Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Acta Biológica Colombiana
Print version ISSN 0120-548X
Acta biol.Colomb. vol.20 no.2 Bogotá May/Aug. 2015
https://doi.org/10.15446/abc.v20n2.42830
Doi: http://dx.doi.org/10.15446/abc.v20n2.42830
Artículo de investigación
EFFECT OF EXOGENOUS PROLINE IN TWO SUGARCANE GENOTYPES GROWN IN VITRO UNDER SALT STRESS
Efecto de la prolina exógena en dos genotipos de caña de azúcar cultivados in vitro bajo estrés salino
Maria Jaislanny L. MEDEIROS1, Marina Medeiros De A. SILVA1, Manuela Maria C. GRANJA2, Gilberto De SOUZA E SILVA JÚNIOR3, Terezinha CAMARA4, Lilia WILLADINO4.
1Universidade Federal de Pernambuco-UFPE. Av. Prof. Moraes Rego, 1235-Cidade Universitária. Recife-PE, Brasil.
2Universidade Federal de Viçosa-UFV. Av. Peter Henry Rolfs, Campus Universitário. Viçosa-MG, Brasil.
3Instituto Federal de Educação de Pernambuco-IFPE. Av. Prof. Luís Freire, 500-Cidade Universitária. Recife-PE, Brasil.
4Universidade Federal Rural de Pernambuco-UFRPE. Rua Dom Manoel de Medeiros, Dois Irmãos. Recife-PE, Brasil.
For correspondence. jaislanny@yahoo.com.br
Received: 27 March 2014; Returned for revision: 15 September 2014; Accepted: 16 December 2014.
Associate Editor: Francisco José Martínez Pérez.
Citation / Citar este artículo como: Medeiros MJL, Silva MMA, Granja MMC, De Souza e Silva Júnior G, Camara T, Willadino L. Effect of exogenous proline in two sugarcane genotypes grown in vitro under salt stress. Acta biol. Colomb. 2015;20(2):57-63. doi: http://dx.doi.org/10.15446/abc.v20n2.42830
ABSTRACT
Sugarcane (Saccharum officinarum L.) production is influenced by various abiotic stresses, including salt stress. Salinity can induce oxidative stress, which in turn damages biomolecules and cellular structures. However, such damage can be avoided or minimized by the enzymatic and non-enzymatic defense systems of plants. The aim of the study was to determine the effect of exogenous proline on Na+ and K+ content and on the activity of catalase, ascorbate peroxidase and peroxidase in two sugarcane genotypes (RB931011 and RB872552) grown in vitro under salt stress. The plants were grown for 20 days with or without 100 mM NaCl and exposed to 20 mM proline for varying period of time. The data were subjected to ANOVA factorial (two genotypes and five treatments), and the mean values were compared by Tukey test at a 5 % probability. Under salinity stress, both genotypes exhibited membrane integrity reduction, reduced total soluble protein content and unaltered or increased endogenous proline content. Exogenous proline reduced Na+ accumulation in a manner proportional to the exposition period at the amino acid. In both genotypes, antioxidant enzymes activity increased with the addition of NaCl. In conclusion, RB931011 genotype showed higher proline accumulation and increased in activity of the antioxidant enzymes, indicating better salt stress tolerance than in RB872552 genotype.
Keywords: antioxidant enzymes, oxidative stress, Saccharum officinarum L.
RESUMEN
La producción de caña de azúcar (Saccharum officinarum L.) está influenciada por varios factores abióticos desfavorables, como el estrés salino. La salinidad puede inducir el estrés oxidativo, que causa daño a las biomoléculas y a las estructuras celulares. Sin embargo, estos daños se puede evitar o minimizar por los sistemas de defensa enzimático y no enzimáticos de las plantas. El objetivo del estudio fue determinar el efecto de la prolina exógena en el contenido de Na+ e K+, y la actividad de la catalasa, ascorbato peroxidasa y peroxidasa en dos genotipos de caña de azúcar (RB931011 y RB872552) cultivados in vitro bajo estrés salino. Las plantas se cultivaron durante 20 días en presencia o ausencia de NaCl 100 mM y expuestos a prolina 20 mM durante un periodo de tiempo variable. Los datos fueron sometidos a ANOVA factorial (dos genotipos y cinco tratamientos), y las medias se compararon mediante la prueba de Tukey al 5 % de probabilidad. Bajo estrés salino, ambos genotipos mostraron reducción en la integridad de la membrana, disminución de las proteínas solubles totales y el mantenimiento o incremento en el contenido de prolina endógena. La prolina exógena reduce la acumulación de Na+ en proporción al período de exposición al aminoácido. En ambos genotipos, la actividad de las enzimas antioxidantes se incrementó con la adición de NaCl. En conclusión, el genotipo RB931011 mostró mayor acumulación de prolina y un mayor aumento de la actividad de la catalasa, ascorbato peroxidasa y peroxidasa, lo que indica una mejor tolerancia al estrés salino en comparación con el genotipo RB872552.
