Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Acta Biológica Colombiana
Print version ISSN 0120-548X
Acta biol.Colomb. vol.19 no.3 Bogotá Sept./Dec. 2014
https://doi.org/10.15446/abc.v19n3.42195
Artículo de investigación
http://dx.doi.org/10.15446/abc.v19n3.42195
MORPHOLOGY AND CONSERVATION OF THE MESOAMERICAN SLIDER (Trachemys venusta, EMYDIDAE) FROM THE ATRATO RIVER BASIN, COLOMBIA
Morfología y conservación de la tortuga hicotea Mesoamericana (Trachemys venusta, Emydidae) del río Atrato, Colombia
CLAUDIA P. CEBALLOS1, Ph. D.; WILLIAM A. BRAND2, Ecol.
1 Grupo Centauro. Escuela de Medicina Veterinaria, Facultad de Ciencias Agrarias, Universidad de Antioquia. Carrera 75 n.° 65-87, of. 47-122, Medellín, Colombia. claudiaceb@gmail.com
2 Corpouraba. Calle 92 n.° 98-39, Turbo, Antioquia, Colombia. ecobrand23676@gmail.com
Author for correspondence: Claudia P. Ceballos, claudiaceb@gmail.com
Received 20th February 2014, first decision 14th May 2014, accepted 05th June 2014.
Citation / Citar este artículo como: CEBALLOS CP, BRAND WA. Morphology and conservation of the mesoamerican slider (Trachemys venusta, Emydidae) from the Atrato River basin, Colombia. Acta biol. Colomb. 2014;19(3):483-488
ABSTRACT
The phylogenetic relationships of the Mesoamerican Slider, Trachemys venusta, that inhabits the Atrato River basin of Colombia have been controversial as three different names have been proposed during the last 12 years: T. v. venusta, T. v. uhrigi, and T. ornate venusta. Using a group of sliders that was confiscated by the regional authority we compared the morphology of T. venusta distributed in the Atrato River with the morphology of the subspecies putatively distributed in Colombia. We found that the Colombian Mesoamerican slider has an overall smaller size, different plastral inter-scute seam formula, and different head, carapace and plastron coloration patterns. In addition, we also report the poor health condition of these individuals that have endured this illegal trade. We underscore an urgent need for further studies of individuals indigenous to Colombia to better understand the phylogenetic relationships of T. venusta throughout its distributional range, along with a more effective control of the illegal turtle trade in the Urabá region of Colombia.
Keywords: conservation, dermatitis, health, taxonomy, traffic.
RESUMEN
Las relaciones filogenéticas de la tortuga hicotea mesoamericana, Trachemys venusta, que habita la cuenca del río Atrato en Colombia ha sido controversial dado que tres subespecies diferentes han sido propuestas en los últimos 12 años: T. v. venusta, T. v. uhrigi, y T. ornate venusta. En este estudio se usó un grupo de tortugas hicoteas que fue decomisado por la autoridad ambiental para documentar su morfología y compararla con la reportada para la subespecie supuestamente distribuida en Colombia. Nosotros encontramos que la hicotea Mesoamericana colombiana es más pequeña, tiene una fórmula de las suturas de los escudos plastrales diferentes, y patrones de coloración de la cabeza, caparazón y plastrón diferentes. Adicionalmente, reportamos el pobre estado de salud de estos individuos que han soportado las condiciones de este mercado ilegal. Resaltamos la urgencia de realizar estudios de esta especie que incluyan especímenes nativos de Colombia para comprender mejor las relaciones filogenéticas de T. venusta en todo su rango de distribución, así como el realizar un control más efectivo del tráfico ilegal de tortugas en la región del Urabá colombiano.
Palabras clave: conservación, dermatitis, salud, taxonomía, tráfico.
