Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Ingeniería e Investigación
Print version ISSN 0120-5609
Ing. Investig. vol.35 no.1 Bogotá Jan.Apr. 2015
https://doi.org/10.15446/ing.investig.v35n1.44761
DOI: http://dx.doi.org/10.15446/ing.investig.v35n1.44761
M. Santander1 and L. Valderrama2
1Mario Santander Muñoz. Ingeniero en Metalurgia, Universidad de Atacama, Chile. Doctor Ingeniero, Universidad Federal de Rio Grande do Sul, Porto Alegre, Brazil. Affiliation: Professor in Departamento de Metalurgia, Universidad de Atacama, Chile. E-mail: mario.santander@uda.cl
2Luis Valderrama Campusano. lngeniero en Metalurgia, Universidad de Atacama, Chile. Doctor lngeniero, Universidad Federal de Rio Grande do Sul, Porto Alegre, Brazil. Afilliation: Professor in Departamento de Metalurgia, Universidad de Atacama, Chile. E-mail: luis.valderrama@uda.cl
How to cite: Santander, M., & Valderrama, L. (2015). Dissolved Air Flotation of arsenic adsorbent particles. Ingeniería e Investigación, 35(1), 36-42. DOI: http://dx.doi.org/10.15446/ing.investig.v35n1.44761
ABSTRACT
The removal of arsenic from synthetic effluent was studied using the adsorbent particle flotation technique (APF) and dissolved air flotation (DAF). A sample of an iron mineral was used as adsorbent particles of arsenic, ferric chloride as coagulant, cationic polyacrylamide (NALCO 9808) as flocculants, and sodium oleate as collector. Adsorption studies to determine the pH influence, contact time, and adsorbent particles concentration on the adsorption of arsenic were carried out along with flotation studies to determine the removal efficiency of adsorbents particles. The results achieved indicate that the adsorption kinetic of arsenic is very rapid and that in range of pH’s from 2 to 7 the adsorption percentages remain constant. The equilibrium conditions were achieved in 60 minutes and about 95% of arsenic was adsorbed when used an adsorbent concentration of 2 g/L and pH 6.3. The maximum adsorption capacity of adsorbent particles was 4.96 mg/g. The mean free energy of adsorption (E) was found to be 2.63 kJ/mol, which suggests physisorption. The results of the flotation studies demonstrated that when synthetic effluents with 8.9 mg/L of arsenic were treated under the following experimental conditions; 2 g/L of adsorbent particles, 120 mg/L of Fe(III), 2 mg/L of Nalco 9808, 20 mg/L of sodium oleate, and 40% of recycle ratio in the DAF, it was possible to reach 98% of arsenic removal and 6.3 NTU of residual turbidity in clarified synthetic effluent.
Keywords: Adsorption particles flotation, arsenic removal, Dissolved Air Flotation.
RESUMEN
La remoción de arsénico desde un efluente sintético fue estudiada usando la técnica de flotación de partículas adsorbentes (FPA) y flotación por aire disuelto (FAD). Una muestra de mineral de hierro fue utilizada como partículas adsorbentes de arsénico, cloruro férrico como coagulante, poliacrilamida catiónica (NALCO 9808) como floculante y oleato de sodio como colector. Fueron realizados estudios de adsorción para determinar la influencia del pH, tiempo de contacto y concentración de partículas adsorbentes sobre el porcentaje de adsorción de arsénico y estudios de flotación para determinar la eficiencia de remoción de las partículas adsorbentes. Los resultados alcanzados indican que la cinética de adsorción de arsénico es muy rápida y que en el rango de pH’s desde 2 a 7 el porcentaje adsorción permanece constante. Las condiciones de equilibrio fueron alcanzadas en 60 minutos y alrededor de 95% del arsénico fue adsorbido cuando fue utilizada una concentración de partículas adsorbente de 2 g/L y pH 6,3. La máxima capacidad de adsorción de las partículas adsorbentes fue 4,96 mg/g. La energía media de adsorción (E) fue 2,63 kJ/mol lo cual sugiere que el mecanismo de adsorción es fisisorción. Los resultados de los estudios de flotación demuestran que cuando fueron tratado efluentes sintéticos con 8,9 mg/L de arsénico bajo las siguientes condiciones experimentales, 2 g/L de partículas adsorbentes, 120 mg/L de Fe(III), 2 mg/L de Nalco 9808, 20 mg/L de oleato de sodio y 40% de reciclo en la FAD, se removió 98% del arsénico y la turbidez residual en el efluente sintético clarificado fue 6,3 NTU.
