Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Biomédica
Print version ISSN 0120-4157On-line version ISSN 2590-7379
Biomédica vol.29 no.4 Bogotá Oct./Dec. 2009
1 Division of Pediatrics Infectious Diseases, Department of Pediatrics, University of Texas Medical School at Houston, Houston, TX, USA.
2 Molecular Genetics and Antimicrobial Resistance Unit, Universidad El Bosque, Bogotá, D.C., Colombia
Recibido: 07/10/08; aceptado:18/05/09
Staphylococcus aureus is a well recognized pathogen with global distribution. In recent years community-associated, methicillin-resistant S. aureus has emerged as an increasing cause of severe infections among adults and children. Herein, a case is reported of a previously healthy, 19-month-old male, who presented with empyema necessitans and acute osteomyelitis due to a community-associated, methicillin-resistant, S. aureus strain. This report highlights the evolving epidemiology of S. aureus, as important pathogen in the community as well as the hospital setting, and the importance of establishing appropriate guidelines for diagnosis, management and surveillance of this public health problem.
Key words: empyema, osteomyelitis, Staphylococcus aureus, pneumonia, vancomycin.
Staphylococcus aureus es uno de los principales patógenos a nivel mundial. Durante los útimos años se ha reportado un incremento en el número de casos de S. aureus resistente a la meticilina adquiridos en la comunidad, tanto en niños como en adultos de los Estados Unidos y de otras partes del mundo.
En el presente trabajo reportamos un caso de empiema necessitans y osteomielitis aguda en un niño de 19 meses de edad previamente sano. El presente reporte resalta la cambiante epidemiología de S. aureus tanto en la comunidad como en el medio hospitalario y la importancia de establecer guías apropiadas para el diagnóstico, el tratamiento y la vigilancia de este relevante problema de salud pública.
Palabras clave: empiema, osteomielitis, Staphylococcus aureus, neumonía, vancomicina.
Methicillin-resistant Staphylococcus aureus (MRSA) is an important, globally distributed, nosocomial pathogen. Recently, numerous cases of community associated MRSA (CA-MRSA) strains, which differ phenotypically and genotypically from the nosocomial isolates, have been reported in adult and pediatric populations. Most published CA-MRSA cases present potentially life threatening infections, such as soft tissue infections, pneumonia and endocarditis. Herein, a case is reported of a previously healthy infant with empyema necessitans and acute osteomyelitis due to CA-MRSA strain. In a review of the literature, appropriate tools are identified for CA-MRSA diagnosis and management.
Case definition
The case definition used for distinguishing CA-MRSA from healthcare-associated MRSA (HA-MRSA) was an individual with MRSA infection that met all of the following criteria: 1) the MRSA diagnosis was made in the outpatient setting or by positive culture for MRSA within 48 hours after admission to the hospital, 2) no medical history of MRSA infection, 3) no medical history in the past year of hospitalization, surgery or permanently inserted catheters or medical devices that penetrate the skin.
Case report
A previously healthy 19-month-old, African American male was seen at a community hospital emergency room (Houston, TX) because of a five-day history of fever and chest tenderness. A diagnosis of left side pneumonia was made and the patient was discharged, with on azithromycin and acetaminophen.
Three days later he returned to the emergency room with persistent fever, pain over his left chest wall and worsening of respiratory symptoms. He was given albuterol, atrovent and one dose of ceftriaxone and vancomycin; later, he was transferred to the Hermann Hospital (Houston TX). No history of previous hospitalizations, antibiotic use, or sick contacts were documented.
The patient appeared to be in moderate distress; he had a temperature of 98°F, a pulse of 168 beats per minute and a respiratory rate of 60 per minute. Chest examination revealed a 3 x 4 cm mass in the left chest wall between the midclavicular and axillary lines, approximately between the third and fifth intercostal spaces. Breath sounds over the entire left side of the chest were decreased. The remainder of the examination was unremarkable.
Laboratory data showed a white blood count of 8,200 with 15% neutrophils, 13% bands, 42% lymphocytes and 11% lymphocytes. Hemoglobin was 11.8 g/dl, hematocrit 35.3% and platelet count of 328,000 mm3.
A chest radiograph showed complete opacification of the left hemithorax consistent with pneumonia and effusion (figura 1). A contrast computed tomography scan of the chest showed a large left empyema with total collapse of the left upper lobe and extension into the left chest wall. A diagnosis of empyema necessitans was made on the basis of the scan and clinical presentation; subsequently, a left thoracospic decortication was performed with removal of fibrin-purulent exudates, and ceftriaxone plus vancomycin were administrated.