INTRODUCTION
The Mesoamerican Slider turtle, Trachemys venusta (Seidel, 2002) (Emydidae, Deirochelyinae), is widely distributed in Central and South America, ranging from Mexico to Colombia. Because of its wide distribution and diversity of forms its taxonomy has been debated (Castaño-Mora, 1992; Seidel, 2002; McCord et al., 2010; TTWG, 2012). Three subspecies were initially proposed: T. v. cataspila, T. v. grayi and T. v. venusta (Seidel, 2002), with the latter being the only one distributed in Colombia. Eight years later three new subspecies were proposed based on their biogeography and morphology: T. v. iversoni, T. v. panamensis, and T. v. uhrigi, (McCord et al., 2010) with the later corresponding to the one distributed in Colombia. These subspecies have been provisionally listed as valid pending genetic analysis since 2010 (TTWG, 2010), a situation that has not changed since then (TTWG, 2011; TTWG, 2012). More recently in 2012 the phylogeny of most sliders (Trachemys spp.) from Central and South America was reviewed (Fritz et al., 2012), but this time using mitochondrial and nuclear DNA. This study included the two taxa, T. v. uhrigi and T. v. venusta, both of which had been previously reported as present in Colombia. Surprisingly, this study found that these two subspecies did not cluster with the other subspecies of Trachemys venusta as expected, but instead they clustered with a different species, Trachemys ornata. This finding suggests that the taxon distributed in Colombia, regardless of the subspecies name, may belong to a different species. In fact, in this study the authors proposed to rename T. venusta uhrigi as T. ornate venusta. It must be considered however that the specimen of T. v. uhrigi used by Fritz et al., (2012) came from Honduras, not from Colombia, thus whether this new name applies to the taxon naturally found in Colombia still remains unknown. In addition to these gaps in the phylogeny of T. venusta, previous authors have already called attention tothe lack of ecological information on the slider turtle from the Atrato River basin of Colombia (Medem, 1962; Rueda-Almonacid et al., 2007; Bock et al., 2012). To date there is only one report published from Colombia that describes aspects of its morphology and reproduction (Medem, 1962). The conservation status of this species is Vulnerable in Colombia (Castaño-Mora, 2002), it has not been evaluated globally (IUCN, 2012), and is considered Data Deficient in South America by the Tortoise and Freshwater Turtle Specialist Group (Bock et al., 2012).
Herein we report the morphology of T. venusta from the Atrato River basin, and compare it with the morphology reported in the literature for the subspecies putatively distributed in Colombia (Medem, 1962; Seidel, 2002; McCord et al., 2010). For this purpose we used a group of 89 turtles confiscated in the region by the regional authority. The bad health condition of these animals suggests the harsh circumstances that they endured during that time. The illegal trade of Trachemys turtles has been documented worldwide including Colombia (Ceballos and Fitzgerald, 2004; Gong et al., 2009; Martínez and Botero, 2011; Bock et al., 2012). In fact, in a retrospective study on reptile pathologies at Universidad Nacional, in Bogotá, Colombia, during the last 37 years, most cases were on dead turtles (54.3 % of cases, n=101), and of these Trachemys sp. was the species most common (40.6 %, n=41) (Martínez and Botero, 2011). The origins of most turtles were confiscations or were voluntary given to the Wildlife Rescue and Rehabilitation Unit (URRAS). Thus in this study we also report the bad health condition of these confiscated turtles thus adding another threat to the conservation of this species.
MATERIALS AND METHODS
On March 8, 2011, a group of 89 Mesoamerican Slider turtles, Trachemys venusta (Seidel, 2002), were confiscated by the local environmental authority, Corpourabá, assisted by the local environmental police. These turtles were found in a truck, and the driver reported that the turtles came from "Kilómetro 40", located in the corregimiento Lomas Aisladas, municipality of Turbo, department of Antioquia, Colombia (7°39'12"N, 76°57'37"W). This region is surrounded by several creeks (e.g. Nandí, Zumbadora, Manatí, León) that drain into the Atrato River, and this river into the Caribbean Sea in the Gulf of Urabá.
After the confiscation, the sliders were taken to the wildlife reception center ("Estación de Paso") of Corpourabá located in the municipality of Carepa (Antioquia). On March 12 and 13, 2011, the linear carapace length and width (LCL, LCW), linear plastron length and width (LPL, LPW), and curved carapace length and width (CCL, CCW) of each specimen (n = 89 plus one turtle confiscated a few days before) were measured. Linear measurements were taken with a digital electronic caliper (mm), and curved measurements with a metric tape (mm). Linear and curved carapace widths were measured at the periphery of the seam, specifically between marginal scutes six and seven. The sliders were also weighed (g), marked by the notching technique (Cagle, 1939), and photographed with a digital camera attached to a tripod.
We did not notice evidences of sexual differences among the individuals of this group (e.g., tail size, claws length, or snout size), suggesting either that: that there were all females in this group, there were females and young males, or that sexual dimorphism was not evident. For this reason we do not analyze the data by sex.