Palabras clave: Flotación de partículas adsorbentes, remoción de arsénico, flotación por aire disuelto.
Received: August 4th 2014 Accepted: October 14th 2014
Introduction
The minerals processing involve the use of large amounts of water which generates contaminated effluents (Santander et al., 2011). The pollutants associated with these effluents can include toxic ions, such as arsenic (Rubio, 1998). Common methods for removing arsenic from aqueous waste streams are precipitation with lime, iron salts or sodium hydrosulphide (Valenzuela & Sanchez, 2001, Gunther & Wanner, 1991). The solvent extraction and ion exchange techniques have also been used (Nishimura et al., 1998).
Arsenic can be removed from liquid effluent as ferric arsenate FeAsO4·2H2O by co-precipitation with iron (Fe(III))(Papassiopi et al., 1996). For arsenic precipitation as ferric arsenate, first it is necessary to oxidize the arsenic from a trivalent to a pentavalent state using ozone or hydrogen peroxide (Nishimura & Tosawa, 1988). Ferric arsenate is soluble, but the solubility decreases rapidly when the iron/arsenic ratio increases (Krause & Ettel, 1989). The solubility of ferric arsenate can be reduced in several orders of magnitude when the amount of iron present in the solution is bigger than four times, the stoichiometric amount required, and pH is between 3.0 and 7.0 (Valenzuela & Sanchez, 2001, Cui et al., 2014). At pH 5, the arsenic remaining in solution is about 8 mg/L at a Fe/As molar ratio of 2.0 (Krause & Ettel, 1989). Increasing the Fe/As ratio to 2 results in a significant reduction of arsenic in the solution to almost 0.15 mg/L. Further reduction of arsenic to about 0.02 mg/L can be obtained with a Fe/As ratio of 5.0 (Robins, 1987; Robins, 1990).
Sodium hydrosulfide is used in the removal of arsenic as arsenic sulfides to form AS2S3, species of very low solubility at pH below 5; over this value of pH the arsenic residual concentration increases abruptly from 18 mg/L to complete dissolution (Klamp and Wanner, 1991). The main disadvantage of this method is the formation of hydrogen sulfide acid, a toxic gas.
The arsenic can also be removed by means of the solvent extraction, ionic exchange and adsorption techniques (Sharma & Sohn, 2009). Among other materials that have been used as adsorbents are found the activated carbons (Nagarnaik et al., 2003); Zeolites (Onyango et al., 2003); industrial product and waste (Mohan & Chander, 2006; Brunori et al., 2005; Bhakat et al., 2007); oxides minerals (Saha & Bains, 2005; Wilkie & Hering, 1996; Zhang & Blowers, 2005); biosorbents (Kadukova & Vircikova, 2005; Ghimire et al., 2006).
The solvent extraction, ionic exchange and adsorption techniques have several disadvantages. For example, in the solvent extraction technique the arsenic is transferred from an aqueous solution to an organic solvent (Kerfoot, 1978, Baradel et al., 1986). In the ionic exchange and adsorption techniques, the regeneration of the resin and/or adsorbent produces a solution contaminated with arsenic (Mohan & Pittman, 2007); both organic solvent and solution contaminated with arsenic are considered hazardous waste (Leist et al., 2000).
