INTRODUCTION
Extravillous cytotrophoblast (EVT) is an integral tissue of the placenta which invades the uterine endometrium and the maternal spiral arteries, remodeling them (Lunghi et al., 2007; He et al., 2017). An adequate EVT invasion is vital for blastocyst implantation and successful pregnancy development. When this process is modified, some pathologies occur such as preeclampsia, intrauterine growth restriction, and gestational trophoblastic diseases. This invasive process requires the expression and activation of extracellular proteases, highlighting matrix metalloproteinases (MMPs) and urokinase-type plasminogen activator (uPA). An increase in the expression of MMPs and uPA is a uniform phenomenon in diverse cancer types, and it is correlated with tumor progression and metastasis (Friedl and Wolf, 2003; Turunen et al., 2017; Mahmood et al., 2018). Amongst MMPs, MMP2 and nine have been the most characterized enzymes in cancer cases, and MMP9 plays a relevant role in invasive trophoblast processes (Staun-Ram et al., 2004; Cohen et al., 2006; Zhang et al., 2018). On the other side, it has been demonstrated that the activator system of plasminogen uPA plays an important role in the regulation of invasion and migration of trophoblast (Lala and Chakraborty, 2003; Zheng et al., 2018). Even though EVT cells behave as metastatic (Ferretti et al., 2007), their invasion in vivo is transitory, spatially confined and highly regulated by a plethora of factors (Cohen et al., 2006; Menk horst et al., 2016; Li and Shao, 2017), including the transforming growth factor beta (TGF-β), which has negative effects on trophoblast proliferation, migration and invasion (Chakraborty et al., 2002; Lunghi et al., 2007; Yi et al., 2018) and promote its differentiation (Cheng et al., 2018).
TGF-β causes a significant reduction in secretion and activity of uPA, and induces the expression and secretion of its inhibitor PAI-1 and the tissue inhibitor of metalloproteases TIMP-1 and -2, which inhibit the extracellular matrix degradation performed by the MMPs (Lala et al., 1998). It has been seen that TGF-β reduces the expression and secretion of MMP9 in primary cultures of trophoblast isolated from first-trimester placenta (Meisser et al., 1999), in explants of first-trimester placenta (Lash et al., 2005), in the choriocarcinoma cell line JEG-3 (Karmakar and Das, 2002) and in the trophoblast cell line NPC (Zhao et al., 2006). Nevertheless, other studies in primary cultures of the first-trimester cytotrophoblast have demonstrated that TGF-β stimulates expression and secretion of MMP9 (Shimonovitz et al., 1996). The immortalized first-trimester EVT cell line HTR-8/SVneo was established and described as a model that partially responds to the regulatory actions of TGF-β, with a decrease in secretion and activity of uPA without affecting the invasion level (Graham et al., 1993), being proposed as a model for the study of human placental function and tumor progression (Lala et al., 2002). However, more recent works show that TGF-β promotes invasion of HTR-8/SVneo cells in advanced culture passages, involving expression changes in members of its signaling pathway (Yanzhen et al., 2014). Furthermore, TGF-β 1 upregulated cadherin-11 expression in HTR-8/SVneo through activating SMAD2 and SMAD3 signaling pathways inducing epithelial to mesenchymal transition (Cheng et al., 2018). This apparent contradiction in the effect of TGF-β on protease expression and cellular invasion of the trophoblast requires additional studies that consider the expression over time and in vitro culture conditions, specifically the treatment in presence or absence of fetal bovine serum (FBS).
In previous studies with HTR-8/SVneo cells cultured for 24 hours with or without fetal bovine serum (0 %, 5 %, and 10 %), we have seen a change in protein profiles which suggests a metabolic adaptation due to serum absence (Novoa-Herran et al., 2016).
Additionally, it has been seen that the effects of IGF-I and insulin on the invasion of cellular trophoblast models are serum-dependant (Mandl et al., 2002).
Considering the above, it is relevant to study the effects of TGF-β on the expression of MMP9 and uPA in different culture conditions and over time, since expression of these proteases is an inflection point in the regulation of EVT invasion. In this work, transcriptional expression of uPA and MM9, as a response to different doses of TGF-β, was evaluated by means of quantitative PCR in HRT-8/SVneo cell line, in presence or absence of 0.5 % FBS and over time; possible transcription factors and involved signaling pathways as well, were identified by a bioinformatic analysis.
