Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Biomédica
Print version ISSN 0120-4157On-line version ISSN 2590-7379
Biomédica vol.31 no.4 Bogotá Oct./Dec. 2011
ARTÍCULO ORIGINAL
1Instituto de Biología, Universidad de Antioquia, Medellín, Colombia
2Unidad de Biología Celular y Molecular, Corporación para Investigaciones Biológicas, Medellín, Colombia
3Escuela de Medicina, Universidad de Antioquia, Medellín, Colombia
Author contributions:
Orville Hernández contributed to both the design and development of laboratory assays, analysis of data and writing themanuscript.
Diana Tamayo and Isaura Torres contributed to development of fungal .growth, RNA extraction, cDNA synthesis and Real time PCRlaboratory assays.
Angela Restrepo and Juan G. McEwen contributed to the writing and correction of the manuscript.
Ana María García contributed to the design and development of the project, data analysis and writing and correction of themanuscript.
Recibido: 02/05/11; aceptado:22/08/11
Introduction: Paracoccidioidomycosis is an endemic systemic mycosis caused by Paracoccidioides brasiliensis, a thermally dimorphic fungus that in tissues and cultures at 37°C grows as a yeast while at lower temperatures (less than 24°C) it becomes a mold; however the genes that rule these processes and their expression are poorly understood.
Objective: This research focused on the kinetic expression of certain genes in P. brasiliensis throughout the dimorphic process, one that involves the transition from the mycelium to yeast forms and the germination from the yeast to mycelium form.
Materials and methods: A real-time quantitative polymerase chain reaction (RT-qPCR) was optimized to measure the expression of ten genes connected with diverse cellular functions including cell synthesis and wall structure, oxidative stress response, heat shock response, metabolism, proteins’ processing, solute transport across the cell membrane and signal transduction pathways at different time points during the mycelia to yeast transition, as well as in the yeast to mycelia germination processes.
Results: Genes involved in cell synthesis and wall structure, metabolism and signal transduction were differentially expressed and highly up-regulated during the yeast to mycelia germination process; on the other hand, genes involved in heat shock response, cell synthesis and wall structure were highly up-regulated during the mycelia to yeast transition process. The remaining genes were differentially regulated during both processes.
Conclusion: In this work the up-regulation of certain genes involved in the morphological changes occurring in P. brasiliensis yeast and mycelia forms were confirmed, indicating that these biological processes play an important role during the host-pathogen interactions, as well as in the fungus adaptation to environmental conditions
Key words: Paracoccidioides, paracoccidioidomycosis, yeasts, mycelium, germination, gene expression.
Análisis de la cinética de expresión de genes durante la transición de micelio a levadura y la germinación levadura a micelio en Paracoccidioides brasiliensis
Introducción. La paracoccidioidomicosis es una micosis sistémica causada por el hongo termodimorfo Paracoccidioides brasiliensis. En tejidos y cultivos a 37°C crece como levadura, mientras que a temperaturas menores de 24°C crece como un moho. Sin embargo, se conoce poco sobre los genes que regulan estos procesos.
Objetivo. Se evaluó la cinética de expresión de algunos genes en P. brasiliensis mediante el proceso de dimorfismo incluida la transición del micelio a levadura y de la germinación de levadura a micelio.
Materiales y métodos. Se optimizó una PCR cuantitativa en tiempo real (RT-qPCR) para medir la expresión de diez genes relacionados con diversas funciones celulares que incluyeron: síntesis de pared, respuesta al estrés oxidativo, respuesta al choque térmico, metabolismo, procesamiento de proteínas, trasporte de solutos a través de membranas y transducción de señales, todo ello a diferentes tiempos durante la transición de micelio a levadura, así como de la germinación de levadura a micelio.
Resultados. Se encontró que los genes relacionados con síntesis de pared, metabolismo y transducción de señales, se expresaban de manera diferencial y con regulación positiva durante la germinaciónlevadura a micelio, mientras que algunos genes relacionados con respuesta a choque térmico y a síntesis de pared estaban sobreexpresados en la transición de micelio a levadura. Los genes restantes se regularon de manera diferencial en ambos procesos.
Conclusiones. En este trabajo se confirma la regulación positiva de algunos genes relacionados con los cambios morfológicos de las fases levadura y micelio en P. brasiliensis, procesos biológicos que juegan un papel de importancia durante la interacción huésped-parásito y durante la adaptación del hongo al ambiente, respectivamente.
