Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Investigación y Educación en Enfermería
Print version ISSN 0120-5307
Invest. educ. enferm vol.32 no.2 Medellín May/Aug. 2014
ARTÍCULO ORIGINAL / ORIGINAL ARTICLE/ ARTIGO ORIGINAL
Assessment of distress and quality of life of cancer patients over the course of chemotherapy
Prevalencia del distrés y de la calidad de vida de los pacientes con cáncer a lo largo de su tratamiento con quimioterapia
Avaliação do distresse e da qualidade de vida dos pacientes com câncer ao longo de seu tratamento com quimioterapia
Cristiane Decat Bergerot1; Tereza Cristina Cavalcanti Ferreira de Araujo2
1Psychologist, Ph.D. Centro de Câncer de Brasília (CETTRO), Brasilia, Brasil. email: crisdecat@yahoo.com.br.
2Psychologist, Ph.D. Professor Universidade de Brasília. Researcher, Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil. email: araujotc@unb.br.
Receipt date: Jul 22, 2013. Approval date: Feb 10, 2014.
Article linked to research: Projeto de Tese sobre Avaliação de Distress em Oncologia.
Subventions: none.
Conflicts of interest: none.
How to cite this article: Bergerot CD, Araujo TCCF. Assessment of distress and quality of life of cancer patients over the course of chemotherapy. Invest Educ Enferm. 2014; 32(2): 216-224.
ABSTRACT
Objective. Assess the prevalence of distress and quality of life of cancer patients over the course of chemotherapy. Methodology. Longitudinal prospective study addressing 200 patients. The Distress Thermometer (DT) and Functional Assessment of Chronic Illness Therapy-General (FACT-G), as indicators of distress and quality of life, were applied at three points in time during chemotherapy: the first day (T1), half way through the treatment (T2), and last day of medication (T3). Results. The average age was 56.8 years old, and 70% were women while the most frequent types of cancer included breast (30%) and hematological (22%) cancers. The number of patients with a high level of distress statistically decreased over time (T1=41.5%, T2=8.0% and T3=2.5%); consequently, quality of life scores improved (T1=85.6%, T2=90.4% and T3=92.0%). Patients with moderate to severe distress experienced worse quality of life. Distress, type of cancer and disease stage significantly impacted quality of life. Conclusion. There was a reduction in the time of impact from the side effects of chemotherapy in the patients as a consequence of adapting to the diagnosis and treatment. Continuous assessment of the needs of patients is essential to ensuring integral and humanized care, contributing to improved oncological nursing.
Key words: anxiety; quality of life; health services; oncologic nursing; neoplasms.
RESUMEN
Objetivo. Evaluar la prevalencia del distrés y de la calidad de vida de los pacientes con cáncer a lo largo de su tratamiento quimioterápico. Metodología. Estudio longitudinal prospectivo de 200 pacientes. Se aplicaron los instrumentos Termómetro de Distress (TD) y FACT-G (Functional Assessment of Chronic Illness Therapy-General) como indicador de la calidad de vida en tres etapas del tratamiento con quimioterapia: primer día (T1), mitad del tratamiento (T2) y último día de medicación (T3). Resultados. Media de edad 56.8 años; 70%, mujeres; los tipos de cáncer más frecuentes fueron los de mama (30%) y los hematológicos (22%). Mientras la proporción de pacientes con alto nivel de distrés tuvo una disminución estadísticamente significativa a lo largo del tratamiento (T1=41.5%, T2=8.0% y T3=2.5%), la calidad de vida aumentó (T1=85.6%, T2=90.4% y T3=92.0%). Los pacientes con alto distrés presentaron peor calidad de vida. Se constató también que el distrés y el tipo y estadío del cáncer interfieren significativamente en la calidad de vida. Conclusión. Hay una reducción en el tiempo del impacto de los efectos colaterales de la quimioterapia en el paciente, como consecuencia de la adaptación al diagnóstico y al tratamiento. Es indispensable que se haga evaluación continua de las necesidades del paciente con el fin de asegurar su atención integral y los cuidados humanizados, contribuyendo al mejoramiento de la enfermería oncológica.
Palabras clave: ansiedad; calidad de vida; servicios de salud; Enfermería Oncológica; neoplasias.
