Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Acta Biológica Colombiana
Print version ISSN 0120-548X
Acta biol.Colomb. vol.20 no.1 Bogotá Jan./Apr. 2015
https://doi.org/10.15446/abc.v20n1.40764
Artículo de investigación
In vitro EVALUATION OF THE ANTIBACTERIAL AND MODULATORY ACTIVITIES OF CORN AND SOY FIXED OILS
Evaluación in vitro de la actividad antibacteriana y moduladora de aceites de maíz y soja
Emanuele N. TAVARES1, Jacqueline C. ANDRADE1, Cícera Datiane De M. OLIVEIRA1, Saulo R. TINTINO1, Micheline A. LIMA2, Henrique D.M. COUTINHO1.
1 Universidade Regional do Cariri-URCA. Av. Cel. Antonio Luiz, 1161. Crato, CE, Brazil.
2 Universidade Federal da Paraíba, UFPB. Cidade Universitária, Campus I. Phone:. João Pessoa, PB, Brazil.
For correspondence. hdmcoutinho@gmail.com.
Received 29th November 2013, Returned for revision 9th April 2014, accepted 7th May 2014.
Citation / Citar este artículo como: Tavares EN, Andrade JA, Oliveira CDM, Tintino SR, Lima MA, Coutinho HDM. In vitro evaluation of the antibacterial and modulatory activities of corn and soy fixed oils. Acta biol. Colomb. 2015;20(1):27-30 .doi: http://dx.doi.org/10.15446/abc.v20n1.40764.
ABSTRACT
Nowadays, it is widely known that resistant pathogenic strains are becoming one of the greatest health problems related to human sciences. It is also known that fatty acids can present antimicrobial activity. The corn and soy oils are rich in fatty acids. In this regard, this work aimed to evaluate antibacterial and modulatory activity of these two industrial fixed oils. Both tests were performed using the microdilution method to Minimum inhibitory concentration under 1024 mg/ml. The aminoglycosides activity was enhanced by combining corn oil using E. coli 27. One of the mechanisms that might explain the synergism toward the corn oil, in part, might be due to the hidrophobic nature of the saturated and unsaturated lipids present in the sample. The results indicate that fixed oils might play a role as an alternative way to potentiate of the antimicrobial drugs.
Keywords: antibacterial activity, corn oil, modulation, soy oil.
RESUMEN
Hoy en día, es ampliamente conocido que las cepas patógenas resistentes a antibioticos se están convirtiendo en uno de los mayores problemas de salud en el campo de la ciencias de la salud. También se sabe que los ácidos grasos pueden presentar actividad antimicrobiana. Los aceites de maíz y de soja son ricos en ácidos grasos. En este sentido, esta investigacion dirigida para evaluar la actividad antibacteriana y modulador de estos dos aceites fijos usados industrialmente. Ambas pruebas se realizaron utilizando el método de microdilución de concentración inhibitoria mínima en 1,024 mg/ml. La actividad de los aminoglucósidos se mejoró mediante la combinación de aceite de maíz usando E. coli 27. Uno de los mecanismos que podrían explicar la sinergia hacia el aceite de maíz, en parte, podría ser debido a la naturaleza hidrofóbica de los lípidos saturados e insaturados presentes en la muestra. Los resultados indican que los aceites fijos podrían desempeñar un papel como una forma alternativa para potenciar de los fármacos antimicrobianos.
Palabras clave: actividad antibacterial, aceite de soja, aceite de maíz, modulación.
INTRODUCTION
The knowledge of the phenomena of the resistance against physical and chemical agents by the microorganisms is in history since the use of antimicrobial drugs (Walter et al., 2000). Nowadays, science knows that this resistance is acquired practically in all specie of bacteria, knowing these mechanisms in details about the resistance and molecular manifestation (Cunha et al., 1998; Walter et al., 2000). It is also known that the emergence of resistant and pathogenic strains is becoming a huge problem of public health.
In this regard, actually the pharmaceutical industry is searching new strategies to provide new antimicrobial drugs. One of the most common strategies used is change the structural formula of the drugs already in the market with the intention to potentiate them or recover their initial efficiency before the development of the bacterial resistance (Chartone-Souza, 1998).
It became known that lipids may present antimicrobial activity likewise the ones that are synthesized by the skin (Drake et al., 2008). It is well known also that essential oils present antimicrobial activities and sometimes modulate the antibiotic activity, as well as also known that fixed oils are rich in triacylglicerol esters and also are composed by saturated and insaturated fatty acids presente antimicrobial activity or maybe modulators of antibiotic activity (Coutinho et al., 2010; Cabral et al., 2013).
