Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Acta Biológica Colombiana
Print version ISSN 0120-548X
Acta biol.Colomb. vol.17 no.1 Bogotá Jan./Apr. 2012
PRESENCE OF ASPERGILLUS AND OTHER FUNGAL SYMBIONTS IN COFFEE BEANS FROM COLOMBIA
Presencia de Aspergillus y otros simbiontes fúngicos en granos de café procedentes de Colombia
MIGUEL ÁNGEL GAMBOA-GAITÁN1, M.Sc.
1 Departamento de Biología. Universidad Nacional de Colombia. Carrera 30 # 45-03. Bogotá, D.C., Colombia. magamboaga@unal.edu.co
Presentado 5 de julio de 2010, aceptado 2 de junio de 2011, correcciones 15 de febrero de 2012.
ABSTRACT
Fungi are common inhabitants of plants and plant-derived products. Some of these fungal species are potentially dangerous to human health since they are able to produce chemical substances that alter normal physiological activity. There are no studies about natural mycoflora associated with coffee beans in Colombia, and nothing is known about the presence and abundance of toxigenic fungal species in Colombian coffee. In this study 5,000 coffee beans were studied by plating them on potato-based artificial culture medium and it was shown that potentially toxigenic fungal taxa (mostly from genera Aspergillus, Fusarium, Penicillium), are currently found in Colombian coffee beans. This is true for all steps of coffee processing, from berries in trees to toasted grains, including packed coffee ready for retail in supermarkets. Results show that the distribution of these fungi is not random among different steps of coffee processing, which means that some steps are more vulnerable to infection with some fungi that others. The convenience of establishing a program devoted to detect fungi and/or mycotoxins in Colombian commodities, specially coffee, is discussed here.
Key words: Commodities, food microbiology, coffee, mycotoxins.
RESUMEN
Los hongos son comúnmente encontrados tanto en plantas como en sus productos, bien sea para uso humano o animal. Algunos de tales hongos son potencialmente peligrosos para la salud porque producen compuestos químicos fisiológicamente activos, como alcaloides y toxinas. En Colombia no hay estudios sobre la micoflora naturalmente asociada a granos de café, ni sobre la presencia de especies toxígenas en dicho producto. En este estudio se tomaron muestras de 5.000 granos de café en diferentes estadios de su procesamiento, encontrando que taxones fúngicos potencialmente toxígenos, tales como Aspergillus, Fusarium y Penicillium, son comúnmente encontrados en todos los estadios del procesamiento de café. El estudio incluyó muestras desde el fruto en el árbol hasta café tostado y empacado listo para la venta en supermercados. Los resultados muestran que la distribución de estos hongos no es al azar entre los diferentes pasos en el procesamiento del café, lo que significa que algunos pasos son más vulnerables a la infección con hongos que otros. Se discute la conveniencia de establecer un programa dedicado a la detección de hongos o micotoxinas en Colombia y las implicaciones de esto en la salud pública.
Palabras clave: café, micotoxinas, salud pública, microbiología de alimentos.
INTRODUCTION
The presence of fungi on and within plant tissues has been well documented since many years ago. Those living on plant surfaces are referred to as epiphytic fungi, while the ones growing within tissues are named endophytic fungi. This term was first used by de Bary (1866, reviewed by Petrini, 1991), being intended to cover all fungi found within plants, including plant pathogens. Today, nevertheless, 'endophyte' is applied only to those microorganisms living in the interior of plants that produce no evidence of disease (Carroll, 1988; Petrini, 1991; Gamboa and Bayman, 2002; Backman and Sikora, 2008). Since fungi are in healthy plants, mycologists wondered if they were also able to grow in commodities derived from those plants. Research showed that, in fact, foods and feeds are commonly infected with both endoand epiphytic fungi (e.g. King-Jr. et al., 1979; Batista et al., 2003).