Palabras clave: enzimas antioxidantes, estrés oxidativo, Saccharum officinarum L.
INTRODUCTION
Sugarcane (Saccharum officinarum L.) is a major crop species in Brazil, largely reflecting the high sugar and ethanol production in the country (Dibax et al., 2013). Brazil is the largest sugarcane producer worldwide, with production of 589 million tons in the 2012/2013 harvest (CONAB, 2013). Sugarcane is traditionally cultivated in humid forest zones and in the northeastern shore region of Brazil, but the cultivation area has expanded into semi-arid regions.
Irrigated agriculture in semi-arid zones frequently leads to soil salinity problems, and about half of the existing irrigation systems worldwide are affected by salinization. Salinity reduces plant growth due to its osmotic and ionic effects on the soil solution. These effects ultimately result from complex interactions among various morphological, physiological and biochemical processes (Munns et al., 2006), including inhibition of protein synthesis and changes in ion balance, water status, mineral nutrition, stomatal behavior and photosynthetic efficiency (Shaheen and Hood-Nowotny, 2005).
Stress caused by NaCl excess induces metabolic changes in plants and initiates oxidative stress, characterized by the overproduction of reactive oxygen species (ROS), which are highly reactive and toxic and cause damage to proteins, lipids, carbohydrates and DNA. The ROS comprises both free radical (O2•-, superoxide radicals; OH•, hydroxyl radical and HO2•, perhydroxy radical) and non-radical (molecular) forms (H2O2, hydrogen peroxide and 1O2, singlet oxygen) (Gill and Tuteja, 2010). Stress-induced ROS accumulation is counteracted by the prevention or elimination of ROS formation and by ROS scavenging performed by both enzymatic (Silva et al., 2014) and non-enzymatic antioxidants (Keunen et al., 2013).
Plant cells are protected from the toxic effects of ROS through the activity of antioxidant enzymes, such as catalase (CAT – EC 1.11.1.6), ascorbate peroxidase (APX – EC 1.11.1.11) and peroxidase (POD – EC 1.11.1.7), as well as by synthesis and accumulation of osmoprotectants such as proline (Pro) (Reddy, 2004; Gill and Tuteja, 2010). In fact, increased of exogenous proline may favor salt stress tolerance (Ashraf and Foolad, 2007). Pro has been proposed to act as an osmoprotectant, a protein stabilizer, a metal chelator, an inhibitor of lipid membrane peroxidation, maintaining the membrane integrity, and as a ROS scavenger (Trovato et al., 2008). Therefore, proline is not only an important redox signaling molecule, but also an effective quencher of ROS formed under salt stress conditions in plants (Ashraf and Foolad, 2007).
The aim of the present study was to evaluate the effects of exogenous proline on Na+ and K+ content and on antioxidant enzyme activity in two sugarcane genotypes (RB931011 and RB872552) grown in vitro under salt stress conditions.
MATERIALS AND METHODS
The plants were provided by the Center for Strategic Technologies of the Northeast (CETENE). The genotypes evaluated were RB931011, which exhibits water stress tolerance, excellent performance in sandy soils and rapid vegetative growth, and RB872552, which exhibits high agricultural productivity and high sugar production (RIDESA, 2010).
The MS medium (Murashige and Skoog, 1962) used for plant cultivation was supplemented with 30 g L-1 sucrose and 0.3 mg L-1 BAP (6-benzyl aminopurine). The pH was adjusted to 5.8, and the medium was autoclaved for 20 minutes at 120 °C. The two genotypes plants were grown in glass flasks containing 30 mL MS medium and incubated for eight weeks. The cultures were maintained in a growth chamber under white fluorescent light with a photon flux density of 50 µmol m-2 s-1, a 16/8 light/dark regime and a temperature of 25 ± 2 °C.