Oils are lipophylic substances originated from animal or vegetable kingdom formed by triacylglicerol esters besides to have another components in a smaller proportion, as mono and diacylglicerols, free fatty acids, glycopeptides, proteins, phosfolipids, steroids and vitamins (Gambarra Neto, 2008).
Soy oil is one of the most utilized worldwide. Considered healthy for containing unsaturated fatty acids in a large amount and presents a high level of linoleic (omega 6), oleic (omega 9) and linolenic acid (omega 3) (Mendonça, 2008; Yunes, 2010). While the corn oil, extracted from the corn grain, owns a favorable composition in terms of essential fatty acids like linoleic, oleic, palmitic, stearic and linolenic acids. It is considered to be of high quality (Mittelmann et al., 2006).
In this context, this work intend to evaluate the in vitro antimicrobial and modulatory activity of the industrialized corn and soy oils, due to the lipophilic character caused by the composition of saturated and unsaturated fatty acids.
MATERIAL AND METHODS
Plant Fixed Oils
The soy oil and corn oil used in this work were industrialized (Salada Brand®). The solution used in the tests was prepared to 100mg/mL dissolved in 1ml of dimetilsulfoxide, diluted in distillted water up to a concentration of 1024 mg/mL.
Bacterial Material
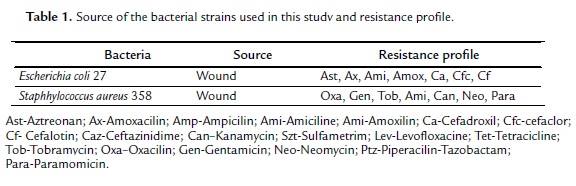
Bacterial strains used were of E. coli ATCC10536, S. aureus ATCC25923 and P. aeruginosa ATCC15442 to determine the minimum inhibition concentration and multiresistant strains of E. coli 27 and Staphylococcus aureus 358 for the test of modulation, resistant to several antibiotics (Table 1). The strains were maintained in heart infusion agar (HIA, Difco laboratories Ltda.). Before the assay, the strains were cultivated under 37#ºC in heart brain infusion (BHI,difco Laboratories Ltda.).
Drugs
The action of the antibacterial drugs utilized were aminoglycosides such as: amicacine, gentamycin, canamycin, neomycin (Sigma co., St. Louis, USA) to a concentration of 5000 mg/mL. All solutions were prepared according to the maker indications.
Antibacterial Activity Test and Antibiotic Activity Modulation
The minimum inhibitory concentration, the last concentration in which no growth is observed was determined in micro dilution assay. The inoculum was diluted in BHI 10% reaching 105 UFC/mL. Portions of BHI in amounts of 100 mL were separated in plaque containing 96 pits. After that serial micro dilution took place in 100 mL solution of soy and corn oils altering from 1024-2 mg/mL. The plaques were taken to the incubator for 24h under 35oC (Javadpour et al., 1996). The MIC was registered with the minor concentration capable to inhibit the growth.
To verify if the extract would modify the antibiotic action toward the strain tested, has been followed the work of Coutinho et al., (2008), was used a concentration sub-inhibitory (MIC/8). 100 mL of BHI was distributed, inoculum and solution of the oils of soy and corn in each pit following the alphabetic order in the plaque. Then, 100 mL of the drug was added to the first pit, proceeding with the micro dilution series, in a proportion of 1:1. The concentrations of aminoglycosides altered gradually from 5000-6 mg/mL. The revelation of bacterial MIC and modulation activity were done using a dye called resazurin. All assays were performed in triplicate.
RESULTS
The minimum inhibitory concentration (CIM) determined for both oils was ≤ 1024 mg/mL, not presenting antibacterial activity clinically relevant against tested strains.