Palabras clave: Paracoccidioides, paracoccidioidomicosis, levaduras, micelio, germinación, expresión génica.
Paracoccidioidomycosis is one of the most important endemic systemic mycosis in Latin America, affecting most countries within the area, especially Brazil, where the largest number of cases have been reported. Colombia, Venezuela, Ecuador and Bolivia follow in incidence albeit at an important distance (1,2). In paracoccidioidomycosis the primary infection site is represented by he lungs but dissemination to other organs occurs frequently (3). Two distinctive clinical forms are found, the chronic and the acute/subacute (4,5), with predominance of adult males as in females the inhibitory action of their hormones hinders the micelium to yeast transition process during the initial stages of the infection (6,7). The disease is caused by Paracoccidioides brasiliensis, a thermally dimorphic fungus that in tissues and cultures at 37°C appears as round to oval yeast cells of variable size (6 to 40 µm) and in the environment or in vitro at temperatures lower than 24°C, it changes its morphology to the mold form which is composed of thin, septated hyphae capable to producing asexual propagules, microconidia (<5 µm), when cultured under starvation conditions in minimal media (2). Conidia and mycelial fragments are considered the infectious particles. Once inhaled, they reach the alveoli where at the host’s tissues temperature (37°C) change from its saprophytic form to the parasitic multiple budding yeast form capable of producing disease (8). On the same way, the germination process from the yeast to the mycelium form is an important process during P. brasiliensis life cycle since this allows the fungus to grow under different environmental conditions allowing its return to the natural habitat to re-acquire in this way, its infectious capacity (9).
A similar process occurs in other dimorphic fungi such as Histoplasma capsulatum and Blastomyces dermatitidis (3,10).
Previous studies have reported the over-expression of genes involved in diverse cellular pathways during the mycelium to yeast transition in P. brasiliensis, including metabolism, signal transduction, energy, protein and fat synthesis, cellular transport, biogenesis of cellular components, cell wall and membrane remodeling and genes encoding putative virulence factors (11-13). Additionally, in a study on the conidium to yeast transition process, some genes were expressed after 48 hours on transition and shown to be involved in different biologic processes such as metabolism, cell wall synthesis and signal transduction: some of these were considered specific for this process (14). Other studies have focused on the morphological aspects during yeast to mycelium germination (15,16) and the conidium to mycelium germination (17), but the molecular mechanisms of these processes have been poorly explored. A single paper has been published in which an EST library was obtained after 96 hours during conidial germination that showed some hypothetical and unknown proteins involved in this process (18).
In the present study with P. brasiliensis, we analyzed the expression profile of 10 genes at different time-points during the first 96 h of the mycelium to yeast transition, as well as during the first 120 h of the yeast to mycelium germination, some of which had been previously described in relation with pathogenic and transition processes (11,12,14). All the selected genes were involved in a diverse range of cellular functions in P. brasiliensis and some of them were described as expressed during the conidium to yeast transition (14), with exception of Kex2, found by Venancio to be associated to the transition process as a serine-protease involved in protein fate, with an important role during pathogenesis (19-21), glucan synthases α (αGS) and glucan synthases 1 (GS1), proteins of importance in fungal cell wall synthesis (22-25) and AOX as important protein controlling oxidative stress (26). In this study we showed that HSP90 and αGS presented very high expression during the mycelium to yeast transition, Prr1, GS and GDh were over expressed during the yeast to mycelium germination and the other genes studied were differentially regulated in both processes. Altogether, the detailed analysis of the expression profile of this group of genes will bring forth relevant information on the dynamics of transition and germination processes as a whole, and help in selecting which pathways could be targeted for future studies intended to evaluate the pathogenic capacity of P. brasiliensis.
Materials and methods
Fungal growth mycelium to yeast transition and yeast to mycelium germination
Paracoccidioides brasiliensis mycelial form (strain ATCC 60855) was regularly grown in the modified Synthetic McVeigh Morton (SMVM) medium plus thiamine 1 % (27). For morphological transition assays, cultures in their exponential growth and with viability above 97 % were used. Cell concentration and viability were determined using a Neubauer chamber and ethidium bromide-fluorescence staining procedures, respectively (28).