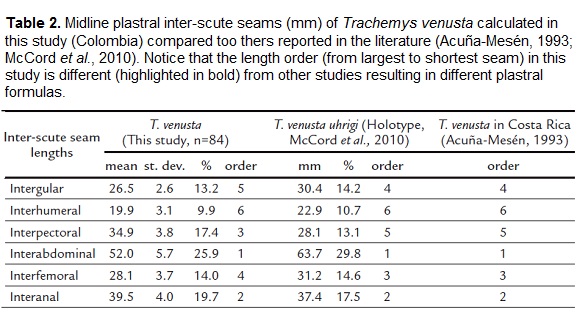
The plastral formula of the midline inter-scute seams (mm) was calculated using the digital photographs of the plastrons and the software TPS dig (Rohlf, 2001). Every seam average was calculated to determine the order of magnitude (from longest to shortest): abdominal (abd), anal (an), pectoral (pect), gular (gul), femoral (fem) and humeral (hum).
Because the nesting season of T. venusta in the locality where turtles originated, Lomas Aisladas, starts in December (Medem, 1962), turtles were palpated in their inguinal region to determine if there were gravid females. Other diagnostic media were unavailable to confirm reproductive state; however, inguinal palpation is a simple and effective method to determine female reproductive status in river turtles and land tortoises (73.3 to 90.4 % accuracy) (Andreu and Villamor, 1989; Keller, 1998; Bertolero and Marin, 2005). Two veterinarians palpated each turtle independently, so turtles were classified as gravid only when both agreed. Finally, after recording all these data, Corpouraba released the turtles into the National Natural Park Los Katíos.
RESULTS
Morphometry
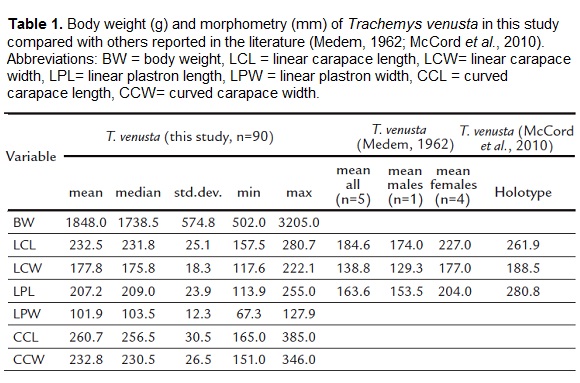
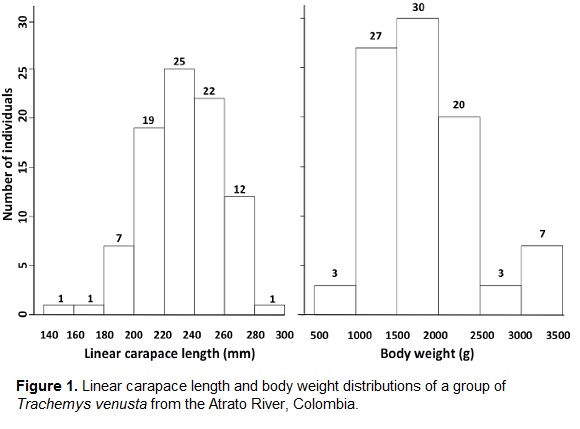
Body measurements and weights are summarized in table 1 and figure 1. Interestingly, the size classes of this group are distributed normally, with the most common size (LCL) class being between 220 and 240 mm (average: 232 mm), and only one individual above 280 mm. Body weight distribution was similar, with the majority of the individuals weighing between 1500 and 2000 g (average: 1848 g). The largest individual was also the heaviest, with 28,066 mm of LCL and 3205 g.
The midline plastral inter-scute seams were measured and the resulting formula was: abd> an >pect>gul> fem > hum (Table 2). This formula is different from: abd> an > fem >gul>pect> hum, reported for the T. v. uhrigi holotype (McCord et al., 2010) and T. venusta in Costa Rica (Acuña-Mesén, 1993).
