Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Ingeniería e Investigación
Print version ISSN 0120-5609
Ing. Investig. vol.33 no.3 Bogotá Sept./Dec. 2013
J. P. Zamora Aguas1, W. A. Adarme2, M. D. Arango Serna3
1 Juan Pablo Zamora Aguas. BSC in Industrial Engineering, specialization in Engineering Project Management, Universidad Distrital Francisco José de Caldas, Colombia. MSc Industrial Engineering student, Universidad Nacional de Colombia, Colombia. Affiliation: Universidad Nacional de Colombia, Colombia. E-mail: jpzamoraa@unal.edu.co
2 Wilson Adarme Jaimes. BSc in Industrial Engineering, Universidad Industrial de Santander, Colombia. Specialization in Production Management, Universidad Pedagógica y Tecnológica de Colombia, Colombia. MSc in Engineering, Universidad del Valle, Colombia. PhD in Engineering-Industry and Organizations, Universidad Nacional de Colombia, Colombia. Affiliation: Full-time profesor and researcher at the Universidad
Nacional de Colombia, Colombia. E-mail: wadarmej@unal.edu.co
3 Martín Dario Arango Serna. BSc in Industrial Engineering, specialization in Finance, Universidad de Antioquia, Colombia. MSc in Systems Engineering, Universidad Nacional de Colombia (Medellín), Colombia. PhD in Engineering, Universidad Politécnica de Valencia, Spain. Affiliation: Full-time professor and researcher, Universidad Nacional de Colombia (Medellín), Colombia. E-mail: mdarango@unal.edu.co
How to cite: Zamora, J., Adarme, W., Arango, M., Supply risk analysis: applying system dynamics to the Colombian healthcare sector., Ingeniería e Investigación, Vol. 33, No. 3, December 2013, pp. 76 - 81.
ABSTRACT
This paper addresses supply risk in the oncological medicine supply chain (SC) in Colombia. A system dynamics model was developed for assessing supply risk impact on SC operation and performance. The costs of risk and logistics operation were evaluated in current conditions and in a risk mitigation scenario. The model was based on a systematic analysis of supply networks, mainly concerning the reduction of overall SC costs and improving service, quality and opportunity performance index. This article has resulted from research into SC risk management (SCRM) carried out by the Universidad Nacional de Colombia's SEPRO research group.
Keywords: supply risk, supply chain, oncology drugs, system dynamics.
RESUMEN
En este artículo se aborda la problemática del riesgo de aprovisionamiento en la cadena de suministro (CS) de medicamentos oncológicos en Colombia. Se desarrolla un modelo de dinámica de sistemas para evaluar el impacto del riesgo de aprovisionamiento, sobre las condiciones de operación y desempeño de la CS. Se valoran los costos del riesgo y de la operación logística en las condiciones actuales y en un escenario con mitigación del riesgo. Las contribuciones del modelo se basan en el análisis sistémico de las redes de suministro, principalmente, en la reducción de los costos globales de la cadena y en la mejora de los índices de desempeño en oportunidad, calidad y servicio. Este artículo es el resultado de la investigación de gestión del riesgo en la cadena de suministro (SCRM - SupplyChainRisk Management), realizada por el grupo de investigación SEPRO de la Universidad Nacional de Colombia.
Palabras clave: riesgo de aprovisionamiento, cadena de suministro, medicamentos oncológicos y dinámica de sistemas.
Received: April 11th 2013 Accepted: October 7th 2013
Introduction
Supply chain (SC) risk refers to the possibility of shortages or disruptions ocurring regarding materials, product and service flows in the SC. This implies the possibility and effect of a mismatch between supply and demand which might affect customers' needs and expecttions regarding products and services and SC performance, thereby causing overcosts and demanding greater effort from an organisation (Khan & Burnes, 2007).
Interruption on any SC level can affect an enterprise in terms of product delivery, customer service and operation continuity (Juttner, Peck, & Christopher, 2003).