MATERIALS AND METHODS
Cell line and culture conditions
The immortalized cell line of human extravillous trophoblast HTR-8/SVneo was donated by Dr. Ángela Cadavid (Universidad de Antioquia, Colombia). This cell line was developed from a culture of first-trimester human placenta explant and was immortalized by transfection with a cDNA that codes for the simian virus 40 (SV40) large T-antigen (Graham et al., 1993). These cells exhibit a high proliferation index and share several phenotypic similarities with parental HTR-8 cells, including invasive abilities in vitro. Cells were manipulated from passage 15 to 20 (own records) and cultured at 37 °C in humidified atmosphere with 5 % CO2 in RPMI 1640 medium (Sigma Chemical Co.) supplemented with 10 % FBS (Gibco, Invitrogen), 2 mM L-glutamine (Gibco, Invitrogen), and 40 μg/mL gentamicin (Genfar). Cells were seeded in 60 mm culture dishes and cultured in supplemented medium for one to two days until 60-80 % confluency was achieved, were starved overnight in serum-free medium, after that stimulus was performed in the mentioned doses and times.
Treatment
Cells were stimulated with transforming growth factor β isoform 1 (hBA-112, Santa Cruz Biotechnology Inc., Receptor grade) in a concentration of 10 ng/mL and for 0, 6, 12, 18 and 24 hours. Additionally, TGF-β1 in concentrations of 0.1, 1, 5, 10 and 20 ng/mL was used. Different concentrations up to 20 ng/mL as indicated for each experiment. These treatments were performed in serum-free RPMI medium or concomitant with 0. 5% FBS using serum-free RPMI medium or with 0.5 % FBS as control, respectively.
RNA extraction
Each culture dish was treated with 1 mL of TRIzol reagent (Invitrogen, USA) according to the instructions of the manufacturer. The cells were lysed with the reagent, generating a monophasic solution composed of phenol-guanidium isothiocyanate, and extracting total RNA in the aqueous phase by phase-partitioning with chloroform. RNA was precipitated with isopropanol, washed with 75 % ethanol in RNase-free water and solubilized in 30 μL of RNase-free water. The concentration and purity of the obtained RNA were determined by spectrophotometry (Pharmacia Biotech Ultraspec, 2000) and its integrity was verified by agarose gel electrophoresis.
cDNA synthesis by retro-transcription
The reaction was carried out in a Perkin Elmer PC100 thermocycler (Perkin Elmer, USA). Each reaction was prepared in a volume of 20 μL, mixing 2 μg of RNA with 0.50 mM dNTPs and 50 ng of random primers in a first step. This mixture was heated to 65 °C for 5 min, cooled and then 40 units of RNase inhibitor, 4 μL of 5X transcription buffer, 5 mM dithiothreitol (DTT) and 200 U of SuperScript III RT (Invitrogen) were added. Afterward, the mixture was homogenized and centrifuged, proceeding with the following program: 25 °C for 10 minutes, 50 °C for 50 minutes, 70 °C for 15 minutes and preservation at 4 °C.
Evaluation of expression by real-time PCR
The cDNA was diluted 1:10 with RNase-free water. Additionally, a pool composed by the mixture of1 μL of cDNA of each sample was prepared, at different serial dilutions (1:5, 1:25, 1:125, 1:625, 1:3125) and a calibration curve for the genes 18S (Hrs 18S), β-actin, PLAU (uPA) and MMP9 was built. Real-time PCR reactions were performed in a BioRad CFX96 thermocycler (BioRad Laboratories Inc), using a reaction mixture: 1 μL of cDNA (1:10 dilution or calibration curve dilution as corresponds), 1.5 μL of Master Mix iQTM SYBR® Green (BioRad Laboratories Inc), 0.35 μL of RNase-free water and 0.15 μL of 10 μM gene-specific primers for each gene. The primers were designed based on the reported entries in GenBank and the sequences in direct (D) and reverse (R) sense were: MMP9: D: AACCAATCTCACCGACAGG, R: CGACTCTCCACGCATCTC;PLAU (uPA): D: AGCAGAGACACTAACGACTTCAG, R: CTTACTCACACTTACACTCACAGC; Hrs-18S: D: ATGTGGTGTTGAGGAAAGC, R: TACTGGCGTGGATTCTGC;β-actin: D: GCGTGACATTAAGGAGAAG,R:GAAGGAAGGCTGGAAGAG. Forty cycles were performed with 10 s at 95 °C, 20 s at 56 °C and 20 s at 72 °C. The fluorescence threshold (Ct) was calculated with the software CFX Manager version 1.6 (BioRad Laboratories Inc) and a melting curve was made once the last amplification cycle finished as a criterion to detect unspecificity and dimer formation. Each determination was performed by triplicate, obtaining the relative expression normalized to genes 18S and β-actin used as reference genes, accordingly to the data treatment and error propagation suggested by Hellemans et al., (2007). Finally, the normalized relative expression (NRQ) and the obtained standard deviation (SD) was re-scaled, normalizing the data to each biological control and following the propagation of error formulas.