RESUMO
Objetivo. Avaliar a prevalência de distresse e a qualidade de vida dos pacientes com câncer ao longo de seu tratamento quimioterápico. Metodologia. Estudo longitudinal prospectivo de 200 pacientes. Aplicaram-se os instrumentos Termômetro de distresse (TD) e FACT-G (Functional Assessment of Chronic Illness Therapy-Geral) como indicador da qualidade de vida em três etapas do tratamento com quimioterapia: primeiro dia (T1), metade do tratamento (T2) e último dia de medicação (T3). Resultados. Média de idade 56.8 anos, 70% foram mulheres e os tipos de câncer mais frequentes foram os de mama (30%) e os hematológicos (22%). Enquanto a proporção de pacientes com alto nível de distresse teve uma diminuição estatisticamente significativamente ao longo do tempo de duração do tratamento (T1=41.5%, T2=8.0% e T3=2.5%), a qualidade de vida aumentou (T1=85.6%, T2=90.4% e T3=92.0%). Os pacientes com alto distresse apresentaram pior qualidade de vida. Constatou-se também do que o distresse e o tipo e estado do câncer interferem significativamente na qualidade de vida. Conclusão. Há uma redução no tempo do impacto dos efeitos colaterais da quimioterapia no paciente, como consequência da adaptação ao diagnóstico e ao tratamento. É indispensável que se faça avaliação contínua das necessidades do paciente com o fim de assegurar seu atendimento integral e os cuidados humanizados, contribuindo ao melhoramento da enfermagem oncológica.
Palavras chaves: ansiedade; qualidade de vida; serviços de saúde; enfermagem oncológica; neoplasias.
INTRODUCTION
The oncological literature acknowledges that many important changes occur both in the patient's physical and psychosocial states with the event of a cancer diagnosis and during the treatment.1-3 Considering the specificity of experiences that take place in such circumstances, the National Comprehensive Cancer Network (NCCN), in 1999, proposed the use of the term distress to describe "a multifactorial unpleasant emotional experience of a psychological (cognitive, behavioral, emotional), social, and/or spiritual nature that may interfere with the ability to cope effectively with cancer, its physical symptoms and its treatment."4 It is noteworthy that distress does not merely imply intense suffering5 - conceived as a natural response among those facing a severe disease4 - rather it comprises more severe clinical conditions that may lead to a psychiatric disorder.6 It is then recommended that cancer patients be systematically assessed to prevent harm and promote well-being.4
In Canada, since 2006, distress has been included as the sixth vital sign to monitor cancer patients.7 According to Ashbury, Findlay, Reynolds and McKerracher, patients not treated for symptom-related distress used five times more frequently the healthcare units , twice as frequently for the emergence department, and were more frequently prescribed third- and fourth-line chemotherapy.8 Numerous studies addressing quality of life in the field of oncology have been conducted in the last six decades.9-11 More specifically in regard to distress, some studies have already verified that quality of life is affected by its symptoms.6,12-14 Lung cancer is more frequently associated with moderate to severe distress and low quality of life.15 In regard to breast cancer, fluctuation was observed in different phases of treatment, while quality of life was perceived as more positive before chemotherapy and distress reduced twelve months after treatment; no differences were found in terms of age and type of chemotherapy protocol.16 Zabora et al. compared 14 types of cancer, among 4,496 patients, and obtained the following classification of decreasing levels of distress: lung, brain, Hodgkin's lymphoma, pancreas, lymphoma, liver, head and neck, adenocarcinoma, breast, leukemia, melanoma, colon, prostate and gynecological cancer.17
In short, evidence has been gathered from more than one decade that reaffirms the need to broaden knowledge concerning with distress and quality of life in order to guide care practices.6,9-11,18 Systematic assessment in oncological nursing is required to ensure the delivery of integral and humanized care in all phases of the cancer experience.16,18-20 That is, accurate understanding of patients enables nursing professionals to better assist patients to cope with the disease, adhere to treatment, and also optimizes human and material resources availables.9-16 For Costa-Requena, Rodríguez and Fernández-Ortega, the assessment of distress performed by a properly trained nurse during the initial phases of monitoring contributes to offer a high quality of cancer care.16
Thus, given the relevance of this subject, an investigation was performed with the general objective to assess distress and quality of life among cancer patients over the course of chemotherapy.
