Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Salud Uninorte
Print version ISSN 0120-5552On-line version ISSN 2011-7531
Salud, Barranquilla vol.26 no.1 Barranquilla Jan./June 2010
ARTÍCULO DE REVISIÓN / REVIEW ARTICLE
REVISIÓN BÁSICA / BASIC REVIEW
Nondisjunction and chromosomal anomalies
La no disyunción y las anomalías cromosómicas
Mostapha Ahmad1, Silvera-Redondo C.1, Muna Hamdan Rodríguez2
1 Grupo de Investigación Genética y Medicina Molecular, Universidad del Norte, Barranquilla (Colombia). mahmad@uninorte.edu.co
2 Departamento de Enfermería, Universidad del Norte, Barranquilla. munahamdan85@gmail.com
Correspondencia: Universidad del Norte, Km 5 via a Puerto Colombia, Barranquilla (Colombia).
Fecha de recepción: 2 de febrero de 2010
Fecha de aceptación: 15 de marzo de 2010
Resumen
La no segregación es el fracaso de los cromosomas homólogos en separarse correctamente durante la meiosis. Esto resulta en la producción de gametos que contienen una cantidad de cromosomas mayor o menor a la encontrada en una célula normal. Consecuentemente, el individuo puede desarrollar una trisomía o monosomía. La no disyunción puede ocurrir en meiosis I o meiosis II de la división celular, es una causa de diversas condiciones médicas anormales, incluyendo el Síndrome de Down (trisomía del cromosoma 21), Síndrome de Patau (trisomía del cromosoma 13), Síndrome de Edward (trisomía del cromosoma 18) y Síndrome de Turner (la presencia de un solo cromosoma X). A pesar de que es la causa de numerosos trastornos genéticos, aún no se conoce su etiología exacta y el proceso en el cual se lleva a cabo. La no disyunción se origina en el mayor de los casos de errores en la meiosis II materna, sin embargo, la meiosis paterna y la meiosis I materna influyen en ella. La edad materna se considera como un factor de riesgo de las trisomías, igual que la alteración de la recombinación y otros factores que pueden afectar la segregación cromosó-mica, tal como la genotoxicidad y translocaciones cromosómicas. Esta revisión se realizará con base en artículos publicados entre 2003 y 2009 en ISI Web, Science Direct, PUBMED, SPRINGER y SCIELO; se interpretará y analizará en ella los resultados de estos estudios que lograron demostrar conclusiones importantes y sobresaltaron factores interesantes que pueden ser el punto de partida para próximas investigaciones.
Palabras clave: No disyunción, monosomía, trisomía, miosis, mitosis, recombinación, síndrome, genotoricidad, mutación cromosómica.
Abstract
Nondisjunction is the failure of homologous chromosomes to disjoin correctly during meiosis. This results in the production of gametes containing a greater or lesser chromosomal amount than normal ones. Consequently the individual may develop a trisomal or monosomal syndrome. Non disjunction can occur in both Meiosis I and Meiosis II of the cellular division. It is a cause of several abnormal medical conditions, including Down's syndrome (trisomy of chromosome 21), Patau's Syndrome (trisomy of chromosome 13), Edward's Syndrome (trisomy of chromosome 18) and Turner's Syndrome (the presence of only one X chromosome). It is also the main cause of many genetic disorders, however its origin and process remains vague. Although it results in the majority of cases from errors in the maternal meiosis II, both paternal and maternal meiosis I do influence it. The maternal age, is considered a risk factor of trisomies, as well as recombination alterations and many others that can affect the chromosomal segregation, such as genotoxicity and chromosomal translocations. We will review the results of previously realized studies between the years 2003 and 2009, found in ISI WEB, PUBMED, SCIENCE DIRECT,SPRINGER LINK and SCIELO, that led to important conclusions and highlighted interesting factors that can be the starting point to future investigation.
Key words: Nondisjunction, monosomy, trisomy, Meiosis, Mitosis, Recombination, Syndrome, genotoxicity, chromosomal mutation.
INTRODUCTION
Investigators have been studying the aneuploids human conditions after the description of the first case of trisomy 21 (Down syndrome) on 1959 by Lejeune and Patricia Jacob. They focused their investigation and studies on the genesis of this abnormal condition developed by humans. Recently, aneuploidy was also confirmed experimentally as a dominant mutator, independent of gene mutation in other eukaryotes, including Drosophila, yeast, plants, and mice. These studies were centered in searching about three basic things; the frequency of aneuploid conditions, the reason of gaining (extra) or loosing (less) chromosomes and the nondisjunctional mechanism that gives rise to aneuploid conditions. Despite the technological development and the number of realized studies in aneuploid genesis, investigators still don't have clear answers to the second and the third interrogates, however the first one has been answered. The frequency of aneuploid conditions which is amazingly common and clinically important in our species is estimated in approximately 5% of clinically recognized pregnancies (1). Therefore we will try to recombine all the results of previous studies, in order to reach a conclusion that defines the causes of chromosomal nondisjunction and answer the question wheather maternal age is its only trigger or there are still other factors behind it?
HOW DOES NONDISJUNCTION OCCUR?