A study presented by Sanchez-Rodrigues & Naim (2010) observed that 60% of the most frequently occurring problems which can have the greatest effect on SC risk were related to delays, information availability, demand knowledge, delivery restrictions and SC coordination. Others having lesser effect on SC performance were related to infrastructure rigidity, operating costs, SC integration and technology.
Sc risk is presented by internal and external or enviromental factors including control, supply, demand and environmental risks (Christopher, 2005). Macro, safety, political, competitive and resource risks have been distinguished in similar classification (Manuj & Mentzer, 2008).
Supply risk
SC risk is defined as the probability of an incident associated with inbound supply from individual supplier failures or the supply market occurring, in which its outcome results in purchasing firm inability to meet customer demand or causes threats to customers' lives and safety (Zsidisin, 2003). This kind of risk has implications throughout the SC affecting system productivity levels, inventory and cycle times (Eltantawy, Fox, & Giunipero, 2009).
The danger of shortages and interruptions jeopardise SC objetives, especially regarding product availibility, quality conditions and total costs. Risk management action must thus be orientated towards understanding sources of supply regarding risk and state the best ways of managing this (Wu, Blackhurst, & Chidambaram, 2006).
Context and problems of risk regarding the oncological drug SC
It has been estimated that more than 12 million new cancer cases appear in the world each year (IARC/WHO, 2008), most (80%) appearing in developing countries (Ministerio de la Proteccion Social de la República de Colombia, 2010); 62,660 new cancer cases ocurred in Colombia in 2008 (IARC/WHO, 2008). According to the last Basic Indicators for Monitoring Millenium Development Objectives report, Colombia's mortality rate was 76.5 per 100,000 inhabitants; more than 34,000 people died in 2010 due to cancer (PAHO/MPS/INS, 2010).
Colombian pharmaceutical policy shows that some of the main problems in the Colombian health system are related to poor supply and availability of essential medicines. Regarding pharmaceutical policy, it was stated that only 63% of all insured people attended by the system and who were prescribed medicine (in 2008) received the complete formulation, 15.8% received it poorly and 21.2% did not received anything at all (Consejo Nacional de Política Económica y Social - DNP, 2012).
Problems regarding medicine supply, availability and supply is a critical Sc risk affecting Colombian patients and the population in general, due to public health, social and economic implications for the country.
A shortage of medicines affects the continuity of oncological treatment, thereby endangering patients' health due to adjustments and changes in treatment shemes. At international level, it has been identified that oncological medicine shortages have forced treating physicians to set application priorities on patients and changes in treatment. For example, capecitabin may be substituted for 5-flourouracil in adjuvant therapy for colorectal cancer, altough this kind of treatment option has not been proved regarding a cure for cancer (Chapner, 2011).
A shortage of medicines forces the search for substitutes thereby causing overcosts due to the difference in prices for available medicine on the market (United Nations, 2013).
A shortage of active agents has ocurred in Colombia, such as 5-fluorouracyl, azatioprine, citarabin, chlorambucyl, doxorubicin, mercaptopurin, metotrexate and tioguanine. This situation has caused increased costs for the Colombian health system due to administrative weakening in contingency operations when searching for medicines on the market and purchasing decisions concerning trading agents having diferent levels of intermediation.
Research methodology
The study was focused on healthcare institutions (HCI) which are enabled to provide clinical oncologicy services (Diagnosis Support and Therapeutical Complementation Group code 709, according to the Checking Procedures for Conditions for Evaluating Oncological Services' Handbook, Ministerio de Salud y Protección Social, 2012) identified from HCI special registration data (Ministerio de Salud y Proteccion Social, 2012).
Such HCI in Colombia providing clinical oncology services includes 217 hospitals and specialised clinical centres. A sample of 42 HCI was taken, according to the scope of data collection planning and programming pertinent resources. Sunter's method was used for selecting the HCI, using proportional likelihood for sampling concerning HCI size and number of oncology patients attended (Särndal, Swensson, & Wretman, 1992).
Selecting drugs
Doxorrubicine's active compounds were defined as study subject medicine in this research, considering Colombian Health and Social Protection Ministry information concerning such active principles which had a regular supply pattern during the past year, even though a shortage in the system was reported.