Were established five treatments with varying stress conditions: T0 (control group), without NaCl and without proline; T1, plant immersion in 20 mM proline for 24 hours prior to culture in medium containing 100 mM NaCl; T2, plant immersion in 20 mM proline for 72 hours prior to culture in medium containing 100 mM NaCl; T3, culture in medium containing 100 mM NaCl; and T4, culture in medium containing 20 mM proline and 100 mM NaCl. The plants were subjected to these treatments for 20 days, then frozen with liquid nitrogen and stored at -20 °C until achievement the chemical and biochemical analysis.
Membrane integrity was assessed by measuring electrolyte leakage using a conductivity meter (LFT 613T, Schott Geratie). Leaf segments were maintained in 30 mL distilled water in a test tube for 24 hours, prior to the first measurement of free conductivity (L1). The total conductivity was performed one hour later in test tubes that were immersed in a 100 °C water bath (L2). The percentage of membrane damage (PD) was estimated using the following equation: % PD = (L1/L2) x 100 (Costa et al., 2011).
The total soluble proteins content (Bradford, 1976) and proline content (Bates et al., 1973) were quantified using a delta absorbance spectrophotometer, 595 and 520 nm, respectively. The Na+ and K+ ion contents were determined using a flame photometer (B462, Micronal) (Malavolta et al., 1997).
The enzyme extract was prepared by homogenizing 0.1 g fresh matter in 4 mL 0.1 M sodium phosphate as buffer (pH 6.5) with 0.05 g polyvinylpyrrolidone (PVP). The homogenate was centrifuged at 10,000 x g at 4 °C for 10 minutes. The activity of the following antioxidant enzymes was measured: CAT (Berrs and Sizer, 1952), APX (Nakano and Asada, 1981) and POD (Kar and Mishra, 1976), using a delta absorbance spectrophotometer, 240, 290 and 470 nm, respectively.
A completely randomized 2 x 5 factorial design was used for the experiment (two genotypes and five different treatments with or without NaCl and exposed to 20 mM proline for varying period of time). There were 20 replicates per treatment, with each replicate consisting of five plants per flask. The data were subjected to ANOVA, and the mean values were compared by Tukey test at a 5 % probability, using the ASSISTAT program.
RESULTS
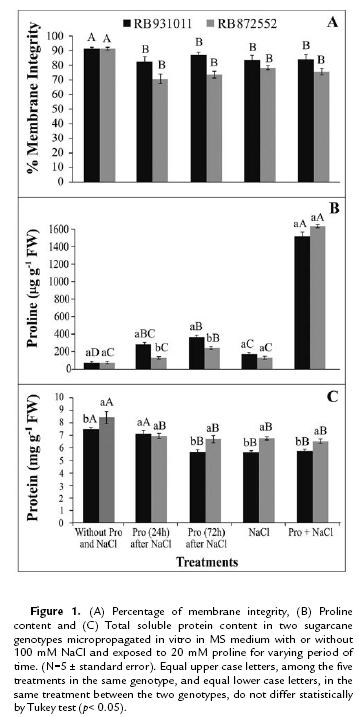
Salt stress compromised membrane integrity in both genotypes (Fig. 1A). Furthermore, proline failed to minimize the membrane damage caused by NaCl, regardless of the method used for of this amino acid application. Salinity stress induced maximum injury percentages of 19 % in the RB872552 genotype and only 8 % in RB931011.
Adding proline to the culture medium increased the endogenous proline content by more than 10-fold in both genotypes (Fig. 1B). With the exception of plants immersed in Pro for 72 hours, RB872552 plants did not accumulate proline in response to the addition of NaCl to the culture medium. In contrast, RB931011 plants exhibited an increase in endogenous proline content under all salinity treatment conditions when compared to control (without Pro and NaCl). For both genotypes, the total soluble protein content was highest in plants cultivated without NaCl (Fig. 1C).