Morphological comparison
The coloration and stripe patterns of the head, carapace and plastron of the Colombian T. venusta individuals (Fig. 2) do not match the morphological description of T. v. uhrigi (McCord et al., 2010). The postorbital stripe is the main stripe in the Colombian individuals, which is orange rather than yellow and at least 5x wider than that reported for T. v. uhrigi. The primary orbitocervical stripe has similar width in both taxa, but it is not the widest in the Colombian animals as it is in T. v. uhrigi. The color of the iris in the Colombian individuals is yellow, while in T. v. uhrigi it is bluish-green. The carapace of Colombian individuals is dark and the pattern seems to fade with size (thus presumably with age as well). The vertebral scutes have a dark solid blotch, with only one orange ring around the blotch, and do not exhibit additional incomplete rings as in T. v. uhrigi. The plastron in Colombian turtles exhibits a tear-like rounded and dark pattern, which is completely different from the swirling pattern of straight lines described for T. v. uhrigi.
Health assessment
We assessed the health status of the confiscated animals (n=90). We found 16 individuals with dermatitis in the plastron including erythema and ulcers, and 15 individuals had "X" marks on the carapace likely done with a sharp object. Four individuals had structural deformations in the carapace (hump-like), and one individual had lost the dermis of the entire carapace, which was replaced with a large burn-like scar. One individual was missing a phalanx of the left rear foot, and another had its carapace pierced in the eleventh left marginal scute.
The turtles were also palpated to determine if they were gravid. We estimated that at least 33 (36.6 %) individuals were gravid, however we did not confirm it with any other technique. The average body weight and LCL of these presumably gravid females were 1816 g (range = 1,100-3,160 mm) and 231 mm (range = 177-279 mm), respectively.
Two turtles died after the seizure, and were found to be gravid during necropsy. One of them had four fully developed eggs, though the number of eggs of the second female was not recorded. Morphometry of these two gravid females were: 1945 and 2220 g body weight, and 267 and 320 mm LCL, respectively.
DISCUSSION
Phenotype
The state of knowledge of Trachemys venusta distributed in Colombia is almost nonexistent. The only known report from Colombia is anecdotal and 50 years old (Medem, 1962). All other scientific literature on this species comes from populations in other countries like México (Alvarez del Toro, 1982; Vogt, 1990), Costa Rica (Acuña-Mesén, 1993; Savage, 2002), and Panamá (Moll and Legler, 1971). Our data on these confiscated sliders suggests that the average T. venusta inhabiting Colombia is smaller than sliders reported elsewhere. This is 231.7 mm LCL (this study) and 184.6 mm (Medem, 1962), compared to 262 mm for T. v. uhrigi (McCord et al., 2010) and up to 350 mm for females (Seidel, 2002). The maximum LCL in this report (280.7 mm) is similar to the maximum LCL previously reported for Colombia: 296 mm (Castaño-Mora, 1992; Castaño-Mora, 2002). This same individual also attained the heaviest weight, 3205 g.
The order in the relative size of the scutes of the plastron is different from what is reported in the literature for T. venusta. The first and second largest scute seams do not vary, but the third and fourth largest seams correspond to the pectoral (17.4 % of LPL) and femoral scutes (14 % of LPL). In other studies, the third and fourth largest scute seams are the femoral (14.6 %) and the gular (14.2 %) (Acuña-Mesén, 1993; McCord et al., 2010). The interpectoral seam is not as variable as the interabdominal or interanal seams (see standard deviations in Table 2); however it may also be explained by sexual shape differences, or even plastic responses due to environmental differences as shown in other aquatic turtles (Ceballos and Valenzuela, 2011; Ceballos et al., 2014). Thus, because all of these external features described in this group of Colombian T. venusta are different from those of T. venusta in other populations, we highlight the importance of including native individuals from Colombia in making additional studies on the phylogeny of this species with such a wide geographic distribution.
Conservation
The illegal trade of this species has already been reported (Castaño-Mora, 1992; Castaño-Mora, 2002; Bock et al., 2012). In her report, Castaño-Mora (1992) reported observations from the local people of thousands of T. venusta from the lowlands of the Atrato River being traded in sacs around the time of Holy Week. The locals also mentioned that the authorities retained a truck full of these turtles in Chigorodó in 1982 (Castaño-Mora, 1992), and 30 years later, the illegal trade of this species is still going on (this report).One of the main problems with the illegal trade is the uncertainty of the true site of origin of confiscated animals. The reported site of origin of the confiscated sliders in this study is the municipality of Lomas Aisladas, which along with Bajirá and Chigorodó have been reported as a reproductive site for this species (Castaño-Mora, 1992), and thus it is not surprising there is illegal trade of T. venusta in this region. As Castaño-Mora (1992) commented, this species has survived such excessive and sustained extraction for decades perhaps due to the richness of water bodies, and the remoteness and undeveloped infrastructure of this region. We strongly suggest reinforcing turtle trade surveillance especially in the municipalities of Lomas Aisladas, Bajirá and Chigorodó, before and during the Holly Week to diminish this unmanaged extraction of T. venusta.