To induce the mycelium to yeast transition process, mycelial cultures were incubated under constant agitation in 500 ml Erlenmeyer flasks at 36.5°C±0.5 with 250 ml of Brain Heart Infusion broth (Becton Dickinson and Company Sparks, MD 21152 USA) plus 1 % glucose (14). Twenty-five ml of this growth medium were used to obtain RNA samples and to evaluate microscopically the transition process prior to RNA extraction. To induce yeast to mycelia germination, yeast cultures grown under the same conditions were incubated at 19 °C±1°C. Twentyfive ml of this growing medium were used to obtain RNA sample and to evaluate germination process microscopically prior to RNA extraction.
RNA extraction and cDNA synthesis
Total RNA was obtained by maceration of the cells with liquid nitrogen followed by the addition of TRIzol ® (Invitrogen, Carlsbad, CA, USA) at different time points of the mycelium to yeast transition (0, 1, 3, 6, 12, 24, 48, 72 and 96 hours), as well as of yeast to mycelium germination process (0, 1, 3, 6, 12, 24, 48, 72, 96 and 120 hours). Total RNA was treated with DNase I (Invitrogen, Carlsbad, CA, USA) and tested through conventional PCR using β-tubulin primers to confirm absence of chromosomal DNA contamination (13). cDNA was synthesized using 2 µg of total RNA with SuperScript III reverse transcriptase according to the manufacturer’s instructions (Invitrogen, Carlsbad, CA, USA).
Real time PCR assays
We measured the expression level of 10 genes involved in different P. brasiliensis cellular functions and described as being expressed during de conidium to yeast transition by Garcia in 2010, (14), with the exception of Kex2 (21), GS1, αGS and AOX. All of them had previously been describe in the Broad Institute P. brasiliensis genomic data base (http://www.broadinstitute.org/annotation/genome/paracoccidioides_brasiliensis). The Broad Institute accession code and references for each gene are listed in table 1. RT-qPCR was performed using SuperScriptTM III Platinum® Two-Step qRT-PCR Kit with SYBR® Green, according to the manufacturer’s instructions (Invitrogen, Carlsbad, CA, USA). The CFX96 Real-Time PCR Detection System (Bio- Rad Laboratories Inc.) was used to measure the expression level. The variations in mRNA expression were calculated using 2ÎÎCT formula, where ÎÎCT is the difference between the target gene and β-tubulin (βâTub) as housekeeping gene, in accordance with previous studies performed by several authors (13,29). Primers were designed using P. brasiliensis sequence data available at the Broad Institute Internet site (http://www.broadinstitute.org/annotation/genome). The primer sequences are listed in table 1. Three biological replicas were carried out for the evaluation of both the mycelium to yeast transition and the yeast to mycelium germination processes and three different qRT-PCR runs for each biological replica (Bio-Rad cat No 2239441) were determined. Data were analyzed with the SPSS statistics 17.0 with ANOVA. A p value less than or equal to 0.05 was considered statistically significant.
Results
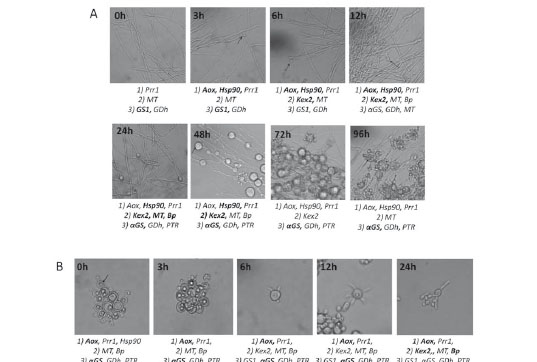
As it concerns morphological changes, when the mycelium was incubated at 37°C, its cell wall became thicker and after 3 hours small internal swellings began to be visualized. In the next 48 hours, these swellings increased in size until acquiring the intracellular vacuoles (lipid vesicles) that characterize P. brasiliensis yeast cells and after 96 hours, multiple budding cells were observed (see arrows in figure 1A). On the other side, the incubation of yeast cells at 18°C stimulated the production of germ tubes from the first 6 hours on transition; these germ tubes became elongated and produced hyphae with loses of intracellular inclusions (see arrows in figure 1B).
RT-qPCR was successfully completed for all evaluated genes, determining the expression level of each one during the mycelium to yeast transition and yeast to mycelium germination processes.