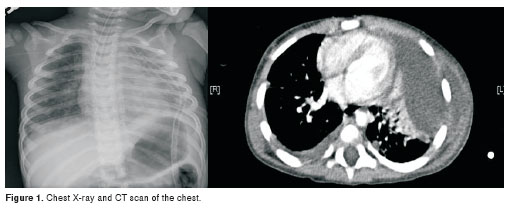
Given the history of bilateral pulmonary nodules compatible with septic emboli, two echocardiograms were performed and were negative for endocarditis. Blood cultures at admission and cultures from the purulent fluid extracted from lungs and from the subcutaneous and chest wall collections were positive for S. aureus infection which was resistant to oxacillin and erythromycin. The isolate was sensitive to clindamycin, gentamycin, rifampin,trimethoprim-
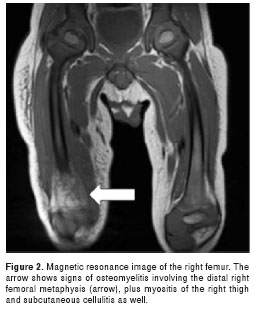
During his hospitalization, local swelling and tenderness in the right thigh was found and thrombosis of right femoral and popliteal vein was documented; therefore, anticoagulation therapy with heparin was initiated. Additionally, magnetic resonance imaging showed early stages of acute osteomyelitis of right distal femur (figura 2); therefore, vancomycin was continued for 36 days, followed by oral clindamycin. The patient was discharged after resolution of the lung infection and maintained on oral clindamycin to complete the treatment for osteomyelitis. Anticoagulation with warfarin was continued for three months. At follow up, the patient appeared completely recovered.
Discussion
Empyema necessitans is a rare clinical condition, which was initially recognized by Gullan De Baillon in 1640 (1). This complication of empyema is characterized by the extension of pus from the pleural cavity into the thoracic wall to form a mass of purulent fluid in the adjacent soft tissues. The most common sites of extension are the anterior chest wall, esophagus and mediastinum and alternative areas of extension include breast, diaphragm, retroperitoneum, and groin (2). Empyema necessitans is usually reported in adults; and rarely in pediatric patients (1-3). The majority of the cases are associated with Mycobacterium tuberculosis, Streptococcus pneumoniae, S. aureus, Streptococcus milleri, Pseudomonas cepacia, and Mycobacterium avium-intracellulare. Non infections etiologies include lymphomas, bronchogenic carcinoma, mesothelioma and sarcomas (1,4-5). Two cases of empyema necessitans due to CA-MRSA have been reported in otherwise healthy children (5,6).
Currently, CA-MRSA is one of the most common causes of pneumonia in all age groups (7-10). These cases are characterized by high fever, hypotension, leucopenia and multilobar infiltrates that usually progress into abscess/empyema. Metastatic pulmonary infections have become more frequent among infants with invasive staphylococci disease, especially among those with bone and joint infections (similar to the current report) (11). Furthermore, an increased frequency of deep venous thrombosis (DVT) associated with bone infections has been reported (11). Therefore, when the history and physical examination suggest metastatic disease, laboratory and diagnostic imaging evaluation are recommended for confirmation.
However, routine performance of echocardiography for individuals with S. aureus bacteremia is controversial (12-14). Nevertheless, evaluation of children for endocarditis seems prudent when blood culture results are persistently positive or when findings consistent with pulmonary emboli are noted on chest radiograph images (15).
The primary approaches to CA-MRSA infections management include surgical drainage, along with cultures for bacterial identification and drug susceptibility testing, followed by adequate antimicrobial therapy (16). The primary therapy in the management of empyema necessitans includes aggressive drainage of abscesses or other purulent collections by thoracotomy, decortications or wide surgical drainage, and adequate antimicrobial therapy.
The specific choice of antistaphylococcal agent depends on the site of the infection and the susceptibility of the isolates to antimicrobials. Empirical therapy with intravenous vancomycin and oxacillin is recommended. Once sensitivities are known and MRSA infection is confirmed, vancomycin is the recommended therapy, if the infection is due to methicillin-susceptible S. aureus, oxacillin is the drug of choice (17).
