INTRODUCTION
Lipid vesicles are closed structures organized in phospholipid bilayers with an internal aqueous compartment. Its use in therapy has occurred for the last 40 years, with its first applications being in the cosmetics area. Products based on these vesicles include liposomes and niosomes, which are considered essential representatives in the controlled release of substances (Khan et al., 2015; Suzuki et al., 2017).
Since its creation to the present day, lipid vesicles are used as colloidal systems for the study of cellular biochemical phenomena, with a significant part of the understanding regarding cellular biology being given through these systems (Perrier et al., 2017). These are constituted by amphiphiles organized in closed compartments in the presence of water due to the orientation of their polar groups through hydrophobic interactions and van der Waals forces. The vesicles formed may have one or more phospholipid bilayers, these being characterized as unilamellar or multilamellar (Khan et al., 2015).
Among these vesicles, liposomes and niosomes stand out for being suitable molecule carriers with high bioavailability, biocompatibility, and biodegradability (Andreu and Arruebo, 2018). The use ofthese systems covers areas such as cosmetics, food, agriculture, genetics and pharmaceuticals, explicitly aimed at improving product quality through increased stability and durability (Fathima etal., 2016). Pharmacologically, the interest in these systems lies in the similarities they share with biological membranes, where their physical and chemical properties allow easier drug passage through barriers within the body, thus providing better targeting in drug delivery (Letchford and Burt, 2007).
Liposomes and niosomes are considered relatively safe lipid nanocarriers since they are produced using natural origin products, such as phosphatidylcholines and cholesterol. This composition possesses advantages over other nanocarriers, such as polymer nanoparticles, carbon nanotubes, or gold nanoparticles, which may demonstrate some degree of toxicity (Lim et al., 2012). Studies have shown the importance of substance encapsulation in lipid vesicles to combat reactive oxygen species, moreover, these vesicles can be used as models in studies with antioxidants which especially combat lipid peroxidation (Giri et al., 2016; Barroso et al., 2018). These among other studies have increased the interest in the development of vesicle nanostructures aimed at overcoming obstacles encountered in the delivery of drugs.
The great impasse in the development of phospholipid carriers is finding an ideal formulation with high stability, biocompatibility and, once encapsulated, the substance has its activity potentiated, remaining stable for an extended period. Considering the difficulties encountered in lipid vesicle development, this review sought to gather current information regarding the methods used for its development, including its main components and information on the most current behavior of each one when used in different formulations. This work reports a detailed description of the development of each method, also highlighting some advantages and disadvantages between the methods, citing studies that used the different methodologies for the development of different lipid vesicles. Data were included that show the importance of formulations currently in the medical area, evidencing current studies performed with vesicles liposomes and niosomas as promising in this area. It was concluded with a general approach to the components and methods used for formulation.
METHODOLOGY
This research was carried out using the databases Science Direct, Pubmed and Scielo using the terms "Lipid vesicles", "Nonionic surfactant", "Phosphatidylcholine", "Cholesterol", "Zeta potential" and "Transition temperature phase" either alone or in combination. The research was conditioned to journals in the health sciences, biotechnology and nanotechnology areas, directing the terms for titles, abstracts, and keywords. For the composition, 90 articles were selected after the application of the criteria.
Major lipid vesicles and applications
Liposomes
Liposomes are lipid vesicles formed by mixtures of amphipathic lipids arranged in bilayers in an aqueous medium (Santos and Castanho, 2002). Because of their similarity to biological membranes, liposomes were initially used as a model for membrane studies, however, they soon began to have medical and pharmacological applications, especially in substance transport. This shared similarity with biological membranes permits advantages to the vesicles, such as cellular interactions, endocytosis, and fusion (Chorilli et al., 2004). Another advantage is the encapsulation of drugs, which allows a reduction in side effects, by reducing the required dose and the consequent drug accumulation at the administration site.