METHODOLOGY
This descriptive and qualitative study was authorized by the Institutional Review Board of the College of Health, University of Brasilia. All the patients agreed to participate in the study by signing free and informed consent forms. The sample was composed of 200 patients who began their treatments in the Brasilia Cancer Center (CETTRO), a private and specialized service located in the Federal District, Brazil. The following instruments were applied: a) socio-demographic questionnaire specifically developed to obtain data characterizing the study's sample, composed of five items (age, gender, marital status, education, type of cancer, and disease stage); b) the Distress Thermometer (DT), proposed by the NCCN,4 adapted and validated to Portuguese21, assess the level of distress in a scale ranging from 0 (no distress) to 10 (extreme distress); and c) Functional Assessment of Chronic Illness Therapy-General (FACT-G), measure quality of life scores, indicated for adult patients with cancer. It was translated to and validated for Portuguese22 and contains 27 items assessing four domains of 'well-being' (physical, social/family, emotional and functional) on a four-point Likert scale.
The procedures for data collection were performed at three different points of the chemotherapy: the first day (T1); half way through the treatment (T2); and at the last day of chemotherapy infusion (T3). After each assessment, the results were discussed between the health team in order to identify emergency needs and specific demands that would require adjustments in the planned therapeutic regimen. Data were submitted to descriptive statistical analysis to characterize the sample in regard to socio-demographic and clinical variables. The Chi-square test was used to verify associations between distress and quality of life and also to determine the likelihood of occurrence and level of significance. ANOVA Repetitive Measures (ANOVA-RM) was performed among the independent variables (distress and quality of life) and socio-demographic data (age, gender, marital status, education, type of cancer and disease stage) to verify significant relationships among the variables.
RESULTS
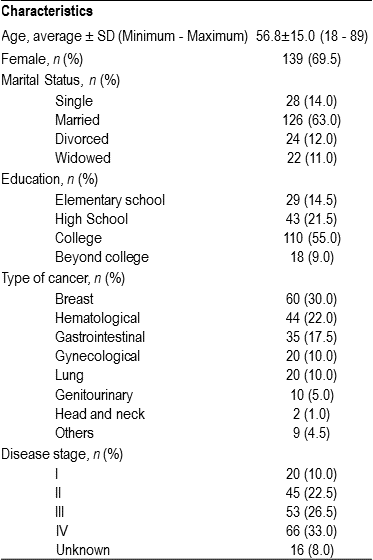
The interval between T1, T2 and T3 was of five months on average. There was a sample loss of 6.5% at T2 and of 3.8% at T3 due to death or change of treatment facilities. Table 1 presents the main socio-demographic characteristics of the patients in this study: an average age of 56.8 years old, most patients were women (69.5%), married (63%), and had college degrees (55%). The following cancer types stood out from the clinical point of view: breast, hematological and gastrointestinal cancers while 59.5% of the participants had a late disease stage (III and IV).
Table 1.Socio-demographic profile of the participants (n = 200)
In regard to the incidence of distress, 41.5% of the participants reported moderate to severe distress at T1 (DT ≥ 4). Marked and gradual decreases were observed in the subsequent phases: T2=9.1% and T3=4.4%. Table 2 shows the incidence of distress over the course of assessments and also shows increase or decrease in the level of distress at T1, T2 and T3. Five patients did not experience improvement in their symptoms despite clinical follow-up performed by the health team.
Table 2.Incidence of distress over the course of the treatment
When analyzing the relationship between the average level of distress, type of cancer and disease stage we verified that at T1, the patients with late disease stage (III and IV) experienced a higher average level of distress compared to those with a type of cancer with an early disease stage (I and II). It is important to note that patients with lung cancer presented a higher average level of distress when compared with the remaining participants. Additionally, patients with lung cancer with and early disease stage obtained an average level of distress higher than those with a late disease stage. At T2, patients with lung cancer continued to have higher averages, followed by patients with gynecological cancer. In regard to the disease stage, patients with lung cancer maintained the tendency indicated in the previous assessment phase (T1). Contrary to the result observed at T1, cases of gynecological, breast and hematological cancers with an early disease stage were associated at T2 with a higher average compared to similar cases with a late disease stage.
In the last assessment (T3), patients with head and neck cancer, followed by patients with lung cancer, presented an average level of distress higher than those with other types of cancer. In general, patients with an early disease stage reported a higher average in this assessment phase. It is worth mentioning that the average level of distress was higher among patients in a late disease stage of breast and gynecological cancers.