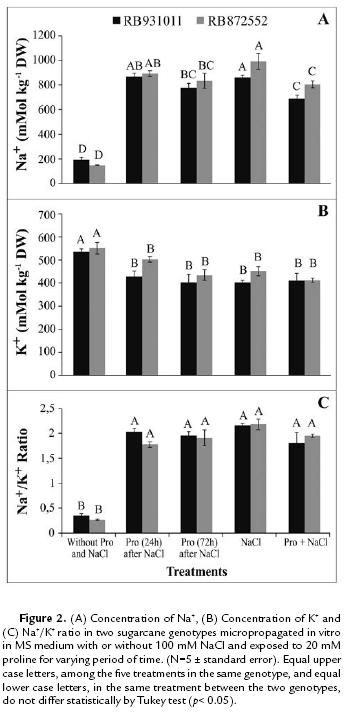
Exogenous proline reduced Na+ accumulation in plants in a manner proportional to the exposition period at the amino acid. Plants subjected to NaCl stress without exogenous proline treatment exhibited the highest Na+ contents (Fig. 2A). In RB872552 and RB931011, Na+ content increased 4-fold and 3.5-fold, respectively, relative to the control. In both sugarcane genotypes analyzed in the present study, reduced K+ content was observed in plants subjected to salt stress relative to the control (Fig. 2B). The combination of increased Na+ and reduced K+ content drastically increased the Na+/K+ ratio (Fig. 2C).
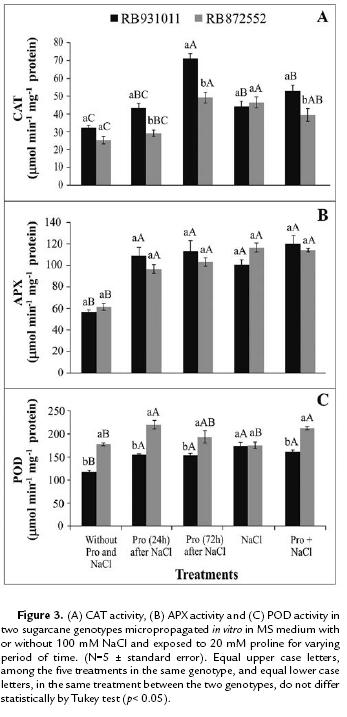
The enzymatic activity of CAT, APX and POD increased in response to salinity stress. Although increased CAT activity was observed in both genotypes when NaCl was present in the culture medium, the increase was greater in the RB931011 genotype than in RB872552 (Fig. 3A). APX activity similarly increased in both genotypes when the plants were subjected to salt stress (Fig. 3B).
Regardless of the presence of proline, the addition of NaCl to the culture medium increased POD activity in the RB931011 genotype. In RB872552, however, higher levels of POD activity were observed even in the control plants, indicating that this genotype is characterized by constitutively high POD activity. POD activity in RB872552 were similar in the control, in plants subjected to NaCl treatment only and in plants pre-treated with proline for 72 hours (Fig. 3C).
DISCUSSION
Electrolyte leakage was found in both of the sugarcane genotypes analyzed, which indicates membrane integrity damage, this leakage has also been observed in wheat leaves subjected to salt stress (Mandhania et al., 2006). Leakage values reflect the ability of a membrane to take up and retain solutes and thus minimize changes in membrane potentials and membrane permeability (Liu et al., 2006). In fact, cellular membrane stability is widely used to differentiate between sensitive and tolerant genotypes (Asrar et al., 2012).
Proline accumulation in response to salinity stress has been observed in several monocot species, such as sorghum (Lacerda et al., 2003), rice (Lima et al., 2004) and sugarcane (García and Medina, 2003). Specifically, sugarcane has been found to accumulate proline in leaves and roots when subjected to 100 mM NaCl treatment for 60 days (García and Medina, 2003). The proline content increase previously observed in RB855156 sugarcane leaf discs under osmotic stress appeared to correspond more to antioxidative defense than to osmotic adjustment mediation (Molinari et al., 2007). Pro has also been implicated in the maintenance of cytoplasmic pH, carbon and nitrogen storage, protein stabilization and ROS scavenging activity (Trovato et al., 2008).
The decreased protein content in plants under salt stress may reflect the inhibition of protein synthesis, increased degradation or the inhibition of amino acid incorporation into proteins, resulting in the accumulation of free amino acids (Piza et al., 2003). Given that the protein content of RB931011 and RB872552 plants decreased while proline content increased or remained unchanged under salt stress conditions, one may conclude that part of the endogenous proline content was attributable to proteolysis or the inhibition of protein synthesis.