Doxorrubicine is used as chemotherapy medicine and is sold in two presentations: 10 mg and 50 mg vials. It is used for treating non-Hodgkin's lymphoma, multiple myeloma (bone marrow cell cancer), acute leukaemia, Kaposi's sarcoma, Ewing's sarcoma, Wilm's tumour and breast, suprarenal crust, endometrium, lung and ovarian cancer, in addition to cancer occurring in other parts of the body (American Cancer Society, 2011).
The SISMED (Ministerio de Salud y Proteccion Social, 2012) database, concerning units sold and prices, reported 229,573 units of doxorrubicine being sold in Colombia during 2011.
Identifying, assessing and analysing risk
SC risk was identified, assessed and analysed using risk management methodology with a systemic focus, as appears in Figure 1. Three stages were considered: contextualisation, assessment and decision-making and feedback. Interaction between these stages led to dealing with the complete cycle of risk management where system performance is assessed as time elapses, allowing management action to be evaluated according to danger level and susceptibility to risk.
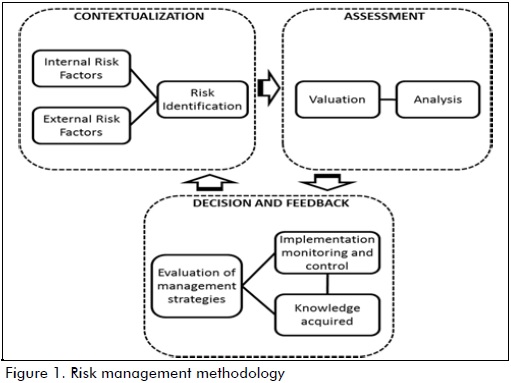
Stages 1 and 2 are dealt with in this research; stage 3 will be researched later on.
Risk relational theory was used for answering key theorical and practical questions regarding the first stage concerning how and why something is considered a risk. At least three conceptual elements concerning risk definition must be considered: an object, the damage and a link based on some kind of causality between the object and the damage (Boholm & Corvellec, 2011). The object becomes a risk factor causing a risk generator agent and can be an internal or external circumstance, natural phenomena or patterns within a social system.
Damage or risk consists of adverse circumstances which can be materialised and linkage is the systemic association between risk objects and objects at risk which can be stronger than others.
Four supply risks were identified concerning conceptualisation (stage 1, Figure 2): supply delay, disruption in supply, differences in quantities received and demand forecast errors. Such risks concerned a definition of internal and external risk factors; depleting a drug's active compounds, affects pharmaceutical industry drug production due to scarcity of raw materials. Drug availability on the market depends on market supply and demand dynamics. Logistics system capacity is associated with response times and quality regarding drug management and delivery. There are also asymmetries of information which are a critical risk factor in SC links resulting from power relationships, competition conditions in the provider market, low coordination between SC agents and limited use of communication and information technologies.
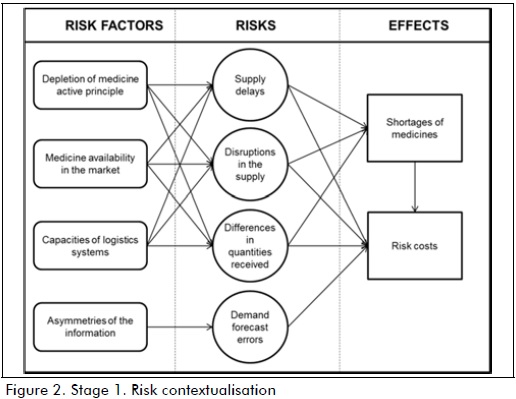
Supply risk implications for the system were summarised into two effects: drug shortage in the SC and risk costs resulting from risk materialisation.
Risk assessment methodology was used in stage 2 (assessment), considering the likelihood of ocurrence and risk impact. Likelihood and impact coring scales were set, which allowed establishing risk areas: severe, major, medium and minor (Figure 3). It was found that disruption risks and delays in supply were rated severe and major; the risk of difference in received numbers was rated major and medium and error risk regarding forecasting demand was rated medium.