Many fungi produce dangerous chemical compounds named mycotoxins (Bennett and Klich, 2003; Zain, 2011). Mycotoxins were noticed after a sad episode in which 100,000 turkey poults were killed by contamination with Aspergillus flavus derived toxins (aflatoxins; Blout, 1961). Since then, the study of toxigenic species has tried to avoid contamination of commodities with those compounds. Plant products in which toxigenic species and mycotoxins are found include cereals, pulses, maize, nuts and coffee (Bayman et al., 2002; Batista et al., 2003; Baydar et al., 2005; Magnoli et al., 2006; Zain, 2011).
Given that coffee is a common beverage worldwide, various studies have been developed in order to investigate the presence of fungal contaminants and the potential importance of mycotoxins in coffee beans (Levi et al., 1974; Taniwaki et al., 1999; Romani et al., 2000; Urbano et al., 2001; Taniwaki, 2005; Vega et al., 2006). Among mycotoxins, ochratoxins are one of the most important groups affecting coffee as shown by some studies (Levi et al., 1974; Romani et al., 2000; Urbano et al. 2001) and aspergilli are definitively involved in contamination with ochratoxins (see review by Bayman and Baker, 2006). Unfortunately, no work in this regard has been performed in Colombia although coffee is an important component of the economy of this country. Understanding the nature of fungal communities associated with coffee beans will help in establishing proper policies for improving harvesting, storage, and handling of this commodity.
This study was directed to know if species of Aspergillus and other potentially toxigenic fungi are associated to fruits and seeds of coffee, covering three points of coffee manufacture: harvest, processing and storage. Since coffee beans are highly manipulated from harvest to retail due to pulp scalping, fermentation, sun drying, toasting, etc. (Rincón-Sepúlveda, 1999), associated microbial assemblages will be constantly changing and a dynamic succession of microbe communities is expected (Silva et al., 2008). In this situation, composition of fungal communities associated with coffee beans will be dependent of environment in which seeds are processed and stored, and if no specificity (fidelity) is found between coffee beans and fungi, their presence and distribution among different steps of coffee processing will be explained only by chance.
MATERIALS AND METHODS
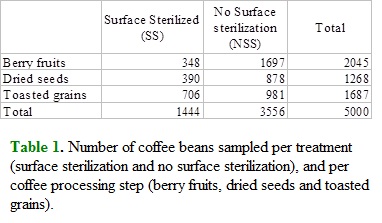
Coffee beans from seven Colombian coffee plantations (La Vega, La Mesa, Bitaco, Pico de Águila, San Antonio del Tequendama, Villeta and Guayabal de Síquima) were screened for aspergilli and other fungi associated with external surface and internal tissues. Bean samples were collected from June 2005 to November 2007 and grouped into the following categories: 1) berries on plants, 2) dried seeds, and 3) toasted grains. A total of 1,444 of sampled coffee beans were surface sterilized (SS) by placing them in ethanol 70% and then flaming. This procedure is intended to destroy any spore or growing microorganism on the external surface of sampled plant tissue.
