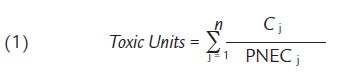Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Acta Biológica Colombiana
Print version ISSN 0120-548X
Acta biol.Colomb. vol.19 no.1 Bogotá Jan./Mar. 2014
https://doi.org/10.15446/abc.v19n1.38461
http://dx.doi.org/10.15446/abc.v19n1.38461
INTEGRATING CHEMICAL AND BIOLOGICAL STATUS ASSESSMENT: ASSEMBLING LINES OF EVIDENCE FOR THE EVALUATION OF RIVER ECOSYSTEM RISK
Integrando el estado químico y biológico: reunión de diversas líneas de evidencia para la evaluación del riesgo en el ecosistema fluvial
ISABEL MUÑOZ, Ph. D.1; SERGI SABATER, Ph. D.2,3
1 Departament d'Ecologia. Universitat de Barcelona. Av. Diagonal, 645, 08028 Barcelona, Spain. imunoz@ub.edu.
2 Institute of Aquatic Ecology, Universitat de Girona, Campus Montilivi, 17071 Girona, Spain. sergi.sabater@udg.edu.
3 Catalan Institute for Water Research (ICRA), Parc Científic i Tecnològic de la Universitat de Girona, Edifici Jaume Casademont, C/ Pic de Peguera, 15, 17003 Girona, Spain. Send correspondence for: Isabel Muñoz, imunoz@ub.edu
Received 15 June 2013, first decision 15 September 2013, accepted 19 November 2013.
Citation / Citar este artículo como: MUÑOZ I, SABATER S. Integrating chemical and biological status assessment: assembling lines of evidence for the evaluation of river ecosystem risk. Acta biol. Colomb. 2014. 19(1):25-34.
ABSTRACT
This paper describes various approaches to evaluating ecological risk in rivers affected by multiple chemical stressors, with emphasis on biofilm and invertebrate community responses. Metrics should be considered as different lines of evidence that, when weighted, form an advanced weight of evidence approach to establishing the environmental risk on a basin scale. Combination of field surveys to obtain observational data of communities, in situ experiments, toxicological sediment tests and a good chemical description of the medium (water and sediment) helps give an integrative view of the chemical and biological state of a river ecosystem. The greater and more distinct the variables used, the greater is our ability to identify the effects of major stressors impairing communities. Weight of evidence is an integrative methodology for tackling the challenge of determining causal relations and applying this knowledge in the decision-making processes of river management.
Keywords: bioassay, chemical indicators, ecological status, weight of evidence.
RESUMEN
Este trabajo describe diferentes aproximaciones para la evaluación del riesgo ecológico en ríos afectados por múltiple contaminación química, con énfasis en la respuesta de las comunidades del biofilm e invertebrados bentónicos. Las métricas descritas se considerarían como diferentes líneas de evidencia que convenientemente ponderadas formarían un sistema de decisión (weight of evidence) para establecer el riesgo ambiental a escala de cuenca. La combinación de trabajo de campo para obtener datos de la comunidad biológica, experimentos in situ, test de exposición en sedimentos, y una buena caracterización química del medio (agua y sedimento), ayuda a tener un resultado integrado del estado biológico y químico del ecosistema fluvial. Cuantas más variables se incorporen, mejor se identificarán los efectos de las presiones químicas en el deterioro de la comunidad. Esta es una metodología integradora que permite abordar el reto de determinar las relaciones causales y aplicar este conocimiento en el proceso de toma de decisión en la gestión de las cuencas fluviales.
Palabras clave: bioensayo, estado ecológico, indicadores químicos, ponderación de evidencias.
INTRODUCTION
Well-preserved fluvial ecosystems conserve natural biodiversity and key ecological processes that provide abilities to respond to moderate disturbances and offer goods and services with interest for humans. However, human activities induce continuous changes to the environment at local and global scales. Many alterations have the potential to compromise the performance of ecosystems (Dudgeon, 2013). Major environmental stressors include, among others, contamination with toxic chemicals, habitat degradation and fragmentation, changes in hydromorphology, nutrient pollution and presence of invasive species. These pressures, occurring alone or in combination, impair the quality of aquatic ecosystems (Ormerod et al., 2010) through changing species diversity and system function.
One important pressure in freshwater systems is the presence of a high number of chemicals, whose occurrence is not included in regular monitoring. Holmstrup (2010) details how synergic interactions between natural stressors and toxicants are common phenomena in river ecosystems mainly in urban and industrialised areas. The potential effects of chemicals extend through several organization levels, from molecules to communities, throughout individuals and populations. Their responses can be seen in metabolic and behavioral changes (Newman and Clements, 2008). At the same time, though low levels of a toxicant may not cause evident effects at the species level in the short term, they can produce long term changes in the whole community (Sumpter et al., 2006).
