practical terms are geographically isolated by a natural barrier, the Amazon jungle. An interesting observation reflected in the dendrogram is the conformation of three large subgroups, which we have called the 1997, 1999 and 2001 subgroups, according to the year in which the majority of the isolates from each subgroup were obtained, with an important degree of proximity, between 66% and 86%. This could suggest that infecting Giardia intestinalis populations, in the same location, may probably undergo changes and diverge in relatively short periods of time (2-4 years). However, this variation might not be necessarily reflected in the genes, since with the employed technique we may detect changes that are result of rearrangement and recombination in non coding sequences.
The differences between strains and their clones suggest the strains are actually a heterogeneous mixture of populations. The source of the genetic diversity in Giardia is unclear, as even the parasites reproductive mechanisms are still being debated. Its reproduction is presumed to be asexual, via a process of binary fission, and genetic diversity is suggested as originating only through mutation and rearrangement events (21-24). Analyses with isoenzymes support this assumption and also clonal selection as the mechanism that generates genetic diversity. However, occasional "outbreaks" of sexual recombination, which are not easily detected, could occur and have more of an impact on extending this genetic diversity (25).
The extensive genetic diversity found here prompts concerns about its origins and meaning. It seems that a considerable length of time would be required for mutations to become evident through techniques other than direct sequencing of the genome. However, for rRNA genes in Giardia, located in telomeric regions of several chromosomes, an estimated rate of rearrangement was reported to be as high as 3% per division and sixty per cent of subcloned lines were found to have discrete rearranged karyotypes (23,24). Further experiments will be needed to confirm if these results with RAPD, a general method, have the same molecular basis as the hypervariation reported in the rRNA genes.
Conflict of interests
The authors declare that they did not incur in any conflict of interests during the present study.
Financial support
This work was supported by the Universidad Nacional de Colombia, the Instituto Nacional de Salud, and the Instituto Colombiano para el Desarrollo de la Ciencia y la Tecnología Francisco José de Caldas, Colciencias, project grant No. 1101-05010060.
Correspondencia:
Moisés Wasserman, Laboratorio de Investigaciones Básicas en Bioquímica, LIBBIQ, Departamento de Química, Facultad de Ciencias, Universidad Nacional de Colombia, Bogotá D.C., Colombia. Teléfono: (571)3165152, fax: (571) 3165165
References
1. Gascón J. Epidemiology, etiology and pathophysiology of travelers diarrhea. Digestion 2006;73 (Supl. 1):102-8. [ Links ]
2. Monis PT, Andrews RH, Mayrhofer G, Ey PL. Genetic diversity within the morphological species Giardia intestinalis and its relationship to host origin. Infect Genet Evol 2003;3:29-38. [ Links ]
3. Nash T. Surface antigen variability and variation in Giardia lamblia. Parasitol Today 1992;8:229-34. [ Links ]
4.Mayrhofer G, Andrews RH, Ey PL, Chilton NB. Division of Giardia isolates from humans into two genetically distinct assemblages by electrophoretic analysis of enzymes encoded at 27 loci and comparison with Giardia muris. Parasitology 1995;111:11-7. [ Links ]
5. Adam RD. The biology of Giardia spp. Clin Microbiol Rev 2001;14:447-75. [ Links ]
6.Homan WL, Mank TG. Human giardiasis: genotype linked differences in clinical symptomatology. Int J Parasitol 2001;31:822-6. [ Links ]
7.Read C, Walters J, Robertson ID, Thompson RC. Correlation between genotype of Giardia duodenalis and diarrhoea. Int J Parasitol 2002;32:229-31. [ Links ]
8.Welsh J, McClelland M. Fingerprinting genomes using PCR with arbitrary primers. Nucleic Acids Res 1990;18:7213-8. [ Links ]
9.Williams JG, Kubelik AR, Kenneth KJ, Rafalski JA, Tingey SV. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res 1990;18:6531-5. [ Links ]
10.Guimaraes S, Sogayar MI, Franco MF. Giardia duodenalis: inter-strain variability of proteins, antigens, proteases, isoenzymes and nucleic acids. Rev Inst Med Trop Sao Paulo 1999;41:45-58. [ Links ]
11.Arévalo A, Duque S, Nicholls RS. Comportamiento de la infección experimental por aislamientos colombianos de Giardia duodenalis en el modelo animal del gerbo (Meriones unguiculatus). Biomédica 2005;25:305-14. [ Links ]
12.Diamond LS, Harlow DR, Cunnick CC. A new medium for the axenic cultivation or Entamoeba histolytica and other Entamoeba. Trans R Soc Trop Med Hyg 1978;72:431-2. [ Links ]
13.Keister DB. Axenic culture of Giardia lamblia en TYI-S-33 medium supplemented with bile. Trans R Soc Trop Med Hyg 1983;77:487-8. [ Links ]
14.Rojas MO, De Castro J, Mariño G, Wasserman M. Detection of genomic polymorphism in Plasmodium falciparum using an arbitrarily primed PCR assay. J Eukaryot Microbiol 1996;43:323-6. [ Links ]
15.Deng MQ, Cliver DO. Rapid DNA extraction methods and new primers for randomly amplified polymorphic DNA analysis of Giardia duodenalis. J Microbiol Methods 1999;37:193-200. [ Links ]
16.Armstrong JS, Gibbs AJ, Peakall R, Weiller G. The RAPDistance Package. 1994. [Consultado: noviembre de 2001]. Disponible en: http://www.rsbs.anu.edu.au/Products&Services/BiotechnologyTransferUnit/rapdistance.asp [ Links ]
17.Nei M, Li WH. Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc Natl Acad Sci USA 1979;76:5269-73. [ Links ]
18.Saitou N, Nei M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 1987;4:406-25. [ Links ]
19.Gillin FD, Diamond LS. Clonal growth of Giardia lamblia trophozoites in a semisolid agarose medium. J Parasitol 1980;66:350-2. [ Links ]
20.Andrews RH, Chilton NB, Mayrhofer G. Selection of specific genotypes of Giardia intestinalis by growth in vitro and in vivo. Parasitology 1992;105:375-86. [ Links ]
21.Tibayrenc M, Kjellberg F, Ayala FJ. A clonal theory of parasitic protozoa: the population structures of Entamoeba, Giardia, Leishmania, Naegleria, Plasmodium, Trichomonas and Trypanosoma and their medical and taxonomical consequences. Proc Natl Acad Sci USA 1990;87:2414-8. [ Links ]
22.Tibayrenc M, Ayala FJ. The clonal theory of parasitic protozoa: 12 years on. Trends Parasitol 2002;18:405-10. [ Links ]
23.Le Blancq SM, Korman SH, Van der Ploeg LH. Frequent rearrangements of rRNA-encoding chromosomes in Giardia lamblia. Nucl Acid Res 1991;19:4405-12. [ Links ]
24.Hou G, Le Blancq SM, E Y, Zhu H, Lee MG. Structure of a frequently rearranged rRNA-encoding chromosome in Giardia lamblia. Nucl Acid Res 1995;23:3310-7. [ Links ]
25.Meloni BB, Lymbery AJ, Thompson RC. Genetic characterization of isolates of Giardia duodenalis by enzyme electrophoresis: implications for reproductive biology, population structure, taxonomy and epidemiology. J Parasitol 1995;81:368-83. [ Links ]