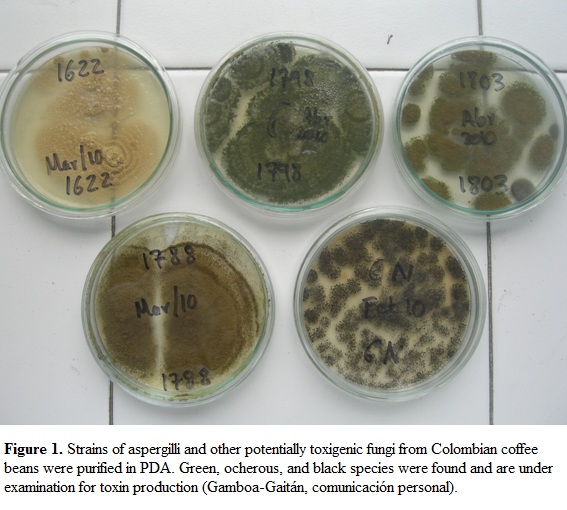
All beans were plated on Petri dishes with salt agar (6% NaCl, 2% agar) with a density of 5 grains/dish. Salt agar was used in order to isolate fungi able to grow in low water potential environments (e.g. Aspergillus and Penicillium). Petri dishes were placed at 25 C, 12 h light and observed each other day for detecting growing molds, that were further isolated in potato dextrose agar (PDA) and malt extract agar (MEA) (both from Scharlau®) as needed. A total of 3,556 beans were plated without surface sterilization (NSS) in the same conditions for a final sample size of 5,000 coffee beans (Table 1). Isolates were separated as morphospecies in individual Petri dishes with PDA and kept for further specific identification. Frequency of each morphospecies was recorded and statistical analysis of goodness of fit was performed ( 2, Zar, 1999). Strains of potentially toxigenic taxa (e.g. A. alliaceus, A. ochraceus, A. flavus, Penicillium spp., Fusarium spp.) (Figura 1), were isolated into pure culture and stored for further identification and screening of toxin production and genetic analysis (Gamboa-Gaitán, personal communication).
RESULTS
From 5,000 sampled coffee beans, 1,433 (28.66%) produced no microorganisms in culture while from the remaining 3,567 (71.34%) beans al least one microorganism was isolated in Petri dishes. This is an almost 3:1 proportion of infected to non-infected coffee beans. Isolated microorganisms included filamentous fungi, bacteria and yeasts, and both epiphytic and endophytic symbionts were found. Isolated microorganisms were classified as toxigenic and non toxigenic according to literature, and from those 3,567 microorganism-positive coffee beans, a total of 2,525 (70.78%) beans produced strains of fungal taxa in which toxigenic species are commonly found (e.g. Penicillium, Aspergillus, Fusarium), while 1,042 (29.22%) coffee beans produced strains of microorganisms not known to produce toxins or unidentified species. This means that roughly a half of total sampled beans were infected with potentially toxigenic microorganisms. Communities of microorganisms from coffee beans were sampled employing two strategies, surface sterilization (SS) and no surface sterilization (NSS). A total of 3,556 (71.12%) beans were NSS and 2,834 of them yielded at least one strain of microorganism. On the other hand, from 1,444 (28.88%) SS beans 733 of them produced microorganisms. According to this 79.70% of NSS beans were associated to microorganisms as contrasted with 50.76% of SS beans. According to Fisher Exact Test this difference was significative.
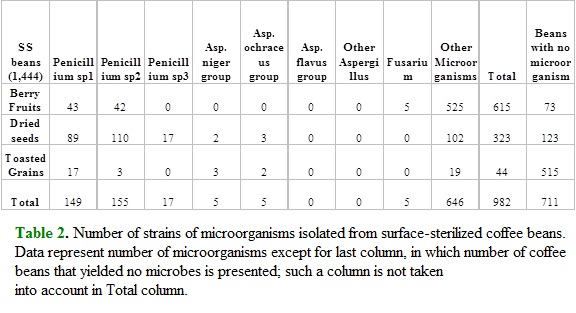
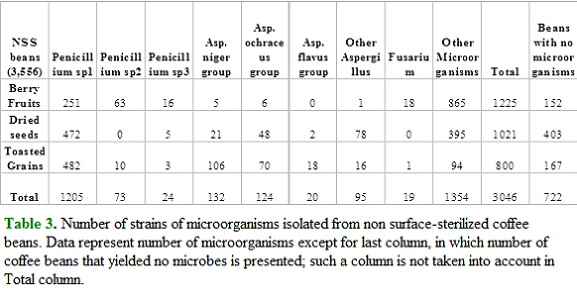
Most conspicuous potentially toxigenic taxa found in this study are Penicillium spp., and isolates from Aspergillus niger group and A. ochraceus group. All of them were found as both endophytes and epiphytes, and for Penicillium sp.1 a general trend of increasing isolates was found from berries to toasted grains (Table 2 and Table 3). Strains of Aspergillus niger and ochraceus groups showed the same trend for NSS beans, although with lower number of isolates. Three species of Penicillium (Table 2 and Table 3) were found as the most common symbionts of coffee beans since at least two of them were always present in beans sampled from the three steps of coffee processing (cherry fruits, dried seeds and toasted granis). Aspergillus was very scarce at the fruit stage and more common at further steps while Fusarium was exclusively found on cherries (Table 2 and Table 3).