In this context of multiple impacts and multiple responses, it is difficult to define cause-effect relationships (Allan and Castillo, 2007). Identifying the multiple stresses in aquatic ecosystems entails having tools that can provide multiple evidences of the associations of observed effects and potential causes. Classic surveillance monitoring (e.g. biomonitoring) often fails to fulfill these criteria, since field sampling is limited to selected biological and chemical quality elements and general measures or indices (for example those determined by Water Framework Directive, WFD in Europe). Additionally, most studies of the effects of toxicants on organisms are either tests under controlled conditions or are separately derived from observations in field studies that only take into account a limited set of stressors (Newman and Clements, 2008).
The Weight of Evidence (WoE) approach has become more and more common in ecological risk assessments. WoE determines environmental impact by weighting multiple Lines of Evidence (LoE) (Chapman et al., 2002). LoE is information of a particular type (e. g. water chemical analyses, toxicity testing, and laboratory or field analyses using organisms) that considers important aspects of the environment (Suter, 2007). The intrinsic uncertainty of environmental data means that analysis of single items of evidence is, in most cases, insufficient to achieve reliable evaluation of a system's impairment. Several lines are needed to adequately assess stressors' exposure and effects. Perhaps the best known approach for combining different LoE is the Sediment Quality Triad (Long and Chapman, 1985), based on a standard combination of three LoE: sediment quality, benthic community structure and sediment toxicity. These 3 selections, relating to chemistry, ecology and toxicity, respectively, are combined and weighted with a WoE assessment for final decision-making. We review various measures that could be included as evidence to combine and score as WoE does, particularly those applicable to rivers affected by multiple chemical hazards, ones in urban or industrial areas or where pressure on the water resources challenges the recovery ability of the river. Since communities respond differently to toxicity pressures, this study includes both biofilm and macroinvertebrate community metrics.
ECOLOGICAL MEASURES
Bioassessment
Ecological assessment measures biological conditions in the ecosystem and evaluates the river's grade of degradation. Benthic algae, macrophytes, phytoplankton, macroinvertebrates or fish have long been used to assess ecological condition in freshwater systems (Birk et al., 2012). The information provided by the biological community can be summarized through several metrics, that may be useful as descriptors of multiple pressures on the river.
These metrics are mainly based on taxonomic richness and composition (e.g. number of species, diversity indices, number of individuals, ratios of some taxa); or on ecological functions (e.g. species traits of the aquatic fauna, habitat preferences, tolerance/intolerance measures and others) (Barbour et al., 1999; Hering et al., 2004). It is generally assumed that an increase in environmental stress is associated with a decrease in diversity caused by the dominance of tolerant species, but this relationship is not smooth and applicable straight down the line (Ricciardi et al., 2009). Depending on the status of the community, increasing levels of stress (e.g. pollution) may result in either an increase or, a decrease of diversity. It is also difficult to discern what value of diversity corresponds to a site if the community is under an environmental or an anthropogenic stress. Therefore, changes in diversity and consequently those metrics derived from it can only be assessed by comparisons between sites along a spatial contamination gradient, regarding to reference conditions or with respect to historical data (Warwick and Clarke 1995). These limitations may reduce the correlation between environmental quality and biodiversity.
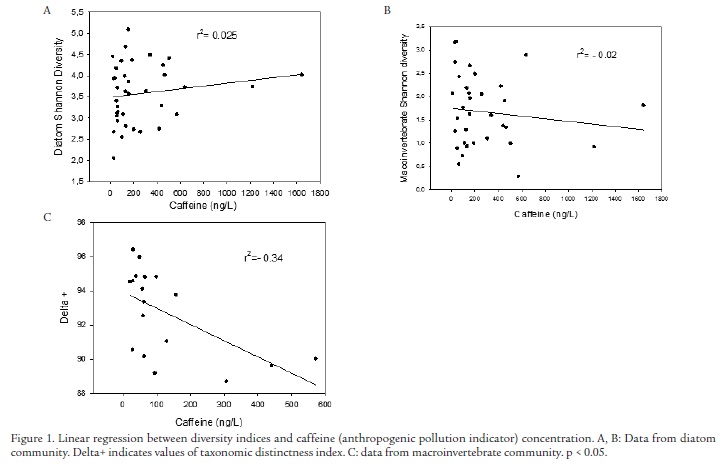
As an example, diversity values from benthic diatoms and macroinvertebrate samples collected in sediments at five sites distributed along pollution gradients in Spanish rivers (Ebro, Júcar, Guadalquivir and Llobregat; see sampling methodology in Navarro-Ortega et al., 2012) have been contrasted with water caffeine concentration, given the latter's role as an indicator of general anthropogenic pollution (Buerge et al., 2003). Shannon-Wiener and taxonomic diversity indices have been calculated. The latter is based not just on species abundance, but also the taxonomic distance between every pair of individuals (Clarke and Warwick, 1998). The Shannon index with the diatom or macroinvertebrate communities (Figure 1 A, C) did not follow the pollution gradient observed in previous studies in these rivers (Muñoz et al., 2009, Navarro et al., 2010, Ricart et al., 2010).