There was a slight increase in the average of quality of life over the course of the assessments: T1=85.6; T2=90.4; T3=92. When the domains "emotional well-being" and "functional well-being" were assessed by FACT-G, a more evident increase was observed, as shown in Table 3.
Table 3.Descriptive values concerning the subscales of quality of life
The correlation between DT and FACT-G was inversely proportional over time (p<0.001); that is, the higher was the level of distress, the worse was the perceived quality of life scores (see Table 4). The descriptive analysis of the data generated by FACT-G, based on the distress incidence configuration proposed in Table 2 shows that the average quality of life of the participants with moderate to severe distress was lower than for those with mild distress. For those experiencing mild distress at T1, the average was even higher when compared to those who had their level of distress reduced in the subsequent assessment, as shown in Table 5.
Table 4.Correlations among quality of life (FACT-G + subscales) and level of distress (DT) over the course of the assessments
* All the probability values were below 0.05.
This correlation was statistically significant at T1, T2 and T3 for all the domains assessed by FACT-G with the exception of data found at T1 in the subscale 'social and family well-being'. For the remaining subscales at T1 and for the consecutive assessments, the correlations were significant (p<0.05): FACT-G (T1: χ2=93.36, T2: χ2=311.58 and T3: χ2=278.39, 'physical well-being' (T1: χ2=45.02, T2: χ2 =95.04, and T3: χ2=152.76), 'social and family well-being' (T1: χ2=13.37, T2: χ2= 47.54 and T3: χ2=106.88, 'emotional well-being' (T1: χ2=82.04, T2: χ2=180.60, and T3: χ2=282.35) and 'functional well-being' (T1: χ2=84.87, T2: χ2=256.73 and T3: χ2=205.83).
Table 5.Average qualify of life based on the configuration of incidences of distress
Note. *MSD: Moderate to Severe Distress (DT ≥ 4) or RMSD: Remained Moderate to Severe Distress (DT ≥ 4)
†MD: Mild Distress (DT ≤ 3) or RMD: Remained Mild Distress (DT ≤ 3)
‡RD: Reduced Distress (DT≤ 3)
§ID: Increased Distress (DT ≥ 4)
The patients with gastrointestinal and genitourinary cancer in an early disease stage (I and II) at T1 showed a higher score of quality of life. Additionally, patients with the disease in an early disease stage reported higher scores of quality of life when compared to those whose disease was in a late disease stage. At T2, the averages of quality of life for early and late breast cancer stage were comparable. The average was slightly higher for patients with gastrointestinal cancer and late disease stage (III and IV) as well for those with hematological and gynecological cancers. Patients with genitourinary or lung cancers in an early disease stage reported a higher quality of life scores compared to those with the same diagnosis in a late disease stage. At T3, patients with breast, hematological or gynecological cancers in a late disease stage obtained an average higher than patients in an early disease stage.
The scores for quality of life were higher among patients who reported mild distress at T1. Additionally, the diagnoses more frequently related to a higher quality of life scores were breast and gynecological cancers. At T2 and T3 the relationship between mild distress and higher quality of life score was maintained. The average quality of life for patients whose level of distress was reduced in these phases of assessment was also higher than for those whose level of distress was maintained or increased.
ANOVA-MR showed a violation of the assumption of Mauchly's Sphericity test in the analysis performed for distress and socio-demographic variables (age, gender, marital status, education, type of cancer and disease stage). The Greenhouse-Geisser test was significant only for distress reduction over time (p<0.001). The analysis of quality of life (FACT-G and subscales) and the sample's socio-demographic characteristics indicated significant interaction in the Greenhouse-Geisser test for: FACT-G and education (p<0.05); FACT-G and type of cancer (p<0.05); 'physical well-being' and type of cancer (p<0.05); 'physical well-being' and disease stage (p<0.05); 'emotional well-being' and education (p<0.01); 'emotional well-being' and type of cancer (p<0.01); 'functional well-being' and marital status (p<0.05); 'functional well-being' and education (p<0.05). Interaction was significant for increased of quality of life over time: FACT-G (p<0.001), 'physical well-being' (p<0.05), 'emotional well-being' and 'functional well-being" (p<0.001).