Genes involved in response to stimuli are differentially up-regulated during morphological changes
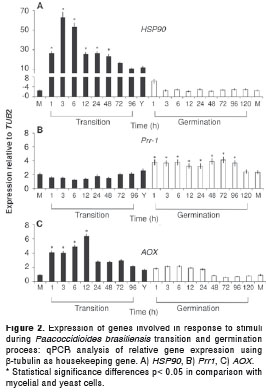
Three genes were involved in the perception and response to stimuli, namely, a heat shock protein (HSP90), a signal transduction gene, a transcription factor (Prr1) and an oxidative stress response protein (AOX). Data analysis indicated that the HSP90 gene expression was not altered during the yeast to mycelium germination time-course maintaining a level between 2 and 4 fold. However, this gene was strongly up-regulated during the mycelium to yeast transition process (more than 60 times fold) and reached its higher level during the first 3 hours but was sustained up to 48h on transition, with significant differences in comparison with the yeast to mycelium germination or mycelia and yeast forms (figure 2A). Whereas, the expression of Prr-1 gene was between 1 and 2.5 times fold during the mycelium to yeast transition without significant alterations, since this gene had higher expression levels (almost 4 fold) all throughout the germination process with meaningful differences in comparison with yeast and mycelium cells (figure 2B). Also, the AOX gene expression was up-regulated during the first 12 hours of the mycelium to yeast transition process (6 fold). Meanwhile, during the yeast to mycelium germination and also in the mycelium form, lower values were observed (figure 2C).
Genes involved in protein modification and fate, played an important role during the morphological changes occurring in Paracoccidioides brasiliensis
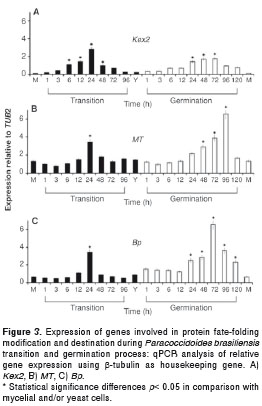
The expression of two genes involved in protein fate (Kex2 and Bp) and one gene related with cell wall synthesis and structure (MT) were evaluated. Kex2 gene expression levels reached their highest values in the middle of the mycelium to yeast transition process (24 h) and in the middle of the yeast to mycelium germination process (24, 48 and 72 h) resulting in a 2 - 3 fold increase. Nonetheless, its expression was lower in yeast cells, in the mycelium and also in the transition processes (figure 3A). The MT gene showed high expression levels (almost 3.5 fold) at 24 hours during the mycelium to yeast transition process while during the yeast to mycelium germination, such gene was up regulated at 48, 72 and 96 hours. Lower levels of expression were observed in mycelial and yeast cells (figure 3B). Also, high expression levels of the Bp gene were noticed at 24 hours of the myceliumto yeast transition (almost 4 fold) was observed; in contrast, during the yeast to mycelium germination, this gene was up regulated with its expression peak occurring at 72 h (more than 6 fold) and with significant differences in comparison with its expression in mycelial and yeast cells (figure 3C).
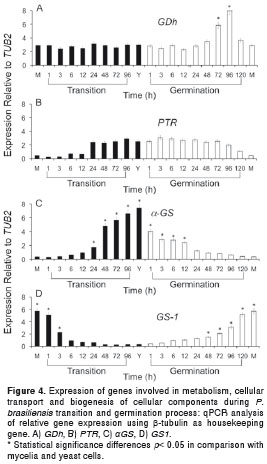
Transition and morphological changes stimulate expression of some genes involved in metabolism, cellular transport and biogenesis of cellular components
Four genes involved in metabolism (GDh), cellular transport - transport facilitation and transport routes (Ptr) and biogenesis of cellular components, such as cell wall synthesis (αGS and GS1, were also evaluated. While GDh was not altered during the mycelium to yeast transition neither in the mycelial or yeast forms, it was expressed at significant levels at 72 and 96 h during the yeast to mycelium germination process (6 and 7.5 times fold, respectively), with significance differences in comparison with the mycelium and yeast forms and the transition process (figure 4A). Also, we observed that the Ptr gene was up regulated after 24h of the mycelia to yeast transition process, and until 96 hours of the yeast to mycelium germination process, with statistically significant higher levels in the yeast form than in the mycelial one (figure 4B). The increase in the αGS gene expression during the mycelium to yeast transition correlated with a decrease in the expression levels of the GS1 gene. On the contrary, an increase in the later GS1 gene level expression at the same time as in the yeast to mycelium germination occurred and was correlated with a decrease in the expression levels of αGS with sustained high levels of αGS in yeast cells and high levels of GS1 in the mycelial form (figure 4C and figure 4D).