However, the relationship with diatoms improves when using the taxonomic distinctness index in the model (Fig 1B). These indices depart from standard Linnaean classification and incorporate information on taxonomic relationships within a sample into an index that measures species dominance. These indices are independent of sample size, and can be used to compare historical or geographical data sets, in which the sampling effort is unequal or unknown. Although results need to be treated cautiously, lower taxonomical distinctness corresponds to higher pollution values in Spanish Mediterranean rivers. Ricciardi et al. (2009) also described the higher proficiency of taxonomy-based indices of diatom communities when associated with the waterborne concentration of the herbicide Diuron.
Guasch et al. (2012) reviewed responses of macroinvertebrates to toxicants and indicated that diversity indices did not always correlated well with chemical pollution. Diversity reduction was often more closely related to changes in habitat and physico-chemical parameters than to the presence of xenobiotics.
Using large-scale pesticide and biomonitoring data Liess et al. (2008) observed that conventional bioassessment indices based on macroinvertebrates, such as BMWP, Saprobic Index, percentage of selected families and species number, did not correlate well with pesticide contamination. This varied significantly with environmental variables such as pH, current velocity and temperature. Damasio et al. (2007) also observed that biotic indexes (Specific Polluosensitivity index (IPS) for diatom, or BMWP for invertebrates) did not respond in the same way than toxicological biomarkers. The use of physiological, biochemical or histological changes in organisms due to the effects of xenobiotics is considered as a biomarker or "early warning systems" (Ernst and Peterson, 1994). When a biomarker showed sublethal effects, these could not be reflected at population level and biotic metrics did not change, being both results complementary for determining the effects of chemical pollution.
The combination of several measures and indices that address different stressors (multi-metric indices) has also been extensively tested for algae, invertebrates, macrophytes and fish (e.g. Karr, 1991; Hill et al., 2000; Furse et al., 2006). Blanco and Bécares (2010) highlighted the complementarily of diatom and invertebrate indices for detecting and evaluating the huge toxicants. These examples illustrate the suitability of using several metrics to track community-level toxicity effects.
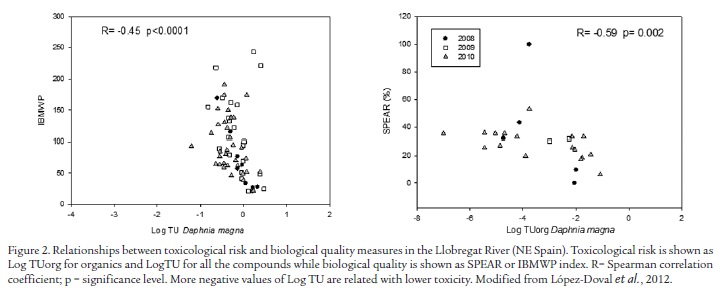
The use of species traits (e.g. size, growth form, morphology, life cycles or behavior) is a more functional community approach than use of biological indices based on tolerance to organic pollution. Species traits account for biological characteristics of organisms; environmental stressors may favor their specific presence. In other words, biological traits provide simple indications on how organisms respond to environmental constraints. For example, the presence of species with aerial respiration mechanisms or hemoglobin is common in systems with high organic pollution, as a strategy to obtain oxygen during anoxic conditions. Some studies ascribe changes in biological traits (e.g. size, growth form, morphology, life cycles, or behavior) to the effects of chemical pollution. Bonada et al. (2006) reviewed that trait-based metrics in freshwater bioassessment were able to discriminate between different types of human impact.The use of multiple species traits in diverse groups of organisms to indicate humancaused stressors has increased recently (e.g. Archaimbault et al., 2010; Statzner and Bêche, 2010). Thus, the species at risk (SPEAR) index is recommended for chemical stress (Liess and von der Ohe, 2005). This index shows the ratio of physiologically-sensitive species in the macroinvertebrate community and, therefore, assesses the effects of organic toxicants on these organisms. As SPEAR is based on biological traits and not on taxonomic units or abundance parameters, its application is not constrained by geographical influences on biological communities. Species are classified and grouped according to their vulnerability to pesticides (or other toxicants), including organisms' sensitivity to toxicants, generation time, migration ability, and the presence of aquatic stages during the time of maximum pesticide application. Species with high generation time and low migration ability are considered at risk due to their low ability to avoid chemical exposure. López-Doval et al. (2012b) observed that the relationship between biological indices and toxicity in the River Llobregat (see below the information for the toxicity units (TU) concept) improved with the use of the SPEAR index (Fig. 2).