Statistical analysis
For simple comparisons between treatments and controls, unpaired Two-tailed T-test was used. For comparison between treatments and times or stimuli doses a two-way analysis of variance (ANOVA) was performed, and Bonferroni test for subsequent analysis (biological replicate, n=3, p<0.05).
Bioinformatic analysis of the promoter region and regulation by transcription factors
The 5' sequences flanking genomic sequences of MMP9 and PLAU (uPA) were analyzed using the PROSCAN v 1.7 software (Dan Prestridge, University of Minnesota; http://www-bimas.cit.nih.gov/molbio/proscan/) applying default parameters. For the sequence of the MMP9 gene, 14654 base pairs were processed and for the sequence of PLAU, 13398 base pairs. The predicted results of the promoter region were complemented with a transcriptional analysis employing Enrich analysis tools (Chen et al., 2013). The transcription factors that bind to the promoter of the genes were obtained from the experimental databases ChIP ChEA 2015 (ChIP-x Enrichment Analysis) and ENCODE TF ChIP-seq 2015 (Encyclopedia of DNA Elements). Transcriptions factors with binding sites in the promoter were inferred against the databases TRANSFAC, JASPAR PWMs, and Genome Browser PWMs. A consensus between the different analysis of the transcription factors that bind to the promoter of MMP9 and/or PLAU was performed.
To determine which of these factors are part of the TGF-β signaling pathway, we used Cytoscape 3.3.0 (Shannon et al., 2003) to link the list of transcription factors in the TGF beta Signaling Pathway network of Wikipathways App (Kutmon et al., 2014).
Protein interaction networks were created using PCVIz of Pathway Commons (Cerami et al., 2011), for TGF-β with MMP9 and uPA, as a group and separately, including interactions that control state change and expression, in order to detect intermediaries that could mediate functional relationships between the cytokine and proteases.
RESULTS
The presence of serum differentially affects the expression of MMP9 and uPA as a response to different doses of TGF-β
The effect of TGF-β on the expression of uPA and MMP9 proteases was evaluated in cells of the immortalized trophoblast-derived cell line HTR-8/SVneo, by RT-PCR in real time, normalizing the results against the 18S ribosomal unit and the β-actin genes, before the specific normalizations of each analysis.
In general, the nature and magnitude of the effect of TGF-β on gene expression change with the dose, the presence of serum and with the evaluation time (Fig. 1). No significant differences were observed among the different TGF-β doses, although this factor affects the expression of each protease, being significant for the expression of MMP9 with serum (Fig. 1B; 0.1 ng/mL at 12 and 24 h, 1 and 5 ng/mL at 12 h) and the expression of uPA without serum (Fig. 1 C; 10 and 20 ng/mL at 24 h) and with serum (Fig. 1 D; 1 and 20 ng/mL at 12 h). Specifically, the treatment of TFG-β with 0.5 % FBS caused a decrease between two and four times the expression of MMP9 at 12 hours, depending on the doses (0 vs 0.1 ng/mL 3.57-fold p=0.0192; vs 1 ng/ml 4.17-fold p=0.0151; vs 5 ng/mL 3.23-fold p=0.0221; vs 10 ng/mL 2.63-fold n.s.), while at 24 hours a decrease higher than five times with the dose 0.1 ng/mL (0 vs 0.1 ng/mL 5.56-fold p=0.0490) was recorded. In the case of uPA we found a two to three times higher increase as a response to TGF-β with FBS at 12 hours (0 vs 1 ng/mL 3.15-fold p=0.0306; vs 20 ng/mL 2.74-fold p=0.0323). When the treatment was performed in the absence of serum, the expression of uPA decreased as a response to TGF-β, being significant at 24 hours (0 vs. 10 ng/mL 2.78-fold p=0.0005; vs. 20 ng/mL 1.79-fold p=0.0062).