Discussion
In P. brasiliensis, the transition process is generally considered as one of the most important steps during pathogenesis and adaptation of the fungus to its host (3), whereas the germination processes is a crucial step for fungal growth and adaptation to environmental conditions with continued value in its biological cycle (9). In both cases molecular regulation is still poorly understood and few studies had reported genes that could possibly be involved in both processes (12,14,18).
The relevance of a gene in a process could be revealed by a quantitative measurement of its expression, whereas higher expression levels could represent its importance in a particular process. This approach has been used before to evaluate genes found in the EST library of P. brasiliensis yeast cells (12). Consequently, measurement of gene expression is an initial step towards understanding gene role during a process. In our study new molecular outlines on the complex mycelium to yeast and yeast to mycelium morphogenetic processes have been revealed by evaluating the kinetic expression profile of some genes that could participate in P. brasiliensis dimorphic processes. Changes in the profile of gene expression during the transition and germination processes are correlated with morphological changes occurring in fungal cells, giving clue times at which individual gene expression switches on or off, leading to production of proteins that rule such morphological changes by acting as modifiers of both structure and fungal metabolism (13).
Three genes were found to enhance expression during the mycelium to yeast transition processes (HSP90, AOX and αGS). Temperature changes represent a kind of environmental stress as fungi and other eukaryotic and prokaryotic cells respond to this stress by inducing synthesis of heat-shock proteins, including HSP90 (30,31). One of the most important features in HSP90 is its ATPase activity which enables chaperone functions of this protein (32). Previous studies in P. brasiliensis have been reported on HSP90 expression in the yeast and mycelial forms, (13,33) but this gene is preferentially expressed in the yeast form (34). In Candida albicans, HSP90 rules the cellular pathway required for transition from yeast to filamentous growth, a crucial feature for virulence in this pathogen (35). In our study, the expression of this gene was strongly up-regulated at the beginning of the transition process, just at the time point when the first changes in the mycelia structure were visualized (figure 1). Similar data were reported by Nicola, et al. (34). This evidence indicates that this protein is essential for thermal adaptation of P. brasiliensis to new environment status resembling host conditions, especially after the first hours of contact and later on in connection with its adaptation as a yeast cell inside the host.
The up-regulation of the AOX gene throughout the mycelia transition could be attributed to the capacity of this gene to reduce the reactive oxygen species (36), elements that can alter the energy status of the yeast cells due to the non-phosphorylative nature of the alternative respiratory pathway (37) produced during a change in cellular metabolism in order to obtain the carbon and energy sources required for an effective transition process. This condition could be of particular relevance during the whole transition and even during the maintenance of the yeast cells in vitro or in tissues, where their own oxidative stress activity makes it necessary a turn on of mechanisms to compensate the morphological changes involved in becoming a yeast cell to ensure its survival in the host´s tissues.
Three genes were up regulated during the yeast to mycelium germination (Prr-1, GDh and αGS). Prr-1 and its homologous in Saccharomyces cerevisiae, Skn7, are recognized for their role in the oxidative stress response (38,39) but their importance in dimorphism has not been reported before. This regulator gene is part of the Two Component Signal System that acts as a response regulator (40). Unless this gene could be expressed during both the mycelium to yeast transition and the yeast to mycelium germination processes, its constant up-regulation mainly during all the germination processes (4 times), making of it a candidate for the initial effectors in the signal transduction pathway in charge of inducing the yeast to mycelia germination.
Glycine dehydrogenase (GDh), present in the yeast EST library published by Goldman, et al. (13), plays an important role in producing ATP (41). We observed a constant over expression of the corresponding GDh gene during morphological changes and mainly at the end of the germination process, indicating that it as a key element in the cell cycle and possibly also by playing an important role in formation and maintenance of P. brasiliensis hyphae.
In dimorphic fungi, αGS and GS1, are important proteins in cell wall structure, αGS in yeast cells and GS1 in mycelial cells (25). During morphological changes occurring in P. brasiliensis cell wall remodeling, a change in the glucan structure occurs going from the beta to the alpha structure during transition while in the yeast cell, changes assist in the evasion from the host immune responses. On the contrary, a change of alpha to beta structure occurs during the germination process (25). These changes cause an alteration in cell shape originated by the exchange of glucans at each stage. We observed a rise in GS1 expression throughout the entire yeast to mycelium germination and of αGS during the mycelium to yeast transition, in accordance with required cell wall structural changes required for mycelium or yeast cellular architecture, respectively.