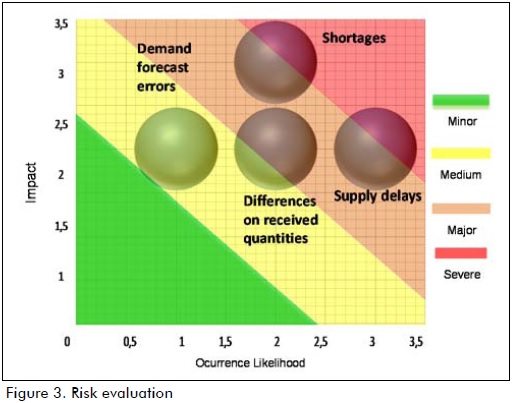
The expression presented by Liu, Xu, Li, Wang & Wu (2012) was used for quantifying the danger of risk:
where:
P(NO) was the likelihood of a negative result, and
L(NO) was loss or effect due to the result.
System dynamics (SD) application
The system dynamics (SD) domain developed at the Massachusetts Institute of Technology (MIT) in 1950 by Jay Forrester is based on the structure and dynamics of complex systems. Control engineering and non-linear dynamic systems theory ststes that SD are used for developing formal models and simulation, leading to capturing complex dynamics for creating a favorable environment for learning and designing policy (Sterman, 2002).
SD combines the theory, methods and philosophy needed for analysing economic , medicine, engineering patterns and other issues (Forrester, 1991).
Causal diagrams are used in system dynamics methodology for representing causal relationships and inter-dependence between variables and Forrester or flow diagrams to represent system status, feedback and decision-making regarding the information so obtained (Sterman, 2000).
Figure 2 resulted from applying the causal diagram representing the current relationship between risk factors, risks and effects.
System dynamics model
The structure of the system based on representation of flows and drug accumulation in the different SC are presented in the system dynamics model. They do not represent a specific node in the SC or a physical place for storage or distribution; instead, they consist of product flows and accumulation in the system.
The danger of risk concerning SC is posed by the risk factor concerning supply, associated with the provider market, drug availability on the market, logistics services and the asymmetry of information in the SC causing alterations in product entry flows, producing instability when programming and attending patients and their consequent costs regarding system operation.
Model structure
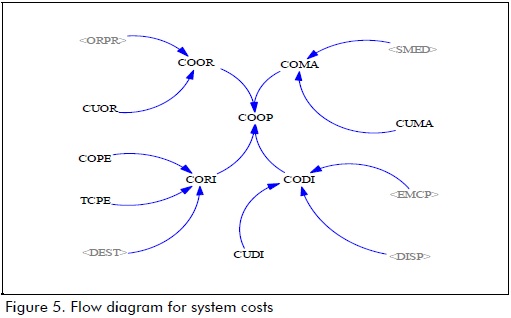
Figures 4 and 5 show the simulation model using Vensim PLE 6.0 b software.
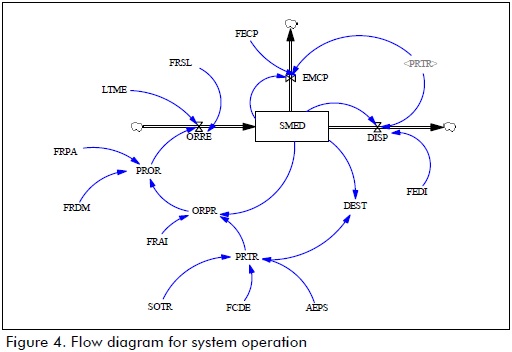
Levels:
SMED: medicine stock in a system (units)
Flows:
ORRE: entrance flow for drugs received in a system (units/month)
DISP: outflow of drugs delivered by drugstore services (units/month)
EMCP: outflow of medicines sent to preparation centres (units/month)
Auxiliary variables:
SOTR: monthly value for added consumption of drugs in units/month
FCDE: correction factor for demand
AEPS: approval time for treatment by a healthcare service provider (EPS) (months)
PRTR: number of programmed treatments by HCI (units/month)
DEST: stock deficit is the difference regarding drug stock in a system regarding demand (units/month).