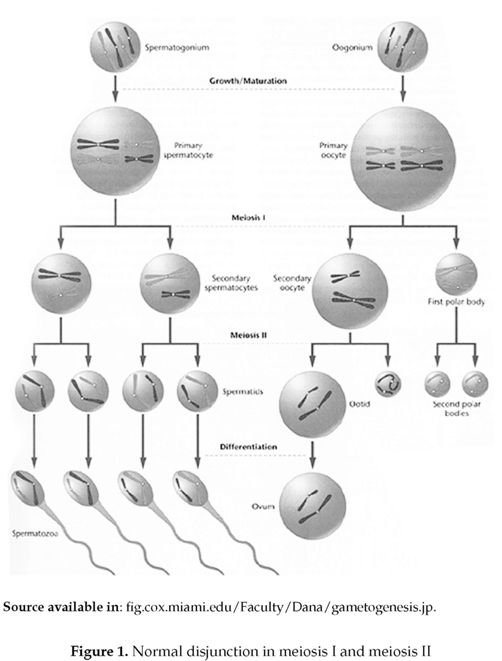
Nondisjunction is the miss segregation of a homologous pair of chromosomes during meiosis (figure 1). It leads to the formation of a new cell with an abnormal amount of genetic material. A number of clinical conditions are the result of this type of chromosomal mutation.
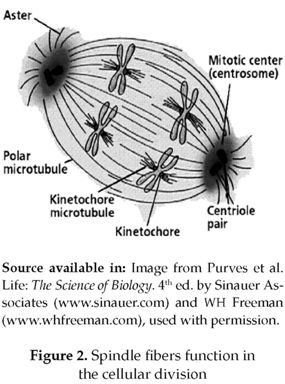
Homologous chromosomes are identical chromosomes that can be observed in pairs, in which 50 °% of this amount is inherited from each parent. Humans have 46 chromosomes, or 23 homologous pairs. Normally, in meiois the homologous chromosomes attach to spindle fibers (figure 2), which connect the 2 centrioles and become aligned at the cell equator. Before the first meiotic division takes place the homologous pairs migrates to the cell's opposite poles by means of the pulling action of the spindle fibers, and upon meiosis completion each gamete will have one copy of every chromosome.
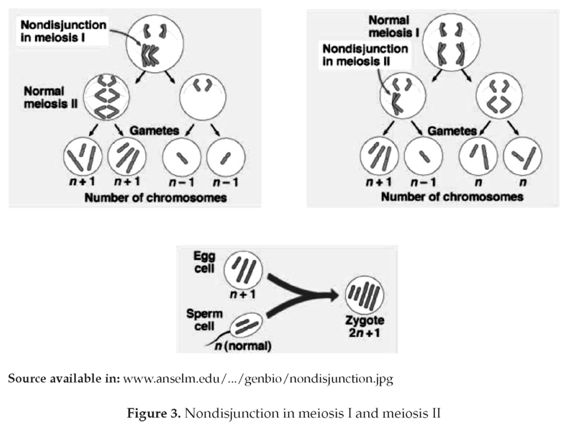
However, this process can suffer some errors that lead to homologous chromosomes separation failure and thus both migrate to the same pole (figure 3). Consequently two types of gametes are produced, one of which has two chromosomal copies, whereas the other lacks one. A zygote which has a chromosome less than the normal diploid amount (2n-1) is called monosomic, and that which has an extra chromosome (2n+1) is trisomic, both conditions may develop severe abnormalities that can be fatal (1).
CAUSES AND FREQUENCY OF NONDISJUNCTION IN HUMANS
Meiosis is a process that consists of a number of check points that regulate cell division in all its phases to ensure that the cells will divide and give rise to new ones correctly without any error. In case an error occurs these regulating points must correct it. One of these check points is the spindle fibers checkpoint which is responsible of three principle steps; formation of these spindle fibers, the attachment of chromosomes to them and the adequate segregation of these chromosomes. When any check point fails in realizing its correct function, it leads to nondisjunction, as a result of the incorrect segregation of the homologous chromosomes.
Despite the insufficient studies about the causes of nondisjunction, it is known to occur more frequently in older cells. This is why older women may give birth to affect off springs due to an aneuploid abnormality more than young ones. The risk of a twenty-years-old mother giving birth to a child with Down syndrome is about one in two thousand and it increases to one in thirty in the case of a forty-five years old woman. This hypothesis depends on a simple elucidation that in older cells the regulating system does not function as in younger cells and as a consequence the cell will lose the control. Thus an older cell undergoing meiosis would be more likely than a younger one to ignore the constraints of the spindle checkpoint and hence give rise to aneuploid cells. This was confirmed by a study done in patients with Down syndrome which demonstrated that the incidence of this syndrome was elevated with increased maternal age. Many specialists recommend that women who become pregnant at age 35 or older must undergo prenatal testing for Down syndrome. The probability of pregnant fewer than 30 to give birth to a baby with Down syndrome is less than 1 in 1,000, but the chance of having a baby with Down syndrome increases to 1 in400 in women who become pregnant at age 35. The likelihood of Down syndrome continues to increase as women ages do, so that by age 42, the chance is 1 in 60 and by age 49, the chance is 1 in 12. But using maternal age alone will not detect over 75% of pregnancies that result in Down syndrome (10).
No disjunction doesn't only relate with maternal conditions, but also with paternal and mitotic conditions. A study verified that trisomy 21 was 90.9% maternal, 4.5% paternal and 4.5% from a mitotic origin; similar to distributions reported previously (4). It also confirmed that nondisjunction doesn't only take place in meiosis II but also in meiosis I (MI: 46.1%, MII: 53.9%).Even though it was established by other studies that MI is 70% and MII is 30% related to Down syndrome, it was reported that what causes it in 88% of cases is the extra copy of chromosome 21 derived from the mother, in 8% of the cases the father provided the extra copy of chromosome 21 and in the remaining 2% Down syndrome is due to mitotic errors; an error in cell division which occurs after fertilization when the sperm and ovum are joined (10). It was also reported in 82 patients with trisomy 13 that the parental origin was determined in every case and in 89% the extra chromosome 13 was of maternal origin with an almost equal number of maternal MI and MII errors (8).

