Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Acta Biológica Colombiana
Print version ISSN 0120-548X
Acta biol.Colomb. vol.20 no.1 Bogotá Jan./Apr. 2015
https://doi.org/10.15446/abc.v20n1.43149
COMPORTAMIENTO DE OVIPOSICIÓN Y CARACTERIZACIÓN DE LA MICROESTRUCTURA CORIONICA DE LOS HUEVOS DE Simulium dinelliii (Joan) (Diptera: Simuliidae)
Oviposition Behavior and Microstructure of the Eggs of Simulium dinellii (Joan) (Diptera: Simuliidae)
Juan S. MANTILLA1, Ligia I. MONCADA2, Nubia E. MATTA 1, Peter H. ADLER3
1 Departamento de Biología, Facultad de Ciencias, Universidad Nacional de Colombia. Cr 30 n. 45-03, edificio 421, laboratorio 126. Bogotá, Colombia.
2 Departamento de Salud Pública, Facultad de Medicina, Universidad Nacional de Colombia. Bogotá, Colombia.
3 School of Agricultural, Forest & Environmental Sciences, Clemson University. Clemson SC, United States.
For correspondence: jsmantillag@unal.edu.co
Received 28t April 2014, Returned for 25th June revisión 2014, accepted 30th July 2014.
Citation / Citar este artículo como: Mantilla JS, Moncada LI, Matta NE, Adler PH. Oviposition behavior and microstructure of the eggs of Simulium dinellii (Joan) (Diptera: Simuliidae). Acta biol. Colomb. 2015;20(1):217-220. doi: http://dx.doi.org/10.15446/abc.v20n1.43149
ABSTRACT
Little is known about the oviposition habits and egg structure of Neotropical members of the Simuliidae family. The oviposition behavior of Simulium dinellii (Joan) was observed near at Otún Quimbaya, Colombia. Small groups of females hovered about 2 cm above the water surface to oviposit. Some of these females had been capture and the eggs were obtained. These eggs were counted, measured and processed by scanning electron microscopy. Subtriangular eggs had a latticework of polygons on the endochorionic surface. Chorionic differences between the eggs of S. dinellii and the few previously studied species suggest potential taxonomic value of egg microstructure.
Keywords: black flies, insect eggs, Otún Quimbaya, Simuliidae.
RESUMEN
El conocimiento sobre los hábitos de oviposición y estructura de los huevos de la familia Simuliidae es muy escaso. En este estudio se observó el comportamiento de oviposición de Simulium dinellii (Joan) en quebradas cercanas al Santuario de fauna y flora Otún Quimbaya, Colombia. El cual consistió en la formación de pequeños grupos de hembras que sobrevolaban a 2 cm de la superficie del agua para ovipositar, algunas de estas hembras fueron colectadas, obteniendo así los huevos, los cuales fueron contados, medidos y procesados por Microscopia Electrónica de Barrido con el fin de caracterizar la estructura endocoriónica. Los huevos presentaron una forma subtriangular, con una entramado poligonal sobre la superficie. Las características morfológicas observadas en los huevos de S. dinellii fueron diferentes a las previamente reportadas en otras especies de la familia Simuliidae, sugiriendo un posible valor taxonómico de la microestructura del huevo.
Palabras clave: jejenes, huevos de insectos, Otún Quimbaya, simúlidos.
The females of most species in the family Simuliidae are hematophagous, acquiring nutrients for egg maturation from vertebrate blood (Crosskey, 1990). Females use visual and chemical cues to select oviposition sites (Golini and Davies, 1988). Oviposition strategies include release of eggs while flying above water and placing eggs in groups while crawling on wetted substrates (Crosskey, 1990). A single female may use both strategies (McCreadie and Colbo, 1991). An alternative classification of oviposition recognizes three categories (Petry et al., 2006) (1) dispersion of small numbers of eggs with little or no gelatinous matrix (Pegoraro, 1993), (2) deposition of eggs in layers within a gelatinous matrix, and (3) oviposition in everal layers without clear boundaries, but with thousands of eggs in a gelatinous matrix originating from multiple females. Pheromones might be involved in mass oviposition (McCall et al., 1997).