FRAI: risk factor regarding asymmetry of information
ORPR: number of drug units ordered by an EPS/HCI (units/month)
FRDM: suppliers' market risk factor
FRPA: risk factor rearding availability of active compound
PROR: provider response to drug purchase order (units/month)
LTME: provider response time (units/month)
FRSL: logistics service risk factor
FEDI: percenatge drugs delivered/supplied
FECP: percentage regarding sending drugs to preparation centres
COOR: ordering cost ($/month)
CUOR: ordering unit cost ($/month)
COMA: inventory maintenance cost ($/month)
CUMA: maintenance unit cost ($/unit)
CODI: distribution cost ($/month)
CUDI: distribution unit cost ($/unit)
CORI: risk cost ($/month)
COPE: penality cost ($/unit)
TCPE: penalty application time (month)
Model formulation
SMED=INTEG(ORRE-DISP-EMCP,SMED) (1)
ORRE= fixed delay (PROR*(1-FRSL),LTME,0) (2)
DISP=if then else (PRTR>=SMED,SMED*FEDI,PRTR*FEDI) (3)
EMCP=if then else (PRTR>SMED,SMED*FECP, PRTR*FECP) (4)
PRTR= fixed delay (SOTR*(1+FCDE),AEPS,0) (5)
DEST=if then else (PRTR>SMED, PRTR-SMED,0) (6)
ORPR=if then else (SMED<PRTR,(PRTR-SMED)* (1-FRAI) , 0 ) (7)
PROR= ORPR * (1- (FRPA+FRDM)) (8)
COOP= CODI+COMA+COOR+CORI (9)
CODI= (DISP+EMCP)*CUDI (10)
COMA= SMED*CUMA (11)
COOR= ORPR*CUOR (12)
CORI= fixed delay (DEST*COPE, TCPE ), 0 ) (13)
Setting simulation and analysis runs
Two settings were assessed by using simulation runs during a 100-month period. The first setting included the risk factors evaluated in the study and their impact on system operation. The results of simulation in this setting (Figure 6) showed how entrance flow pattern was affected by risk factors, producing deficits during different periods due to differences between demand for and overall supply of drugs.
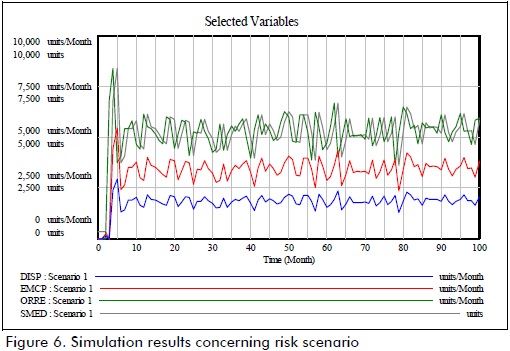
The difference between stock level and treatment request in the system highlighted the problem of supply risk affecting SC. Outflow analysis led to observing system response during simulation time frames.
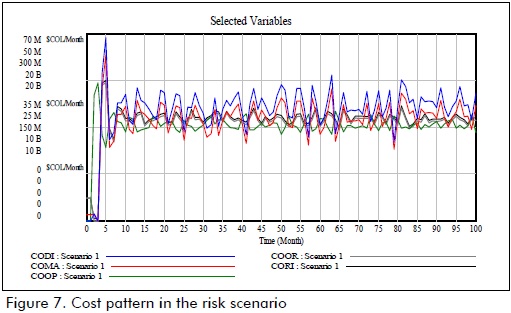
The cost pattern in the initial setting is presented in Figure 7 where the high penalty cost is reflected by the current condition of supply risk.
The second setting showing the hypothetical reduction of risk factors in supply was considered by applying risk mitigation and management strategies focused on policy definition and negotiation rules with market provider and agent coordination, where logistics service operators and statement, suitability of information systems for management and healthcare system regulating entities were included. This provided cross-support for the SC and provided reliable information for decision-making.