Field Experiments (in situ Bioassays) Single species' or communities' in situ assays determine directly the effects of pollutants. These in situ bioassays attach greater relevance to the natural situation, especially in the contamination scenario. Detection of effects can be obtained more rapidly (hours to days) than analyses of pollutant effects on community structure (months to years) (Maltby et al., 2002). For primary producers in situ bioassays were developed basically for phytoplankton and biofilms (Courtney and Clements, 2002). Translocations of communities from unpolluted to polluted sites are useful for testing changes in community structure and function. Corcoll et al. (2012) used a translocation experiment to demonstrate metal effects on biofilm photosynthetic efficiency, diatom biovolume and IPS index (Specific Polluosensitivity index). Proia et al. (2013) found relationships between emerging pollutants (pharmaceutical, mainly antibiotics) and algal and bacterial biofilm compartments. For invertebrate species, in situ bioassays basically expose them to natural conditions in specially designed cages. The more common endpoints measured with invertebrates include leaf consumption in shredders like Gammarus (Maltby et al., 2002), body mass growth for Chironomus (Chappie and Burton, 1997), post-exposure algal grazing rates for Daphnia (Barata et al., 2007), and mollusc reproduction (Schmitt et al., 2011, De Castro-Catala et al., 2013). Other in situ bioassays have included snails, caddisfly and stonefly larvae (Pestana et al., 2009; Gust et al., 2010), and fish (Orrego et al., 2005). Besides, these bioassays have also been developed in tropical ecoregions using surrogate European species (Moreira et al., 2010; Lopes et al., 2011).
The combination of biomarker and toxicological in situ responses with multimetric indices based on community species assemblages has made it possible to identify causal agents impairing river biota that are exposed chronically to multiple environmental factors. Recently, different LoE combining biomarkers, in situ bioassay and community based indexes effectively separated stressors affecting species assemblages from those having sub lethal effects on specific groups of invertebrates (Damasio et al., 2007).
CHEMICAL MEASURES: QUANTIFYING THE RISK ASSOCIATED WITH TOXIC MIXTURES
For well-defined mixtures of toxicants in fluvial systems, i.e. those with a well-defined qualitative and quantitative composition, the toxic unit (TU) approach (e.g. Altenburger and Greco, 2009) may be used to quantify the toxic stress caused by a mixture of pollutants. A Toxic Unit (TU) is defined as the ratio of the exposure concentration of a compound to the effect concentration measured as Predicted No-Effect Concentrations (PNEC) for a specific medium (e.g. water) and standard test organisms (such as Daphnia magna, invertebrate; Selenastrum capricornutum, algae; and Pimephales promelas, fish), covering all trophic levels. The resulting three values indicate the respective risks for the aquatic biota. The TU concept is based on additive toxicity of the compounds that considers toxicity of a mixture will be equivalent to that expected from a simple summation of the toxicities of the individual chemicals present in the sample. To estimate the toxicity of the mixture, the TU for each constituent in the mixture of substances are added together (Equation 1).
When the TU in the mixture equals one or is greater than one, the mixture is expected to be above the risk threshold. This approach was recently recommended by the European Commission (2011) in their technical guidance for attaining environmental quality standards in mixtures.
High values of TU for invertebrates at monitoring sites on various European rivers correlated, negatively with the SPEAR index (Liess and von der Ohe, 2005; von der Ohe et al., 2009), indicating that potential effects from chemical exposure were higher than expected. Ginebreda et al. (2010) described inverse linear correlation between the Hazard Quotient (similar to TU) for pharmaceuticals and diversity, based on the invertebrate community. Consequently, toxic stress should be considered equally for the assessment of ecological status, by applying basic mixture toxicity concepts to different biological components.
The multisubstance potentially affected fraction of species (msPAF) directly quantifies the expected loss of species, also taking into account mixture toxicity (Posthuma et al., 2002; De Zwart and Posthuma, 2005). This is a predictive model that attributes impairment in biological condition to multiple causes. The toxic pressure at a site is a modeled value, obtained by combining local monitoring data on chemical concentrations with a train of models that subsequently take into account bioavailability, Species Sensitivity Distributions (SSD), and mixture toxicity. The resulting measure covers all trophic levels (algae, invertebrates and fish) using acute LC50 data and includes both organic and metal compounds. The method makes use of both the concentration addition and the response addition models to express the potentially affected fraction (PAF) of taxa for single compounds, and the multi-substance PAF (msPAF) for mixtures (De Zwart and Posthuma, 2005).
These works showed the need for extensive chemical prospecting. Including large number of substances in the analysis would allow a more refined calculation of the chemical risk. Moreover, the incorporation of emerging compounds analysis in routine monitoring could assess their potential effects in the environment.
TOXICITY: SEDIMENT CONTACT EXPERIMENTS
The contaminant concentrations in sediments may be several orders of magnitude higher than in the overlying water. Although certain chemicals are strongly adsorbed to sediment, they may still be available for biota, thus causing a risk to benthic organisms. This risk can be evaluated by exposing organisms to contaminated whole sediment and determining whether this sediment is potentially harmful to aquatic organisms. Because these tests measure biological responses directly, they account for interactive toxic effects of complex contaminant mixtures in sediment. The amount of accumulated chemicals is determined by sediment and chemical characteristics; and the uptake route, by the characteristics of the organism, such as its size or feeding behavior (Leppänen and Kukkonen, 2006). In addition, the sensitivities of species to different chemical types may vary and the organism may have different sensitivities at different life stages. This variability means that no single species is optimal for all assessments of sediment toxicity. It is therefore recommended to use toxicity test batteries with species from different trophic levels, habitats and endpoints in order to cover different exposure routes and sensitivities to toxicants, and to represent the natural population as extensively as possible (Chapman et al., 2002; Tuikka et al., 2011).