All species of Penicillium isolated from surface-sterilized coffee beans were more abundant in dried seeds than in berry fruits and toasted grains (Table 2), and the same trend was found for Penicillium sp1 from non surface-sterilized coffee beans (Table 3). Aspergillus niger and ochraceus groups were most abundant in non surface-sterilized toasted grains as compared with berries and dried seeds (Table 3).
Statistical analysis of goodness of fit (chi-square), were performed to data in Table 2 and Table 3. In Table 2 only Penicillium sp1. and sp2. were sufficiently abundant for statistical analysis and null hypothesis (random distribution of microorganisms among coffee beans (treatments)) was rejected in both cases. For no surface sterilized (NSS) beans, only Penicillium sp3. showed an even distribution among coffee beans.
In summary, many microorganisms are associated as both epiphytes and endophytes to coffee beans from Colombian plantations, and those microbes are found at every stage of coffee processing in different proportions. Some important potentially toxigenic fungal isolates are common among them.
DISCUSSION
Fungi are microorganisms commonly found as both endoand epiphytic symbionts of plants (Petrini, 1991) and their products (Sieber et al., 1988; Bayman et al., 2002; Batista et al., 2003). Many of those fungi are able to produce metabolites physiologically active in vertebrates named mycotoxins (Alexopoulos et al., 1996; Bennett and Klich, 2003). These substances can dramatically alter animal and human physiology by damaging important organs as kidney and liver, and are even able to induce immunological suppression and cancer (Squire, 1981; Bennett and Klich, 2003; Al-Anati and Petzinger, 2006). The posibility these toxins are contaminating foods and feeds is an important stimulus to study the presence of fungi in plants and plant-derived commodities.
Many recent studies show that cereals, almonds and many other foods that include wine and coffee beans hold a diverse array of fungi that potentially produce mycotoxins (Sieber et al., 1988; Fisher et al., 1995; Silva et al., 2000; Batista et al., 2003). Almost no samples from Colombian coffee were studied in this regard (e.g. Vega et al., 2006), but all of them were positive for potentially toxigenic fungi. The present study is the first one developed with an extensive sample set of Colombian coffee beans obtained from different farms in which all steps of coffee processing are performed. Previous work involving coffee and fungi was developed in Brazil (Urbano et al., 2001; Taniwaki, 2005), and Thailand (Pitt et al., 1994), and those studies showed that coffee is a common substrate for many strains of fungi, including potentially toxigenic ones.
Our research employed two sampling strategies: surface sterilization (SS) and no surface sterilization (NSS; Table 1). Surface sterilization procedure here employed is a method of eliminating spores and growing microorganisms from plant epidermis, and is currently used when sampling populations of microorganisms living within plant tissues, here referred to as endosymbionts or endophytes. This means that microbes isolated from SS beans are considered endosymbionts of coffee, while strains from NSS beans are preferentially epiphytes. Data here show that some aspergilli, Penicillium and Fusarium are found as both endoand epiphytes of coffee beans, being the epiphytic state more abundant (Table 2; Table 3). It is clear that abundance is highly variable among different processing steps, which can be interpreted as differential abilities to exploit diverse substrates, given that nature of coffee beans change dramatically during processing from berries in trees to stored, toasted grains. Differential substrate preference of fungi is a common theme in mycology (Carroll and Petrini, 1983).