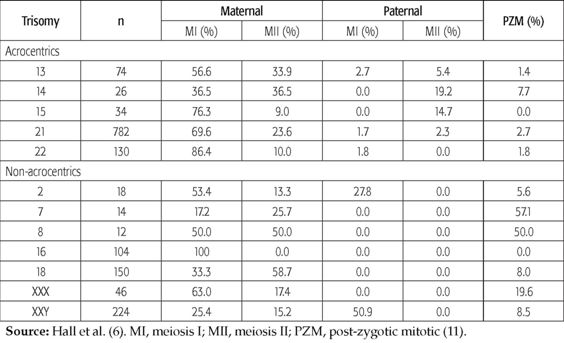
Until here we achieved to demonstrate that nondisjunction is from maternal, paternal and mitotic origin, the distinctive difference are the frequencies and percentages as we can see briefly in the table below.
Adapted from Hall et al. (6). MI, meiosis I; MII, meiosis II; PZM, post-zygotic mitotic (11). The direct exposure to high levels of genotoxic gaseous and particulate substances from the engines combustion used in motor vehicles is required by certain type of occupations. These occupational exposures may convert into an important cause of many illnesses, usually through chromosomal changing mechanisms that include strand breakage, deletions, sister chromatid exchange and non-disjunction. To determine the effect of occupational exposure in gasoline station attendants and traffic enforcers, the micronucleus test was used in three groups: gasoline station attendants, traffic enforcers and a group of control. A study showed no relation between MNC frequency and any of the factors such as age, smoking habits, alcohol habits and working period. This was further confirmed in the multiple regression analysis which showed that only occupational exposure was a good predictor of MNC frequency. The results of this study suggest that gasoline station attendants and traffic enforcers, compared to the control individuals, are at a greater risk of chromosomal damage. For the assessment of chromosomal damage, the study, development, and standardization of tests are recommended for public institutions concerned with matters regarding environmental quality and community health (12).
It was also shown in six males(13), carriers of Robertsonian translocation that higher incidences of aneuploid sex chromosomes in spermatozoa were found in three Rob translocation carriers, which indicated that the ICE (interchromosomal effect) on sex chromosome is likely in some male carriers of Rob translocations. This study suggests that genetic counseling is important for the carriers of Rob translocations. In order to maximize the chances of normal pregnancy, they highly recommend that normal or balanced embryo should be selected for transfer by preimplantation genetic diagnosis analysis of translocation chromosome, accompanied with a preimplantation genetic screening for sex chromosome aneuploidy (13).
On the other hand meiotic origin and the stage of non-disjunction of the extra X chromosomes in two sisters with 47, XXX chromosomal complements were studied (14) and demonstrated that the lack of recombination in the X chromosomes suggests a possible maternal genetic defect leading to an erratic recombination at MI. This information may contribute to further understanding of mechanisms leading to X chromosome non-disjunction and may assist in counseling of families with this chromosomal rearrangement (14).
CHROMOSOMAL MUTATION AND ITS CLASSIFICATION
Chromosomal mutations are variations from the wild-type condition in either chromosome structure or chromosome number. Variation in chromosome number includes aneuploids, which do not involve whole sets of chromosomes (genomes) but only parts of a set (genome) (aneu-uneven; ploid-unit). They may be of the following types: Monosomy; diploid organisms which lack one chromosome of a single homologous chromosome pair are called monosomics and have the genomic formula 2n -1. A monosomic produces two kinds of gametes (n) and (n-l), as the single chromosome missing a pairing partner may travel to either pole during meiosis. In plants n -1 gametes remain non-functional, whereas in animals they result in genetic imbalance which is manifested by high mortality or reduced fertility. Nullosomy; diploid organisms which have lost a pair of homologous chromosomes are called nullosomics and posses the genomic formula 2n-2. The nul-losomics exhibit reduced vigor, fertility and survivability, but polyploidy nullosomics such as nullosomic hexaploid, having (6n-2) survive to maturity due to the genetic redundancy in polyploidy. Trisomy; diploid organisms which have one extra chromosome are called trisomies. They have the chromosomal formula 2n+1. In a trisomic, one of the chromosomal pairs has an extra member, forming a trivalent structure during meiosis. During anaphase, two chromosomes travel to one pole and the third to another. Thus, two types of gametes n + 1 and n are resulted. Trisomy has variable effects on the phenotype of the organism. In humans trisomy of autosome 21 causes Down syndrom. Tetrasomy, it results when one chromosome of a diploid organism is present in quadruplicate. Tetrasomics have the chromosomal formula 20+2. During meiosis a quadrivalent is formed by extra chromosomes and segregation of chromosomes occurs like autotetraploids. Double Trisomy; in a diploid organism when two different chromosomes are represented in triplicate, double trisomy results. A double trisomic has the chromosomal formula 2n+1+1. Although euploidy (eu-true or even; ploid-unit) designates genomes containing whole sets of chromosomes, it is very important to distinguish between aneuploid conditions mentioned previously and mixoploidy (mosaics), which refers to the presence of two cell lines, one diploid and the other polyploid. Though polyploidy in humans is not viable, mixoploidy has been found in live adults and children. Mixoploidy consists of two types: diploid-triploid mixoploidy, in which some cells have 46 chromosomes and others have 69. Diploid-tetraploid mixoploidy, is characterized by cells having 46 chromosomes and others having 92. Euploids are organisms that posses balanced set or sets of chromosomes or genomes in any number, in their body cells. Euploidy is of the following types: Monoploidy, in this case organisms have one genome (n) in their body cells. When monoploidy occurs in gametes (sperms and eggs) it is termed as haploidy. Diploidy, is characterized by two genomes (2n) in each somatic cell of diploid organisms. Most animals and plants are diploids. Diploidy is related with fertility, balanced growth, great vigorosity, adaptability and survivability of the diploid organisms. Polyploidy is the condition where organisms have more than two genomes. Among plants and animals, polyploidy occurs in a multiple series of 3, 4, 5, 6, 7, 8, etc., of the basic chromosome or genome number and thus causes triploidy, tetraploidy, pentaploidy, hexaploidy, heptaploidy, octaploidy, respectively. Ploidy levels higher than tetraploid are not commonly encountered in natural populations, but our most important crops and attractive flowers are polyploidy, e.g., wheat (hexaploid, 6n), strawberries (octaploid, 8n), many commercial fruit and attractive plants, liver cells of man, etc. Variation in chromosomal structure includes deletion (loss), duplication (gain), inversion (a segment of a chromosome is reversed end to end), and translocation (exchange segments). These mutations in chromosomal structure are caused by unequal crossover and/or abnormal segregation of chromosomes during mitosis. Unbalanced chromosomal rearrangement has loss or gain of genetic material, which may causes phenotype disorders or diseases. Balanced chromosomal rearrangement may also cause mutations through changes in gene expression.