Clindamycin is an antibiotic option for the treat-ment of serious infections caused by CA-MRSA. However, CA-MRSA strains that are susceptible to clindamycin but resistant to erythromycin may have the phenotype of in vitro inducible macrolide-lincosamide-
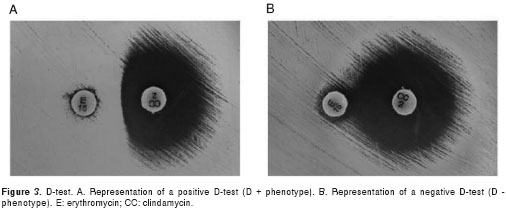
TMP-SMX can be an ideal agent for the management of uncomplicated CA-MRSA infections; this agent is available in oral and intravenous formulations and has excellent tissue penetration. The current review led to the conclusion that TMP-SMX may be effective for S. aureus infections with a low bacterial burden. However, for severe staphylococcal infections with high bacterial loads, such as tricuspid valve endocarditis, bacteremia, pneumonia, and bone and joint infections, TMP-SMX is not recommended as a monotherapy (17).
Rifampin distributes well into many body tissues and may act synergistically with other antibiotics (e.g. vancomycin). Hence, it is a useful supportive therapy in treating deep-seated staphylococcal infections such as endocarditis, osteomyelitis and empyema (19). Linezolid was equal to vancomycin for the treatment of infections caused by resistant gram-positive bacteria. It has the ability to inhibit toxin production in PVL-producing strains, as does clindamycin, fusidic acid and rifampin (20).
Daptomycin, tigecycline, ceftobiprole, oritavancin, telavancin and dalbavancin are potential treatment options, but data supporting use in children is limited.
Currently, the majority of the CA-MRSA pediatric infections in the United States and Colombia are caused by the USA300 and USA400 strains (previously MW2) (19,20, In: American Society for Microbiology. Abstracts of the 47th Inter-Science Conference on Antimicrobial Agents and Chemotherapy. Abstract C2-145. p.101).
CA-MRSA infections are widely disseminated in the world. The new strain causes a wide number of life-threatening infections (skin and soft tissue infections, necrotizing pneumonia, acute osteomyelitis, py omisitis, endocarditis and meningitis) in patients with no apparent risk factors for MRSA acquisition (21).
The prevalence of CA-MRSA varies geogra-phically. According to a study in 2005 from the Center for Disease Prevention and Control, CA-MRSA comprised 8-20% of all MRSA isolates. An annual disease incidence of 25.7/100,000 was recorded in Atlanta, Georgia compared with a rate of 18/100,000 in Baltimore, Maryland. The incidence was higher among children <2 years old (22).
In Colombia, the first report of CA-MRSA was of two healthy adults who presented with severe skin and soft infections in 2005 (23). Subsequently, multiple cases of bloodstream and soft tissue infections were indentified in adults and children from Cali and Cartagena (American Society for Microbiology. In: Abstracts of the 47th Inter-Science Conference on Antimicrobial Agents and Chemotherapy. Abstract C2-145. p. 101).
In Houston, Texas, the number of CA-MRSA infections has increased markedly over the past decade. The most recent report from Division of Pediatrics Infectious Diseases, Department of Pediatrics, University of Texas Medical School at Houston showed that 67% of CA-MRSA infections among hospitalized children were abscesses and complicated pneumonias (24).
The capacity of CA-MRSA to cause severe infections is probably due to multiple cell surface-associated and secreted virulence factors. The Panton-Valentine leukocidin (PVL) induces polymorphonuclear cell death by necrosis or by apoptosis and it has been strongly associated with necrotizing pneumonia and osteomyelitis (25,26). The arginine catabolic mobile element (ACME) allows staphylococci to grow and survive within the host, whereas the oligopeptide permease (the second element of the ACME) contributes to nutrient uptake, quorum sensing, pheromone transport, chemotaxis, eukaryotic cell adhesion, binding of serum components, and expression of virulence determinants (26). Enterotoxins K and Q (superantigens) lead to the typical clinical findings of CA-MRSA infections such as fever, hypotension, multiple organ failure and disseminated intravascular coagulation (26). The collagen adhesion protein leads to greater adherence to host tissue, especially collagen (11). Additionally, a group of secreted peptides in CA-MRSA have the ability to lyse human neutrophils, thereby compromising a major cellular defense against CA-MRSA (27).