As an alternative treatment method to these techniques, adsorbent particle flotation (APF) has been recognized as an effective technique for treating aqueous solutions contaminated with heavy metals (Féris et al., 2004). The APF technique involves the adsorption of the dissolved metals on the adsorbing particles (Rubio et al., 2002). A surfactant acting as a collector is then added; it is adsorbed on the particles and makes them hydrophobic (Santander et al., 2009). Subsequently, the adsorptive particles are removed by microbubbles using the flotation technique. APF has been used to remove arsenic and other toxic metals from synthetic and industrial effluents (Zouboulis et al., 1992; Zouboulis, 1995; Rubio and Tessele, 1997; Zouboulis et al., 2003). The APF technique using synthetic goethite as adsorbent particles has demonstrated to be an effective process to remove As(V) (Matis et al., 1997). The APF technique also proved to be an effective process to remove mercury, arsenic, and selenium from effluent produced in the gold cyanidation circuit; removal efficiencies over 98% were reported (Tessele et al., 1998).
Few studies have been reported on the application of the APF technique to remove trivalent arsenic using natural minerals and dissolved air flotation; for this reason the objectives of this study were to (1) evaluate the effectiveness of an ore that contains iron oxide, silicon oxide and amorphous hydrous ferric oxide in arsenic adsorption from synthetic effluents and to (2) examine the feasibility to remove the arsenic adsorbent particles from synthetic effluents by dissolved air flotation.
Experimental
Materials
Synthetic solution with variable concentrations of arsenic was prepared by dissolving a predetermined amount of As2O3 of technical grade in de-mineralized water. The pH was adjusted with sodium hydroxide (NaOH) or hydrochloric acid (HCl) of analytic grade as needed for each test, and registered with a HANNA HI 8424 pH meter. Ferric chloride was used as coagulant while Nalco 9808 was used as flocculant and sodium oleate was applied as an adsorbent particles collector. De-mineralized water was used to prepare the reagents. Before and after the adsorption and flotation studies, arsenic concentration was determined with the hydride generation technique in combination with a GBC 908 atomic absorption spectrophotometer. Turbidity of clarified synthetic solution was measured with a HANNA Model HI 98703 Turbidity Meter. Mineral ore collected in iron ore deposits located in the Atacama Region-Chile was used as adsorbent particles. The adsorbent particles sample was grinded until it reached a size of 100% -74 micrometers, homogenized, and divided with the purpose of obtaining representative samples for its characterization and for carrying out the adsorption and flotation tests. The crystalline species were identified by X-ray diffraction (XRD) with a Bruker D8 Advance diffractometer, whereas the chemical composition was determined by X-ray fluorescence with a Siemens SRS 3000 spectrometer. A morphological characterization sample of the adsorbent particles was carried out in a scanning electron microscope (SEM), LEO brand, model 420. The surface area of the adsorbent particles sample was determined by a BET-N2 surface area analyzer.
Adsorption studies
The adsorption tests were conducted at room temperature using a variable concentration of adsorbent particles, a fixed volume of synthetic effluent (600 mL), and a constant stirred speed (600 rpm). The adsorbent particles were put in contact with synthetic solution containing variable concentrations of arsenic, between 4 and 10 mg/L according to the value predetermined for each test. The adsorption of arsenic on adsorbent particles was determined as a function of pH (between 2 and 12), contact time (between 0 and 360 min) and adsorbent particles concentration (between 0.25 to 3 g/L).
In all the adsorption tests, a beaker of 1000 mL with four baffles and mechanic stirrer variable speed (Stuart Scientific model SS3) were used to search the optimal conditions by which adsorbent particles adsorbed the arsenic from the synthetic effluent.
Once the adsorption tests were finished, the adsorbent particles were separated from the synthetic effluent by filtration with Whatman N°42 filter paper. The filtrate was analyzed for residual arsenic and the pH was measured. The adsorption efficiency (R %) was determined as arsenic adsorption relative percentage and it is expressed as:
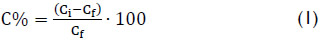
where Ci, is the arsenic concentration in synthetic effluent before the adsorption test and Cf, is the arsenic concentration in synthetic effluent after the adsorption test.