Its classification is made according to the number of lamellae present in the vesicle, these being characterized as unilamellar (a single lamellae) or multilamellar (two or more lamellae); in terms of size, these may be large or small unilamellar vesicles, as well as large or small multilamellar vesicles (Garcia, 2014). As an advantage, small and multilamellar liposomes are more spherical and have a higher rigidity; these characteristics permit beneficial mechanical properties during cellular absorption. A liposome formulated with phosphatidylcholine and cholesterol is rapidly eliminated from the bloodstream. A strategy to tackle this problem is in the production of liposomes with a size less than 100 nm and the use of conjugated polymers on their membrane such as PEGs (polyethylene glycol) which can form a protective layer in the bilayer (Fontes et al., 2018).
Liposomes can encapsulate several substances with different solubilities. Thus, the location of such substances depends on their characteristics, especially the solubility between the components of the liposome. Thus, lipophilic substances are inserted into the hydrophobic part of the membrane while the hydrophilic substances remain in the aqueous core, and amphiphiles become part of the liposome membrane (Muller and Landfester, 2015). An example of the localization of hydrophobic compounds can be seen with eugenol derivatives, which when inserted into the aqueous phase tend to migrate slowly into the bilayer, each component having a different penetration and surface bilayer attachment time (Saha and Verma, 2018).
At first, liposomes in the medical area were only seen as suitable molecule carriers capable of promoting their release in a slow and controlled way, from there it was discovered that the liposomal membrane is able to protect these substances from external agents such as light, moisture, and oxygen, increasing its durability (Assis et al., 2012). Doxil® was the first liposomal-based drug formulated, an anticancer drug that entered the market in 1995; after this a multitude of pharmacological formulations that are commercially available appeared, as well as many papers frequently published with liposomal formulations (Rahnfeld et al., 2018).
Some studies have shown the ability of liposomes to increase the physicochemical stability of compounds such as retinoic acid and rutin, where the increased stability is related to increased durability of the formulation (Babazadeh et al., 2017). Besides, liposomes can reduce the toxicity of compounds and increase their action time in the organism (Araújo et al., 2017). The utilization of liposomes in the treatment of inflammatory dermal diseases through the encapsulation of anti-inflammatory compounds such as naringin, potentializing its activity and reducing its side effects, is another example of liposomal use (Pleguezuelos-Villa et al., 2018)
The controlled delivery of substances by liposomes can be improved by adding components that direct the liposomes to the site of action. The study by Poh et al. (2018) shows that folate-targeted liposomes specifically bind to folate receptor-positive cells accumulating at the site of inflammation, with a better response in biological activity. Liposomes increase the penetration of substances in the stratum corneum by increasing the fluidity of the bilayer through lipid disturbance (Imam et al., 2017), thus being considered a delivery system that was succeeding the transdermal route (Abdel-Hafez et al., 2018).
Substances with low water solubility have their therapeutic applications limited; however, when encapsulated in liposomes such substances may improve their bioavailability, as seen in the study by Yingyuad et al. (2018), which encapsulated the calothrixin B compound and found significant improvements in chancre treatment.
In a study with capsaicin used to verify the reduction in liver oxidative stress, it was verified that the activity of this substance was also potentiated when it is was encapsulated (Giri et al., 2016). Although liposomes are the first systems produced for the controlled delivery of substances, they have disadvantages, such as high instability, which promotes vesicle aggregation (Gálvez et al., 2011). This feature is related to the composition and the method used for vesicle formulation, where a solution can be the addition of diethylphosphate which decreases aggregation by electrostatic repulsion between the molecules (Shehata et al., 2016).
Niosomes
Niosomes are lipid vesicles formed from the self-assembling of non-ionic surfactants by hydrophobic interactions in aqueous media (Gutiérrez et al., 2016). One or more lipid bilayers represent them, these being characterized as unilamellar or multilamellar (Khan et al., 2015). Its size is generally between 80 and 200 nm, depending on the method used for its formulation. The most commonly used surfactants for the production of niosomes are polysorbates (Tween 60 and 80) and sorbitans (Span 60 and 80) (Yeom et al., 2014). Wang et al. (2016a), argue that formulations based on Tween 80 are pharmacologically more acceptable because they allow for improved molecule stability.