The determinants of methicillin resistance in S. aureus are mediated by the gene mecA, which has been localized on the chromosome of S. aureus. The mecA gene encodes a penicillin-binding protein (PBP2a), which has low affinity for all beta-lactam antibiotics (28). The mecA gene is localized in a mobile genetic element called staphylococcal cassette chromosome (SCC). This element has been classified in six different types (I-VI) according with the combination of the chromosome recombinase genes (28). The type I, II and III SCCmec have been mainly described in nosocomial strains, whereas type IV, V and VI have been described in the CA-MRSA strains (28). Normally, the SCCmec IV-VII harbor lower determinants of resistance. This explains why CA-MRSA isolates remain susceptible to a wide range of antibiotics such as erythromycin, clindamycin, gentamycin, rifampin, TMP-SMX, vancomycin, and tetracycline.
Prevention is a powerful strategy, and recognition of a population at risk is the first step in control the emergence of this new threat. Most CA-MRSA infections have been identified in populations that have extreme physical contact such as kindergarten children, incarcerated or homeless people, intravenous drug users and athletes. Consequently, keeping skin injuries clean, short fingernails and changing personal hygiene materials may assist in the prevention of CA-MRSA infections (29,30).
A second strategy for individuals in a high risk environment is to bathe twice a week for 15 minutes in water with regular strength Clorox® (one teaspoon per gallon of water). This disinfectant appears to reduce rates of recurrent infections in at-risk populations with a history of skin of soft tissue infections (29). Currently, CA-MRSA is not a reportable disease; however, if an outbreak is suspected, the serious nature of the disease recommends that public health authorities be alerted.
Conclusion
CA-MRSA has emerged as an important pathogen that produces a significant number of severe infections with metastatic complications in otherwise healthy individuals. The management procedures include rapid diagnosis, appropriate incision and drainage (when needed) and vigorous antimicrobial therapy.
There are no conflicts of interest.
No financial support
Correspondencia:
Gloria P. Heresi, Pediatric Infectious Diseases, Department of Pediatrics, The University of Texas Health Science Center at Houston, 6431 Fannin, MSB 6.132a, Houston, TX 77030, USA. Phone: (713) 500 5715, fax: (713) 500 5688 gloria.p.heresi@uth.tmc.edu
1. Mizell KN, Patterson KV, Carter JE. Empyema necessitatis due to methicillin-resistant Staphylococcus aureus: case report and review of the literature. J Clin Microbiol. 2008;46:3534-6. [ Links ]
2. Freeman AF, Ben-Ami T, Schulman ST. Streptococcus pneumonia empyema necessitatis. Infect Dis J. 2004;23:177-8. [ Links ]
3. Ahmed SI, Gripaldo RE, Alao OA. Empyema necessitans in the setting of pneumonia and parapneumonic effusion. Am J Med Sci. 2007;333:106-8. [ Links ]
4. Denlinger CE, Egan TM, Jones DR. Acquired systemic-to-pulmonary arteriovenous malformation secondary to Mycobacterium tuberculosis empyema. Ann Thorac Surg. 2002;74:1229-31. [ Links ]
5. Stallworth J, Mack E, Ozimek C. Methicillin-resistant Staphylococcus aureus empyema necessitatis in an eight-month-old child. South Med J. 2005;98:1130-1. [ Links ]
6. Moore FO, Berne JD, McGovern TM, Ravishankar S, Slamon NB, Hertzog JH. Empyema necessitatis in an infant: a rare surgical disease. J Pediatr Surg. 2006;41:e5-7. [ Links ]
7. Chen CJ, Huang YC, Chiu CH, Su LH, Lin TY. Clinical features and genotyping analysis of community-acquired methicillin-resistant Staphylococcus aureus infections in Taiwanese children. Pediatr Infect Dis J. 2005;24:40-5. [ Links ]
8. Ma XX, Galiana A, Pedreira W, Mowszowicz M, Christophersen I, Machiavello S, et al. Community-acquired methicillin-resistant Staphylococcus aureus, Uruguay. Emerg Infect Dis. 2005;11:973-6. [ Links ]
9. Bratu S, Eramo A, Kopec R, Coughlin E, Ghitan M, Yost R, et al. Community-associated methicillin-resistant Staphylococcus aureus in hospital nursery and maternity units. Emerg Infect Dis. 2005;11:808-13. [ Links ]