The three remaining genes, Kex2 involved in processing of proteins, MT important for posttranscriptional manosilation of proteins essential to cell synthesis and wall structure, and involved in protein fate-folding modification and destination; and Ptr, related to cellular transport, transport facilitation and transport routes (42-44), were over expressed in both processes, mycelium to yeast transition and yeast to mycelium germination while the expression of these genes was low in yeast and mycelium forms. This expression could be correlated with the high production of proteins required for morphological changes, and the later adaptation to new environmental conditions. The over expression of these genes stimulate changes in the structural and functional profiles of proteins. These proteins could be correlated with changes in cell walls and in the activation of metabolic pathways needed fpr adaptation to new environmental conditions such as temperature changes involved in thermal dimorphism, among others (20,45,46).
The up-regulation of certain genes involved in the morphological changes corresponding to the mycelial and yeast forms were noticed. All these biological processes should play a crucial role in transition development as this is closely related to host-pathogen interactions and also to germination, a process that is fundamental in environmental adaptation. These results establish a foundation for the understanding of the dimorphic process, and for the characterization of molecular mechanisms that will contribute to understanding the role of some genes in the morphological changes and establishment of a pathogenic process. These genes could be functionally evaluated using knockdown technology (already established for P. brasiliensis) (47) to recognize it importance as virulence and adaptation factors.
Conflicts of interest
The authors declare not have conflict of interest in the present work.
Acknowledgement
This work was supported by COLCIENCIAS, Colombia (project no. 2213-343-19183), the Corporación para Investigaciones Biológicas and the Instituto de Biología of the Universidad de Antioquia.
The National Doctoral Program of COLCIENCIAS 2008 supported Orville Hernández. We thank Fernando Rodrigues from the School of Health Sciences, University of Minho, Braga, Portugal.
Corresponding author: Ana María García, Carrera 72A N° 78B-141, Medellín, Colombia Telephone: (574) 403 5950; fax: (574) 441 5514 agarcia@cib.org.co
References
1. Restrepo A, McEwen JG, Castaneda E. The habitat of Paracoccidioides brasiliensis: how far from solving the riddle? Med Mycol. 2001;39:233-41. [ Links ]
2. Restrepo A, Tobón A. Paracoccidioides brasiliensis. In: Mandell B, Bennett J, Dolin R, editors. Mandell, Douglas and Bennett´s Principles and Practice of Infectious Diseases. 7th ed. Philadelphia: Churchill Livingston, Elsevier; 2009. p. 3357-64. [ Links ]
3. Klein BS, Tebbets B. Dimorphism and virulence in fungi. Curr Opin Microbiol. 2007;10:314-9. [ Links ]
4. Restrepo A, Tobón A, Agudelo C. Paracoccidioidomycosis. In: Hospenthal A, Rinaldi M, editors. Infectious Diseases: diagnosis and treatment of human mycoses. Totowa, New Jersey: Humana Press; 2008. p. 331- 42. [ Links ]
5. Brummer E, Castaneda E, Restrepo A. Paracoccidioidomycosis: an update. Clin Microbiol Rev. 1993;6:89-117. [ Links ]
6. Loose D, Stover E, Restrepo A, Stevens D, Feldman D. Estradiol binds to a receptor-like cytosol binding protein and initiates a biological response in Paracoccidioides brasiliensis. Proc Natl Acad Sci USA. 1983;80:7659-63. [ Links ]
7. Stover E, Schar G, Clemons K, Stevens D, Feldman D. Estradiol-binding proteins from mycelial and yeast-form cultures of Paracoccidioides brasiliensis. Infect Immun. 1986;51:199-203. [ Links ]
8. McEwen J, Bedoya V, Patino M, Salazar M, Restrepo A. Experimental murine paracoccidiodomycosis induced by the inhalation of conidia. J Med Vet Mycol. 1987;25:165-75. [ Links ]