Test organisms used to evaluate the toxicity of freshwater sediments include between others, bacteria (e.g. Vibrio fischeri), algae (Pseudokirschneriella subcapitata), macrophytes (Lemna minor), nematodes (Caenorhabtidis elegans), worms (Tubifex tubifex), microcrustaceans (Daphnia spp.), insects (Chironomus riparius), and fish (Pimephales promelas), as well as different embrionary phases and levels of biological organization (e.g. genotoxicity tests). The choice of the test organism has a major influence on the ecological relevance, success and interpretation of the test. Organism selection should be representative of the studied environment to easily extrapolate results to natural conditions or comparable with results in literature.
Tuikka et al. (2011) and Wolfram et al. (2012) reported the use of the sediment contact test for six species from different biological groups to analyse the risk of polluted sediment in European rivers in densely inhabited areas. This was used as an additional metric of the biological quality elements listed by the WFD. Their results were integrated into a hierarchical approach, in which different LoE were scored in a WoE proposal that was comprehensible to non-experts.
Additionally, toxicity tests on sediments spiked with known concentrations of contaminants can be used to establish cause-and-effect relationships between chemicals and responses, but the behavior of contaminants in spiked sediments cannot automatically be equated with their behavior in fieldcontaminated sediments. López-Doval et al. (2012a) used sediment spiked with pharmaceuticals as a complementary tool to confirm the statistical relationship found in field assessment between community and concentrations of some pharmaceuticals (Muñoz et al., 2009). These small-scale experimental studies clarified large-scale observations effectively.
CONCLUDING REMARKS Rivers are increasingly impaired by multiple physical, chemical and biological stressors. To achieve good ecological quality in fluvial systems requires integrated assessments of all available information, evaluation of the risk associated with each pressure and selection of the most effective management options. However, determining whether toxicant exposure causes ecological impairment is impeded by several factors, especially natural variability and those factors that mask the effects of contaminants. Besides, climate related factors, differences in ecosystem and species sensitivity or land use practices, may determine different responses to pollution in tropical rivers versus temperate ones (Daam and Van den Brink, 2010; Rico et al., 2011).
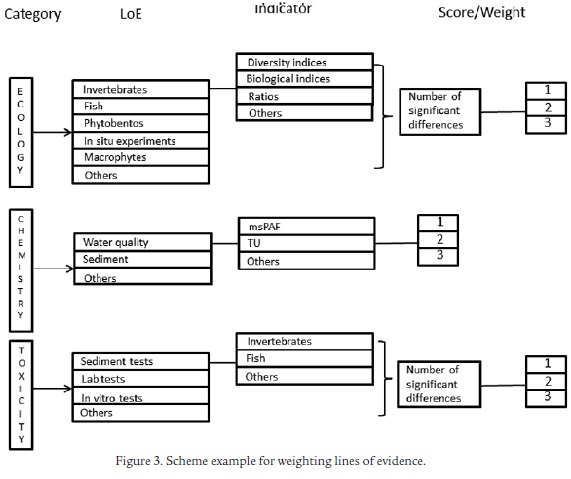
We emphasize that the combination of some of the described approaches can help obtain an integrative view of the chemical and biological state of a river. The greater and more distinct the variables used, the greater will be our ability to identify the effects of major stressors impairing communities. Combining field surveys with in situ and laboratory experiments facilitates different lines of evidence for establishing links between pressures and effects. The results obtained, once appropriately scored and quantitatively weighted (Fig. 3), may help to detect risk levels and to prioritize management action on reaches or on groups of compounds causing the greatest risk. The successful and comparable application of the WoE framework in lakes (McDonald et al., 2007), in soils (Semenzin et al., 2008) and in European rivers (Gottardo et al., 2011; Wolfram et al., 2012), highlights its usefulness as an understandable scheme that can be applied at basin or regional scale.
To be effective, the analysis of risk for the ecological integrity of river ecosystems through WoE methodology needs an interdisciplinary study, to generate diverse and complementary knowledge of causes and effects and, in a further step, to integrate this knowledge into decision-making processes.
ACKNOWLEDGEMENTS
This research was funded by the Spanish Ministry of Economy and Competitiveness SCARCE project (Consolider- Ingenio 2010 program, project: CSD2009-00065).
