Tuberculosis continues to be a public health priority worldwide and its control is part of the sustainable development goals established by the United Nations organization 1. The World Health Organization (WHO) estimated that 10.4 million new tuberculosis cases would occur in 2016 2; in Colombia, 13,871 new tuberculosis cases were registered that same year, of which 840 occurred among indigenous populations 3.
The global “End Tuberculosis Strategy” adopted by WHO as a work approach for member states seeks to provide continuity for tuberculosis control actions after 2015. The strategy considers the protection and promotion of ethics and equity in its principles and fosters differential attention to vulnerable populations including indigenous groups 4. The social and cultural determinants and the immunological factors typical of indigenous peoples are associated with a predisposition for the disease. Risk factors such as overcrowding, malnutrition, and difficulties in accessing healthcare services are linked primarily to the geographic areas that these people inhabit. Additionally, ignorance of the disease has been described 5-7.
According to the Organización Nacional de Indígenas Colombianos, there are 102 indigenous groups in Colombia 8. However, the last national census carried out by the Departamento Administrativo Nacional de Estadística (DANE) registered only 87 indigenous groups; these groups are distributed in 710 reservations located in 27 of the 36 territorial entities of the country 9.
Several studies worldwide have shown high rates of tuberculosis among indigenous peoples, mainly associated with difficulties for accessing health services. The active transmission of Mycobacterium tuberculosis has also been confirmed through molecular epidemiology studies 10-15. The approach outlined in the strategic plan “Colombia libre de tuberculosis, 2010-2015” by the Ministry of Health and Social Protection prioritizes vulnerable populations and favors the recognition of the actual tuberculosis situation in these peoples. The following incidence rates have been reported: More than 200 cases per 100,000 inhabitants for the departments of Cesar and Antioquia, more tan 80 cases per 100,000 inhabitants in Nariño and La Guajira, and more than 30 cases per 100,000 inhabitants in Guaviare and Guainía 16,17. These rates are well above the national incidence, which has been approximately 25 cases per 100,000 inhabitants over the last ten years 3.
Classical epidemiology has contributed with important advances in the understanding of the determinants associated with the development of tuberculosis. However, during the last few decades, the use of techniques such as spoligotyping and the implementation of molecular epidemiology have strengthened the study of infectious diseases. At present, associations can be determined between genotypes and the manifestation of multidrug resistance, defined as joint resistance to isoniazid and rifampicin 2. These approaches provide accurate information and increase our understanding of the transmission dynamics of pathologies with a public health impact such as tuberculosis 18.
The objectives of this study were to determine molecular epidemiology and phenotypical features of M. tuberculosis isolates from indigenous people in Colombia between 2009 and 2014.
Materials and methods
Epidemiological information
The information (origin of indigenous people, gender, age, occupational profile, possible risk factors, and clinical characterization of tuberculosis) was compiled in an Excel 2013 database. This information was based on 234 unique mycobacterial surveillance forms submitted to the Colombian Instituto Nacional de Salud Mycobacteria Group during the 2009-2014 period by the health secretariats in each department.
These records corresponded to 229 individuals from indigenous groups diagnosed with tuberculosis. Five people contributed two isolates of M. tuberculosis at different times. In some cases, the tuberculosis and HIV databases of the Sistema Nacional de Vigilancia en Salud Pública, Sivigila, were consulted to complement the information.
Clinical isolates
A total of 234 M. tuberculosis clinical isolates from indigenous people diagnosed with tuberculosis between 2009 and 2014 were obtained from the biobank of the Instituto Nacional de Salud Mycobacteria Group. The cryopreserved isolates were recovered in Löwenstein-Jensen solid medium and incubated at 37°C for two to four weeks to obtain bacillary biomass.
Identification of species and drug susceptibility test
Species identification was performed using the immunochromatography test (TB Ag MPT64 Rapid, Cat N° 08FK50), which detected the MPT64 protein and was confirmed by spoligotyping. First and second-line drug sensitivity tests were performed using the automated BACTEC MGIT TM 960™ system according to the conditions previously standardized by the Instituto Nacional de Salud Mycobacteria Group.