LETHAL AND NON-LETHAL HUMAN ANEUPLOID CONDITIONS
Aneuploid conditions are divided into lethal and nonlethal. Lethal aneuploid conditions are related to gene dose and its importance in the development. Each normal cell contains 46 chromosomes (1 pair of sex chromosomes that can be XX in females or XY in males and 22 pairs of autosomal chromosomes). In order to develop appropriately, the cell must contain the correct dose of genes and each gene in its correct position. A change in the gene dose and position can occur by removing or adding chromosomes to the normal set resulting in disproportion and developmental problems. Not all gene duplication or silencing cases are lethal; it is the addition or loss of a chromosome that contains 1000 or more genes, that results in lethality. Down syndrome, caused by trisomy 21 is an example that demonstrates the cell's tolerance to small changes but not to large ones. Chromosome 21 contains a small number of genes for it is one of the smallest chromosomes, thus any change in it will not lead to mayor effects approving why Down syndrome is not a lethal condition. On the contrary, when a large amount of genes contained by large chromosomes is involved, it may be lethal, this occurs mainly with autosomal chromosomes, where as in sex chromosomes it is less probable. The X chromosome can illustrate such condition, despite of its large size only one is involved in the female development. On other hand, the Y chromosome contains a few genes but is indispensable in male development. From these explanations we can understand that in some conditions where the sex chromosomes are involved lean not to be lethal, however, the YO condition is fatal due to the lack of the essential X chromosome.
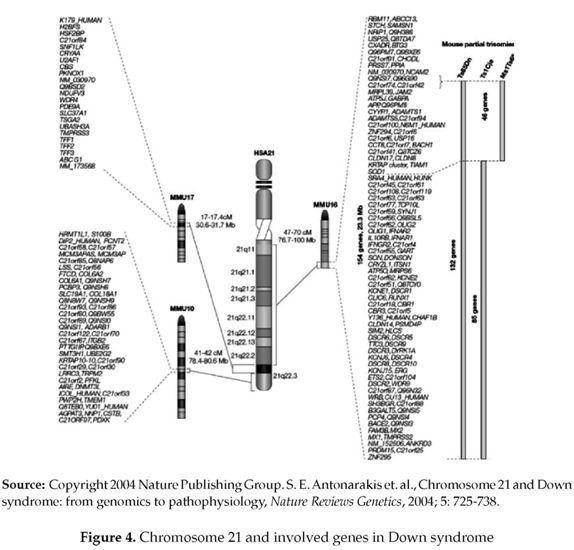
Down syndrome, described for the first time by Jerome Lejeune and Patricia Jacobs in 1959 is the most important non-fatal trisomy in humans. Caused by the presence of an extra copy of chromosome 21(figure 4), it gives rise to an extra set of genes leading to an over expression of the involved ones and an increase in the production of certain products. For most genes, their over expression has little effect due to the body's regulating mechanisms of genes and their products, but those causing Down syndrome appear to be an exception. They are Superoxide Dismutase DOS1 ( its over expression may cause premature aging and decreased function of the immune system and its role in Senile Dementia of the Alzheimer's type or decreased cognition is still speculative)(2), COL6A1( its over expression may be the cause of heart defects) (2), ETS2 ( its over expression may be the cause of skeletal abnormalities) (2), CAFA1( its over expression may be detrimental to DNA synthesis)(2), Cystathione Beta Synthase or CBS and GART (their over expression may disrupt DNA metabolism and repair ) (2), DYRK ( its over expression may be the cause of mental retardation) (2), CRYA1( its over expression may be the cause of cataracts ) (2), IFNAR or the gene coding for Interferon( its over expression may interfere with the immune system as well as other organs)(2). Other genes that also represent suspects in clude APP, GLUR5, S100B, TAM, PFKL, besides few others that are not being mentioned (2). Again, it is important to note that no gene has yet been fully linked to any feature associated with Down syndrome (2).
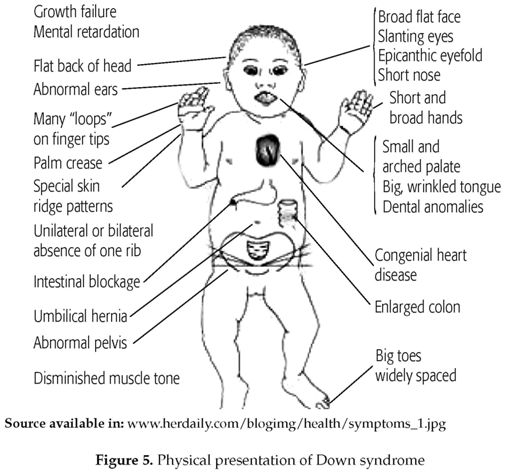
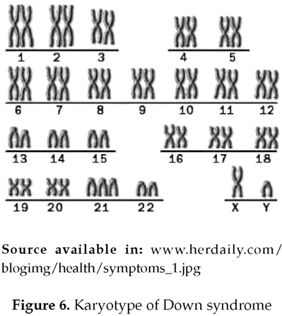
Down syndrome is an example of an au-tosomal trisomy as it involves autosomal chromosome 21 (3). It is reported that trisomy 21(figure 5 & 6) is 90.9% maternal, 4.5% paternal and 4.5% from a mitotic origin (4).