REFERENCES
Allan JD, Castillo MM. Stream Ecology: Structure and Function in Running Waters. Springer, Dordrecht, The Netherlands. 2007. p. 317-357. [ Links ]
Altenburger R, Greco WR. Extrapolation concepts for dealing with multiple contamination in Environmental Risk Assessment. Integr Environ Assess Manag. 2009;5(1):62-68. [ Links ]
Archaimbault V, Usseglio-Polatera P, Garric J, Wasson JG, Babut M. Assessing pollution of toxic sediment in streams using bio-ecological traits of benthic macroinvertebrates. Freshwater Biol. 2010;55(7):1430-1446. [ Links ]
Barata C, Damasio J, Lopez MA, Kuster M, López de Alda M, Barceló D. et al. Combined use of biomarkers and in situ bioassays in Daphnia magna to monitor environmental hazards of pesticides in the field. Environ Toxicol Chem. 2007;26(2):370-379. [ Links ]
Barbour MT, Gerritsen J, Snyder BD, Stribling B. Rapid bioassessment protocols for use in streams and wadeable rivers: periphyton, macroinvertebrates and fish. 2nd ed. EPA 841-B-99-002. US Environmental Protection Agency, Office of Water, Washington D.C. 1999. [ Links ]
Birk S, Bonne W, Borja A, Brucet S, Courrat, Poikane S, et al. Three hundred ways to assess Europe's surface waters: An almost complete overview of biological methods to implement the Water Framework Directive. Ecological Indicators. 2012,18:31-41. [ Links ]
Blanco S, Bécares E. Are biotic indices sensitive to river toxicants? A comparison of metrics based on diatoms and macroinvertebrates. Chemosphere. 2010;79(1):18-25. [ Links ]
Bonada N, Prat N, Resh VH, Statzner B. Developments in aquatic insect biomonitoring: a comparative analysis of recent approaches. Annu. Rev. Entomol. 2006;51: 495-523. [ Links ]
Buerge IJ, Poiger T, Muller MD, Buser HR. Caffeine an anthropogenic marker for wastewater contamination of surface waters. Environ Sci Technol. 2003;37(4):691-700. [ Links ]
Chapman PM, McDonald BG, Lawrence GS. Weight-ofevidence issues and frameworks for sediment quality (and other) assessments. Hum Ecol Risk Assess. 2002;8(7):1489-1515. [ Links ]
Chappie DJ, Burton GA. Optimization of in situ bioassays with Hyalella azteca and Chironomus tentans. Environ Toxicol Chem. 1997;16(3):559-564 [ Links ]
Clarke KR, Warwick RM. A taxonomic distinctness index and its statistical properties. J appl Ecol. 1998;35(4):523-531 [ Links ]
Corcoll N, Bonet B, Morin S, Tlili A, Leira M, Guasch H. The effect of metals on photosynthesis processes and diatom metrics of biofilm from a metal-contaminated river: A translocation experiment. Ecological Indicators. 2012; 18:620-631. [ Links ]
Courtney LA, Clements WH. Assessing the influence of water and substratum quality on benthic macroinvertebrate communities in a metal-polluted stream: an experimental approach. Freshwater Biology. 2002;47(9):1766-1778. [ Links ]
Daam MA, Van den Brink PJ. Implications of differences between temperate and tropical freshwater ecosystems for the ecological risk assessment of pesticides. Ecotoxicology. 2010;19(1):24-37. [ Links ]
Damasio JB, Barata C, Munne A, Ginebreda A, Guasch H, Sabater S, et al. Comparing the response of biochemical indicators (biomarkers) and biological indices to diagnose the ecological impact of an oil spillage in a Mediterranean river (NE Catalunya, Spain). Chemosphere. 2007;66(7): 1206-1216. [ Links ]
De Castro-Catala N, López-Doval JC, Gorga M, Petrovic M, Muñoz I. Is reproduction of the snail Physella acuta affected by endocrine disrupting compounds? An in situ bioassay in three Iberian basins. J Hazard Mater. 2013;263: 248-255. [ Links ]
De Zwart D, Posthuma L. Complex mixture toxicity for single and multiple species: proposed methodologies. Environ Toxicol Chem. 2005;24(10):2665-2676. [ Links ]
Dudgeon D. Anthropocene Extinctions: Global Threats to Riverine Biodiversity and the Tragedy of the Freshwater Commons. In: River Conservation: Challenges and Opportunities. Sabater S, Elosegi A (eds.). BBVA Foundation, 2013 (available at www.fbbva.es/). p129-165. [ Links ]
Ernst WHO, Peterson PJ. The role of biomarkers in environmental assessment. Ecotoxicology. Terrestrial plants. 1994;3(3):180-192. [ Links ]
European Commission. Guidance Document N.º 27 Technical Guidance for deriving Environmental Quality Standards. Common Implementation Strategy for the Water Framework Directive (2000/60/EC); 2011. p. 204. [ Links ]
