Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Acta Biológica Colombiana
Print version ISSN 0120-548X
Acta biol.Colomb. vol.14 no.3 Bogotá Sep./Dec. 2009
FIRST HOST RECORD FOR THE ROOT PARASITE Corynaea crassa (BALANOPHORACEAE)
Primer registro de la hemiparásita de raíz Corynaea crassa (Balanophoraceae)
J. TUPAC OTERO1, 2, MARCELA MORA3, JUAN F. COSTA4
1Departamento de Ciencias Biológicas, Universidad Nacional de Colombia, Palmira, Valle, Colombia.
2Instituto de Estudios Ambientales IDEA, Capítulo Palmira. Universidad Nacional de Colombia. Palmira, Valle, Colombia.
3Department of Biological Sciences, University of Alabama, Tuscaloosa, Alabama 35487. USA.
4Universidad Nacional de San Antonio Abad del Cusco. Cusco-Perú. Dirección para correspondencia: J. Tupac Otero, Departamento de Ciencias Biológicas, Universidad Nacional de Colombia, Palmira, Valle, Colombia. jtoteroo@unal.edu.co
Received 27 March 2009, Revised 2 July 2009 - Acepted 17 September 2009.
ABSTRACT
Corynaea crassa is a root hemiparasitc plant infrequent in nature. There is little information regarding their natural history, in particular their host range. In this study we dig 32 tubers and followed their parasitized roots to determine the species host. We found Bocconia frutescens (Papaveraceae), Verbesina sp. (Asteraceae), Cayaponia sp. (Cucurbitaceae) y Palicourea sp.(Rubiaceae) as natural hosts of C. crassa. This is the first host record available for C. crassa and provides some bases for its conservation in natural habitats.
Key words: Balanophoraceae, Corynaea crassa, Costa Rica, host plant, root parasitism.
RESUMEN
Corynaea crassa es una planta hemiparásita de raíz poco común y de la cual sabemos muy poco acerca de su historia natural y en particular sobre su rango de hospederos. En este estudio excavamos 32 tubérculos de dicha especie y seguimos las raíces que estaban parasitando para determinar su identidad. Encontramos Bocconia frutescens (Papaveraceae), Verbesina sp. (Asteraceae), Cayaponia sp. (Cucurbitaceae) y Palicourea sp. (Rubiaceae). Este es el primer registro de hospederos disponible para la especies y este tipo de información es de gran utilidad para las conservación de C. crassa en hábitats naturales.
Palabras clave: Balanophoraceae, Corynaea crassa, Costa Rica Planta hospedera, parasitismo radical.
INTRODUCTION
The Balanophoraceae is a nonphotosynthetic holoparasite flowering plant family distributed primarily throughout the tropics. They parasite roots of other plants and most of their life cycle occurs underground. The vegetative body of the Balanophoraceae consists of an irregular tissue known as haustorial tuber from which inflorescences develop (Hsiao et ál., 1993).
Although 15 species in the family are found in America (Gómez, 1983), only one Amazonian species has been studied from the ecological point of view (Borchsenius and Olesen, 1990). Here we report observations on the parasitism Corynaea crassa Hook. f., a Neotropical species distributed from Costa Rica to all the Andean countries except Chile (W3 TROPICOS, 2007). Although several collections of this species exist, little is known about their host ranges and levels of specificity and there is no information about its conservation status. In order to provide key factors determining their distribution to search for new populations it is important to know their potential host species. In this study we ask the following questions: (1) which species are the most frequent hosts of C. crassa? and (2) Does it exist a relation between the size of the host’s root and the size of the root parasite?
MATERIAL AND METHODS
Corynaea crassa and the other members in the subfamily Helosidoideae, are characterized by their inflorescence in spadix which developed from subterranean globose to elongated tubers, large umbrella-like bracts enclosing the young spadix forming a tectum, a dense layer of nectar-hairs below the tectum, and male and/or female flowers scattered among the nectar-hairs in more or less distinct patterns (reviewed by Eberwein and Weber, 2004). Their inflorescences have been reporter to be visited by Hymenoptera and Diptera (Gómez, 1983) but no study of the pollination biology of the species has ever been published.