There are clear differences in the frequency of maternal meiotic errors as reported in distinct studies. Ramirez et al. (4 ) reported that 46.1% of these errors originate from meiosis I and 53.9% from meiosis II, on the other hand a previous one encountered that 70% of the errors resulted in meiosis I and 30% in meiosis II. Multiple correspondence analyses showed association of either local recombination events or absence of recombination with specific non-disjunction stages. Recombination patterns reported by Ramirez et al. (4) support the hypothesis that susceptible chiasmate configurations are associated to maternal meiosis I and meiosis II errors. Similarly, it was reported in other studies, using DNA polymorphisms to identify the origin of the extra chromosome 21(5). In the largest meta-analysis study of 807 Down syndrome patients, the parental origin was maternal in 90.7% of cases, paternal in 5.5% and mitotic in the remaining 3.8% (6). Additionally, when pericentromeric microsatellite markers were used to detect the meiotic stage of nondis-junction, 76% of maternal cases were found to occur at meiosis I (MI) and 24% at meiosis II (MII); in the cases of paternal origin, a 1:1 ratio between stages was found, with a slight excess of MII errors (7).
Trisomies can also affect another autosomal chromosomes, such as trisomy 13(Patau syndrome), trisomy 18 (Edwards syndrome), trisomy 15, trisomy 16 and trisomy 22. A study done by Merete B et al (8), which included 82 patients affected by trisomy 13, reported that the parental origin was determined in every case and in 89% the extra chromosome 13 was of maternal origin with an almost equal number of maternal MI and MII errors. There was evidence for elevated maternal age in cases with maternal meiotic origin for born-alive infants. Maternal and paternal ages were elevated in cases with paternal meiotic origin. This comes in contrast to results from a similar study of trisomy 21 non-disjunction, where paternal but not maternal age was elevated (6). Clear evidence for reduced recombination was found in both maternal MI and MII errors where the cause was associated with a significant number of tetrads (33%) that are nullichiasmate, which does not appear to be a feature of chromosome 13 normal meiosis. Merete B et al support in their study (8) the evidence for subtle chromosome-specific influences on the mechanisms that determine non-disjunction of human chromosomes, consistent with the diversity of findings for other trisomies.
GENETIC DISORDERS CAUSED BY ROBERTSONIAN TRANSLOCATION, MOSAICS AND RING CHROMOSOME
Some syndromes result from mosaics or Robertsonian translocation. Robertsonian translocation is a common and significant type of chromosome rearrangement that is formed by fusion of the whole long arms of two acrocentric chromosomes (chromosomes with the centromere near the very end). They are also known as whole-arm or centric-fusion translocations or rearrangements. Robertsonian translocations are named for the American insect geneticist W.R.B. Robertson who first described this form of translocation in the grasshoppers in 1916. One in about 900 babies is born with a Robertsonian translocation making it the most common kind of chromosomes rearrangements known in humans. All five of the acrocentric chromosomes in humans (chromosome number 13, 14, 15, 21 and 22) have been found to engage in Robertsonian translocations. The formation of Robertso-nian translocations was discovered by Hecht and coworkers to be highly nonrandom. In balanced form, a Robertsonian translocation takes place between two acrocentric chromosomes and results in no problems for the person carrying it. But in unbalanced form, Robertsonian translocations produce chromosome imbalance and cause syndroms of multiple malformations and mental retardation. Robertsonian translocations between chromosomes 13 and 14 lead to trisomy 13 (Patau) syndrome. Robertsonian translocations between chromosomes 14 and 21 and between 21 and 22 do also result in other syndroms.
Normally, when an egg and a sperm are joined at conception, a single cell is created with a total of 46 chromosomes. These chromosomes are copied, the copies are separated, and the cell then divides to create two identical "daughter" cells. The chromosomes in these two cells are copied, the copies divide, and four cells are created. These four cells become eight cells. Eight cells become 16 cells, and so on. If nothing disrupts the chromosome replication and separation process, each cell in the body should have the same number of chromosomes that were present in the fertilized egg. However, errors can occur in this replication and separation process. Two mechanisms have been proposed to explain why a child may be born with mosaic Down syndrome. The most likely explanation is that an extra copy of chromosome 21 was present in the egg or the sperm at the time of conception. However, shortly after conception, an error occurred in the chromosome replication and separation process, and the extra copy of the chromosome 21 was not passed on to both cells. In this way, a second cell grouping was created with only 46 chromosomes. If this error in the chromosome replication and separation process occurred at the 4 cell stage, 1/4 of the cells would have 46 chromosomes and 3/4 would have 47 chromosomes. If the error occurred at the 8 cell stage, 1/8 of the resulting cells would have 46 chromosomes and 7/8 would have 47. It is also possible that a child with mosaic Down syndrome inherited a total of 46 chromosomes at the time of conception. If this was the case, then the error in chromosomal separation, which resulted in the formation of a second cell grouping with an extra chromosome 21, occurred early in the baby's development, this is the mosaics which can result also in other syndromes like Patau syndrome, Turner syndrome, etc. On the other hand, many genetics disorders may arise from ring chromosome, which is a chromosome whose arms have fused together to form a ring. A ring chromosome is denoted by the symbol r. Ring chromosomes may form in cells following genetic damage by mutagens like radiation, they may also arise spontaneously during development. Although ring chromosomes are very rare, they have been found in nearly all human chromosomes. Disorders arising from the formation of a ring chromosome include ring chromosome 20 syndrome where a ring formed by one copy of chromosome 20 is associated with epilepsy. Ring chromosome 14 and ring chromosome 13 syndrome are associated with mental retardation and dys-morphic facial features. Ring chromosome 15 is associated with mental retardation, dwarf-ism and microcephaly. Ring formation of an X-chromosome causes Turner syndrome. Symptoms seen in patients carrying ring chromosomes are more likely to be caused by the deletion of genes in the telomeric regions of affected chromosomes, rather than by the formation of a ring structure itself.