Dissolved Air Flotation studies
Previous to the flotation test, suspensions with 2 g/L of adsorbent particles loaded with arsenic were prepared under the optimum conditions predetermined in adsorption studies, i.e., contact time equals 60 min and pH between 6.0 and 7.0. DAF of adsorbent particles was conducted with different recycle ratio (between 10 and 50%) and distinct concentration of coagulant (between 20 and 140 mg/L of Fe(III)). As in the studies with different recycle ratio not all adsorbent particles loaded with arsenic ions were removed. Studies increasing the coagulant dosage and moreover adding flocculant (polyacrylamide cationic) and collector (sodium oleate) to increase the flotation efficiency of the adsorbent particles were carried out. Also, the coagulation, flocculation and hydrophobization of adsorbent particles loaded with arsenic ions were carried out in a beaker of 1000 mL with four baffles and mechanic stirrer variable speed. In the studies with different recycle ratio; once the adsorption finished, it was added 20 mg/L Fe(III) ions and the solution was stirred for five minutes at 600 rpm, then it was transferred immediately to the flotation cell. In the studies with flocculant and collector, once finished the coagulation stage, it was added 2 mg/L of flocculant and stirred at 600 rpm (fast mixing) during one min, subsequently the stirred speed was reduced at 300 rpm (slow mixing) and was stirred again for five minutes. Then 20 mg/L of collector (sodium oleate) was added, and the solution was stirred again for one min, once this stage finished the solution was transferred immediately to the flotation cell.
Flotation tests were conducted in a batch scale dissolved air flotation (DAF) unit consisting of a pressure vessel of 2 L to saturate water with air, and a flotation cell of 1.5 L capacity which was connected by a release needle valve for depressurization and microbubbles formation. In all DAF tests, water was saturated with a constant air flow of 10 L/minute for 60 minutes and at 4 bar pressure. Ten minutes after the addition of water saturated with air, a sample of the clarified synthetic effluent was collected from the bottom. A clarified synthetic effluent was analyzed by its arsenic concentration and residual turbidity. All the tests were carried out at room temperature by duplicate and the average error between replicates was always under 5%.
Results and discussion
Characterization of the adsorbent particles
The adsorbent particles chemical composition determined by X-ray fluorescence analysis demonstrated that the main elements present in the adsorbent particles sample are oxygen with 48.22%, silicon with 31.49% and iron with 12.23%. The X-ray diffraction analysis revealed that the major crystalline constituents are quartz-α, cristobalite-α and magnetite. However, the peaks intensity shows that the quartz-α is the predominant species; this agrees with high silica content analysed in the X-ray fluorescence analysis. The peak intensity of magnetite detected in the XRD analysis is very low considering the amount of iron detected in the XRF analysis. This discrepancy between the two analyses is probably due to the fact that part of the iron is contained in the amorphous hydrous ferric oxide which is not possible to detect in the XRD analysis. The BET surfaces area of adsorbent particles is 13.52 m2/g and the pore diameter average is 6.54 nm. This surface area value is excessively high considering that the main species is quartz-α. The quartz-α surfaces area value of 99.8% purity and grain size from 40 to 70 micrometers is 0.20-0.21 m2/g (Tikhomolova & Urakova, 2002).
The morphology of the adsorbent particles was examined by a Scanning electron micrograph (SEM). The SEM images are shown in Figure 1a) and 1b). Figure 1a) shows that each particle is an aggregate of smaller particles; each of these aggregates has a regular geometric, spherical or ellipsoidal shape and homogenous size of about 100 micrometers. Figure 1b is a magnified view which shows that the particles that form the aggregates are smaller than 2 micrometers.

The morphological characteristics of the adsorbent particles, where each particle is formed by aggregates of particles with size less than 2 micrometers, and the presence of amorphous hydrous ferric oxide could justify the high value at the superficial area.
Adsorption study
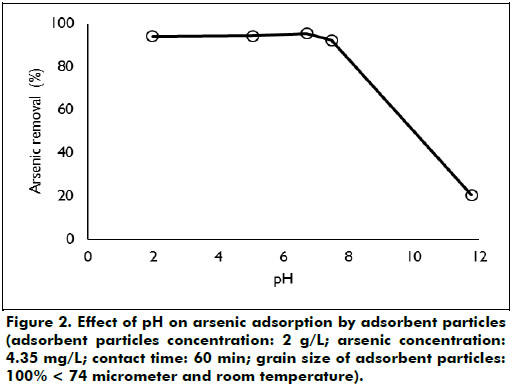
Effect of pH: The adsorption of arsenic by adsorbent particles for different pH values is shown in Figure 2. The adsorption percentage remains constant in the pH interval from 2 to 7. In this pH interval the adsorption percentage is in the order of 95%, an increase in pH above 7 reduces the adsorption percentage to 20% when the pH is 11.8. The results of the pH effect obtained in this study agree with the reports done by other investigators. About 92% of arsenic was adsorbed on the natural laterite in the pH range from 2.0 to 9.8, decreasing to 40% when the pH increases to 12 (Maiti et al., 2007). The adsorption of arsenic on ferrihydrite and hydrous ferric oxide are also independent of pH in the range from 4 to 9.5 pH (Wilkie and Hering, 1996, Jain and Loeppert, 2000).