We carried out this study at Cuericí Biological Station (CBS) in Cordillera de Talamanca, Costa Rica. The CBS is located in San Jose Province, 5 km northeast of Pan-American Highway (9°33’30”N, 83°39’42”W) at 2400-2900 m altitude and occurs in Montane Wet Forest life zone (Moran, 2003). The sampling area was centered in a patch of secondary forest with organic-mater rich soils. For each individual of C. crassa the haustorial tuber diameter at the base of the tuber and the number of inflorescences were recorded. To identify the host plant, we excavated the tubers and followed the host plant root until we found its stem. The diameter of the host root where the haustorial tuber of the parasite is attached was measured.
RESULTS
We found 32 tubers of C. crassa parasitizing four host species: Bocconia frutescens (Papaveraceae), Verbesina sp. (Asteraceae), Cayaponia sp. (Cucurbitaceae) and Palicourea sp. (Rubiaceae) (Table 1). Although we found six additional tubers, we excluded them from the analysis since we could not follow the host roots they were parasitizing in order to identify the host species. Most C. crassatubers were parasitizing roots of Bocconiafrutescens(Papaveraceae; Table 1). All parasitized roots of different host were concentratedon the top few centimeters of the soil (no more than 30 cm deep).

There was no relationship between the diameter of the host root and the size of the tuber(r2= 0.06, p = 0.12). However, when we consider only the tubers parasitizing B. frutescensthe correlation was significant (r2= 0.28, p< 0.005, Fig.1). We also found a positivesignificant correlation between C. crassatuber size and number of inflorescences (r2= 0.46,p< 0.0005, Fig. 2).
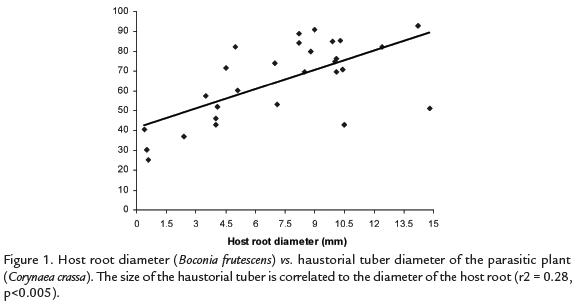
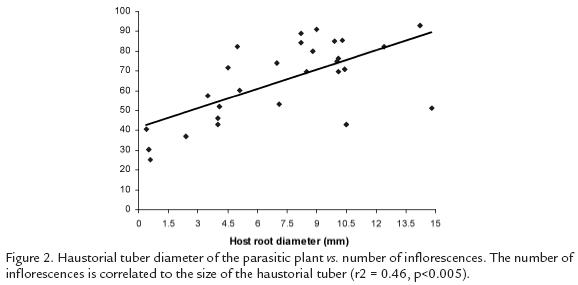
DISCUSSION
Many aspects of the biology of C. crassaare poorly known, due largely to their undergroundlife habit and the seasonal appearance of the inflorescence. The lack of knowledge, the entirelydependence of this species on other plants for its survival, as well as the conversion of manyareas of cloud forest in to agriculture and pastures make the species vulnerable. Parasiticplants may also be limited by the additional suite of factors of host availability, host quality,host resistance to parasitism, and parasite preference (Marvier and Smith, 1996).
In this study we found out that C. crassais parasitizing different host species, suggestingthat this host parasite interaction could be shaping the local plant community. Thiscould be true for other neotropical cloud forest where this parasite system occurs. Nevertheless, the fact that they were parasitizing other hosts from different families suggests not strict host specificity in C. crassa (Fig. 3). These results concur with Werth et ál., 1979, who reported that most plant parasites are not specific to a single host. Similarly, Thesium chinensis (Santalaceae) parasitized 38 plant species in Japan (Suetsugu et ál., 2008). The reduction in host specificity in tropical compared to temperate areas is considered a consequence of the lower relative abundance of individual host species (Hawkins et ál., 1992; Norton and De Lange, 1999). The relative broad host range in C. crassa at CBS can be advantageous as it allows the parasites to grow successfully in different potential hosts encountered. This suggests that plants with superficial or shallow roots will be more susceptible of being parasitized. On the other hand, the factors associated to a higher frequency of parasitism in Bocconia frutescens are not known yet. The fact that B. frutescence had long roots (up to 7 m) could increase the probability of been parasitized. Another possible explanation is related to the high abundance of B. frutescence at the study site. The relative abundance of host species is a key factor determining the degree of host specificity in a parasite (Norton and Carpenter, 1998). If further studies prove that C. crassa shows preferences for certain host species, (e.g. Bocconia frutescens) then conservation efforts should be also addressed to the management of selected host populations.