DISCUSSION
The high incidence of distress at T1 was compatible with data reported in the literature,2-3,23 as well as the inversely proportional relationship between distress and quality of life over the three assessments.12-14 The statistical analyses showed that patients with moderate to severe distress perceived their quality of life to be more negative when compared with those who reported mild distress.
Lung cancer was more frequently associated with moderate to severe distress and low quality of life. These results corroborate a previous study conducted by Anant et al.15 When considering only the variable moderate to severe distress, we observed that at T2 and T3 were those patients diagnosed with gynecological, breast and hematological cancers, with a late disease stage. This finding partially converges with the study conducted by Zabora et al.,17 since lung cancer presented a higher incidence of moderate to severe distress while gynecological cancer the lower incidence. In the same study, hematological cancer occupied the third position, as it is also classified in this study. It was not possible to verify the relationship between distress, type of cancer and disease stage, which may reflect the limited access to information from their illness on the part of Brazilian patients as noted by Faria and Souhami.24
As already described in the literature, patients with lung and head and neck cancers in a late disease stage, reported low quality of life in all assessments.15 When performing ANOVA-MR, we observed that type of cancer and disease stage significantly impact the 'physical well-being', 'emotional well-being' and 'functional well-being' domains. The variable education was positively correlated with quality of life. Significant interference of marital status was observed on 'emotional well-being' and 'functional well-being' subscales. The longitudinal analysis showed that the impact experienced by patients was reduced as well as the coping with the chemotherapy side effects and psychosocial limitations imposed by the experience of cancer experience were improved. The marked and progressive reduction of distress level and increased quality of life scores can been seen as a result of the successful adaptation to the diagnosis and treatment.16 It is worth noting that, according to Akechi et al.25, if distress is not treated, the symptoms may intensify up to six months after the diagnosis, interfering with treatment adherence and consequently, with disease remission phase.
Additionally, the assessment and discussion routine enabled better identification of the difficulties faced by patients and implementation of preventive measures. The well-structured and consistent instruments encouraged communication among different professionals, who began employing similar parameters in their practice. Primarily, the optimization of cancer care contributed for improvement on Oncological Nursing practice. This study, however, presents limitations such as the sample size and its epidemiological representativeness; the fact that this research was conducted in a single oncology service; and the lack of follow-up in the survival phase, after T3. Therefore, we suggest multicenter studies and evaluation of the impact of different chemotherapy and treatments modalities (radiotherapy, surgery and immunotherapy). It is also appropriate in the future to undertake a detailed analysis between those different types of cancer. In short, the evidence obtained in this study reinforces the importance of continuous planning and assessment of care routines to ensure the delivery of integral and humanized care. We recommend systematic measures employing the instruments used in this study in order to increase the effectiveness and efficiency of oncology services.
REFERENCES
1. Loscalzo M, Clark KL, Holland J. Successful strategies for implementing biopsychosocial screening. Psycho-Oncol. 2011; 20(5):455-62. [ Links ]
2. Fallowfield L, Ratcliffe D, Jenkins V, Saul J. Psychiatric morbidity and its recognition by doctors in patients with cancer. Br J Cancer. 2011; 84(8)1011-15. [ Links ]
3. Jacobsen PB, Wagner LI. A new quality standard: the integration of psychosocial care into routine cancer care. J Clin Oncol. 2012; 30:1154-59. [ Links ]