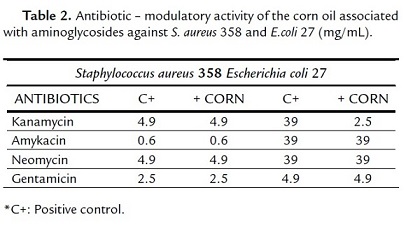
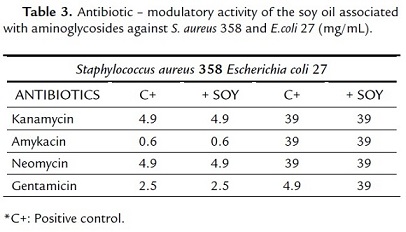
The absence of antibacterial activity clinically relevant, might be due to the fact that microorganisms utilized in the assays are strains that acquire resistance to the mechanism of modification of the membrane. However, this resistance was decreased when the association with the antibiotic took place. Then the association to the corn oil and aminoglycosides presented a synergic effect toward E. coli, where the increase of the antibiotic effect was observed, precisely with kanamycin, eight times fold (Table 2). Being that, with the drug the bacteria was inhibited in a concentration of 39 mg/mL when combined with the product in a concentration went down to 2.5 mg/mL, as can be noticed in table 2. Nevertheless the soy oil was incapable to modify the action of the strain (Table 3).
DISCUSSION
Despite what this work presents, there are reports of another fixed oils with antibacterial activity like the one observed in the seeds of "Camomila", utilizing a different method which is the paper diffusion using strains of Pseudomonas aeruginosa, Escherichia coli and Enterobacter aerogenes, observing the efficacy of the inhibitory effect more than that of the Chloranfenicol used as pattern (Pereira, 2008). Also, there are reports of the antibacterial activity of fixed oils of Ocimum sanctum L., against strains of Staphylococcus aureus, Bacillus oumilus and Pseudomonas aeruginosa, throught the diffusion in cavity method (Singh et al., 2005).
Is already known that the antibacterial activity of the fixed oils is due in part to the presence of fatty acids that may interact with the plasmatic membrane and alter its structure, acting as if it was a surfactant. Such acids as fatty or linoleic like were already cited in papers for acting like antibacterial agents (Kabara et al., 1972).
Several studies related antibacterial activity and modulation activity presented by the vegetable oils (Packer et al., 2007; Pinto, 2010). However, this is the first report of the antibacterial and modulation of the fixed oils of soy and corn, industrialized. Although there are reports of fixed oils from animal sources rich in saturated and unsaturated fat acids between them, linoleic acid (Cabral et al., 2013; Dias et al., 2013).
One of the mechanisms that can explain the synergic effect of the corn oil, in part, is in its hydrophobic nature constituted of saturated and unsaturated lipids. These can interact as a double lipoid layer of cellular membrane causing damages to the microorganism physiology, altering its respiratory chain and energy production, or mainly turning the cell permeate to exaggeration, so a lot of antibiotics would modify the enzyme systems leading to bacterial vital activities to failure (Nicolson et al., 1999; Burt, 2004).
The best result was observed against the Gram-negative bacteria for being majority in the membrane could be that reacted better with the fixed oil. Nevertheless, the antagonic action and the absence of modulation activity of the soy oil could be the fact that some typical component of these products would interact with the antibiotic and made the action impossible (Granowitz et al., 2008).
CONCLUSION
The results indicate that the fixed oils displayed an alternative way to potentiation of the action of the antimicrobial drugs when tested against Gram-negative bacteria. A synergism was observed in combination with tested drugs. These facts open a gateway to curiosity to future papers where this mechanism and activity could be better studied as long as tested in vivo.
ACKNOWLEDGEMENTS
The authors are grateful to the Brazilian research agencies FUNCAP and CNPQ.