Most common symbionts of coffee beans were Penicillium and Aspergillus. Penicillium preferred dried seeds while Aspergillus showed preference for toasted grains (Table 2 and Table 3). Both genera are currently isolated from substrates with low water potential, and this is a problem since means they can survive in stored, dried products, as for example toasted coffee grains. Although Penicillium is used in food industry in curing of ham and salami but mainly as a ripening agent of cheeses such as Roquefort, Camembert and Gorgonzola, many species of this genus produce secondary metabolites with physiological properties, including antibiotics such as penicillin and griseofulvin (Alexopoulos et al., 1996), along with toxic compounds such as ochratoxins, aflatoxins, citrinin, patulin and malformin A (Stolk et al., 1990; Alexopoulos et al., 1996; Bennett and Klich, 2003; Moldes-Anaya et al., 2011).
Although some species of Penicillium are important because they produce penicilliosis, a disease that can affect humans and animals (Beneke and Rogers, 1980), while others are involved in plant disease and postharvest fruit decay (Agrios, 2004), penicillia in commodities are not probably the inoculum source of these problems. Its notorious role seems to be restricted to a potential source of dangerous toxins including ochratoxin A (Vega et al., 2006).
Aspergilli, on the other hand, were found in this study as common endophytes but preferentially as epiphytic symbionts of coffee beans (Table 2 and Table 3). Stored, toasted grains, even from small packets for retail in expensive supermarkets, yielded black, ocherous and flavus (green) aspergilli (Figura 1). All these groups contain species well recognized as toxin producers (Abarca et al., 1994; Varga et al., 1996; Ito et al., 2001; Samson et al., 2004), and their presence in coffee beans must be considered a matter of concern, given that Colombian coffee is commonly consumed by millions of people all around the world.
It is worth noting that fungal presence is an important indicator of potential mycotoxin contamination, but the absence of microorganisms is not enough for discarding the presence of toxins. Many studies demonstrated the presence of mycotoxins in commodities without fungal isolation, as for example pulses, cereals and wine (Blesa et al., 2004a; Blesa et al., 2004a; b; Baydar et al., 2005). This means that the presence of fungal strains is not necessary for a food to be potentially dangerous: if those strains produced toxins at some point of their life histories, toxins can last on substrate well after the mycelium has disappeared. Mycotoxins are quite resistant and it is possible they remain chemically stable in stored commodities for long time. It has been shown that they can even survive extreme heating (Tsubouchi et al., 1987), which is an additional problem to coffee beans.
No coffee beans sampled in this study showed symptoms of disease nor molds growing on them were detected. This means that most probable source of recovered fungi was the spore bank, i.e. the pool of spores resting on or within coffee beans. At this point is not possible to know if such spores were produced by mycelia in situ or if they arrived to beans by wind or any other vector. What we can say is that some fungi are most common at some processing steps, e.g. Penicillium sp2 is much more common in dried seeds than in toasted grains (Table 2), so if we were trying to avoid this particular species maximum handling care should be attained at this processing step.
Mycotoxin research is a field in which low resources are invested, specially in developing countries as Colombia. Many aspects of mycotoxin biology are really unknown, being worth to say that not only biochemical and fungal population topics must be considered when trying to understand mycotoxicosis, given that environmental and climatic factors are very important in allowing the establishment of fungal strains. This is a relevant issue since earth is experiencing significative climatic change that could impact diversity and abundance of molds, as recently pointed out by Russell et al., 2010. As can be seen, in an integrative approach to the study of fungal toxin biology lies the solution.
Solving this problem is important since developed countries are implementing tests directed to detect very low levels of mycotoxins in commodities (CAC, 1998; Walker, 1999), and developing countries should improve quality of their products in this aspect. Import authorities will reject foods and feeds entering Europe and other developed countries if only one small sample out of an entire shipment is positive to higher levels of mycotoxins than allowed. The impact of this politics, when fully implemented, could be disastrous to developing economies since there are no real mechanisms able to detect toxins and agrochemicals in export products from those countries (e.g. Colombia).