Furse MT, Hering D, Brabec K, Buffagni A, Sandin L, Verdonschot PFM. The Ecological Status of European Rivers: Evaluation and Intercalibration of Assessment Methods. Hydrobiologia 2006;566:299-309. [ Links ]
Ginebreda A, Muñoz I, López de Alda M, Brix R, López-Doval JC, Barcelo D. Environmental risk assessment of pharmaceuticals in rivers: Relationships between hazard indexes and aquatic macroinvertebrate diversity indexes in the Llobregat River (NE Spain). Environ Int. 2010;36(2):153-162. [ Links ]
Gottardo S, Semenzin E, Giove S, Zabeo A, Critto A, de Zwart D, et al. Integrated Risk Assessment for WFD Ecological Status classification applied to Llobregat river basin (Spain). Part II â Evaluation process applied to five environmental Lines of Evidence. Sci Total Environ. 2011;409(22):4681-4692. [ Links ]
Guasch H, Bonet B, Bonninneau C, Corcoll N, Lopez-Doval JC, Muñoz I, et al.How to link field observations with causality? Field and experimental approaches linking chemical pollution with ecological alterations. In: Guasch H, Ginebreda A, Geiszinger A eds. Emerging and priority pollutants in rivers. Springer-Verlag Berlin Heildeberg 2012. p. 181-218. [ Links ]
Gust M, Buronfosse T, Geffard O, Mons R, Quéau, H, Mouthon, J. et al. In situ biomonitoring of freshwater quality using the New Zealand mudsnail Potamopyrgus antipodarum (Gray) exposed to waste water treatment plant (WWTP) effluent discharges. Water Res. 2010; 44(15):4517-4528. [ Links ]
Hill B . Herlihy AT, Kaufmann PR, Stevenson RJ, McCormick FH, Johnson CB. Use of periphyton assemblage data as an index of biotic integrity. J North Am Benthol Soc. 2000;19(1):50-67. [ Links ]
Hering D, Moog O, Sandin L, Verdonschot PF. Overview and application of the AQEM assessment system. Hydrobiologia. 2004;516(1):1-20. [ Links ]
Holmstrup M, Bindesbøl AM, Oostingh GJ, Duschl A, Scheil V, Köhler HR et al. Interactions between effects of environmental chemicals and natural stressors: A review. Sci Total Environ. 2010;408(18):3746-3762. [ Links ]
Karr JR. Biological integrity - A long-neglected aspect of waterresource management. Ecol Appl. 1991;28(1):66-84. [ Links ]
Leppänen, MT, Kukkonen,JVK. Evaluating the role of desorption in bioavailability of sediment-associated contaminants using oligochaetes, semipermeable membrane devices and Tenax extraction. Environ Pollut. 2006;140:150-163. [ Links ]
Liess M, von der Ohe PC. Analizing effects of pesticides on invertebrate communities in streams. Environ Toxicol Chem. 2005;24(4):954-965. [ Links ]
Liess M, Schafer RB, Schriever CA. The footprint of pesticide stress in communities-Species traits reveal community effects of toxicants. Sci Total Environ. 2008;406(3):484-490 [ Links ]
Long ER, Chapman PM. A sediment quality triad: Measures of sediment contamination, toxicity and infaunal community composition in Puget Sound. Mar Pollut Bull. 1985; 16:405-415. [ Links ]
Lopes I, Moreira-Santos M, Rendon-von Osten J, Baird DJ, Soares AMVM. Suitability of five cladoceran species from Mexico for in situ experimentation. Ecotox Environ Safe. 2011;74(1):111-116. [ Links ]
López-Doval JC, Kukkonen JVK, Rodrigo P, Muñoz I. Effects of indomethacin and propranolol on Chironomus riparius and Physella acuta. Ecotoxicol Environ Saf. 2012a;78:110-115. [ Links ]
López-Doval JC, De Castro-Català N, Andres-Domenech I, Blasco J, Ginebreda A, Muñoz I. Analysis of monitoring programmes and their suitability for ecotoxicological risk assessment in four Spanish basins. Sci Total Environ. 2012b;440:194-203. [ Links ]
Maltby L, Clayton SA, Wood RM, McLoughlin N. Evaluation of the Gammarus pulex in situ feeding assay as a biomonitor of water quality: Robustness, responsiveness, and relevance. Environ Toxicol Chem. 2002;21(2):361-368. [ Links ]
McDonald BG, deBruyn AMH, Wernick BG, Patterson L, Pellerin N, Chapman PM. Design and application of a transparent and scalable weight-of-evidence framework: An example from Wabamun Lake, Alberta, Canada. Integr Environ Assess Manag. 2007;3:479-483. [ Links ]
Moreira SM, Moreira-Santos M, Rendon-von Osten J, da Silva EM. Ecotoxicological tools for the tropics: Sublethal assays with fish to evaluate edge-of-field pesticide runoff toxicity. Ecotox Environ Safe. 2010;73:893-899. [ Links ]