As noted in this study, reduced Na+ absorption following Pro application has been reported in rice (Sobahan et al., 2009) and in tomato (Bruria, 2003). Thus, exogenous proline absorbed by plants may favor osmotic adjustment processes and lead to reduced inorganic ion accumulation (Bruria, 2003). K+ deficiency can induces metabolic damage. This ion is involved in the ion balances cytoplasm charges, its ability to activate vital enzymatic reactions further contributes to the maintenance of osmotic potential and turgor pressure in cells, and it is essential in protein synthesis (Tester and Davenport, 2003).
Under salt stress, increased enzymatic activity of CAT, APX and POD were observed. These antioxidative enzymes have important roles in eliminating ROS, such as superoxide radicals (O2º-) and hydrogen peroxide (H2O2), which are products of NaCl-induced oxidative stress. ROS damages biomolecules, including DNA, photosynthetic pigments, lipids, proteins and carbohydrates, in a variety of ways (Gill and Tuteja, 2010). In turn, this damage induces several degenerative processes, such as membrane lipid peroxidation and programmed cellular death (Del Rio and Puppo, 2009). As such, increased antioxidative capacity is positively related to salt stress tolerance (Mandhania et al., 2006; Willadino et al., 2011).
Higher CAT activity is frequently observed in salt-tolerant genotypes (Willadino et al., 2011). Furthermore, stress analyses have shown increased susceptibility of CAT-deficient plants to salt and ozone stress (Sharma et al., 2012). CAT has a very high turnover rate but a much lower affinity for H2O2 than APX. This low affinity for H2O2 suggests that CAT may be responsible for the removal of excess ROS generated under stress conditions (Mittler, 2002; Mhamdi et al., 2012).
In general, large increases in APX activity are characteristic of plant varieties with increased NaCl tolerance (Mandhania et al., 2006, Willadino et al., 2011). The affinity of APX (μM range) for H2O2 (Sharma et al., 2012) suggests that APX may be responsible for the fine modulation of ROS for signaling (Mittler, 2002). APX is characterized by high ascorbate specificity and represents the enzyme for the elimination of H2O2 toxicity in the chloroplasts and cytosol of vegetal cells through the oxidation of ascorbate to monodehydroascorbate (Del Rio and Puppo, 2009).
Increased POD activity was previously reported in a salt-tolerant wheat genotype under NaCl treatment at 50 mM and 100 mM (Khan and Panda, 2008). Similar results were observed in a salt-tolerant rice genotype treated with 50 mM, 100 mM and 150 mM NaCl (Mandhania et al., 2006). The increased activity of POD under salt stress conditions highlight the efficiency of the H2O2 removal mechanism, which promotes higher tolerance to NaCl-induced oxidative stress (Agarwal and Pandey, 2004).
CONCLUSION
In both of the sugarcane genotypes analyzed, exogenous proline reduced Na+ accumulation in a manner proportional to the exposition period at the amino acid. Under salt stress, both genotypes exhibited increased CAT, APX and POD activity. However, the increase in activity of the antioxidant enzymes was greater in the RB931011 genotype, which was also found to accumulate higher levels of proline than the RB872552 genotype. Taken together, the antioxidant activity profiles and the lower reduction in membrane integrity in RB931011 indicate a higher salinity tolerance in this genotype.
ACKNOWLEDGEMENTS
The first author is grateful to Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the scholarship received. The authors are grateful to Biofábrica Governador Miguel Arraes–Centro de Tecnologias Estratégicas do Nordeste (CETENE) for giving the plant material used in this study.