So, given that Colombia produces agricultural products intended for humans in both local and foreign markets, programs should be immediately established devoted to know the natural history of toxin-producing fungi and to avoid contamination with them. Although apparently expensive, in the long term these programs will save money by increasing quality of our commodities, especially coffee. Additionally, these programs will allow to measure the impact of mycotoxins in public health, since these compounds are unavoidably (Russell et al., 2010) being consumed in our foods and they are involved in important human diseases as some types of cancer and immunological disorders.
ACKNOWLEDGEMENTS
Thanks to Vicerrectoría de Investigación and Departamento de Biología from Universidad Nacional de Colombia (UN), Bogotá Campus; Department of Biology from the University of Puerto Rico (UPR), Río Piedras Campus; Vicerrectoría Académica from Pontificia Universidad Javeriana (PUJ), Bogotá Campus, and Biology Program from Fundación Universitaria Internacional del Trópico Americano, for providing partial support to this work. Many people provided advise or logistic help, to all of them many thanks: José Enrique García-Arrarás & Paul Bayman (UPR), Mónica Cuervo, Argenis Bonilla, Luz M. Melgarejo, Víctor Cruz & Laura Cerón (UN), Nixon Pérez (PUJ), Luis H. Lotero, Nubia Rodríguez, Fernando Ramos & Pablo Gallo (UN-Palmira), Henry A. Gamboa-G., Liliana Gallo-M., and Jahir Pedreros. Special thanks to coffee farmers from La Vega, La Mesa, Bitaco, Pico de Águila, San Antonio del Tequendama, Villeta and Guayabal de Síquima, who provided coffee beans and access to plantations for sampling.
BIBLIOGRAPHY
ABARCA ML, BRAGULAT MR, CASTELLÁ G, CABAÑES FJ. Ochratoxin Aproduction by strains of Aspergillus niger var. niger. Appl Environ Microb. 1994;60(7):2650-2652. [ Links ]
AGRIOS GN. Plant pathology. 5 ed. Amsterdam : Elsevier Academic Press; 2004. [ Links ]
AL-ANATI L, PETZINGER E. Immunotoxic activity of ochratoxin A. J Vet Pharmacol Ther. 2006;29:79-90. [ Links ]
ALEXOPOULOS CJ, MIMS CW, BLACKWELL M. Introductory mycology. 4 ed. New York: John Wiley & sons, Inc.; 1996. [ Links ]
BACKMAN PA AND SIKORA RA. Endophytes: an emerging tool for biological control. Biol Control. 2008;46:1-3. [ Links ]
BATISTA LR, CHALFOUN SM, PRADO G, SCHWAN RF, WHEALS AE. Toxigenic fungi associated with processed (green) coffee beans (Coffea arabica L.). Int J Food Microbiol. 2003;85:293-300. [ Links ]
BAYDAR T, ENGIN AB, GIRGIN G, AYDIN S, SAHIN G. Aflatoxin and ochratoxin in various types of commonly consumed retail ground samples in Ankara, Turkey. Ann Agr Env Med. 2005;12:193-197. [ Links ]
BAYMAN P, BAKER JL, DOSTER MA, MICHAILIDES TJ, MAHONEY NE. Ochratoxin production by the Aspergillus ochraceus group and A. alliaceus. Appl Environ Microb. 2002;68(5):2326-2329. [ Links ]
BAYMAN P, BAKER JL. Ochratoxins: A global perspective. Mycopathologia. 2006;162:215-223. [ Links ]
BENEKE, ROGERS. Medical mycology manual with human mycoses monograph. 4 ed. Minneapolis: Burgess Publishing Company; 1980. [ Links ]