First-line drug sensitivity tests were performed as follows: 1 μg/ml of rifampicin (R), 0.1 and 0.4 μg/ml of isoniazid (H), 1 and 4 μg/ml of streptomycin (S), and 1 and 7.5 μg/ml of ethambutol (E). In cases with resistance to H and/or R, the tests were performed as follows: 2.5 μg/ml of kanamycin (Km), 1 μg/ml of amikacin (Am), 2.5 μg/ml of capreomycin (CM), and 2 μg/ml of ofloxacin (Ofx).
The interpretation was done using the EpiCenter and TB-eXiST software™ (Becton Dickinson 2009, Springer 2009). The internal quality control of these tests was performed using American Type Culture Collection (ATCC) reference strains and the external control supplied by the Laboratorio Supranacional de Referencia at the Chilean Instituto de Salud Pública de Chile.
Spoligotyping
The DNA extraction process was performed according to van Soolingen, et al.19. The spoligotyping analysis was performed according to Kamerbeek, et al. 20. The polymerase chain reaction (PCR) product was hybridized on a commercial membrane (Isogen-LifeScience, The Netherlands). The hybridization results were visualized by chemiluminescence using the ECL Direct™ commercial kit (Amersham, UK) and developed by exposure to a photosensitive film. DNA from the M. tuberculosis H37Rv and M. bovis BCG P3 reference strains were used as a positive control.
Spoligotyping results were converted into binary and octal codes for the determination of the genetic family and the corresponding Spoligotype International Type (SIT) code using the SpolDB4 international database of the Institute Pasteur de la Guadeloupe (Pasteur Institute of Guadeloupe) 21. The database of the Colombian INS and previous genotyping studies conducted in the country were also consulted for comparisons with circulating spoligotypes 22-27. We calculated the Hunter and Gaston index to determine the discriminating power of spoligotyping 28.
Statistical analysis
We conducted a percentage description of the analyzed variables, as well as a bivariate statistical analysis to determine the possible associations between the sociodemographic variables, the sensitivity patterns for antituberculosis drugs, and the spoligotypes and clusters obtained by spoligotyping. We used the chi-square test or the Fisher’s exact test with a 95% confidence interval and a significance of 0.05 using the statistical package Epi Info 7.0TM. The analyses were performed for 234 clinical isolates.
Ethical considerations
All study procedures were approved by the INS Research Ethics Committee. According to the Resolution 8430, 1993, issued by the Colombian Ministry of Health, this study did not require informed consent as all of the samples used were isolates obtained from the biobank of the INS Mycobacteria Group and no personal information from patients was extracted (i.e., they were anonymous samples).
Results
Sociodemographic characteristics of the indigenous populations
We analyzed isolates from 41 indigenous groups, predominately from the Wayúu (n=30, 13.10%), the Emberá Chamí (n=26, 11.35%), the Awá (n=18, 7.86%), the Nasa Páez (n=15, 6.55%), and the U’wa (n=12, 5.24%) (table 1 and 2).
Table 1 Indigenous groups identified and number of persons who contributed isolates (n=229; 100%)

ND: No data
Table 2 Distribution of indigenous groups per department
ND: No data
*Two pregnant individuals and one individual with chronic obstructive pulmonary disease
** 96 (41.7%) were HIV-negative and 127 (55.5%) had no data on HIV status.
The gender distribution showed that 51.09% (n=117) of the study population was male and 48.91% (n=112) was female. The age ranged from three months to 100 years. The age subgroups were 0 to 4 years, 5.68% (n=13); 5 to 14 years, 5.68% (n=13); 15 to 29 years, 29.7% (n=68); 30 to 44 years, 25.8% (n=59); 45 to 59 years, 16.59% (n=38), and 60 years or over, 16.59% (n=38).
Occupational profile and possible risk factors
The most frequent occupations were homemaker (n=63, 27.51%) and farmer (n=50, 21.83%). Other occupations, such as artisan, shaman, botanist, and rancher, were less frequent. The possible risk conditions associated with the development of tuberculosis are described in table 3.