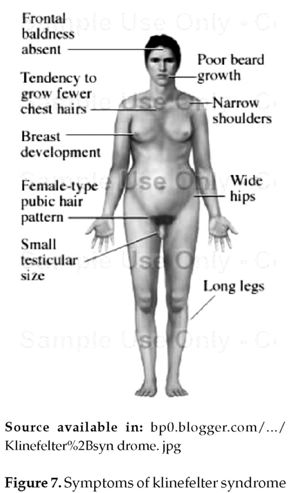
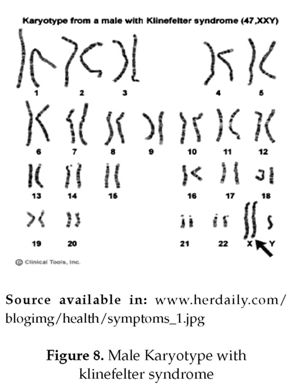
Abnormalities in the autosomal chromosomes is not the only consequence of non disjunction, sometimes the sex chromosomes will also be involved and affect the individual's secondary sexual characteristics and fertility. For example, Klinefelter's syndrome, in which only the gonosomal chromosomes are affected and the autosomal ones are normal. It results from of the fusion of an XY sperm with a normal X egg, or the fusion of a Y sperm with an XX egg. Individuals affected by Klinefelter's syndrome, usually have below-average intelligence (figure 7) (3). 75% of these individuals present the karyotype 47, XXY (figure 8). Approximately 20% of the cases result from chromosomal mosaicism which is the major cause represented in 46, XY/47, XXY. Other variants including 48, XXYY, 48, XXXY, and 49, XXXXY exist in 5% of the cases (3) that were first described by Fraccaro and Lindsten in 1960.
Orit R et al. reported the determination of the meiotic origin and the stage of non-disjunction of the extra X chromosomes in two sisters with 47, XXX chromosomal complement (9). Segregation of the X chromosomes in all family members was analyzed using X-linked short tandem repeat polymorphic (STRP) markers. Densitometric analysis of two STRP markers confirmed that both sisters had three copies of the X chromosome and the extra X chromosomes were maternally derived. Both sisters did not share the same maternal homologue, suggesting that the recurrent trisomy is non-homologous X chromosome-specific. Haplotype analysis demonstrated a reduction to homozygosity for markers examined, covering most of the length of the X chromosomes in both sisters. These findings suggested that the extra X chromosomes have derived from meiotic II non-disjunction following a null transitional meiosis I (MI). A lack of recombination in the X chromosomes of both sisters suggests a possible maternal genetic defect leading to an irregular recombination at MI. This information may contribute to further understanding of mechanisms leading to X chromosome non-disjunction and may assist in counseling of families with this chromosomal rearrangement (9).
Individuals with Turner's syndrome (XO) are females with a single X chromosome. They are sterile, possess underdeveloped secondary sexual characteristics and they are shorter than normal. This condition occurs in about 1 in 2500 female's births worldwide(54).
Females with genetic constitution XXX, on the other hand, have a normal appearance and are fertile, but suffer from a mild mental handicap. Similarly, XYY males have relatively few clinical symptoms and appear phenotypically normal. They are taller than average and may show aggressive behavior and below-average intelligence. Both XXX and XYY conditions usually pass undiagnosed (3).
CONCLUSION
The analysis and interpretation of these results can bold the principal causes of non-disjunction such as, the maternal age, as it elevates the risk of nondisjunction (8), meiotic errors in both phases (I and II) which was demonstrated in many studies(46.1 °% MI, 53.9 M II (6) , paternal and maternal origin(90.9 % maternal and 4.5 % paternal) (4) (90.7% maternal and 5.5 paternal (6) , mitotic errors (4.5 % mitotic origin (4) 3.8 % mitotic origin (6), lack of recombination(9), exposure to toxic substances (12) and as a especial case ; nondisjunction that affect sex chromosomes and has a major incidence in translocation carriers (Robertsonian translocation)(13). Thus the chromosomal nondisjunction doesn't have a unique cause, it is the result of meiotic errors of paternal and maternal origin and many other factors such maternal age, translocations and exposure to toxic substances. There are differences in the frequencies and percentages of the errors incidence as shown throughout the review (11). 50 years of researches about aneuploid conditions, give rise to information about the incidence, frequency, source and the mechanisms in which nondisjunction and aneuploid condition takes place. Nowadays we consider that we have finished the easy part, but still have to search for the difficult one, which is the treatment, prevention and avoiding the occurrence of these abnormalities, so will it be possible?