Such strategies will be defined in a subsequent stage of the study. However, projected values were taken on the basis of a research project hypothesis for assessing a mitigation setting. With this in mind, the simulation model was run with reduced risk factors for assessing the possible impact on system operation and associated costs. Figure 8 compares both settings.
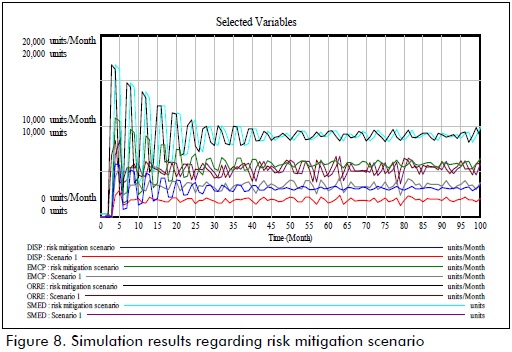
Figure 9 shows operating cost pattern concerning a current setting and a theoretical setting.
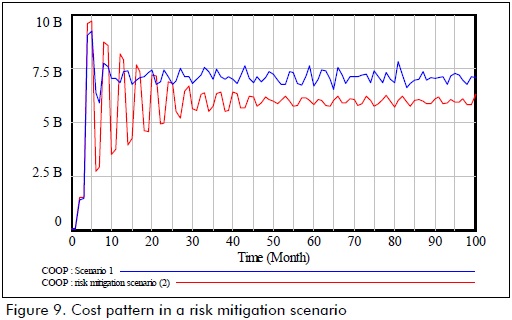
Other more optimistic settings will enable better service and cost indicators. However, the hypothesis considers the complex market dynamics surpassing regulatory control and operation measures in current market conditions in Colombia.
Conclusions
The research has presented fundamental information about the effects of supply risk on the oncological drug supply chain in Colombia, the implications for operating costs and service level indicator, presupposing an effect on the health of cancer patients.
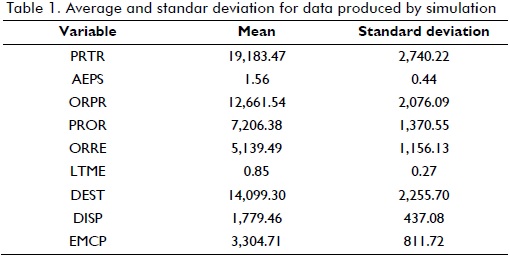
These results show that market response level was 56.91%, thereby implying a deficit in the medication system. Logistics service supply was 71.31%, representing delays in delivery times. It was found that waiting time for receiving treatment was longer than three months in 52% of cases. On average, it is known that only 38 out of every 100 patients requiring drugs will actually receive them in normal conditions.
Risk cost accounted for 70.66% of operating cases. According to the analysis, it was seen that the use of high cost drugs, subtituting doxorrubicine's active compound, caused an increase in costs, therby constituting the main system cost component.
The theoretical setting revealed that shortage ocurring due to breaks in supply became reduced (obviously) where market response level would rise from 56.91% to 85.81%, meaning that cost could be reduced by 21.26% in this setting.
Future research
The results presented in the theoretical setting showed that additional reseach on defining SC risk management strategies were needed for reducing risk and affecting more critical environments, such as system economics and cancer patients' health.
Acknowledgements
This work was financed by the Universidad Nacional de Colombia and Colciencias through a project framework called "A methodological approach for defining rules and policy for coordination and negotiation in the oncological medication supply chain in Colombia."