Table 3 Risk factors for tuberculosis identified in the study population

ND: No data
*Two pregnant individuals and one individual with chronic obstructive pulmonary disease
** 96 (41.7%) were HIV-negative and 127 (55.5%) had no data on HIV status.
Clinical and phenotypical characterization of the isolates
Pulmonary tuberculosis was present in 96.94% (n=222) of the cases, whereas extrapulmonary tuberculosis was present in only 3.06% (n=7) of the cases; 83.84% (n=192) of the individuals were classified as new cases, 10.91% (n=25) were previously treated, and 5.24% (n=12) had no recorded data available. Of the previously treated cases, 32% (n=8) were relapses, 8% (n=2) were failures, and 60% (n=15) were cases in which a category could not be established. Three people (1.31%) were recorded as deceased due to tuberculosis.
The 234 positive cultures were recovered from the following clinical samples: Sputum, 82.1% (n=192); gastric aspirate, 7.7% (n=18); secretion with no origin identified, 3% (n=7); biopsy lymph node, 2.1% (n=5); bronchoalveolar lavage, 1.7% (n=4); cerebrospinal fluid, 0.9% (n=2), and ascitic fluid, peritoneal fluid, and pleural fluid, 1.3% (n=3). No information was recorded for 1.3% (n=3) of the samples.
Bacilloscopy
A total of 26.5% (n=62) of the cases were positive in the bacilloscopy (BK+), 23.5% (n=55) were BK++, and 19.2% (n=46) were BK+++, of which 15.2% (n=7) corresponded to minors (3 to 14 years old). The negative BK samples corresponded to 19.2% (n=45) of the cases, and no information was recorded for 11.1% (n=26) of the samples.
All clinical samples had a positive culture on the Löwenstein-Jensen solid medium. Mycobacterium tuberculosis complex identification was positive in all cases.
Drug resistance test
Of the 234 isolates received, five people contributed two isolates at different times. Only one isolate from each of these cases was considered for the sensitivity profiles.
In total, 192 cases were classified as new cases. Of these, sensitivity test results were obtained for 98.44% (n=189); 91% (n=172) of them were sensitive to all the tested first-line drugs, and 8.9% (n=17) presented resistance to at least one of the drugs tested. Of the 189 isolates analyzed, 3.17% (n=6) were multidrug resistant. Of the previously treated cases (n=25), 64% (n=16) were sensitive to all tested first-line drugs and 36% (n=9) were resistant to at least one drug tested. Of the 25 isolates analyzed, 12% (n=3) was multidrug resistant. Of the cases without classification based on a previous history of treatment (n=12), 91.66% (n=11) were sensitive to all first- line drugs, and the remaining case was multidrug resistant (8.33%).
The multidrug resistant isolates came from the indigenous groups Emberá Chamí (n=3), Emberá (n=3), Awá (n=1) and the samples without data (n=2). All cases that showed resistance to H and/or R were also evaluated with second-line drugs, although none presented resistance.
Spoligotyping
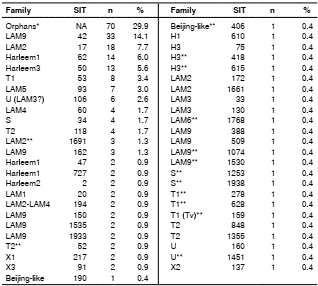
All isolates included in the study (n=234) were confirmed as M. tuberculosis by spoligotyping. No circulation of other members of the M. tuberculosis complex was detected; 164 isolates were distributed in seven genetic families: Latin American-Mediterranean (LAM, 37.2%, n=87), Haarlem (H, 15.8%, n=37), T (8.1%, n=19), U (3.4%, n=8), S (2.6%, n=6), X (2.1%, n=5), and Beijing (0.9%, n=2); a total of 29.9% of the isolates showed orphan spoligotype patterns for genetic families (n=70).