A better understanding of this plant-plant interaction and the ecological role thatC. crassa is playing in the ecosystem is necessary in order to have stronger bases for their successful conservation and restoration. Assuming that bigger tubers are older than smaller ones, those results suggest that C. crassa starts its life cycle parasitizing small diameter roots getting thicker as they age together with the host root. We also founded a positive significant correlation between C. crassa tuber size and number of inflorescences (r2 = 0.46, p< 0.0005, Fig. 2) suggesting that bigger plants can invest more energy in reproduction and presumably also present higher fitness. Since the haustorial tuber diameter is correlated to the host root diameter and the number of inflorescences is determined by the size of the tuber, we could infer that the host diameter is also a good predictor of the number of inflorescences. More studies with larger sample size are required taking into account the frequency of all potential host plants in a given area to determine if high frequency of parasitism reflects indeed host preference in C. crassa.
ACKNOWLEDGEMENTS
We acknowledge the staff at Cuericí, Biological Station for their suport. A. Fargi commented the manuscript and made useful suggestion to the sampling procedure. DIPAL, and El Grupo de Investigación en Orquídeas from Universidad Nacional de Colombia, Sede Palmira supported the participation of JT Otero.
REFERENCES
BORCHSENIUS F, OLESEN JM. The Amazonian root holoparasite Lophophytum mirabile (Balanophoraceae) and its pollinators and herbivores. J Trop Ecol. 1990;6:501-505.
[ Links ]EBERWEIN RK, WEBER A. Exorhopala ruficeps (Balanophoraceae): morphology and transfer to Helosis. Botanical Journal of the Linnean Society, 2004;146:513-517.
[ Links ]GÓMEZ LD. Parasitic plants. En D. H. Janzen [ed.], Costa Rican Natural History. University of Chicago Press, Chicago, Illinois, USA. 1983. p. 292-298.
[ Links ]HAWKINS BA, SHAW MR, ASKEW RR. Relations among assemblage size, host specialization, and climatic variability in North American parasitoid communities. Am Nat. 1992;139:58-79.
[ Links ]HSIAO SC, MAUSETH JD, GÓMEZ LD. Growth and anatomy of the vegetative body of the parasitic angiosperm Heliosis cayennensis (Balanophoraceae). Bulletin of the Torrey Botanical Club. 1993;120:295-309.
[ Links ]MARVIER MA, SMITH DL. Conservation implications of host use rare parasitic plants. Conserv Biol. 1996;11: 839-848.
[ Links ]MORAN R. Low-Trunk Epiphytic Ferns on Tree Ferns Versus Angiosperms in Costa Rica. Biotropica. 2003;35:48-56.
[ Links ]NORTON DA, CARPENTER MA. Mistletoes as parasites; host specificity and speciation. Trends Ecol Evol. 1998;13:101-105.
[ Links ]NORTON DA, DE LANGE PJ. Host specificity in parasitic mistletoes (Loranthaceae) in New Zealand. Funct Ecol. 1999;13:552-559.
[ Links ]SUETSUGU K, KAWAKITAAND A, KATO M. Host range and selectivity of the hemiparasitic plant Thesium chinense (Santalaceae). Ann Bot. 2008;102:49-55.
[ Links ]W3 TROPICOS/ MISSOURI BOTANICAL GARDEN. Nomenclatural Database. 2007. Available for Internet
[ Links ]WERTH CR, VAIRD WV, MUSSELMAN LJ. Root parasitism in Schoepfia Schreb.(Olacaeeae). Biotropica. 1979;11:140-143.
[ Links ]