Eggs are covered by an external filamentous or lamellar matrix (exochorion) that promotes adhesion to the substrate, protects the eggs, and provides food for newly hatched larvae (Goldie, 1982; Crosskey, 1990). Beneath the matrix is a waxy layer that prevents desiccation, followed by a tough envelope, the endochorion (Goldie, 1982).
Although 64 species of simuliids have been documented in Colombia (Mantilla et al., 2013; Adler and Crosskey, 2014), little is known about their oviposition habits. We describe the oviposition behavior and structure of the eggs of S. dinellii.
Samples were collected from 21 to 29 of December of 2011, from one stream near the road between the Otún Quimbaya wildlife refuge (4°43’N 75°33’W, 1885 m a.s.l.) and the El Cedral aquaculture station (4°42’N 75°31’W, 2119 m a.s.l.) in Risaralda State, Colombia. The region is part of the high sub-Andean forest belt (Cuatrecasas, 1958) or low humid montane rainforest (Holdridge, 1982); it is cold and humid with a mean annual temperature of 16.8°C and rainfall of 2638.5 mm. The streams were 30–40 cm wide and 10–100 cm deep with temperatures of 11.1–16.8°C (15.1±2.21) and pH of 7.06–8.34 (7.55±0.51).
Female simuliids flying over the streams were captured with a mouth aspirator and individually transferred to 15mL plastic Falcon tubes moistened with water from the same stream. Oviposition was induced by shaking the tubes. Eggs were stored on damp towels in ambient conditions for 1–2 days to promote maturation. Females and eggs then were fixed in 80% ethanol along with pupae from the same streams. Female flies were dissected and identified using the keys of Coscarón and Coscarón-Arias (2007). Ten eggs were dehydrated in an ethanol series followed by hexamethyldisilazane (Brown, 1993), mounted on a metal stub with double-sided adhesive tape, coated with gold, and imaged with an Hitachi scanning electron microscope Model TM-3000 at 15kV in a vacuum.
All ovipositing females were S. dinelliii, despite pupae of S. dinellii, S. ignescens Roubaud, and S. roquemayu was observed in the same stream. Oviposition behavior at one site, a 30-cm waterfall with no direct sunlight, began with slow circular flight of four–seven females above the waterfall. All females approached the water to drop their eggs while in flight. Similar oviposition behavior occurs in the related S. ochraceum complex (Rodríguez-Pérez, 2003).
The process took several seconds, with multiple iterations, and was observed for 3 days from 11:00 to 15:00; females did not oviposit before or after this window. The timing of oviposition typically is guided by light intensity, at least in some Nearctic species, occurring predominantly in the evening or morning (Corbet, 1967; Imhoff and Smith, 1979). According to Rodríguez-Pérez et al.,(2003), some species of Neotropical black flies, such as members of the S. ochraceum complexand S. dinellii, deposit eggs in midday.
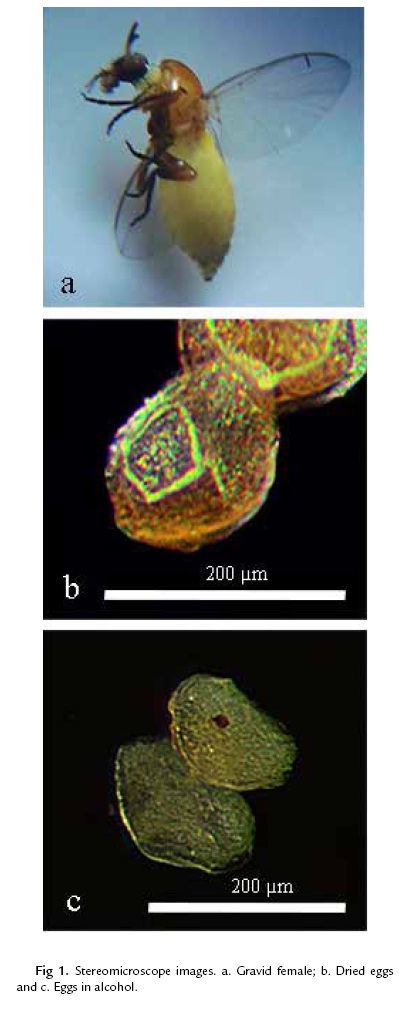
Females (n =13) (figure 1a) collected in flight immediately oviposited on the wet sides of the plastic tubes in which they were placed. Each female deposited 1 or 2 eggs per minute, pausing for about 2 minutes each time before repeating the process. The eggs were covered in a gelatinous matrix. The number of eggs produced in the tubes per female was 127– 140, with an average of 129 (n = 12). The number of eggs produced by simuliids varies from 20 to more than 1100, depending on the species, larval nutrition, water temperature during larval development, size of the female, and quantity and quality of blood consumed (Crosskey, 1990; Adler et al., 2004). The total number of eggs produced by females of S. dinellii is still unknown. Although the number of eggs deposited in the plastic tubes is within the range of that recorded by Takaoka (1981) for the S. ochraceum complex captured while ovipositing, we assume that the females might have deposited some eggs before they were captured, so that we do not have the information on the total number of eggs produced.