BENNETT JW, KLICH M. Mycotoxins. Clin Microbiol Rev. 2003;16(3):497-516. [ Links ]
BLESA J, BERRADA H, SORIANO JM, MOLTÓ, MAÑES J. Rapid determination of ochratoxin A in cereals and cereal products by liquid chromatography. J Chromatog A. 2004a;1046:127-131. [ Links ]
BLESA J, SORIANO JM, MOLTÓ, MAÑES J. Concentration of ochratoxin A in wines from supermarkets and stores of Valencian Community (Spain). J Chromatog A. 2004b;1054:397-401. [ Links ]
BLOUT WP. Turkey "X" disease. Turkeys. 1961;9(52):55-58,61,77. [ Links ]
BUCHELI, P, C KANCHANOMAI, I MEYER, A PITTET. Development of ochratoxin A during robusta (Coffea canephora) coffee cherry drying. J Agr Food Chem. 2000;48:1358-1362. [ Links ]
CAC. Codex Alimentarius Commission. CX/FAC 99/14. Position paper on Ochratoxin A. Food and Agriculture Organization of the United Nations, Rome. 1998 [cited Aug 11 2011]. Available in: http://www.who.int/fsf/Chemicalcontaminants/ochratoxinpp99_14.pdf. [ Links ]
CARROLL GC. Fungal endophytes in stems and leaves: from latent pathogen to mutualistic symbiont. Ecology. 1988;69:2-9. [ Links ]
CARROLL GC, PETRINI O. Patterns of substrate utilization by some fungal endophytes from coniferous foliage. Mycologia. 1983;75:53-63. [ Links ]
FISHER PJ, PETRINI LE, SUTTON BC, PETRINI O. A study of fungal endophytes in leaves, stems and roots of Gynoxis oleifolia Muchler (Compositae) from Ecuador. Nova Hedwigia. 1995;60:589-594. [ Links ]
KING-JR AD, HOCKING AD, PITT JI. Dichloran-rose bengal medium for enumeration and isolation of molds from foods. Appl Environ Microb Microbiology. 1979;37:959-964. [ Links ]
ITO Y, PETERSON SW, WICKLOW DT, GOTO T. Aspergillus pseudotamarii, a new aflatoxin producing species in Aspergillus section Flavi. Mycol Res. 2001;105(2):233-239. [ Links ]
LEVI CP, TRENK HL, MOHR HK. Study of the occurrence of ochratoxin A in green coffee beans. J Assoc Off Ana Chem. 1974;57:866-870. [ Links ]
MAGNOLI C, HALLAK C, ASTORECA A, PONSONE L, CHIACCHIERA S, DALCERO AM. Occurrence of achratoxin A-producin fungi in commercial corn kernels in Argentina. Mycopathologia. 2006;161:53-58. [ Links ]
MOLDES-ANAYA A, RUNDBERGET T, UHLIG S, RISE F,WILKINS AL. Isolation and structure elucidation of secopenitrem D, an indole alkaloid from Penicillium crustosum Thom. Toxicon. 2011;57:259-265. [ Links ]
PETRINI O. Fungal endophytes of tree leaves. In: Andrews JH, Hirano SS, editors. Microbial ecology of leaves. New York: Springer-Verlag; 1991. p. 179-197. [ Links ]
PITT JI, HOCKING AD, BUDHASAMI K, MISCAMBLE BF, WHEELER KA, TANBOON-EK P. The normal mycoflora of commodities from Thailand. 2. Beans, rice, small grains and other commodities. Int J Food Microbiol. 1994;23:35-43. [ Links ]
RINCÓN-SEPÚLVEDA O. Manual para el cultivo del café. In: El cultivo del café, manual práctico. Bogotá, D.C.: Temas de orientación agropecuaria (TOA), número 136; 1999. p. 7-96, [ Links ]
RINNAN R, BÅÅTH E. Differential utilization of carbon substrates by bacteria and fungi in tundra soil. Appl Environ Microb. 2009;75(11):3611-3620. [ Links ]