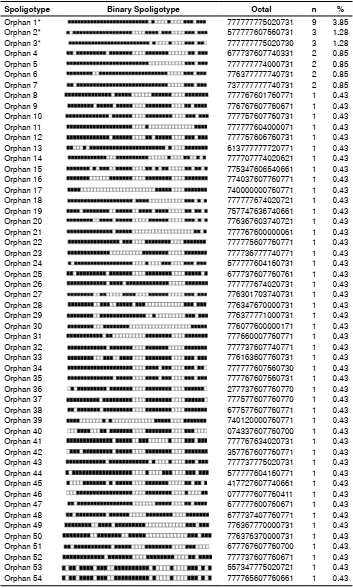
Out of 234 isolates, 102 spoligotypes were identified, of which 47.1% (n=48) were previously described in the SpolDB4 database (table 4) and 52.9% (n=54) were orphan spoligotypes (Supplementary table 1).
Table 4 Distribution of the spoligotypes found in the isolates characterized by spoligotyping (n=234; 100%)
Tv: Tuscany variant; SIT: Spoligotype International Type
*Orphan spoligotypes are described in Appendix 3.
** Spoligotypes reported for the first time in Colombia
Out of the spoligotypes described in the study, 62% (n=144) had not been reported in previous genotyping studies in Colombia; the spoligotype Beijing- like SIT 406 was of particular interest.
When we compared the proportion of spoligotypes found with the INS database and the information reported in Colombia, we established that the spoligotypes with the greatest circulation among the indigenous population were LAM 9 SIT42, H1 SIT62, H3 SIT50, T1 SIT53, and H1 SIT727.
Clustering by spoligotyping and variable association analysis
The similarity dendrogram showed the presence of 30 clusters consisting of two to 37 isolates for a total of 162 isolates, while 72 isolates were not clustered (Supplementary figure 1).
Non-clustered isolates were associated with being a previously treated patient (p=0.011), relapse (p=0.039), orphan spoligotypes (p=<0.001), and belonging to the Beijing family (p=0.014) (table 5).
Table 5 Bivariate analyses of sociodemographic variables and genetic families identified by spoligotyping
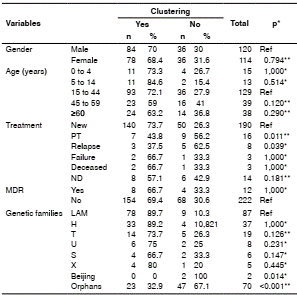
PT: Previously treated; ND: No data; Ref: Reference value
Fisher’s exact test
** Chi-squared test; confidence level: 95 %
The analysis showed an association between the T family (p=0.018) and the X family (p=0.022) for resistance to antituberculosis drugs (table 6). Conversely, an association was detected between the failure of treatment (p=0.005) and the presence of multidrug resistance. The S (p=0.010) and X families (p=0.007) were associated with multidrug resistance. No significant association was found between belonging to a particular indigenous group and drug resistance (table 7).
Table 6 Bivariate analysis of genetic families identified by spoligotyping with first-line drug resistance
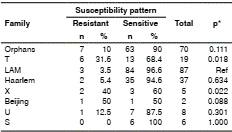
Fisher’s exact test, confidence level: 95 %
Table 7 Bivariate analyses of variables associated with multidrug resistance
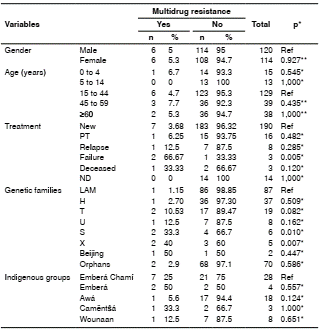
PT: Previously treated; ND: No data; Ref: Reference value
Fisher’s exact test;
** Chi-squared test, confidence level: 95 %
The association of the H family was significant for the Arhuaco (p=0.012) and Camëntŝá (p=0.033) indigenous groups. The U family was associated with the Wounaan (p=0.014) group, and the T family was associated with the Motilón Barí (p=0.002) group (data not presented).