Although niosomes are very similar to liposomes, their composition by non-ionic surfactants allows them to be more stable to oxidation and hydrolysis, having excellent long-term stability and biocompatibility, as well as being cheaper and easy to reproduce formulation (Samed et al., 2017; Somjid et al., 2018). These characteristics allowed niosomes to be rapidly inserted into the market as an alternative to liposomes in the controlled delivery of drugs in the body. Similar to other lipid vesicles, niosomes can incorporate both lipophilic and hydrophilic substances. The lipophilic substances are retained in the bilayer, whereas the hydrophilic substances are in the dissolved in the aqueous compartments (Khan et al., 2015).
The earliest niosome formulations occurred in cosmetics by L'Oreal® and were quickly described as suitable carriers of molecules and food preservatives. These vesicles are considered as an important controlled delivery system in which drugs that are encapsulated may have increased solubility, stability and time to degradation, causing the substances to remain in the body longer (Bansal et al., 2013; Pando et al., 2015). The study by Liang et al. (2016) shows that niosomes can protect the encapsulated substance from intestinal digestion, this characteristic being through the niosome's ability to control the release of substances and the protection against external agents that the vesicle offers. In addition, to increase the time the substance is in the body, niosomes may enhance the biological activities of drugs already used in the market. For example, the Cefdinir® antimicrobial activity against Staphylococcus aureus has been shown to increase when it is incorporated into these vesicles (Bansal et al., 2013).
An increase in the activity of drugs already used in the market by their encapsulation in niosomes allows the reduction of drug concentrations, promoting a decrease in side effects and an increase in the specificity of drug delivery at the target area within the organism (Kanaani et al., 2017). Another study shows that niosomes loaded with Embelin® have an efficient hypoglycemic effect, as well as an antioxidant activity by decreasing lipid peroxidation levels, with these results being related to an improvement in intestinal absorption of the encapsulated drug (Alam et al., 2018)
Niosomes are considered to be interesting to modify the drug dosage system for the treatment of dermatological disorders. Niosome topical application can increase the drug resistance time in the stratum corneum and epidermis while reducing the systemic absorption of the drug, transdermal water loss, and promoting lipid replacement at the same time. The study by Moghddam et al. (2016) has shown that niosomes increase Diacerein® penetration in the deeper layers of the skin. In another study, smaller vesicles are more efficient at penetrating the drug into the skin (Pando et al., 2015). Another benefit of using niosomes is based on the capacity the system has to potentiate the effect of drugs already used in the market, this being proven in the skin absorption study with the anti-fungal agent Itraconazole®, where the drug-containing niosomal formulations have been shown to be more efficient than the drug alone (Alomrani et al., 2015). Figure 1 shows the chemical structure of the compounds encapsulated in liposomes and niosomes cited in this work, and Table 1 shows the medical application of the encapsulated compounds.
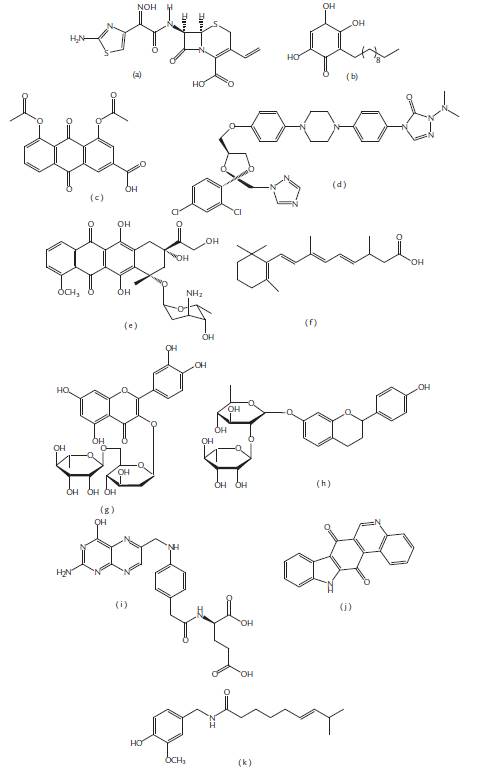
Figure 1 Molecular forms of encapsulated drugs in liposomes and niosomes cited in this work. a.Cefdinir®, b. Embelin ®, c. Diacereina®, d. Itraconazol®, e. Doxil®, f. Ácido retinóico® g. Rutina®, h.- Naringina®, i. Folato®, j. Calothixin B®, k. Capsaicina®.