Muñoz I, López-Doval JC, Ricart M, Villagrasa M, Brix R, Geiszinger A. et al. Bridging levels of pharmaceuticals in river water with biological community structure in the Llobregat river basin (NE Spain). Environ Toxicol Chem. 2009;28(12):2706-2714. [ Links ]
Navarro A; Tauler R; Lacorte S; Barcelo D. Occurrence and transport of pesticides and alkylphenols in water samples along the Ebro River Basin. J Hydrol. 2010;383(1):18-29. [ Links ]
Navarro-Ortega A, Acuña V, Batalla RJ, Blasco J, Conde C, Elorza FJ, et al. Assessing and forecasting the impacts of global chamge on Mediterranean rivers: The SCARCE consolider project on Iberian basins. Environ Sci Pollut Res. 2012;19(4):918-933. [ Links ]
Newman MC, Clements WH. Ecotoxicology: a comprehensive treatment. CRC Press; 2008. p. 13, 193, 359. [ Links ]
Ormerod SJ, Dobson M, Hildrew AG, Townsend CR. Multiple stressors in freshwater ecosystems. Freshwater Biology. 2010;55(1):1-4 [ Links ]
Orrego R, Burgos A, Moraga-Cid G, Inzunza B, Gonzalez M, Valenzuela A et al. Effects of pulp and paper mill discharges on caged rainbow trout (Oncorhynchus mykiss): biomarker responses along a pollution gradient in the Biobio River, Chile. Environ Toxicol Chem. 2006; 25(9):2280-2287. [ Links ]
Pestana JL, Alexander AC, Culp JM, Baird DJ, Soares AM. Structural and functional responses of benthic invertebrates to imidacloprid in outdoor stream mesocosms. Environ Pollut. 2009;157(8-9):2328-2334. [ Links ]
Posthuma L, Traas TP, Suter GW. Species sensitivity distributions in ecotoxicology. Lewis Publishers, Boca Raton, FL. 2002. [ Links ]
Proia L, Osorio V, Soley S, Köck-Schulmeyer M, Pérez S, Barceló D, et al. Effects of pesticides and pharmaceuticals on biofilms in a highly impacted river. Environ Pollut. 2013;178:220-228. [ Links ]
Ricart M, Guasch H, Barceló D, Brix R, Conceiçao MH, Geiszinger A. et al. Primary and complex stressors in polluted mediterranean rivers: pesticide effects on biological communities. J Hydrol. 2010;383(1-2):52-61. [ Links ]
Ricciardi F, Bonnineau C, Faggiano L, Geiszinger A, Guasch H, Lopez-Doval JC et al. Is chemical contamination linked to the diversity of biological communities in rivers? Trends in analytical Chemistry. 2009;28:592-602. [ Links ]
Rico A, Waichman AV, Geber-Correa R, van den Brink PJ. Effects of malathion and carbendazim on Amazonian freshwater organisms: comparison of tropical and temperate species sensitivity distributions. Ecotoxicology. 2011;20(4):625-634. [ Links ]
Semenzin E, Critto A, Rutgers M, Marcomini A. Integration of bioavailability, ecology and ecotoxicology by three lines of evidence into ecological risk indexes for contaminated soil assessment. Sci Total Environ. 2008;389(1):71-86. [ Links ]
Schmitt C, Streck G, Lamoree M, Leonards P, Brack W, De.Deckere, E. Effect directed analysis of riverine sediments-The usefulness of Potamopyrgus antipodarum for in vivo effect confirmation of endocrine disruption. Aquat Toxicol. 2011;101(1):237-243. [ Links ]
Statzner B, Bêche LA. Can biological invertebrate traits resolve effects of multiple stressors on running water ecosystems?. Freshwater Biology. 2010;55:80-119. [ Links ]
Sumpter JP, Johnson AC, Williams RJ. Modeling effects of mixtures of endocrine disrupting chemicals at the River Catchment Scale. Environ Sci Technol. 2006;40(17):5478-5489. [ Links ]
Suter GW. Ecological risk assessment. CRC press. Boca Raton, USA. 2007. [ Links ]
Tuikka AI, Schmitt C, Hoss S Bandow N, de Zwart D, von der Ohe PC et al. Toxicity assessment of sediments from three European river basins using a sediment contact test battery. Ecotox Environ Safe. 2011;74:123-131. [ Links ]
Von der Ohe PC, de Deckere E, Prϋb; Muñoz I, Wolfram G, Villagrasa M, et al. Towards an Integrated Assessment of the Ecological and Chemical Status of European River Basins. Integr Environ Assess Manag. 2009;5:50-61. [ Links ]
Warwick RM, Clarke K.R. New 'biodiversity' measures reveal a decrease in taxonomic distinctness with increasing stress. Mar Ecol Prog Series. 1995;129: 301-305. [ Links ]
Wolfram G, Höss S, Orendt C, Schmitt C, Adamek Z, Bandow N, et al. Assessing the impact of chemical pollution on benthic invertebrates from three different European rivers using a weight-of-evidence approach. Sci Total Environ. 2012; 438:498-509. [ Links ]