Discussion
Indigenous peoples are among the vulnerable groups defined by the WHO and the Colombian Programa Nacional de Control de Tuberculosis and they are one of the population groups with the highest morbidity and mortality rates associated with tuberculosis 16. According to Cardona, et al.6, some of the factors associated with this vulnerability involve inherent characteristics of these populations, such as their nomadic condition, their settlement in rural areas that hinders timely access to medical services, and the low adherence to the antituberculosis treatment observed in these population groups.
The number of tuberculosis cases recorded in this study represented 5.6% of the total tuberculosis cases in the indigenous population of Colombia according to the Sivigila reports from 2009 to 2014 (n=4,205). In the country, especially in indigenous areas, the most accessible diagnostic method is bacilloscopy (89.5% of tuberculosis cases for 2015) 3 instead of culture. The clinical isolates in this study were recovered from these cultures for the characterization of drug sensitivity and molecular epidemiology analyses.
A progressive increase of more than 13.4-fold was observed in the number of cultures sent to the INS from indigenous communities between 2009 and 2014 (2009, n=7, and 2014, n=94). This increase was made possible by the implementation of memo 058 of 2009 29 and the subsequent publication of the strategic plan “Colombia Free of Tuberculosis, 2010-2015” 30, which established actions against tuberculosis among indigenous peoples. However, the country must strengthen the dissemination of public policies and ensure its compliance. In this case, all respiratory symptomatic individuals belonging to indigenous peoples should undergo more sensitive laboratory tests, such as those based on DNA amplification, culture, and drug sensitivity tests.
Regarding the presence of tuberculosis cases among indigenous groups, the highest number was registered among the Wayúu people whose groups inhabit La Guajira peninsula and share territory with Venezuela, which means there is a continuous passage between the reservations of both countries. In addition to the geographical conditions of the area, other conditions such as the lack of permanent access to drinking water supply contribute to malnutrition and, therefore, to infectious diseases 31,32.
The indigenous group with the second highest number of tuberculosis cases was the Emberá Chamí, which is primarily located in the Colombian coffee-growing region, whose beliefs about the disease have a spiritual origin and where transmission occurs by settlers. In such a context, the treatment is dictated by the shamans. Although the group also resorts to modern medicine, this approach could explain why this area is the second most affected by the disease 17.
The indigenous group with the third highest number of tuberculosis cases was the Awá. This Amerindian group living in the border between Colombia and Ecuador is one of the most vulnerable populations in Colombia 16. Their main settlements are located in the municipalities of Ricaurte, Tumaco, and Barbacoas in the department of Nariño. Little is known about their traditions because they do not allow their culture to be known. Glyphosate fumigations and invasion of their lands by illegal armed groups may have resulted in poverty, shortage of foods of animal origin, and low soil fertility, which have impacted their health conditions 31-33.
The number of tuberculosis cases was similar in women and men contrasting with global reports in which men were the most affected by the disease 2. This difference may be related to deficiencies in the treatment of index cases, late diagnosis, and social factors such as "machismo", as in these groups men decide whether women can go to health care centers when they are sick 34, as well as to women’s low education level hindering them from participating in decision-making, thus making access to diagnosis and timely care difficult 35, and child care, which also limits women’s access to healthcare services. This situation calls for reinforcing prevention, diagnosis, monitoring, and control programs in families to prevent transmission, especially from mothers affected by the disease to underage children.
Regarding age, the group most affected by tuberculosis was between 15 and 29 years. The younger and older age groups had fewer cases. This situation coincided with previous studies conducted by Culqui, et al., whose study on tuberculosis among indigenous peoples from Perú reported that 78.6% of the cases occurred in people of productive age (adolescents and adults), 2.5% in children and infants, and 7% in older adults 12. This distribution may be explained by the displacement that indigenous peoples in productive age are forced to for food or resources, which means they have contact with non- indigenous people and, therefore, with different types of diseases 36.