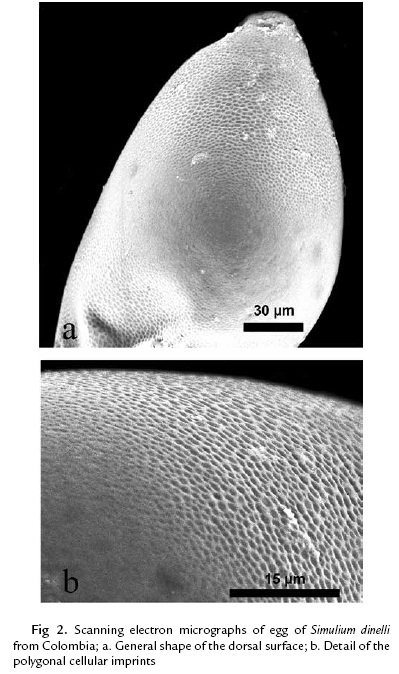
Freshly laid eggs were creamy yellow, darkening as they matured (figure 1b; 1c). The eggs (n = 13) were subtriangular, more so than for S. cormonsi (Moncada et al., 2013), and were 181–193 µm (188±7.5) long and 98–131 µm (108±13.9) wide. The chorionic surface was marked by a latticework of polygonal cellular imprints with salient borders and a slight indentation in the middle of each imprint. The shape and size of the imprints varied, with those nearest the poles larger and better defined, and those in the middle third of the egg becoming imperceptible so that the surface appeared nearly smooth (Fig. 2a; 2b).
The microstructure of the endochorion of S. dinellii contrasts with that of Simulium vittatum (Goldie, 1982), which has smoother eggs with imperceptible microsculpture and a well-developed micropyle. Thus, surface patterns of the endochorion might be characteristic at certain taxonomic levels. The absence of a micropyle is not unique to S. dinellii. The European S. posticatum Meigen also lacks a micropyle, the sperm presumably entering the egg enzymatically (Crosskey, 1990). Egg structure is a diagnostic feature among species of other dipteran families, such as the Culicidae (Estrada et al., 2003), but its potential for distinguishing species of simuliids is unexplored.
ACKNOWLEDGEMENTS
This work was supported by ECOPETROL, Colciencias, and Colombia National University. Logistical support and field accommodations were provided by the Special Administrative Unit of the Colombian National Natural Park system and the aqueduct company Aguas y Aguas of Pereira. JSM was supported for 1 month in the laboratory of PHA by a grant from Colciencias and from the National University of Colombia. We thank C. E. Beard for technical assistance with imaging.