REFERENCES
Agarwal S, Pandey V. Antioxidant enzyme responses to NaCl stress in Cassia angustifolia. Biol Plantarum. 2004;48(4):555-560. Doi: http://dx.doi.org/10.1023/B:BIOP.0000047152.07878.e7 [ Links ]
Ashraf M, Foolad MR. Roles of glycine betaine and proline in improving plant biotic stress resistance. Environ Exp Bot. 2007;59(2):206-216. Doi: http://dx.doi.org/10.1016/j.envexpbot.2005.12.006. [ Links ]
Asrar AA, Abdel-Fattah GM, Elhindi KM. Improving growth, flower yield, and water relations of snapdragon (Antirhinum majus L.) plants grown under well-watered and water-stress conditions using arbuscular mycorrhizal fungi. Photosynthetica. 2012;50(2):305-316. Doi: http://dx.doi.org/10.1007/s11099-012-0024-8. [ Links ]
Bates LS, Waldron RP, Teare ID. Rapid determination of free proline for water-stress studies. Plant Soil. 1973;39(1):205-207. Doi: http://dx.doi.org/10.1007/BF00018060. [ Links ]
Berrs LSJ, Sizer IW. A espectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. J Biol Chem. 1952;195:133-140. [ Links ]
Bradford MM. A rapid and sensitive method for the quantization of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248-254. Doi: http://dx.doi.org/10.1016/0003-2697(76)90527-3. [ Links ]
Bruria H. Influence of exogenous application of proline and glycinebetaine on growth of salt stressed tomato plants. Plant Sci. 2003;165(4):693-699. Doi: http://dx.doi.org/10.1016/S0168-9452(03)00222-X. [ Links ]
CONAB [Online]. Companhia Nacional de Abastecimento. Levantamentos de Safra: levantamento Cana-de-açúcar. Recife, dez. 2013. [Acessed: 2013 dez 12]. Available from: URL: http://www.conab.gov.br/conteudos.php?a=1253&t=. [ Links ]
Costa AS, Nogueira LC, Santos VF, Finger FL, Camara TR, Loges V, et al. Characterization of symptoms of senescence and chilling injury on inflorescences of Heliconia bihai (L.) cv. Lobster Claw and cv. Halloween. Postharvest Biol Tec. 2011;59(1):103-109. Doi: http://dx.doi.org/10.1016/j.postharvbio.2010.08.015. [ Links ]
Del Rio La, Puppo A. Reactive Oxygen Species in Plant Signaling, Signaling and Communication in Plants. New York: Springer-Verlag Berlin Heidelberg; 2009. 25p. [ Links ]
Dibax R, Alcantara GB, Machado MP, Bespalhok-Filho JC, Oliveira RA. Protocol optimization and histological analysis of in vitro plant regeneration of 'RB92579' and 'RB93509' sugarcane cultivars. Cienc Rural. 2013;43(1):49-54. Doi: http://dx.doi.org/10.1590/S0103-84782012005000138. [ Links ]
García M, Medina E. Crecimiento y acumulación de prolina en dos genotipos de caña de azúcar sometidos a salinización con cloruro de sodio. Rev Fac Agron. 2003;20(2):168-179. [ Links ]
Gill SS, Tuteja N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem. 2010;48(12):909-930. Doi: http://dx.doi.org/10.1016/j.plaphy.2010.08.016. [ Links ]
Kar M, Mishra D. Catalase, peroxidase, and polyphenoloxidase activities during pice leaf senescence. Plant Physiol. 1976;57(2):315-319. Doi: http://dx.doi.org/10.1104/pp.57.2.315. [ Links ]
Keunen E, Peshev D, Vangronsveld J, Van Den Ende W, Cuyper A. Plant sugars are crucial players in the oxidative challenge during abiotic stress: Extending the traditional concept. Plant Cell Environ. 2013;36(7):1242-1255. Doi: http://dx.doi.org/10.1111/pce.12061. [ Links ]
Khan MH, Panda SK. Alterations in root lipid peroxidation and antioxidative responses in two rice cultivars under NaCl-salinity stress. Acta Physiol Plant. 2008;30:81-89. [ Links ]
Lacerda CF, Cambraia J, Oliva MA, Ruiz HA, Prisco JT. Solute accumulation and distribution during shoot and leaf development in two sorghum genotypes under salt stress. Environ Exp Bot. 2003;49(2):107-120. Doi: http://dx.doi.org/10.1016/S0098-8472(02)00064-3. [ Links ]
Lima MGS, Lopes NF, Bacarin MA, Mendes CR. Efeito do estresse salino sobre a concentração de pigmentos e prolina em folhas de arroz. Bragantia. 2004;63(3):335-340. Doi: http://dx.doi.org/10.1590/S0006-87052004000300003. [ Links ]