10. Alfaro C, Fergie J, Purcell K. Emergence of community-acquired methicillin-resistant Staphylococcus aureus in complicated parapneumonic effusions. Pediatr Infect Dis J. 2005;24:274-6. [ Links ]
11. Gonzalez BE, Hulten KG, Dishop MK, Lamberth LB, Hammerman WA, Mason EO Jr, et al. Pulmonary manifestations in children with invasive community-acquired Staphylococcus aureus infection. Clin Infect Dis. 2005;41:583-90. [ Links ]
12. Musher DM, Lamm N, Darouiche RO, Young EJ, Hamill RJ, Landon GC, et al. The current spectrum of Staphylococcus aureus infection in a tertiary care hospital. Medicine (Baltimore).1994;73:186-208. [ Links ]
13. Lesens O, Hansmann Y, Brannigan E, Remy V, Hopkins S, Martinot M, et al. Positive surveillance blood culture is a predictive factor for secondary metastatic infection in patients with Staphylococcus aureus bacteremia. J Infect. 2004;48:245-52. [ Links ]
14. Abraham J, Mansour C, Veledar E, Khan B, Lerakis S. Staphylococcus aureus bacteremia and endocarditis: the Grady Memorial Hospital experience with methicillin-sensitive S. aureus and methicillin-resistant S. aureus bacteremia. Am Heart J. 2004;147:536-9. [ Links ]
15. Valente AM, Jain R, Scheurer M, Fowler VG Jr, Corey GR, Bengur AR, et al. Frequency of infective endocarditis among infants and children with Staphylococcus aureus bacteremia. Pediatrics. 2005;115:e15-9. [ Links ]
16. Grayson ML. The treatment triangle for staphylococcal infections. N Engl J Med. 2006;355:724-7. [ Links ]
17. Le J, Lieberman JM. Management of community-associated methicillin-resistant Staphylococcus aureus infections in children. Pharmacotherapy. 2006;26:1758-70. [ Links ]
18. Siberry GK, Tekle T, Carroll K, Dick J. Failure of clindamycin treatment of methicillin-resistant Staphylococcus aureus expressing inducible clindamycin resistance in vitro. Clin Infect Dis. 2003;37:1257-60. [ Links ]
19. Adra M, Lawrence KR. Trimethoprim-sulfamethoxazole for treatment of severe Staphylococcus aureus infections. Ann Pharmacother. 2004;38:338-41. [ Links ]
20. Dumitrescu O, Badiou C, Bes M, Reverdy ME, Vandenesch F, Etienne J, et al. Effect of antibiotics, alone and in combination, on Panton-Valentine leukocidin production by a Staphylococcus aureus reference strain. Clin Microbiol Infect. 2008;14:384-8. [ Links ]
21. Chambers HF. The changing epidemiology of Staphylococcus aureus? Emerg Infect Dis. 2001;7:178-82. [ Links ]
22. Fridkin SK, Hageman JC, Morrison M, Sanza LT, Como-Sabetti K, Jernigan JA, et al. Methicillin-resistant Staphylococcus aureus disease in three communities. N Engl J Med. 2005;352:1436-44. [ Links ]
23. Alvarez CA, Barrientes OJ, Leal AL, Contreras GA, Barrero L, Rincón S, et al. Community-associated methicillin-resistant Staphylococcus aureus, Colombia. Emerg Infect Dis. 2006;12:2000-1. [ Links ]
24. Ochoa TJ, Mohr J, Wanger A, Murphy JR, Heresi GP. Community-associated methicillin-resistant Staphylococcus aureus in pediatric patients. Emerg Infect Dis. 2005;11:966-8. [ Links ]
25. Baba T, Takeuchi F, Kuroda M, Yuzawa H, Aoki K, Oguchi A, et al. Genome and virulence determinants of high virulence community-acquired MRSA. Lancet. 2002;359:1819-27. [ Links ]
26. Diep BA, Gill SR, Chang RF, Phan TH, Chen JH, Davidson MG, et al. Complete genome sequence of USA300, an epidemic clone of community-acquired meticillin-resistant Staphylococcus aureus. Lancet. 2006;367:731-9. [ Links ]
27. Wang R, Braughton KR, Kretschmer D, Bach TH, Queck SY, Li M, et al. Identification of novel cytolytic peptides as key virulence determinants for community-associated MRSA. Nat Med. 2007;13:1510-4. [ Links ]
28. Hanssen AM, Ericson Sollid JU. SCCmec in staphylococci: genes on the move. FEMS Immunol Med Microbiol. 2006;46:8-20. [ Links ]
29. Nicolle L. Community-acquired MRSA: a practitioner!s guide. CMAJ. 2006;175:145. [ Links ]
30. Kaplan SL. Community-acquired methicillin-resistant Staphylococcus aureus infections in children. Semin Pediatr Infect Dis. 2006;17:113-9. [ Links ]