Health
In addition to this unsustained trade, the overall poor health condition reflects the bad conditions that turtles suffer. The ulcerative lessions found in the plastrons are likely associated to overcrowding, injuries due to harsh floors, and bad water quality (Granados et al., 2013). Another lesion observed was a burn scar covering the entire carapace of an individual. Forest burning to expand the cattle frontier in the lower Atrato River region was previously mentioned as a threat for this species (Bock et al., 2012), and thus it is likely that such a fire may have been the cause of the scar in this individual.
CONCLUSIONS
In this study we reviewed the morphology of a group of sliders T. venusta confiscated in the Atrato River, Colombia, and found it smaller, with a different plastral inter-scute seam formula, and different body coloration patterns than the subspecies putatively distributed in Colombia, T. v. uhrigi. For this reason we strongly suggest further studies on the phylogenetic relationships of T. venusta throughout its distributional range including Colombia. This taxon, regardless of its designation, has been illegally harvested in this region of the country for several decades now and thus we call attention to a more effective control of such illegal trade in the Urabá region of Colombia.
ACKNOWLEDGMENTS
We thank the environmental police from Apartadó, Colombia, for logistic help with the confiscation of turtles, A. M. Ceballos for help during reproductive assessments, and A. M. Ceballos, G. Lopera, E. Graciano, J. M. Pedroza, and O. Morales for help during fieldwork. We also thank the Dirección de Regionalización, and the Sustainability Program 2013-2014 of the Universidad de Antioquia for research support to C. P. Ceballos.
REFERENCES
Acuña-Mesén RA. Las tortugas continentales de Costa Rica. 1 ed. San Pedro de Montes de Oca, Costa Rica: Editorial ICER; 1993. p. 45-52. [ Links ]
Alvarez del Toro M. Los reptiles de Chiapas, México. Chiapas, México: Instituto Zoológico del Estado; 1982. p. 12. [ Links ]
Andreu AC, Villamor C. Calendario reproductivo y tamaño de puesta en el galápago leproso, Mauremys leprosa (Schweigger, 1812) en Doñana, Huelva. Doñana. Acta Vertebr. 1989;16:167-172. [ Links ]
Bertolero A, Marin A. Efficacy of inguinal palpation for detecting oviductal eggs in Hermann's tortoise, Testudo hermanni. Amphib Reptil. 2005;26(4):523-526. DOI: 10.1163/156853805774806232. [ Links ]
Bock BC, Páez VP, Castaño-Mora OV. Trachemys venusta. In: Páz VP, Morales-Betancourt MA, Lasso CA, Castaño-Mora OV, Bock BC, editors. Biología y Conservación de las Tortugas Continentales de Colombia. Serie Editorial Recursos Hidrobiológicos y Pesqueros Continentales de Colombia. Instituto de Investigación de Recursos Biológicos y Pesqueros Alexander von Humboldt. Bogotá, D.C.; 2012. p. 292-297. [ Links ]
Cagle FR. A system of marking turtles for future identification. Copeia. 1939;3:170-173. [ Links ]
Castaño-Mora OV. Informe final del proyecto "Las tortugas de Colombia". II Fase. Universidad Nacional-Colciencias. Bogotá; 1992. p. 101-105. [ Links ]
Castaño-Mora OV, editor. Libro Rojo de Reptiles de Colombia. Instituto de Ciencias Naturales, Universidad Nacional, Ministerio del Medio Ambiente, Conservación Internacional-Colombia. Bogotá, D.C.; 2002. p. 102-105. [ Links ]
Ceballos CP, Fitzgerald LA. The trade in native and exotic turtles in Texas. Wildlife Soc B. 2004;32(3):881-892. [ Links ]
Ceballos CP, Hernandez P OE, Valenzuela N. Divergent sex-specific plasticity in long-lived vertebrates with contrastingsexual dimorphism. Evol Biol. 2014;41:81-98. DOI: 10.1007/s11692-013-9249-0. [ Links ]