Principal components
Phosphatidylcholine
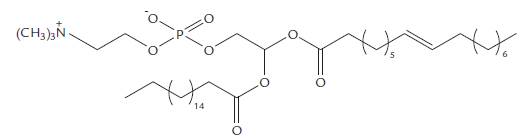
Phosphatidylcholines are the most widely used phospholipids in the production of lipid vesicles and have its highest use in the cosmetics area as emulsifiers. They consist of natural or animal origin lipid mixtures, such as soy or egg, respectively. Its chemical structure displays a glycerol skeleton, esterified at positions 1 and 2 (Kolbina et al., 2017). These lectins are part of the composition of biological membranes which may be present in blood plasma as lipoprotein constituents; Figure 2 shows the structure of phosphatidylcholine.
Phosphatidylcholines are considered biocompatible and biodegradable because they do not present a charge at physiological pH, thus being important in the long-term stabilization of emulsions. In addition, saturated and short-chain hydrocarbon phosphatidylcholines are considered to be more potent for the production of emulsifiers (Bruxel et al., 2012). The main characteristic of phosphatidylcholines which justifies their success in the production oflipid vesicles is that they are amphiphilic, which in turn allows the self-organization and formation of bilayers in aqueous media, mimicking biological membranes (Mertins et al., 2008). The formation of the various structures occurs according to the specific properties of these phospholipids, usually forming micelles (Fricker et al., 2010).
Their ability to mimic biological membranes allows these lectins to be used as a model for the study of lipid peroxidation through photoinduced oxidation using compounds such as pterin (Thomas et al., 2016). Liposomes prepared with saturated phosphatidylcholines limit the incorporation of hydrophobic drugs due to membrane rigidity, an alternative to tackle this is the incorporation of medium or long chain triglycerides which promote greater fluidity and increase the amount of the encapsulated drug (Hong et al., 2015). The increase in membrane permeability can also be improved by the addition of ce-tocopherol, a compound which tends to reduce the inclination angle of phospholipid hydrocarbon chains promoting a more vertical orientation with the passage from a gel phase to a normal phase (Quinn, 2012).
Soy-based phosphatidylcholine liposomes are more resistant to oxidation due to the lower degree of instauration present in the molecule compared to other phospholipids (Rudolphi-Skorska et al., 2017). The lipophilic substances that are incorporated can increase their solubility by 200 to 500 times, which permits phosphatidylcholine-based liposomes to be suitable for the study of lipophilic substances (Cagno et al., 2011). In addition, these phospholipids have a relatively high phase transition temperature, which promotes a more stable vesicular formulation (Kitayama et al., 2014).
Cholesterol
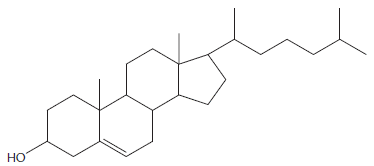
Cholesterol is characterized by the presence of a steroid ring, it has in its chemical structure 27 carbons, constituted by a hydrocarbon chain. Cholesterol is found in all animal biological membranes. The incorporation of cholesterol into lipid vesicle formulations plays an important role in the organization, stability, dynamics, and function of membranes. Thus, it is important to understand the role of this component in different formulations. Cholesterol localization in lipid vesicle membranes occurs near the lipid-water interface with the acyl group in the hydrocarbon region of the bilayer. This arrangement modulates membrane properties by interactions with lipid acyl chains (Marquardt et al., 2016). Cholesterol is used in lipid vesicle formulations to increase the mechanical stability of the bilayer affecting its elasticity. The study by Somjid et al. (2018) shows that surfactant-only niosomal formulations, eg. Span 60, are vaguer; however, when cholesterol is added, there is an interaction between the water and the main surfactant groups leading to the higher mechanical resistance of the system. When the cholesterol concentration has increased the vesicles, tend to increase in size as well as become more rigid with less fluid membranes. This characteristic directly interferes with the release of the encapsulated substance as a more rigid system promotes a decreased or longer drug delivery time (Kaddah et al., 2018). Figure 3 shows the structure of cholesterol.