Conflicto de interés: ninguno
Financiación: Universidad del Norte
REFERENCES
1. Benjamin L. "Genes VIII & IX", Oxford, New. York: Oxford University Press. Goodenough U, "Genetics", Hold Saunders International. ... Genes VIII:-Benza-min L. 1st ed. 2003, Oxford University. 4. Genome:-T.A. Brown, Jhon Wiley & S. 6BT.2- BIOSTATISTICS. UNIT ... [ Links ]
2. Len L, MD. FAAP, Trisomy 21 (Down syndrome: Health issues), News and information for parents and professionals. Published 2003. [ Links ]
3. Strachan, Tom, and Andrew P. Read. Human Molecular Genetics. Oxford: U.K.: Bios Scientific Publishers; 2005. [ Links ]
4. Nelson J, Ramirez et al. Parental origin, nondisjunction, and recombination of the extra chromosome 21 in Down syndrome: a study in a sample of the Colombian population. Biomédica [online] 2007;27 (1): 141-148. [ Links ]
5. Diego D, Garcia M, Trujillo M, González C, Rodriguez de A, Ayuso C et al. Application of quantitative fluorescent PCR with short tandem repeat markers to the study of aneuploidies in spontaneous miscarriages. Hum Reprod 2005; 20:1235-43. [ Links ]
6. Petersen M, Mikkelsen M. Nondisjunction in trisomy 21: origin and mechanisms. Cytogenet Cell Genet 2003; 91:199-203. [ Links ]
7. Savage A, Petersen M, Pettay D, Taft L, Allran K, Freeman S, et al. Elucidating the mechanisms of paternal non-disjunction of chromosome 21 in humans. Hum Mol Genet 2003; 7:1221-7. [ Links ]
8. Merete B, Andrew C , Jens H, Hans E, Claes A. Brandt, Mads Bak, Claus H. Non-disjunction of chromosome 13. Published by Oxford University Press; 2007. [ Links ]
9. Orit R, Todd B, Thomas R. Reduced recombination in maternal meiosis coupled with non-disjunction at meiosis II leading to recurrent 47, XXX. Chromosome Research 2004; 12: 125 - 132. [ Links ]
10. National Institute Of Child Health And Human Development. Facts about Down syndrome. Published on 15 august 2008. [ Links ]
11. Terry H, Heather Hall and Patricia Hunt, The origin of human aneuploidy: where we have been, where we are going. Oxford University Press; 2007. [ Links ]
12. Hallare A, Gervasio M. Monitoring genotoxicity among gasoline station attendants and traffic enforcers in the City of Manila using the micronucleus assay with exfoliated epithelial cells. Published online: 20 August 2008, Springer Science Business Media B.V. 2008. [ Links ]
13. Yongjian C, Jin H, Ping L, Jie Q. Analysis of meiotic segregation patterns and interchromosomal effects in sperm from six males with Robertsonian translocations. Published online: 27 July 2007, Springer Science + Business Media, LLC 2007. [ Links ]
14. Orit R, Todd B, Thomas R. Reduced recombination in maternal meiosis coupled with non-disjunction at meiosis II leading to recurrent 47, XXX. Chromosome Research 2004;12: 125 - 132. [ Links ]
15. Alberts, Bruce, et al. Molecular Biology of the Cell. 3rd ed. New York: Garland Publishing; 2008 [ Links ]
16. Bugge M, Collins A, Hertz J, Eiberg H, Lundsteen C., Brandt C et al. Non-disjunction of chromosome 13. Hum. Mol. Genet 2007. [ Links ]
17. Lamb N, Sherman S, Hassold T. Effect of meiotic recombination on the production of aneuploid gametes in humans. Cytogenet. Genome Res. 2005; 111:250 - 255. [ Links ]
18. Lamb N, Shaffer J., Feingold E., Sherman S. Association between maternal age and meiotic recombination for trisomy 21. Am. J. Hum. Genet. 2005; 76:91 - 99. [ Links ]
19. Sherman S, Lamb N, Feingold E. Relationship of recombination patterns and maternal age among non-disjoined chromosomes 21. Biochem. Soc. Trans 2006; 34:578 - 580. [ Links ]
20. Sun F, Oliver M, Liehr T, Starke H, Turek P, Rademaker A. Variation in MLH1 distribution in recombination maps for individual chromosomes from human males. Hum. Mol. Genet 2006; 15:2376 - 2391. [ Links ]
21. Topping D, Brown P, Judis L, Schwartz S, Seftel A, Thomas A, Hassold T. Synaptic defects at meiosis I and non-obstructive azoospermia. Hum. Reprod 2006; 21:31713177. [ Links ]
22. Sun F, Greene C, Turek P, Ko E, Rade-maker A, Martin R. Immunofluorescent synaptonemal complex analysis in azoospermic men. Cytogenet. Genome Res 2005; 111:366 - 370. [ Links ]
23. Sun F, Turek P, Greene C, Ko E, Rademaker A, Martin R. Abnormal progression through meiosis in men with nonobstructive azoospermia. Fertil. Steril 2007; 87:565 - 571. [ Links ]
24. Martin R. Meiotic chromosome abnormalities in human spermatogenesis. Reprod. Toxicol 2006; 22:142 - 147. [ Links ]
25. Lenzi M, Smith J, Snowden T, Kim M, Fishel R, Poulos B. Extreme heterogeneity in the molecular events leading to the establishment of chiasmata during meiosis I in human oocytes. Am. J. Hum. Genet 2005; 76:112 - 127. [ Links ]
26. Tease C, Hartshorne G, Hulten M. Altered patterns of meiotic recombination in human fetal oocytes with asynapsis and/or synaptonemal complex fragmentation at pachytene. Reprod. Biomed. Online 2006; 13:88 - 95. [ Links ]
27. Koehler K, Schrump S, Cherry J, Hassold T, Hunt P. Near-human aneuploidy levels in female mice with homeologous chromosomes. Curr. Biol 2006; 16: 579 - 580. [ Links ]