Ceballos CP, Valenzuela N. The role of sex-specific plasticity in shaping sexual dimorphism in a long-lived vertebrate, the snapping turtle Chelydra serpentina. Evol Biol. 2011;38(2):163-181. DOI: 10.1007/s11692-011-9117-8. [ Links ]
Fritz U, Stuckas H, Vargas-Ramirez M, Hundsdorfer AK, Maran J, Packert M. Molecular phylogeny of Central and South American slider turtles: implications for biogeography and systematics (Testudines: Emydidae: Trachemys). J Zoolog Syst Evol Res. 2012;50(2):125-36. DOI: 10.1111/j.1439-0469.2011.00647.x. [ Links ]
Granados JL, Moreno OG, Brieva CI. Lesiones ulcerativas cutáneas en tortugas dulceacuícolas. Rev Med Vet Zoot. 2013;60(1):61-70. [ Links ]
Gong S-P, Chow AT, Fong JJ, Shi H-T. The chelonian trade in the largest pet market in China: scale, scope and impact on turtle conservation. Oryx. 2009;43(02):213-216. [ Links ]
IUCN. The IUCN Red List of Threatened Species. Version 2012.2 2012. Available from: http://www.iucnredlist.org. Downloaded on 14 July 2014. [ Links ]
Keller C. Assessment of reproductive state in the turtle Mauremys leprosa: a comparison between inguinal palpation and radiography. Wildlife Res. 1998;25(5):527-531. DOI: 10.1071/wr97013. [ Links ]
Martínez LS, Botero L. Estudio retrospectivo de la casuística de reptiles en el laboratorio de patología veterinaria de la Universidad Nacional de Colombia entre los años 1971 y 2008. Memorias de la Conferencia Interna en Medicina y Aprovechamiento de Fauna Silvestre, Exótica y no Convencional. 2011; p. 11-44. [ Links ]
McCord WP, Joseph-Ouni M, Hagen C, Blanck T. Three new subspecies of Trachemys venusta (Testudines: Emydidae) from Honduras, northern Yucatán (Mexico), and Pacific coastal Panama. Reptilia. 2010;71:39-49. [ Links ]
Medem F. La distribución geográfica y ecología de los Crocodylia y Testudinata en el Departamento del Chocó. Rev Acad Colomb Cienc Exact Fis Nat. 1962;11(14):279-303. [ Links ]
Moll EO, Legler JM. The life history of a neotropical slider turtle, Pseudemys scripta (Schoepff), in Panama. 1971; p. 102. [ Links ]
Rohlf FJ. TpsDig software, version 1.31. Department of Ecology and Evolution. State University of New York at Stony Brook, Stony Brook, New York,USA. 2001. [ Links ]
Rueda-Almonacid JV, Carr JL, Mittermeier RA, Rodríguez-Mahecha JV, Mast RB, Vogt RC, Rhodin AGJ, Ossa-Velásquez Jdl, Rueda JN, Mittermeier CG. Las tortugas y los cocodrilianos de los paises andinos del trópico. Serie de guías tropicales de campo N#°. 6. Conservación Internacional. Editorial Panamericana, Bogotá, Colombia; 2007. p. 538. [ Links ]
Savage JM. The amphibians and reptiles of Costa Rica: a herpetofauna between two continents, between two seas. University of Chicago Press; 2002. p. 934. [ Links ]
Seidel ME. Taxonomic observations on extant species and subspecies of slider turtles, genus Trachemys. J Herpetol. 2002;36(2):285-292. [ Links ]
TTWG. Turtles of the world, 2010 update: Annotated checklist of taxonomy, synonymy, distribution, and conservation status. Chelon Res Monogr. 2010;5:000.85-000.164. [ Links ] [ Links ]
TTWG. Turtles of the world, 2012 update: Annotated checkllist of taxonomy, synonymy, distribution, and conservation status. Chelon Res Monogr. 2012;5:000.243-000.328. [ Links ]
Vogt RC. Reproductive parameters of Trachemys scripta venusta in southern Mexico. In: Gibbons JW, editor. Life history and ecology of the slider turtle. Washington, D.C: Smithsonian Institution Press; 1990. p. 162-168. [ Links ]