In contrast, when cholesterol is added to phospholipid membranes at physiological concentrations, it can regulate the rigidity of the membrane while allowing its fluidity due to its high lateral mobility (Arriaga et al., 2017). The increase in the physical stability of the vesicles occurs because cholesterol inclusion promotes an increase in the distance between the charged head groups within the membrane and may also maximize the contact between neighboring hydrocarbon chains (Kuo et al., 2018). Concerning its participation in membrane fusion, its essential role in lipid membrane aggregation depending on its concentration and temperature has been demonstrated; however, this ability to form fusogenic mixtures is not always linked to destabilization in the vesicle bilayer (Ibarguren et al., 2015). Cholesterol used in a 1/1 molar ratio forms a more stable vesicle with a higher capacity for encapsulation. This characteristic is related to cholesterol's ability to decrease the permeability ofthe bilayer and consequently decrease the rate of degradation by factors such as light, pH and temperature (Ravaghi et al., 2016). The vesicles present high negative zeta potential values, providing electrostatic repulsion and relatively stable nanoemulsion (Gunes et al., 2017).
Nonionic surfactants
Surfactants are organic structures composed of amphiphiles, that is, organic structures characterized by two regions, one polar and hydrophilic and another apolar and hydrophobic. Non-ionic surfactants, derived from polyoxyethylene and polyoxypropylene, are especially used in the preparation of lipid vesicles because they have a balance between the hydrophilic and lipophilic regions. The most common examples are polysorbates and sorbitans (Silva et al., 2016).
The use of nonionic surfactant for lipid vesicle formulation is one of the alternatives to overcome drawbacks encountered in traditional formulations which are based only on phospholipids. These compounds confer the vesicle biocompatibility, stability, flexibility, and suit the already used methods. Surfactants can interact with vesicular membranes and alter surface tension by increasing solubility since surfactants can be inserted into the bilayer (Chabanon and Rangamani, 2018).
Polysorbates are the most widely used non-ionic surfactants because they can form vesicles without the presence of cholesterol. This is due to the presence of a larger hydrophobic fraction in the molecule, therefore having a lower solubility in water, which promotes the organization in the colloidal system (Junyaprasert et al., 2012). An example is polyoxyethylene sorbitan-80 monooleate (Tween 80) which is a widely accepted surfactant in pharmacological formulations, this owing to the presence of a polyoxyethylene chain which balances the hydrophilic and lipophilic domains (Kaur and Mehta, 2017). The study by Alomrani et al. (2015) showed that delivery of the Itraconazole® drug in the skin and stratum corneum, is increased when the compound is incorporated into niosomes prepared with Tween 80, increasing the antifungal effect of the drug.
When the concentration of the nonionic surfactant is increased in the formulation, it promotes an increase in the hydrophobic interactions with the drug; an example is the incorporation of curcumin, where a stronger bond between the compound and the lipid membrane can occur promoting high stability of the formulation (Raval et al., 2018; Wang and Gao, 2018). Junyaprasert et al. (2012) have shown that Tween-60-based niosomes are efficient carriers in the dermal delivery of ellagic acid. This characteristic is related to the low capacity that Tween 60 has in solubilizing structures based on phospholipids, which confers to this molecule potential for the formation of lipid vesicles for substance transport (Lim and Lawrence, 2004).
Another widely used surfactant for the preparation of lipid vesicles are the sorbitans (Span 60 and Span 80). It has been shown that vesicles prepared with Span 80 are more flexible, and their membrane is more fluid compared to liposomes prepared with phospholipids only (Hayashi et al., 2011). In a study on the release of resveratrol, it has been found that Span 60-based formulations promote the release of the compound more slowly than when compared to Span 80-based formulations (Saha and Verma, 2018).