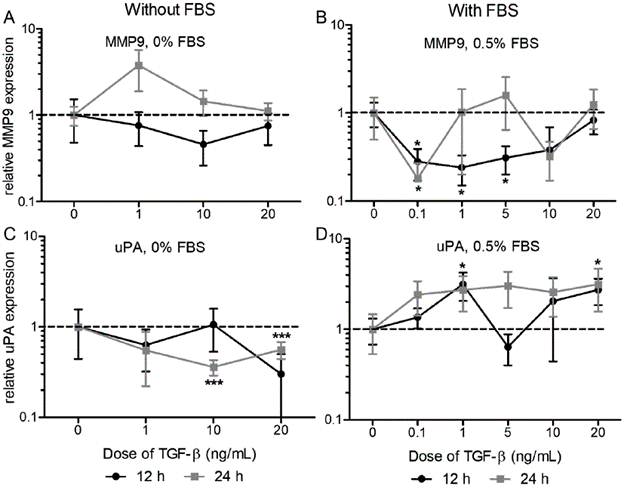
Figure 1 Effect of TGF-β on the expression of MMP9 and uPA. We evaluated the response to TGF--β doses until 20 ng/mL at 12 and 24 hours, without serum (A and C) or with 0.5% FBS (B and D) on the expression of MMP9 and uPA respectively. Relative expression to 18S and β-actin genes, normalized against control (TGF-β 0 ng/mL). Unpaired Two-tailed T-test compared to control (n=3 p>0.05, ** p<0.001). Relative expression axis in logarithmic scale.
When evaluating the influence of serum on the effect of the treatment, the expression of these genes as a response to this cytokine differed significantly between with or without FBS (Fig. 2), specially for uPA which presented an expression profile in response to TGF-β that was clearly distinctive between with or without serum treatment and independent on the dose or the evaluated time. Noticeably we observed that presence or absence of serum modified in a significant manner the nature of the effect of TGF-β, changing from decreasing the expression of uPA in absence of serum to an increase in presence of 0.5 % FBS both at 12 hours (Fig. 2 C, 1 ng/mL t=3761 p<0.01; 20 ng/mL t=3641 p<0.01) and at 24 hours (Fig. 2 D, 1 ng/mL t=3.196 p<0.05; 10 ng/ mL t=3.255 p<0.05; 20 ng/mL t=3.783 p<0.01). In the case of MMP9, at 12 hours the TGF-β negatively affected the expression, consistently and homogeneously, independent on the presence of serum; but at 24 hours TGF-β promoted the expression in the absence of serum while diminished it with serum (Fig. 1 A and B, Fig. 2 B 1 ng/mL t=4.152 p<0.01), observing a concomitance between treatment and serum as in the case of uPA, but with opposite effects.
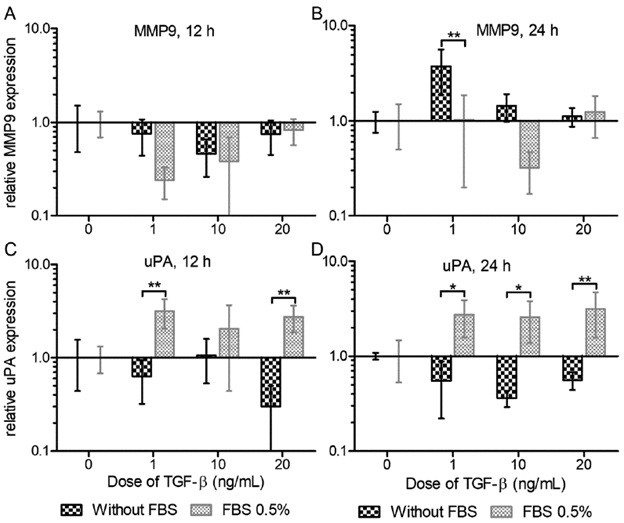
Figure 2 Effect of TGF-β relative to the presence of serum on the expression of MMP9 at 12 (A) and 24 hours (B), and of uPA at 12 (C) and 24 (hours). We evaluated TGF-β doses of 1, 10 and 20 ng/mL at 12 and 24 hours, concomitant treatment with or without 0.5% FBS. Relative expression to 18S and β-actin genes, normalized against control (TGF-β 0 ng/mL). Two-way ANOVA test to compare between with and without conditions (n=3, *p<0.05, ** p<0.01, *** p<0.001). The relative expression axis is presented in Log 10 scale.
Expression kinetics: The effect of TGF-β on the expression of MMP9 and uPA as a function of time
To evaluate the expression dynamics of the genes MMP9 and PLAU (uPA) over time, both in a basal form and as a response to TGF-β, HTR-8/SVneo cells were cultured in RPMI medium with 0.5 % FBS, with or without 10 ng/mL TGF-β and mRNA was extracted every 6 hours leading to analysis times of 0, 6, 12, 18 and 24 hours. The employed dose was 10 ng/mL, widely used in the literature. Cells cultured for the same time in medium with 0.5 % FBS were used as a control. Initially, to carry out the statistical analysis, data were compared and normalized against the respective control for each time. Finally, the relative expression was normalized against zero time to obtain the kinetics showed in Figure 3.