4. National Comprehensive Cancer Network. Distress management. Clinical practice guidelines. J Natl Compr Canc Netw. 2003; 1(3):344-74. [ Links ]
5. Block SD. Assessing and managing depression in the terminally ill patient. ACP-ASIM End-of-life care consensus panel. American College of Physicians - American Society of Internal Medicine. Ann Intern Med. 2000; 132(3):209-18. [ Links ]
6. Grassi L, Indelli M, Marzola M, Maestri A, Santini A, Piva E, et al. Depressive symptoms and quality of life in home-care-assisted cancer patients. J Pain Symptom Manage. 1996; 12(5):300-7. [ Links ]
7. Bultz BD, Groff SL, Fitch M, Blais MC, Howes J, Levy K, et al. Implementing screening for distress, the 6th vital sign: a Canadian strategy for changing practice. Psycho-Oncol. 2011; 20(5):463-9. [ Links ]
8. Ashbury FD, Findlay H, Reynolds B, McKerracher K. A Canadian survey of cancer patients' experiences: are their needs being met? J Pain Symptom Manage. 1998; 16:298-306. [ Links ]
9. Gotay CC. Quality-of-life assessment in cancer. In: Miller SM, Bowen DJ, Croyle RT, Rowland JH (Eds.). Handbook of cancer control and behavioral science: a resource for researchers, practitioners, and policymaker. Washington, DC: American Psychological Association; 2009. p.115-28. [ Links ]
10. Faller H, Schuler M, Richard M, Heckl U, Weis J, Küffner R. Effects of psycho-oncologic intervention on emotional distress and quality of life in adult patients with cancer: systematic review and meta-analysis. J Clin Oncol. 2013; 31(6):782-93. [ Links ]
11. Costa Neto SB, Araujo TCCF. Qualidade de vida do enfermo oncológico: um panorama sobre o campo e suas formas de avaliação. In Carvalho VA, Franco MHP, Kovács MJ, Liberato RP, Macieira RC, Veit MT, et al (Eds.). Temas em Psico-Oncologia. São Paulo, SP: Summus; 2008. p.195-208. [ Links ]
12. Bultz BD, Johansen C. Screening for distress, the 6th vital sign: where are we, and where are we going? Psycho-Oncol. 2011; 20(6):569-71. [ Links ]
13. Andritsch E, Dietmaier G, Hofmann G, Zloklikovits S, Samonigg H. Global quality of life and its potential predictors in breast cancer patients: and exploratory study. Support Care Cancer. 2007; 15:21-30. [ Links ]
14. Dapueto JJ, Servente L, Francolino C, Hahn EA. Determinants of quality of life in patients with cancer. Cancer. 2005; 103:1072-81. [ Links ]
15. Anant M, Randeep G, Ashutosh PJ, Manisha B, Hemraj P, Charu M, et al. Quality of life measures in lung cancer. Indian J Cancer. 2005; 42(3):125-32. [ Links ]
16. Costa-Requena G, Rodríguez A, Fernández-Ortega P. Longitudinal assessment of distress and quality of life in the early stages of breast cancer treatment. Scand J Caring Sci. 2013; 27:77-83. [ Links ]
17. Zabora J, Brintzenhofeszoc K, Curbow B, Hooker C, Piantadosi S. The prevalence of psychological distress by cancer site. Psycho-Oncol. 2001; 10:19-28. [ Links ]
18. Costa Neto SB, Araujo TCCF. Calidad de vida de los portadores de neoplasia de cabeza y de cuello en fase diagnóstica. Rev Colomb Psicol. 2005; 14:53-63. [ Links ]
19. Manrique FMC, Pedraza HMP. Prevalence of nursing diagnosis in people with oncology treatment. Invest Educ Enferm. 2008; 26(2):212-7. [ Links ]
20. Porto AR, Thofehrn MB, Amestoy SC, Gonzáles RIC, Oliveira NA. The essence of interdisciplinary practice in palliative care delivery to cancer patients. Invest Educ Enferm. 2012; 30(2):231-9. [ Links ]
21. Decat CS, Laros JA, Araujo TCCF. Termômetro de distress: validação de um instrumento breve para avaliação diagnóstica de pacientes oncológicos. Psico-USF. 2009; 14(3):253-60. [ Links ]
22. Arnold BJ, Eremenco E, Chang CH, Odom L, Ribaudo JM, Cella DF. Development of a single portuguese language version of the functional assessment of cancer therapy general (FACIT-G) scale. Qual Life Res. 2000; 9(3):316. [ Links ]
23. Carlson LE, Waller A, Mitchell AJ. Screening for distress an unmet needs in patients with cancer: review and recommendations. J Clin Oncol. 2012; 30:1160-77. [ Links ]
24. Faria SL, Souhami L. Communication with the cancer patient. Information and truth in Brazil. Ann N Y Acad Sci. 1997; 809(1):163-71. [ Links ]
25. Akechi T, Okuyama T, Akizuki N, Azuma H, Sagawa R, Furukawa TA, et al. Course of psychological distress and its predictors in advanced non-small cell lung cancer patients. Psycho-Oncol. 2006; 15:463-73. [ Links ]











 text in
text in