In contrast, vesicles formed with Span 80 are suitable antigen carriers and when lyophilized obtain stability of up to nine months (Freixeiro et al., 2017). The high stability of vesicles prepared with Span 80 is confirmed in the study by Hua and Liu (2007), where it was shown that formulations with Span 80 and PEG 400 are highly stable and can remain without changes in their morphology and size for roughly a year, this occurring due to the hydrophilicity regulation that the PEG molecule offers, while the Span 80 molecule has a high hydrophobic capacity which makes it less conducive to vesicles formation.
An increase in surfactant concentration leads to a decrease in the size of the vesicle, as well as reducing aggregation and increasing the stability of the formulation by increasing the charge, while the entrapment efficiency of the substance depends not only on the surfactant but also on the structure of the substance to be encapsulated (Bnyan et al., 2018).
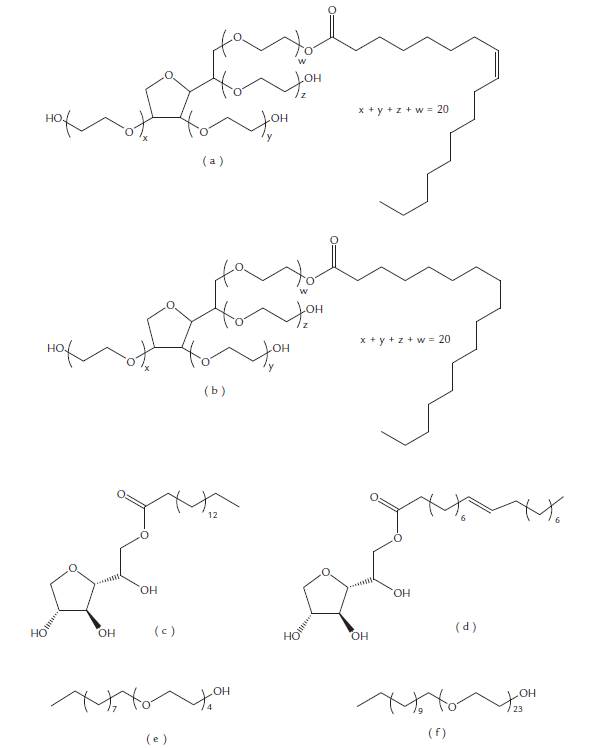
Another nonionic surfactant used in formulations is the Brij family, whose molecule contains an oxyethylene group hydrophilic chain and a hydrophobic group composed of hydrocarbons. These are considered unusual for lipid vesicle formulations because they have low toxicity and are good solubilizers (Ribeiro et al., 2012). Liposomes prepared with short chain Brij (Brij30 and Brij35) have low encapsulation potential, whereas formulations prepared with Brij52, and Brij56 have an incorporation capacity greater than 80 %, yet they have an ideal substance release profile (Tagami et al., 2011). Niosomes prepared with Brij 72 and Span 20 demonstrate a rapid blood clearance capacity system; however, when PEG is added to the membrane the vesicles remain in the blood system longer (Alomrani et al., 2015). Figure 4 shows the chemical structures of the most used non-ionic surfactants for lipid vesicle formulations.
Physico-chemical aspects
The characterization of the vesicle is an essential factor that can influence the stability, durability, targeting, location, accumulation, and elimination of the encapsulated substance, having a direct influence on the behavior of the nanoparticle within the biological system. One of the most important physicochemical factors to be analyzed even before vesicle production is the phase transition temperature in which the phospholipids are to be used in the formulation. This parameter is directly linked to membrane fluidity and stability since the transition temperature is an important tool for the understanding of several colloidal systems (Matsuki et al., 2017). Generally, temperature variation occurs when a component is added to the vesicular membrane, this being influenced by the degree and the branching point of the hydrated phospholipid vesicles (Mitchell et al., 2016). The incorporation of substances such as sodium cholate decreases the transition temperature of the formulation, making it more flexible (Kanaani et al., 2017).