References
American Cancer Society., 2011. Consulted on January 20th 2013, http://www.cancer.org/ [ Links ]
Boholm, Å., Corvellec, H., A relational theory of risk., Journal of Risk Research, Vol. 14, No. 2, 2011, pp. 175-190. [ Links ]
Chabner, B., Drug Shortages - A Critical Challenge for the Generic-Drug Market., The New England Journal of Medicine, Vol. 365, No. 23, 2011, pp. 2147-2149. [ Links ]
Christopher, M., Logistics and Supply Chain Management. Creating Value-Adding Networks., 3 ed., Great Britain, Prentice Hall, 2005. [ Links ]
Consejo Nacional de Política Económica y Social - DNP., Documento CONPES Social 155 - Política Farmaceútica Nacional., Colombia, August, 2012. [ Links ]
Eltantawy, R. A., Fox, G. L., Giunipero, L., Supply management ethical responsibility: reputation and per-formance impacts., Supply Chain Management: An International Journal, Vol. 14, No. 2, 2009, pp. 99-108. [ Links ]
Forrester, J. W., System Dynamics and the Lessons of 35 Years., In K. B. De Greene, The Systemic Basis of Policy Making in the 1990s, 1991. [ Links ]
Hohenemser, C., Kates, R., Slovic, P., A Causal Taxonomy., In R. Kates, C. Hohenemser, J. Kasperson (Ed.), In Perilous progress: Managing the hazards of technology., Westview Press, 1985, pp. 67-89. [ Links ]
IARC/WHO., GLOBOCAN 2008 (IARC) Section of Cancer Information., 2008. Consulted on May 1st 2012, de International Agency for Research on Cancer / World Health Organization: http://www.iarc.fr [ Links ]
Juttner, U., Peck, H., Christopher, M., Supply chain risk management: outlining an agenda for future research., International Journal of Logistics Research and Applications, Vol. 6, No. 4, 2003, pp. 197-210. [ Links ]
Kaiser, J., Shortages of Cancer Drugs Put Patients, Trials at Risk., Science, Vol. 332, No. 6029, 2011, pp. 523-523. [ Links ]
Liu, Z., Xu, J., Li, Y., Wang, X., Wu, J., Using system dynamics to study the logistics outsourcing cost of risk., Kybernetes, Vol. 41, No. 9, 2012, pp. 1200 -1208. [ Links ]
Manuj, I., Mentzer, J., Global supply chain risk management strategies., International Journal of Physical Distribution & Logistics Management, Vol. 38, No. 3, 2008, pp. 192 - 223. [ Links ]
Ministerio de la Protección Social., Plan Nacional para el control del cancer en Colombia 2010-2019., Bogotá, 2010. [ Links ]
Ministerio de Salud y Protección Social., SISMED., 2012. Consulted on 2012, http://www.sispro.gov.co [ Links ]
Ministerio de Salud y Protección Social., Manual de procedimientos para la verificación de condiciones de habilitación de servicios de oncología., 2012. Consulted on October, 2th 2012, http://www.minsalud.gov.co [ Links ]
Ministerio de Salud y Protección Social., Registro Especial de Prestadores de Servicios de Salud., 2012. Consulted on July 31th 2012, http://201.234.78.38/habilitacion/ [ Links ]
Naciones Unidas., Oficina del Alto Comisionado de las Naciones Unidas para los Derechos Humanos., 2013. Consulted on January 25 2013, http://www.hchr.org.co/ [ Links ]
OPS/MPS/INS., Indicadores Básicos 2010 Situación de Salud en Colombia., Bogotá, 2010. [ Links ]
Sanchez-Rodrigues, V., Naim, A. P., Naim, M. M., Evaluating the causes of uncertainty in logistics operations., The International Journal of Logistics Management, Vol. 21, No. 1, 2010, pp. 45-64. [ Links ]
Särndal, C.-E., Swensson, B., Wretman, J., Model Assisted Survey Sampling., Springer Series in Statistics, New York, Springer - Verlag, 1992. [ Links ]
Sterman, J., Business dynamics: systems thinking and modeling for a complex world., Boston, Irwin/McGraw-Hill, 2000. [ Links ]
Sterman, J., System Dynamics: Systems Thinking and Modeling for a Complex World., ESD Internal Symposium, MIT, 2002. [ Links ]
Wu, T., Blackhurst, J., Chidambaram, V., A model for inbound supply risk analysis., Computers in Industry, Vol. 57, No. 4, 2006, pp. 350-365. [ Links ]