REFERENCES
Adler PH, Crosskey RW. World blackflies (Diptera: Simuliidae): a comprehensive revision of the taxonomic and geographical inventory. 2014. 122 p. Available in : http://www.clemson.edu/cafls/biomia/pdfs/blackflyinventory.pdf (Accessed 8 March 2014). [ Links ]
Adler PH, Currie DC, Wood M. The black flies (Simuliidae) of North America, Cornell University Press, Ithaca, NY; 2004. p. 230. [ Links ]
Brown BV. A further chemical alternative to critical-point drying for preparing small (or large) flies. Fly Times. 1993;11:10. [ Links ]
Corbet PS. The diel oviposition periodicity of the black fly, Simulium vittatum. Can J Zool. 1967;45:583-584. Doi: 10.1139/z67-070. [ Links ]
Coscarón S, Coscarón Arias CL. Neotropical Simuliidae (Diptera: Insecta). In: Adis J, Arias JR, Rueda-Delgado G, Wantzen KM, editors. Aquatic Biodiversity in Latin America. Volume 3. Pensoft, Sofia, Bulgaria; 2007. p. 415-416. [ Links ]
Crosskey RW. The natural history of blackflies. John Wiley & Sons Ltd., Chichester, Englan; 1990. p. 447-457. [ Links ]
Cuatrecasas J. Aspectos de la vegetación natural de Colombia. Rev Acad Col Cs Ex Fis Nat. 1958;10(40):221-268. [ Links ]
Estrada DA, Quiñoes ML, Sierra DM, Calle DA, Ruiz F, Erazo HF, et al. Utilidad de la morfología de los huevos como un método indirecto para identificar Anopheles benarrochi Gabaldón, Cova García & López, Anopheles oswaldoi (Peryassu) y Anopheles rangeli Gabaldón, Cova García & López, (Diptera: Culicidae) en Putumayo, Colombia. Biomedica. 2003;23(4):388-95. Doi: 10.7705/biomedica.v23i4.1232 [ Links ]
Goldie P. Progress toward cryopreservation of black fly eggs: in vitro experiments and ultrastructure observations (M. S. thesis). Cornell University, Ithaca, New York. 1982. p. 198. [ Links ]
Golini VI, Davies DM. Oviposition of black flies. In: Kim KC, Merritt RW, editors. Black flies: ecology, population management, and annotated world list. Pennsylvania State University, University Park, Pennsylvania; 1988. p. 261-275. [ Links ]
Holdridge LR. Ecología basada en zonas de vida. Instituto Interamericano de Cooperación para la Agricultura, San Jose Costa Rica; 1982. p.216. [ Links ]
Imhof JE, Smith SM. Oviposition behaviour, egg-masses and hatching response of the eggs of five Nearctic species of Simulium (Diptera: Simuliidae). Bull Entomol Res Research. 1979;69(03):405-25. Doi: 10.1017/S0007485300018939. [ Links ]
Mantilla JS, Moncada LI, Matta NE, Adler PH. Two new species of black flies (Diptera: Simuliidae) from the High Andes of Colombia. Zootaxa. 2013;3700(3):423-34. Doi: 10.11646/zootaxa.3700.3.6. [ Links ]
McCall PJ, Heath RR, Dueben BD, Wilson MD. Oviposition pheromone in the Simulium damnosum complex: biological activity of chemical fractions from gravid ovaries. Physiol Entomol. 1997;22:224-230. [ Links ]
McCreadie JW, Colbo MH. Spatial distribution patterns of larval cytotypes of the Simulium venustum/verecundum complex (Diptera: Simuliidae) on the Avalon Peninsula, Newfoundland: factors associated with occurrence. Can J Zool. 1991;69(10):2651-9. Doi: 10.1139/z91-373. [ Links ]
Ligia MA, Mantilla S, Lotta I, Matta N, Adler PH. Simulium cormonsi Wygodzinsky (Diptera: Simuliidae) in Colombia: First record, egg and larval descriptions, and bionomics. Proc Entomol Soc Wash. 2013;5(1):103-6. Doi: 10.4289/082.115.0101. [ Links ]
Pegoraro RA . Ciclo biológico de Simulium (Chirostilbia) pertinax Kollar, 1832 (Diptera: Simuliidae). An Soc Entomol Bras. 1993;22:29-37. [ Links ]
Petry F, Lozovei AL, Chahad-Ehlers S. Eggs fertility and biological aspects of the life cycle in natural populations of Simulium (Diptera, Simuliidae). Brazil Arch Biol Technol. 2006;49(5):799-805. [ Links ]
Rodríguez-Pérez MA, Valdivieso-López NL, McCall PJ. Aggregated oviposition in Simulium ochraceum s. l. (Diptera: Simuliidae), an important Neotropical vector of Onchocerca volvulus. Ann Trop Med Parasito. 2003;l(97):203-207. Doi: 10.1179/000349803235001534.
Takaoka H. Seasonal occurrence of Simulium ochraceum, the principal vector of Onchocerca volvulus in the southeastern endemic area of Guatemala. Am J Trop Med Hyg. 1981;30(5):1121-32. [ Links ]