9. Bagagli E, Theodoro RC, Bosco SM, McEwen JG. Paracoccidioides brasiliensis: phylogenetic and ecological aspects. Mycopathologia. 2008;165:197-207. [ Links ]
10. Casadevall A, Steenbergen JN, Nosanchuk JD. ´Ready made´ virulence and ´dual use´ virulence factors in pathogenic environmental fungi--the Cryptococcus neoformans paradigm. Curr Opin Microbiol. 2003; 6:332-7. [ Links ]
11. Bastos KP, Bailao AM, Borges CL, Faria FP, Felipe MS, Silva MG, et al. The transcriptome analysis of early morphogenesis in Paracoccidioides brasiliensis mycelium reveals novel and induced genes potentially associated to the dimorphic process. BMC microbiol. 2007;7:29. [ Links ]
12. Nunes LR, Costa de Oliveira R, Leite DB, da Silva VS, dos Reis Marques E, da Silva Ferreira ME, et al. Transcriptome analysis of Paracoccidioides brasiliensis cells undergoing mycelium-to-yeast transition. Eukaryot Cell. 2005;4:2115-28. [ Links ]
13. Goldman GH, dos Reis Marques E, Duarte Ribeiro DC, de Souza Bernardes LA, Quiapin AC, Vitorelli PM, et al. Expressed sequence tag analysis of the human pathogen Paracoccidioides brasiliensis yeast phase: identification of putative homologues of Candida albicans virulence and pathogenicity genes. Eukaryot Cell. 2003;2:34-48. [ Links ]
14. Garcia AM, Hernandez O, Aristizabal BH, De Souza Bernardes LA, Puccia R, Naranjo TW, et al. Gene expression analysis of Paracoccidioides brasiliensis transition from conidium to yeast cell. Med Mycol. 2010;48:147-54. [ Links ]
15. Queiroz-Telles F. Paracoccidioides brasiliensis: Ultrastructural findings. In: Franco M, Restrepo A, Del Negro G, editors. Paracoccidioidomycosis. Boca Raton, FL.: CRC Press; 1994. p. 27-48. [ Links ]
16. Ramirez Martinez J. Paracoccidioides brasiliensis: conversion of yeastlike forms into mycelia in submerged culture. J Bacteriol. 1971;105:523-6. [ Links ]
17. Restrepo BI, McEwen JG, Salazar ME, Restrepo A. Morphological development of the conidia produced by Paracoccidioides brasiliensis mycelial form. J Med Vet Mycol. 1986;24:337-9. [ Links ]
18. Garcia AM, Hernandez O, Aristizabal BH, Cano LE, Restrepo A, McEwen JG. Identification of genes associated with germination of conidia to form mycelia in the fungus Paracoccidioides brasiliensis. Biomedica. 2009;29:403-12. [ Links ]
19. Torres FA, Vilaca R, Pepe De Moraes LM, Reis VC, Felipe MS. Expression of a kexin-like gene from the human pathogenic fungus Paracoccidioides brasiliensis in Saccharomyces cerevisiae. Med Mycol. 2008;46:385-8. [ Links ]
20. Venancio EJ, Daher BS, Andrade RV, Soares CM, Pereira IS, Felipe MS. The kex2 gene from the dimorphic and human pathogenic fungus Paracoccidioides brasiliensis. Yeast. 2002;19:1221-31. [ Links ]
21. Venancio EJ, Kyaw CM, Mello CV, Silva SP, Soares CM, Felipe MS, et al. Identification of differentially expressed transcripts in the human pathogenic fungus Paracoccidioides brasiliensis by differential display. Med Mycol. 2002;40:45-51. [ Links ]
22. Enderlin CS, Selitrennikoff CP. Cloning and characterization of a Neurospora crassa gene required for (1,3) beta-glucan synthase activity and cell wall formation. Proc Natl Acad Sci USA. 1994;91:9500-4. [ Links ]
23. Rappleye CA, Eissenberg LG, Goldman WE. Histoplasma capsulatum alpha-(1,3)-glucan blocks innate immune recognition by the beta-glucan receptor. Proc Natl Acad Sci USA. 2007;104:1366-70. [ Links ]
24. Rappleye CA, Engle JT, Goldman WE. RNA interference in Histoplasma capsulatum demonstrates a role for alpha- (1,3)-glucan in virulence. Mol Microbiol. 2004;53:153-65. [ Links ]
25. Sorais F, Barreto L, Leal JA, Bernabe M, San-Blas G, Nino-Vega GA. Cell wall glucan synthases and GTPases in Paracoccidioides brasiliensis. Med Mycol. 2010;48:35-47. [ Links ]
26. Van Aken O, Giraud E, Clifton R, Whelan J. Alternative oxidase: a target and regulator of stress responses. Physiol Plant. 2009;137:354-61. [ Links ]