Liu JH, Nada K, Honda C, Kitashiba H, Wen X, Pang X, et al. Polyamine biosynthesis of apple callus under salt stress: importance of the arginine decarboxylase pathway in stress response. J Exp Bot. 2006;57(11):2589-2599. Doi: http://dx.doi.org/10.1093/jxb/erl018. [ Links ]
Malavolta E, Vitti GC, Oliveira SA. Avaliação do estado nutricional das plantas: princípios e aplicações. 2 ed. Piracicaba: Potafos; 1997. 309 p. [ Links ]
Mandhania S, Madan S, Sawhney V. Antioxidant defense mechanism under salt stress in wheat seedlings. Biol Plantarum. 2006;50(2):227-231. Doi: http://dx.doi.org/10.1007/s10535-006-0011-7. [ Links ]
Mhamdi A, Noctor G, Baker A. Plant catalases: Peroxisomal redox guardians. Arch Biochem Biophys. 2012;525(2):181-194. Doi: http://dx.doi.org/10.1016/j.abb.2012.04.015. [ Links ]
Mittler R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002;7(9):405-410. Doi: http://dx.doi.org/10.1016/S1360-1385(02)02312-9. [ Links ]
Molinari HBC, Marur CJ, Daros E, Campos MKF, Carvalho JFRP, Bespalhok-Filho JC, et al. Evaluation of the stress-inducible production of proline in transgenic sugarcane (Saccharum spp.): osmotic adjustment, chlorophyll fluorescence and oxidative stress. Physiol Plantarum. 2007;130(2):218-229. Doi: http://dx.doi.org/10.1111/j.1399-3054.2007.00909.x. [ Links ]
Munns R, James RA, Läuchli A. Approaches to increasing the salt tolerance of wheat and other cereals. J Exp Bot. 2006;57(5):1025-1043. Doi: http://dx.doi.org/10.1093/jxb/erj100. [ Links ]
Murashige T, Skoog F. A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plantarum. 1962;15(3):473-497. Doi: http://dx.doi.org/10.1111/j.1399-3054.1962.tb08052.x [ Links ]
Nakano Y, Asada K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981;22(5):867-880. [ Links ]
Piza IMT, Lima GPP, Brasil OG. Atividade de peroxidase e níveis de proteínas em plantas de abacaxizeiro micropropagadas em meio salino. Rev Bras Agrocienc. 2003;9(4):361-366. [ Links ]
Reddy AR, Chaitanya KV, Vivekanandan M. Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. J Plant Physiol. 2004;161(11):1189-1202. Doi: http://dx.doi.org/10.1016/j.jplph.2004.01.013. [ Links ]
RIDESA-Rede Interuniversitária para o Desenvolvimento do Setor Sucroalcooleiro. Catálogo nacional de variedades "RB" de cana-de-acúcar. Daros E, Oliveira RA, Zambon JLC, Bespalhok-Filho JC, editors. Curitiba; 2010. 136p. [ Links ]
Shaheen R, Hood-Nowotny R. Effect of drought and salinity on carbono isotope discrimination in wheat cultivars. Plant Sci. 2005;168(4):901-909. Doi: http://dx.doi.org/10.1016/j.plantsci.2004.11.003. [ Links ]
Sharma P, Jha AB, Dubey RS, Pessarakli M. Reactive Oxygen Species, Oxidative Damage, and Antioxidative Defense Mechanism in Plants under Stressful Conditions. J Bot. 2012;2012:1-26. Doi: http://dx.doi.org/10.1155/2012/217037. [ Links ]
Silva MMA, Ulisses C, Medeiros MJL, Granja MMC, Willadino L, Camarea T. Antioxidant enzymes activity in embryobenic and non-embryogenic tissues in sugarcane. Acta biol. Colomb. 2014;19(2):203-210. [ Links ]
Sobahan AS, Arias CR, Okuma E, Nukamura Y, Mori IC, Murata Y. Exogenous proline and glicynebetaine suppress apoplastic flow to reduce Na+ uptake in rice seedlings. Biosci Biotechnol Biochem. 2009;73(9):2037-2042. Doi: http://dx.doi.org/10.1271/bbb.90244 [ Links ]
Tester M, Davenport R. Na+ tolerance and Na+ transport in higher plants. Ann Bot. 2003;91(5):503-527. Doi: http://dx.doi.org/10.1093/aob/mcg058. [ Links ]
Trovato M, Mattioli R, Costantino P. Multiple roles of proline in plant stress tolerance and development. Rendiconti Lincei. 2008;19:325-346. Doi: http://dx.doi.org/10.1007/s12210-008-0022-8. [ Links ]
Willadino L, Oliveira-Filho RA, Silva-Junior EA, Gouveia-Neto A, Camara TR. Estresse salino em duas variedades de cana-de-açúcar: enzimas do sistema antioxidativo e fluorescência da clorofila. Rev Ciên Agron. 2011;42(2):417-422. Doi: http://dx.doi.org/10.1590/S1806-66902011000200022. [ Links ]