As regards bacilloscopy, 42.7% of the individuals had ≥++ results indicating a late diagnosis, which can reflect health care access difficulties and answer to the dispersion of communities and the lack of knowledge about the disease among other factors 31. Studies conducted by Ribeiro, et al., and Gaviria, et al.,37,38 found a relation between late tuberculosis diagnosis and disease-associated social stigma, as well as insufficient knowledge of health professionals regarding the identification of respiratory symptoms in a timely manner.
The proportion of multidrug resistant isolates cannot be compared with that found worldwide or in national annual reports. According to WHO 2, multidrug resistance is expected in 3.3% of new cases. In 2007 in Colombia, 3.2% of new tuberculosis cases in the general population were multidrug resistant tuberculosis while in tuberculosis previously treated cases it was 20%. Any number of cases is indicative of the failure of treatment, the transmisión of primary drug-resistant strains, and lack of adherence and follow-up. The presence of resistant isolates in a vulnerable population such as the indigenous populations should prompt the implementation of programmatic actions to control tuberculosis sensitivity and develop shorter, less toxic, and more effective treatment regimens to avoid second-line treatments.
Since tuberculosis control and eradication are closely linked to the knowledge about the disease, the social, cultural, economic, and biological aspects contributing to the persistence of the bacillus in the community must be elucidated. In this setting, molecular epidemiology has positioned itself as a useful tool for the study of transmission dynamics by implementing techniques such as spoligotyping 39.
In this study, the genetic families most frequently found by spoligotyping among indigenous peoples were in descendent order LAM, Haarlem, and T, a finding consistent with previous reports for the Colombian population 22-27. However, the high number of orphan spoligotypes recorded is of particular interest for public health. Another important consideration is the presence in the municipality of Buenaventura of the Beijing family due to its virulence, strong association with multidrug resistance, and extensive drug resistance 24,26,27,40-42.
Buenaventura is the most important port in Colombia and probably it was the gateway of this genotype from Asia into the country. Our results confirmed a new Beijing spoligotype (Beijing-like SIT 406) circulating in Colombia; this is the second report of this spoligotype in Latin America 43. The specific location was Tumaco.
The 14 spoligotypes we identified by spoligotyping that had not been previously reported in Colombia were the following: [LAM2 SIT 1691, T2 SIT 52, H3 SIT 418, H3 SIT 615, LAM 6 SIT 1768, LAM9 SIT 1074, LAM9 SIT 1530, S SIT 1253, S SIT 1938, T1 SIT 278, T1 SIT 628, T1 (Tuscany variant) SIT 159, U SIT 1451, and Beijing-like SIT 406], as well as 51 orphan spoligotypes without prior descriptions in the international SpolDB4 database nor in national studies 22-27. These spoligotypes could be circulating exclusively among the indigenous populations, which calls for more in-depth studies. Only three of the orphan patterns identified in the indigenous Risaralda and Putumayo groups had previously been described in Colombia circulating in the departments of Putumayo, Cauca, and in Bogotá 23-24. The presence of these spoligotypes among indigenous populations most likely responds to social factors such as the armed conflict and the search for a better quality of life, which has generated a migratory process to different places in the country 44.
According to the similarity dendrogram, 69.2% of the spoligotypes were clustered suggesting active tuberculosis transmission, especially among the Wayúu, Emberá Chamí, U’wa, and Awá groups. The use of spoligotyping as the sole genotyping method results in low discriminatory power; therefore, the frequency of transmission was most likely overestimated. However, the spoligotyping discrimination index in this study (0.963) should be considered because it indicates discrimination between unrelated isolates, which was close to 1.0, i.e., higher 28.
Although one of the limitations of this study was the low number of isolates analyzed, it is the first at the national level to characterize a large number of isolates from Colombian indigenous populations and the first approach to M. tuberculosis circulating genotypes. Our results provide partial evidence of the actual situation of tuberculosis among Colombian indigenous peoples, which can serve decision-makers in taking actions aimed at putting an end to the tuberculosis epidemic as set out in WHO global guidelines for 2035 2. However, molecular analyses should be done using MIRU-VNTR or Whole Genome Sequencing methodologies to increase the discriminatory power and provide insights into the actual situation of tuberculosis transmission among indigenous peoples in Colombia.