Another critical factor to be analyzed is the surface charge (zeta potential) and particle size since these characteristics can affect the stability of the formulation. The zeta potential has a direct influence on the surface charge density of the vesicle, and its determination can provide the orientation of compounds in the vesicle as amino acids (Disalvo and Bouchet, 2014; Morini et al., 2015). When the zeta potential is reduced, it means that the vesicle has low or no surface charge and cannot repel itself, which can lead to aggregation, this potential being an indicator of the colloidal system stability (Santos and Castanho, 2002).
Ravaghi et al. (2016) showed that the zeta potential of niosomes is influenced by the type of surfactant used, and preparations with Span 60 have better potential values and longer stability time. The amount of cholesterol used in the formulation also influences these values, having an inverse correlation between the amount of cholesterol and the potential value, with zeta potential values ± 30 mV or higher being considered ideal (Tahara et al., 2018). Potential values can be improved by the addition of charge-inducing agents such as diethylphosphate which are ideal for formulations which do not have a zeta potential higher than 30 mV and are unstable, as they induce vesicle stability by preventing aggregation (Khan et al., 2016).
Some physical-chemical factors influence zeta potential values, one of the most important being pH where obtaining high or low values are correlated with an acidic or basic pH, respectively. Ionic strength is also important, where its increase tends to decrease potential values. The study of these factors is related to the stability of the colloidal system (Bhattacharjee, 2016).
Techniques for obtaining liposomes and niosomes
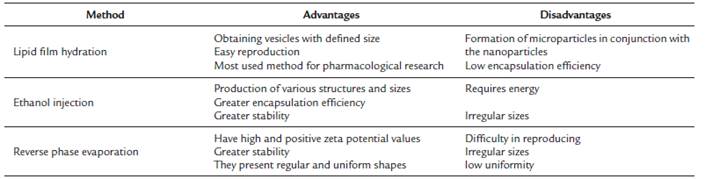
Techniques to produce niosomes and liposomes are diverse and lead to the formation of different vesicles, with different sizes and lamellae quantity. Some techniques are more prominent in laboratories, as they are considered more accessible to reproduce and safer. It is worth noting that in all methods, some of these components are used: lipids such as phosphatidylcholines (soy or egg), sterols such as cholesterol and non-ionic surfactants, either in combination or in different formulations (Chorilli et al., 2006). Thus, they also need some energy either by shaking, sonication or centrifugation. The use of energy is necessary to produce uniform sizes. Generally, the amount of the components is decided from several tests with different molar ratios, until the desired formulation is obtained. The methods listed below indicate how the techniques are developed in most lipid vesicle research.
Lipid film hydration
This method was developed by Bangham et al. (1965), it is one of the most used for lipid vesicle formulations. Over the years it has undergone several adaptations aimed at improving it. Initially, a solution containing phosphatidylcholine, cholesterol, and a non-ionic surfactant such as Tweens or polysorbates should be prepared. These components should be diluted in a mixture of organic solvents (chloroform/ethanol). From this solution, a lipid film will be obtained by evaporating the solvent with the aid of a vacuum pump. It is important that all the solvent is evaporated. The film should be hydrated with water or buffer solution for a period in which the film is fully released from the walls of the flask (approximately 30 minutes). It is necessary to use some energy for the formation of lipid vesicles, where sonication is usually used for 5-10 min or vortexing (Abaee and Madadlou, 2016).