REFERENCES
Burt S. Essential oils: their antibacterial properties and potential applications in foods –a review. Int J Food Microbiol, 2004;94(3):223-253. Doi: 10.1016/j.ijfoodmicro.2004.03.022. [ Links ]
Coutinho HDM, Costa JGM, Lima EO, Falcão-Silva VS, Siqueira-Júnior JP. In vitro interference of Momordica charantia in the resistance to aminoglycosides. Pharm Biol. 2009;47(11):1056–1059. Doi:10.3109/13880200902991540. [ Links ]
Coutinho HDM, Matias EFF, Santos KKA, Tintino SR, Souza CES, Guedes GMM, et al. Enhancement of the norfloxacin antibiotic activity by gaseous Contact with the essential oil of Croton zehntneri. J of Young Pharm. 2010;2(4):362-364. Doi: 10.4103/0975-1483.71625. [ Links ]
Cabral MES, Dias DDQ, Sales DL, Oliveira OP, Teles DA, Sousa JGG D, et al. Evaluations of the Antimicrobial Activities and Chemical Compositions of Body Fat from the Amphibians Leptodactylus macrosternum Miranda-Ribeiro (1926) and Leptodactylus vastus Adolf Lutz (1930) in Northeastern Brazil. Evid Based Complement Alternat Med. 2013;2013:1-7. Doi: 10.1155/2013/913671. [ Links ]
Chartone-Souza E. Bactérias ultra-resistentes: uma guerra quase perdida. Ciên Hoj. 1998;23(138):27-35. [ Links ]
Cunha BA. Antibiotic resistance. Drugs of Today (Barc). 1998;34(8):691-698. [ Links ]
Dias DDQ, Cabral MES, Sales DL, Paiva OO, Teles DA, Sousa JGG, et al. Chemical Composition and Validation of the Ethnopharmacological Reported Antimicrobial Activity of the Body Fat of Phrynops geoffroanus Used in Traditional Medicine. Evid Based Complement Alternat Med. 2013;2013:1-4. Doi: 10.1155/2013/715040. [ Links ]
Drake DR, Brogden KA, Dawson DV, Wertz PW. Thematic review series: skin lipids. Antimicrobial lipids at the skin surface. J Lipid Research. 2008;49:4-11. [ Links ]
Gambarra Neto FF. Classificação de óleos vegetais utilizando voltametria de onda quadrada e métodos quimiométricos. Dissertação de mestrado. Universidade Federal da Paraíba, João Pessoa; 2008. p. 64-51. [ Links ]
Granowitz EV, Brown RB. Antibiotic adverse reactions and drug interactions. Crit Care Clin. 2008;24(2):421–442. Doi: 10.1016/j.ccc.2007.12.011. [ Links ]
Javadpour MM, Juban MM, Lo WC, Bishop SM, Alberty JB, Cowell SM, et al. De novo antimicrobial peptides with low mammalian cell toxicity. J Med Chem. 1996; 39(16):3107–3113. [ Links ]
Kabara JJ, Swieczkowski DM, Conley AJ, Truant JP. Fatty acids and derivatives as antimicrobial agents. Antimicrob Agents Chemother. 1972;2(1):23-28. Doi: 10.1128/aac.2.1.23. [ Links ]
Mendonça MA, Borgo LA, Araújo WMC, Novaes MRCG. Alterações físico-químicas em óleos de soja submetidos ao processo de fritura em unidades de produção de refeição no Distrito Federal. Comu Ciên Saúd. 2008;19(2):115-122. [ Links ]
Mittelmann A, Miranda Filho JB, Lima GJMM, Hara-Klein C, Silva RM, Tanaka RT. Análise dialélica do teor de óleo em milho. Rev Bras de Agrociên, 2006; 12(2):139-143. [ Links ]
Nicolson K, Evans G, O'toole PW. Potentiation of methicillin activity againstmethicillin-resistant Staphylococcus aureus by diterpenes. FEMS Microbiol Lett. 1999;179(2):233–239. Doi: 10.1016/S0378-1097(99)00417-6. [ Links ]
Packer JF, Luz MMS. Método para avaliação e pesquisa da atividade antimicrobiana de produtos de origem natural. Braz J Pharmacogn. 2007;17:102-107. [ Links ]
Pereira NP. Estudo fitoquímico do óleo da semente de [camomilla recutita (l.) Rauschert] camomila, com avaliação de propriedades físico-químicas, biológicas e Funcionais em emulsões. Tese de doutorado. Universidade Federal do Paraná, Curitiba; 2008. p. 79-140. [ Links ]
Pinto DMLP. Avaliação da atividade antibacteriana do óleo essencial e extrato de Minthostachys setosa (Briq.) Epling. Dissertação de mestrado. Universidade de São Paulo, São Paulo; 2010. p. 28-51. [ Links ]
Singh S, Malhotra M, Majumdar DK. Antibacterial activity of Ocimum sanctum L. fixed oil. Indian J Exp Biol. 2005;43(9):835-837. [ Links ]
Yunes JFF. Avaliação Dos Efeitos Da Adição De Óleos Vegetais Como Substitutos De Gordura Animal Em Mortadela. Dissertação de mestrado. Universidade Federal de Santa Maria. Santa Maria; 2010. p. 47-51. [ Links ]
Walter T. Bactérias Gram-positivas problemas: resistência do estafilococo, do enterococo e do pneumococo aos antimicrobianos. Rev Soc Bras Med Trop.2000;33(3):281-301. Doi: 10.1590/S0037-86822000000300008. [ Links ]