27. Restrepo A, Jimenez B. Growth of Paracoccidioides brasiliensis yeast phase in a chemically defined culture medium. J Clin Microbiol. 1980;12:279-81. [ Links ]
28. Calich VL, Kipsnis TL, Mariano M, Fava Neto C, Da Silva D. The activation of the complement system by Paracoccidioidis brasiliensis in vitro: Its opsonic efect and possible significance for an in vivo model of infection. Clin Immunol Immunopathol. 1979;12:20-30. [ Links ]
29. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402-8. [ Links ]
30. Jakob U, Buchner J. Assisting spontaneity: the role of Hsp90 and small Hsps as molecular chaperones. Trends Biochem Sci. 1994;19:205-11. [ Links ]
31. Lindquist S, Craig EA. The heat-shock proteins. Annu Rev Genet. 1988;22:631-77. [ Links ]
32. Nadeau K, Das A, Walsh CT. Hsp90 chaperonins possess ATPase activity and bind heat shock transcription factors and peptidyl prolyl isomerases. The Journal of biological chemistry. 1993;268:1479-87. [ Links ]
33. Felipe MS, Andrade RV, Petrofeza SS, Maranhao AQ, Torres FA, Albuquerque P, et al. Transcriptome characterization of the dimorphic and pathogenic fungus Paracoccidioides brasiliensis by EST analysis. Yeast. 2003;20:263-71. [ Links ]
34. Nicola AM, Andrade RV, Dantas AS, Andrade PA, Arraes FB, Fernandes L, et al. The stress responsive and morphologically regulated hsp90 gene from Paracoccidioides brasiliensis is essential to cell viability. BMC Microbiol. 2008;8:158. [ Links ]
35. Shapiro RS, Uppuluri P, Zaas AK, Collins C, Senn H, Perfect JR, et al. Hsp90 orchestrates temperaturedependent Candida albicans morphogenesis via Ras1-PKA signaling. Curr Biol. 2009;19:621-9. [ Links ]
36. Parente JA, Borges CL, Bailao AM, Felipe MS, Pereira M, de Almeida Soares CM. Comparison of transcription of multiple genes during mycelia transition to yeast cells of Paracoccidioides brasiliensis reveals insights to fungal differentiation and pathogenesis. Mycopathologia. 2008;165:259-73. [ Links ]
37. Gessler NN, Aver´yanov AA, Belozerskaya TA. Reactive oxygen species in regulation of fungal development. Biochemistry (Mosc). 2007;72:1091-109. [ Links ]
38. Saijo T, Miyazaki T, Izumikawa K, Mihara T, Takazono T, Kosai K, et al. Skn7p is involved in oxidative stress response and virulence of Candida glabrata. Mycopathologia. 2010;169:81-90. [ Links ]
39. He XJ, Mulford KE, Fassler JS. Oxidative stress function of the Saccharomyces cerevisiae Skn7 receiver domain. Eukaryot Cell. 2009;8:768-78. [ Links ]
40. Stock AM, Robinson VL, Goudreau PN. Two-component signal transduction. Annu Rev Biochem. 2000;69:183-215. [ Links ]
41. Sinclair DA, Dawes IW. Genetics of the synthesis of serine from glycine and the utilization of glycine as sole nitrogen source by Saccharomyces cerevisiae. Genetics. 1995;140:1213-22. [ Links ]
42. Pao GM, Saier MH Jr. Response regulators of bacterial signal transduction systems: selective domain shuffling during evolution. J Mol Evol. 1995;40:136-54. [ Links ]
43. Walmsley AR, Barrett MP, Bringaud F, Gould GW. Sugar transporters from bacteria, parasites and mammals: structure-activity relationships. Trends Biochem Sci. 1998;23:476-81. [ Links ]
44. Steiner HY, Naider F, Becker JM. The PTR family: a new group of peptide transporters. Mol Microbiol. 1995;16:825- 34. [ Links ]
45. Seidah NG, Chretien M. Eukaryotic protein processing: endoproteolysis of precursor proteins. Curr Opin Biotechnol. 1997;8:602-7. [ Links ]
46. Oka T, Hamaguchi T, Sameshima Y, Goto M, Furukawa K. Molecular characterization of protein O-mannosyltransferase and its involvement in cell-wall synthesis in Aspergillus nidulans. Microbiology. 2004;150:1973-82. [ Links ]
47. Almeida AJ, Carmona JA, Cunha C, Carvalho A, Rappleye CA, Goldman WE, et al. Towards a molecular genetic system for the pathogenic fungus Paracoccidioides brasiliensis. Fungal Genet Biol. 2007;44:1387-98. [ Links ]