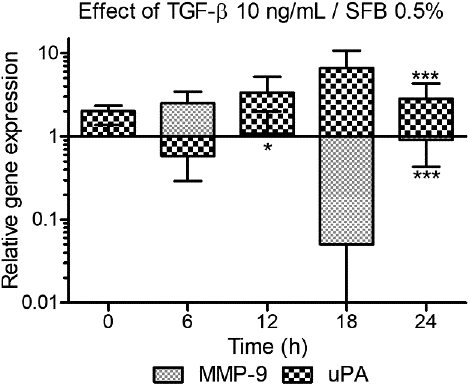
Figure 3 Kinetics of the effect of TGF- β over the expression of MMP9 and uPA. We evaluated the expression of MMP9 and uPA at 0, 6, 12, 18 and 24 hours after treatment with 10 ng/mL TGF-β with 0.5% FBS. Statistical analysis: Two-way ANOVA, comparing the NRQ of the treatment (T = TGF-β against control (C = TGF-β 0 ng/mL, 0.5% FBS) at each time for each gene: (Txh/Cxh) (n=3, *p<0.05, ** p>0.01, *** p<0.001). The bars correspond to the expression relative to the 18S and β-actin genes, normalized against its control (TGF-β 0 ng/mL) at each time and then at time zero: (Txh/Cxh)/(T0h/C0h).
The effect of TGF-β on the expression of MMP9 and uPA over time varied in accordance with the studied gene. When comparing with the corresponding control at each time, TGF-β, in general, decreased the expression of MMP9 being statistically significant at 12 hours (2.63-fold t=3.136 p<0.05) and 24 hours (3.13-fold t=6.228 p<0.001). Unlike uPA, for which TGF-β reduced expression at 6 hours but caused an increase at 12, 18 and 24 hours, being statistically significant in the latter (2.58-fold t=4.676 p<0.001) (data not shown).
Additionally, when normalizing the relative expression over time against expression at zero time, a dual effect on the proteases expression over time was detected, following a sinusoidal pattern, with out-of-phase peaks -coordinated but opposed between the genes. In this way, while at 6 hours TGF-β increased the expression of MMP9 (2.5-fold) and decreased the expression of uPA (1.74-fold), at 18 hours TGF-β increased the expression of uPA (6.61-fold) and decreased the expression of MMP9 (18.34-fold) (Fig. 3).
Analysis of promoters and regulation of transcription factors
The observed differences on the TGF-β transcriptional regulation of the genes under study, could be the result of a differential signaling pathway activation, and therefore, on the corresponding transcription factors. A bioinformatics analysis of the promoters and transcription factors of MMP9 and PLAU (uPA) genes was performed using the Proscan and Enrich tools. MMP9 and PLAU genes are in different chromosomes (ch20q13.12 and chr10q22.2, respectively) and despite a common group of transcription factors was found, most of them were specific for each gene. Figure 4A contains a final representation, indicating the transcription factors that bind, or probably bind, to the promoter of each gene.
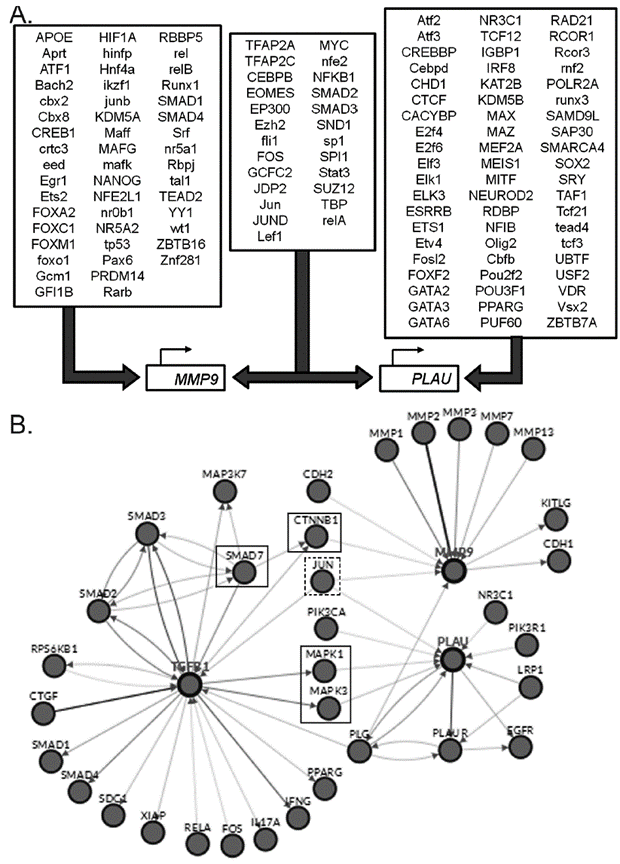
Figure 4 A. Transcription factors with binding sites in of MMP9 and PLAU (uPA) genes promoters. Consensus list created from Proscan v. 1.7 and Enrich analysis (ChIP ChEA 2015, ENCODE TF ChIP-seq 2015, TRANSFAC and JASPAR PWMs and Genome Browser PMWs). B. Analysis of Pathways commons pathways. Obtained network between the TGF-β, MMP9 and PLAU genes, obtained with PCViz and addition of 34 nodes. Blue connections: state changes, green connections: expression. Dotted line frames: connector nodes shared between TGF-β and both proteases. Continuous line frames: connector nodes exclusive to MMP9 or PLAU.