28. Roy A, Matzuk M. Deconstructing mammalian reproduction: using knockouts to define fertility pathways. Reproduction 2006; 131:207 - 219. [ Links ]
29. Bolcun E, Costa Y, Speed R, Taggart M, Benavente R, Rooij D. Cooke H. SYCE2 is required for synaptonemal complex assembly, double strand break repair, and homologous recombination. J. Cell Biol 2007; 176:741- 747. [ Links ]
30. Cherry S, Adelman C, Theunissen J, Hassold T, Hunt P, Petrini J. The Mre11 complex influences DNA repair, synapsis, and crossing over in murine meiosis. Curr. Biol 2007; 17:373 - 378. [ Links ]
31. Kuznetsov S., Pellegrini M., Shuda K., Fernandez O., Liu Y., Martin B., et al. RAD51C deficiency in mice results in early prophase I arrest in males and sister chromatid separation at metaphase II in females. J. Cell Biol 2007; 176:581 - 592. [ Links ]
32. Bannister L, Pezza R, Donaldson J, de Rooij D, Schimenti K, Camerini O, Schimenti J. A dominant, recombination-defective allele of Dmc1 causing male-specific sterility. PLoS Biol 2007; 5: 105. [ Links ]
33. Murchison E, Stein P, Xuan Z, Pan H, Zhang M, Schultz R, Hannon G. Critical roles for Dicer in the female germline. Genes Dev 2007; 21:682 - 693. [ Links ]
34. Hodges C, Revenkova E, Jessberger R, Hassold T, Hunt P. SMC1beta-deficient female mice provide evidence that cohesins are a missing link in age-related nondisjunction. Nat. Genet 2005; 37:1351 - 1355. [ Links ]
35. Yuan L, Liu J, Hoja M, Wilbertz J, Nordqvist K, Hoog C. Female germ cell aneu-ploidy and embryo death in mice lacking the meiosis-specific protein SCP3. Science 2003; 296:1115 - 1118. [ Links ]
36. Warburton D, Dallaire L, Thangavelu M, Ross L, Levin B, Kline J. Trisomy recurrence: a reconsideration based on North American data. Am. J. Hum. Genet 2004; 75:376 - 385. [ Links ]
37. Christianson R, Sherman S, Torfs C. Maternal meiosis II nondisjunction in trisomy 21 is associated with maternal low socioeconomic status. Genet. Med 2004; 6:487 - 494. [ Links ]
38. Freeman S, Allen E, Oxford C, Tinker S, Druschel C, Hobbs C, et al. The National Down Syndrome Project: design and implementation. Public Health Rep 2007; 122:62 - 72. [ Links ]
39. Hunt P, Koehler K, Susiarjo M, Hodges C, Ilagan A, Voigt R, Thomas S, Bisphenol A exposure causes meiotic aneuploidy in the female mouse. Curr. Biol 2003; 13:546553. [ Links ]
40. Nayernia K, Lee J, Drusenheimer N, Nolte J, Wulf G, Dressel R, et al. Derivation of male germ cells from bone marrow stem cells. Lab. Invest 2006; 86:654 - 663. [ Links ]
41. Qing T, Shi Y, Qin H., Ye X, Wei W, Liu H, Ding M, Deng H. Induction of oocyte-like cells from mouse embryonic stem cells by co-culture with ovarian granulosa cells. Differentiation (2007). [ Links ]
42. Daley G.Q. Gametes from embryonic stem cells: a cup half empty or half full? Science 2007; 316:409 - 410. [ Links ]
43. Administration on Developmental Disabilities Administration for Children and Families U.S. Department of Health and Human Services Mail Stop: HHH 300F 370 L'Enfant Promenade S.W. Washington, DC 20447 (202) 690-6590 (2003). [ Links ]
44. Conde C. 'Supermayor' uses firm hand to clean up Manila. International Herald Tribune Asia-Pacific, Mar 21, 2005. [ Links ]
45. Benites C, Amado L, Vianna R, Martino M. (2006). Micronucleus test on gas station attendants. Genetic & Molecular Research; 5: 45 - 54. [ Links ]
46. Ray M, Basu C, Mukherjee S, Roychowdhury S, Lahiri T. (2005). Micronucleus frequencies and nuclear anomalies in exfoliated buccal epithelial cells of firefighters. International Journal of Human Genetics; 5: 45 - 48. [ Links ]
47. Wang X, Spitznagel E, et al. Primary and secondary transcriptional effects in the developing human Down syndrome brain and heart». Genome Biol 2005; 6 (13): R107. doi:10.1186/gb-2005-6-13-r107. PM ID 16420667. [ Links ]
48. Leshin L. "Trisomy 21: The Story of Down Syndrome". http://www.ds-health.com/trisomy.htm. Retrieved 2006-05-21. [ Links ]
49. Rahmani Z, Blouin J, Créau R, Goldberg N, et al. "Down syndrome critical region around D21S55 on proximal 21q22.3". Am J Med Genet Suppl 2003; 7: 98-103. doi: 10.1002/ajmg.1320370720. PMID 2149 984. [ Links ]
50. Chandra S. 2006 July 6. "Down syndrome traced to one gene". The Scientist. http://www.the-scientist.com/news/display/23869/. Retrieved 2006-07-11. [ Links ]
51. Song W, Sternberg L, Kasten C, et al. (December 2003). "Isolation of human and murine homologues of the Drosophila minibrain gene: human homologue maps to 21q22. 2 in the Down syndrome "critical region". Genomics 38 (3): 331 - 9. doi: 10.1006/geno.0636. PMID 8975710. [ Links ]
52. Online 'Mendelian Inheritance in Man' (OMIM) V-ETS Avian Erythroblastosis virus E26 Oncogene Homolog 2 -164740 (2004). [ Links ]
53. Sumarsono S, Wilson T, Tymms M, et al. "Down's Syndrome-like skeletal abnormalities in Ets2 transgenic mice". Nature 2003; 379: 534 - 537. doi:10.1038/379534a0. PMID 8596630. [ Links ]
54. Michèle M.M. Mazzocco. The cognitive phenotype of Turner syndrome: Specific learning disabilities. International Congress Series, vol.1298, October 2006, pp.83-92. [ Links ]