ROMANI S, SACCHETTI G, CHAVES LÓPEZ C, PINNAVAIA GG, DALLA ROSA M. Screening on the occurrence of ochratoxin A in green coffee beans of different origins and types. J Agr Food Chem. 2000;48:3616-3619. [ Links ]
RUSSELL R, PATERSON M Y LIMA N. How will climate change affect mycotoxins in food? Food Res Int. 2010;43:1902-1914. [ Links ]
SAMSON RA, HOUBRAKEN JAMP, KUIJPERS AFA, FRANK JM, FRISVAD JC. New ochratoxin A or sclerotium producing species of Aspergillus section Nigri. Stud Mycol. 2004;50:45-61. [ Links ]
SIEBER T, RIESEN TK, MÜLLER E, FRIED PM. Endophytic fungi in four winter wheat cultivars (Triticum aestivum L.) differing in resistance against Stagonospora nodorum (Berk.) Cast. & Germ. = Septoria nodorum (Berk.) Berk. J Phytopathol. 1998;122:289-306. [ Links ]
SILVA CF, BATISTA LR, ABREU LM, DIAS ES, SCHWAN RF. Succession of bacterial and fungal communities during natural coffee (Coffea arabica) fermentation. Food Microbiol. 2008;25:951-957. [ Links ]
SILVA CF, SCHWAN RF, DIAS ES, WHEALS AE. Microbial diversity during maturation and natural processing of coffee cherries of Coffea arabica in Brazil. Int J Food Microbiol. 2000;60:251-260. [ Links ]
SQUIRE RA. Ranking animal carcinogens: a proposed regulatory approach. Science 1981;214:877-880. [ Links ]
STOLK AC, SAMSON RA, FRISVAD JC, FILTENBORG O. The systematics of the terverticillate penicillia. In: Samson RA & Pitt JI, editors. Modern concepts in Penicillium and Aspergillus classification. New York, Plenum Press; 1990. p. 121-137. [ Links ]
TANIWAKI MH, PITT JI, URBANO GR TEXEIRA AA, DE LEITAO MF. Fungi producing ochratoxin A in coffee. In: ASIC, 18th Colloque. Association Scientifique Internationale du café. Helsinki; 1999. p. 239-247. [ Links ]
TANIWAKI MH. An update on ochratoxigenic fungi and ochratoxin A in coffee. In: Hocking AD, Pitt JI, Samson RA, Thrane U, editors. Advances in food mycology. Amsterdam: Springer; 2005. p. 189-202. [ Links ]
TSUBOUCHI H, YAMAMOTO K, HISADA K, SAKABE Y, UDAGAWA S. Effect of roasting on ochratoxin A level in green coffee beans inoculated with Aspergillus ochraceus. Mycopathologia. 1987;97(2):111-115. [ Links ]
URBANO GR, TANIWAKI MH, DE LEITAO MF, VICENTINI MC. Occurrence of ochratoxin A-producing fungi in raw brazilian coffee. J Food Protect. 2001;64(8):1226-1230. [ Links ]
VARGA J, KEVEI E, RINYU E, TÉREN J, KOZAKIEWICZ Z. Ochratoxin production by Aspergillus species. Appl Environ Microb. 1996;62(12):4461-4464. [ Links ]
VEGA FE, POSADA F, PETERSON SW, GIANFAGNA TJ, CHAVES F. Penicillium species endophytic in coffee plants and ochratoxin A production. Mycologia. 2006;98(1): 31-42. [ Links ]
WALKER R. Mycotoxins of growing interest: Ochratoxins. Third joint [ Links ]
FAO/WHO/UNEP International Conference on Mycotoxins, Tunis. 1999. [cited Aug 11 2001]. Available in: http//www.fao.org/WAICENT/FAOINFO/Economic/ESN/mycoto/papers%5Cmyco5b.pdf. [ Links ]
ZAIN ME. Impact of mycotoxins on humans and animals. J Saudi Chem Soc. 2011;15:129-144. [ Links ]
ZAR JH. Biostatistical analysis. 4 ed. Upper Saddle River, NJ, USA: Prentice Hall; 1999. [ Links ]