Linking these transcription factors with the TGF-β pathway reported by Wikipathway (Fig. 5), some common elements as Fos/Jun were observed. Fos/Jun form a multimeric complex with Smad2-3/Smad4, as well as Myc, EP300, Sp1, and JunD, which coordinate the expression of both proteases. Among the reported transcription factors for PLAU but not for MMP9, are included CREBBP, Ets1, and E2F4, highlighting ATF2, atf3 and MEF2A, which are activated by phosphorylation by MAPK14. As for MMP9, SMAD4, TP53, and JunB are reported as regulators of this gene but not of PLAU (Fig.4A), suggesting they only follow the TGF- β/SMADs canonic pathway (Fig. 5).
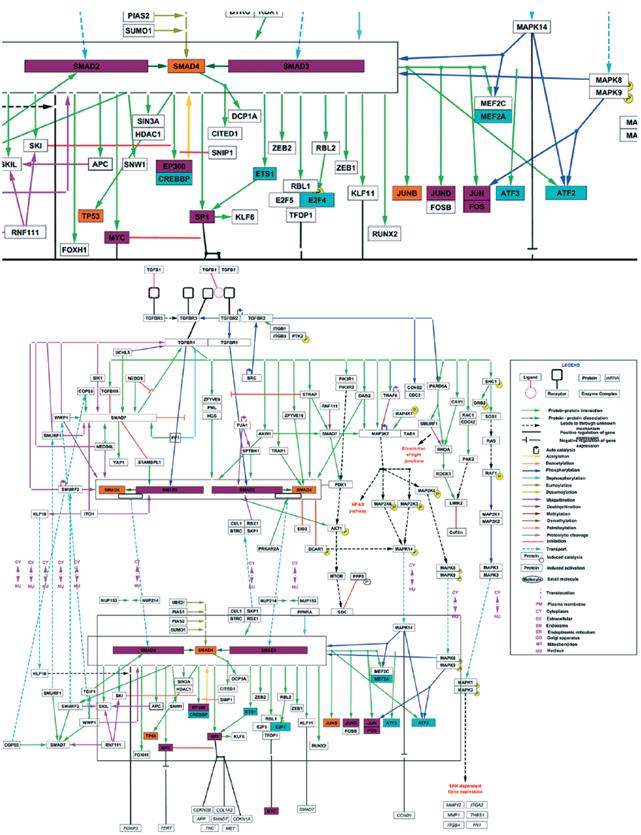
Figure 5 TGF-β signalling pathway and transcription factors involved in the expression of MMP9 and PLAU (uPA). Inferred transcription factors that regulates the expression of MMP9 and PLAU (purple nodes), only MMP9 (orange nodes) or only PLAU (blue nodes), belonging to the TGF-β signalling pathway reported by WikiPathways.
Using the Pathway Commons an analysis was performed to evaluate the interaction network between TGF-β and MMP9 or uPA, as well as the network between the cytokine and both proteases (Fig. 4B). The role of Jun as a common transcription factor for both proteases was confirmed. We observed in the network from TGF-β to MMP9, that SMAD7 - that mediates the signaling from the ligand connects to CTNNB1 or β-catenin, a key downstream component of the canonic Wnt pathway. In the case of uPA network, a remarkable interaction was observed between TGF-β and members of the MAPK pathway as MAPK1 and MMAPK3, reinforcing the results obtained, when crossing the transcription factors that regulate uPA in an exclusive form, with the TGF-β pathway from WikiPathways.