A disadvantage of this method is the formation of microparticles in conjunction with the nanoparticles. This problem can be solved by agitation, which promotes the homogenization and uniformity of the particles. Another impasse is the amount of hydrophilic substance to be encapsulated, knowing that a large part of these can be dissolved in the aqueous medium in which the vesicle was formulated (Eloy et al., 2014). Liposomes prepared by this method are important in studies with anticancer substances because they allow a small amount of the drug to be adequate in up to 99 % in the diminution of cancerous cells. They can promote the targeting of substances and can pass through barriers such as the blood-brain barrier (Singh, 2018; Sun et al., 2018; Zhao et al., 2018)
Ethanol injection
First described by Batzri and Korn (1973) as an alternative for the preparation of small unilamellar vesicles, ethanol injection is currently used for the development of several structures with different sizes from adaptations in the number of components used (Charcosset et al., 2015). A surfactant mixture with ethanol is injected with a syringe, approximately 130 ml/h in aqueous solution at 40 °C under constant stirring at up to 5 000 rpm with the aid of a homogenizer. The vesicle formation occurs at the time of contact between the organic and the aqueous solution; however, vigorous agitation is necessary to obtain narrower and uniform sizes. After roughly five minutes, the ethanol should be removed with the aid of a rotary evaporator under reduced pressure. The vesicles remain in the aqueous phase and may require some energy, by shaking or sonication (Santos and Castanho, 2002). This is one of the simplest and most widely used methods because it is easily reproducible.
Niosomes prepared by this method have higher encapsulation efficiency when compared to the film hydration method; also, the encapsulated substance has a higher penetration capacity when applied to the skin (Saha and Verma, 2018). Toniazzo et al. (2017) have shown that quercetin-loaded liposomes formulated by this method exhibit considerable stability and zeta potential of -17 mV, such characteristics are typical of small vesicles with excellent stability. The small vesicle size obtained by this method is confirmed in work by Liang et al. (2016), which obtained sizes of 60 nm.
Reverse phase evaporation
The surfactants and lipids are diluted in an organic solvent mixture (ethyl ether, chloroform, ethanol or methanol) in different molar ratios. A buffer solution and/or water are added to this mixture and sonicated for ten minutes. The solvent is evaporated with the aid of a rotary evaporator under reduced pressure, so that there is gel formation which must then be rehydrated with an aqueous solution. Non-dissociation of the viscous gel after contact between the vesicles requires high agitation to separate and obtain lipid vesicles (Ojeda et al., 2016).
Niosomes prepared by this method has a nano-scaled size that is appropriate for gene delivery, and a high and positive zeta value potentials, which ensures the stability of the vesicles (Attia et al., 2018). The formulation of conventional liposomes by this method promotes a higher encapsulation potential when compared to the film hydration and ethanol injection methods, and the formed vesicles have ideal zeta potential with regular and uniform forms (Wang et al., 2016b). In another study, the efficiency of curcumin entrapment in liposomes prepared by this method was demonstrated, increasing the release of the substance in chitosan-covered liposomes (Cuomo et al., 2018). An increase in the release of the drug Gemcitabine® was also observed by incorporation into liposomes with laser treatment. The production of vesicles by this method is efficient in obtaining formulations with potential for controlled release of substances useful for the treatment of different diseases (Fuse et al., 2018). Table 2 shows the advantages and disadvantages of these methods.
General considerations
Finding an ideal formulation requires investigation of various parameters, such as hydration medium used, the ideal molar ratio between components, bilayer composition and characteristics of the component to be encapsulated (Sohrabi et al., 2016). It was observed that phosphatidylcholine is the most used phospholipid in liposomal and niosomal formulations because it is considered biocompatible and non-toxic. In order to improve the characteristics of the formulation, some types of nonionic surfactant may be added; the use of two different surfactants may improve the stability and increase the encapsulation efficiency of nasal formulations (Basiri et al., 2017). Cholesterol participation is essential for the determination of membrane fluidity or rigidity, in addition to acting as a preservative by increasing the durability of the vesicle. The analysis of the different methods shows that vesicles prepared by film hydration or ethanol injection do not differ in their physicochemical characteristics, do not present cytotoxicity, and have near stability time. The reverse phase method is ideal for small multilamellar formulations with high controlled release potential. The finding of an ideal formulation is observed through physical-chemical analyzes such as zeta potential and encapsulation potential. This article proposes that the production of lipid vesicles depends on some factors, such as the amount of cholesterol and the presence of agitation. It is being these two essential factors in obtaining vesicles more uniform and stable.
The use of lipid vesicles has a promising future in the controlled delivery of drugs contributing significantly to the development of nanomedicine.