DISCUSSION
In the present work, we found that the presence of fetal bovine serum (0.5 % FBS) affects in a significant way the nature of the effects of TGF-β on the expression of uPA in cells of the HTR-8/SVneo trophoblast-derived cell line. Treatment with TGF-β in conjunction with 0.5 % FBS increased the expression of uPA at 12 and 24 hours, while in the absence of serum, the expression was decreased. In the case of MMP9, we also observed a serum-dependent response at 24 hours, but in an inversely way: in the absence of serum TGF-β increased the expression of MMP9, whereas in the presence of serum a variable response was observed. On the other hand, at 12 hours a decrease was observed in MMP-9 expression, without an apparent effect of serum. This finding suggests that the transcriptional regulation of uPA can be more dependent on the presence of serum, and therefore on the environmental conditions, in comparison with MMP9 expression. In addition to the influence of serum, our results also showed that the effects of TGF-β In the expression of MMP9 and uPA changed with time, evidencing a dual effect of TGF-β with sinusoidal kinetics. A coordinated regulatory pattern for both genes was obtained, but with out-of-phase peaks, due to the opposed effects of TGF-β: while at 6 hours post-stimuli, TGF-β inhibits the expression of uPA and increases the expression of MMP9, at 12 hours the effect is the opposite. This phenomenon could be explained in part, by the activation of different signaling pathways and their corresponding transcriptional mediators operating at their kinetics. Another factor may arise from a possible modification of the culture conditions, due to nutrient uptake and conditioning of the medium with autocrine factors that altogether modify the characteristics of the culture medium. These changes could influence the response to TGF-β, generating other input signals, which explain the changes in response to TGF-β due to the presence of serum and over time, highlighting the influence of microenvironment or cellular context.
Studies on the effects of insulin and IGF-I, acting separately or in synergy with the serum, on the secretion of chorionic gonadotropin beta (hCG), proliferation and invasion of BeWo, JAR and JEG-3 choriocarcinoma cell lines, a strong synergistic effect with serum was revealed (Mandl et al., 2002). Other studies have demonstrated that a high glucose concentration inhibits the activity of uPA and in vitro invasion of HTR-8/SVneo cells, with no observed effects on the levels of MMP2, MMP9, PAI-1 or uPAR (Belkacemi et al., 2005). The observed dependence of the signaling of TGF-β on the culture conditions and its versatility, can be explained based on its dual behavior, as tumor suppressor in certain cell types and as pro-metastatic in many invasive tumors (Pardali and Moustakas, 2007), and whose effects greatly depend on the cellular context (Ikushima and Miyazono, 2010).
The trophoblastic tissue has been considered as "pseudo-malignant" or "physiologically metastatic" because its cellular abilities (Ferretti et al., 2007; Li and Shao, 2017) and similar to solid tumors, the placenta has several trophoblast subtypes, which vary with the gestational stage (Liu et al., 2018); even more, the HTR-8/SVneo cell line after immortalization by transfection with SV40 virus large T-antigen (Graham et al., 1993) can present additional features, being proposed as a tumor progression model (Lala et al., 1998). In spheroid formation assay, HTR-8/ SVneo spheroids showed trophoblast progenitor cell-like characteristics and are suggested that these spheroids contain the progenitor cell characteristic of repopulating activity in a near in vivo model (Weber et al., 2013). Nevertheless, TGF-β has been studied only as a tumor suppressor in these models, based on its classic anti-proliferative, anti-invasive and anti-migratory actions (Chakraborty et al., 2002; Lala et al., 2002), without considering its dual effect according to the biological context in which it performs its actions. Taking into account the HTR-8/SVneo cell line characteristics and its origin, as well as the dual role or dichotomy of TGF-β, it is not surprising to observe the opposed effects of this factor on the expression of MMP9 and uPA, key proteases in the invasive process of both trophoblast (Lala and Chakraborty, 2003; Staun-Ram et al., 2004; Cohen et al., 2006; Zheng et al., 2018) and metastatic tumors (Friedl and Wolf, 2003; Mahmood et al., 2018). Even though the presence of 0.5 % FBS can modify the cellular status and therefore the response capacity to exogenous factors, based on the performed study, it is not possible to discern the role that FBS plays.
Until now, no systematic studies about the expression of these two genes in response to TGF-β over time, or about the influence of serum during the treatment had been performed. In this work, we found a differential response to TGF-β as a function of time, for both uPA and MMP9 genes, exhibiting dual kinetics, which partially agrees with the literature. In general, it has been seen that TGF-β decreases the secretion and activity of uPA in the HTR-8/SVneo cell line (48 h, serum-free ExCell medium) (Graham et al., 1993) and in first-trimester placenta explants (24h, serum-free ExCell 300 medium) (Lash et al., 2005), simultaneously decreasing the secretion and activity of MMP9 in primary first-trimester cytotrophobast cultures (4 days, serum-free DMEM medium) (Meisser et al., 1999), and in placenta explants (24 h, serum-free ExCell 300 medium) (Lash et al., 2005), and confirming a reduction in the expression and activity of uPA and MMP9 in the choriocarcinoma cell line JEG-3 (10 ng/mL, 24 h) (Karmakar and Das, 2002).
Nevertheless, in primary first-trimester cytotrophobast cultures we observed an increase in the expression of MMP9 at 8 hours and its activity at 12 hours, as a response to TGF-β1 (10 and 20 ng/mL, serum-free RPMI 1640 medium) (Shimonovitz et al., 1996); similarly, an increase in MMP9 and 2 as a response to TGF-β was observed in early cytotrophobast cultures and the choriocarcinoma cell line BeWo (Yudate et al., 1996). This agrees with an increase in the expression of MMP9 at 6 hours in 0.5 % FBS medium or at 24 hours in serum-free medium, highlighting the duality of TGF-β and its context-dependent actions.
The disparity in the previous results can be due not only to the specific culture conditions -such as cellular model, medium type, the presence of serum, dose and stimulus time- but also to the type of technique used for transcript measuring. In the present study, the genic expression was evaluated in a time range from 6 to 24 hours by qPCR, which despite being implemented as a relative comparison, has a much higher sensibility and allows to detect small changes in transcript abundance, making possible to determine that TGF-β has a different effect at short times (6 hours). At longer culture times, the medium is conditioned by trophoblast-secreted factors that could have an autocrine effect, increasing the complexity in both the signaling and the results interpretation.
The difference in the TGF-β transcriptional regulation on each gene may be explained in part by the different sets of transcription factors found for each gene, especially those belonging to the TGF-β signaling pathway, as Smad4, TP53 and JUNB for MMP9, and CREBBP, Ets1, E2F4, ATF2, ATF3 and MEF2A for PLAU. This difference can also be due to a differential signaling activation, as the bioinformatic analysis suggests. It is necessary to explore possible crossed signaling events, including co-activation of the MAPK pathway. Previously it was seen that TGF-β promotes invasion of HTR-8/SVneo cells in advanced culture passages, involving a reduction in the expression of T(RI, Smad3, and Smad4, increase of Smad7 (Yanzhen et al., 2014), and induction of expression of MMP2 through Smad2, with no Smad 4 involvement (Lin et al., 2006), and more recently TGF-β was reported to induce invasion in JEG-3 cells and up-regulates the MMP-9 expression (Huang et al., 2017). Cellular context-specific factors whose action depends on environmental conditions can generate signaling dynamics and spatial-temporal regulations (Kholodenko et al., 2010). Further studies besides the activation of MPK and TGF-/SMADS pathways can explain the phenomena observed here, confirming the formulated hypotheses and bioinformatic inferences. Additionally, it is necessary to verify the expression at the protein level and furthermore, verify the secretion and extracellular activity of these enzymes to corroborate a possible temporal regulation, like the one we propose.
The invasion of the maternal endometrium by EVTs is a key process (He et al., 2017) whose deregulation, seen as hypoinvasiveness or hyperinvasiveness, lead to preeclampsia or trophoblastic neoplasms, respectively (Chakraborty et al., 2002). Being TGF-β a main regulatory factor in trophoblast physiology, including invasion (Pollheimer and Knöfler, 2005), and taking into account the over-expression of this factor in preeclampsia and hydatidiform mole (Pang and Xing, 2003); understanding the molecular mechanism underlying this regulation is important. The possible temporal regulation and the observed dual effects of TGF-β on the expression of MMP9 and uPA, key proteases in the invasive phenotype, are of remarkable interest and they confer an important role to these enzymes in the invasive process of trophoblastic cells, which is highly regulated in a spatial-temporal way (Menkhorst et al., 2016). These results are in agreement with previous reports about the effects of TGF-β in trophoblast and are compatible with the dual and context-dependent TGF-β role in cancer (Ikushima and Miyazono, 2010). Additionally, novel results were obtained, as the temporal regulation of the expression of MMP9 and uPA, involving opposed effects of TGF-β; and remarkably, the influence of serum on the nature and effect of TGF-β on the expression of uPA, a concomitance not previously studied in this model.