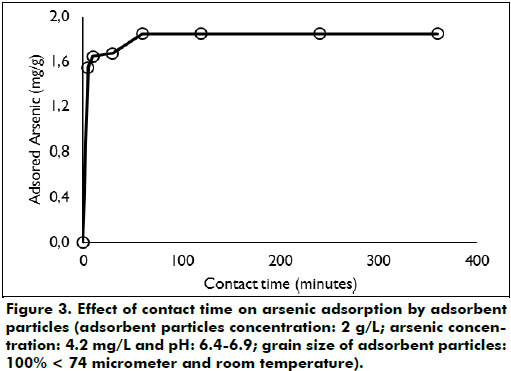
Effect of contact time: The effect of contact time on arsenic ad-sorption is shown in Figure 3. The adsorption kinetics clearly demonstrates that the maximum adsorption capacities of 1.85 mg/g and the equilibrium conditions were achieved with contact times of 60 min; longer contact times do not increase the ad-sorption capacities. This result is consistent with another study showing that the adsorption kinetic of arsenic on iron oxide coated cement (IOCC) was also very fast, during the first 20 min the equilibrium conditions were achieved (Kundu & Gupta, 2007). On the contrary, adsorption kinetic of arsenic on natural siderite is slower, to reach equilibrium conditions 194 h were needed (Guo et al., 2007).
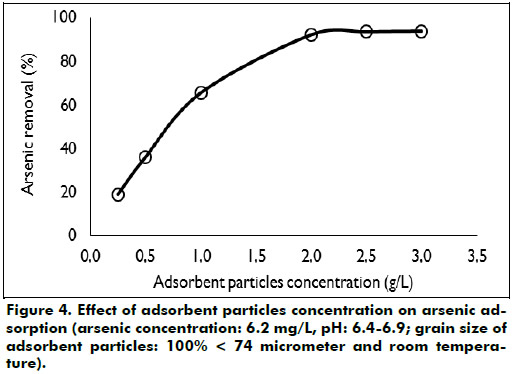
Effect of adsorbent particles concentration: Figure 4 shows that an increase in the adsorbent particles concentration from 0.25 to 2 g/L increases the arsenic adsorption. This rise may be attributed to the increase in the adsorption surface owing to the increase in the adsorbent particles dose. Concentration greater than 2 g/L does not increase significantly the arsenic adsorption.
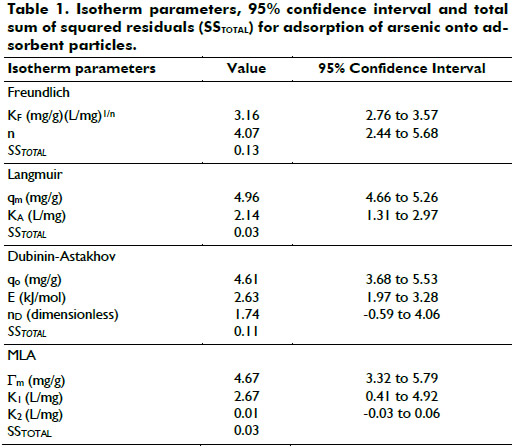
Adsorption isotherms: Adsorption models were used to determine the adsorption capacity of arsenic onto adsorbent particles and to diagnose the nature of adsorption. The results obtained on the adsorption of arsenic were analyzed using the well-known models given by Freundlich, Langmuir and Dubinin-Astakhov. The isotherms data were obtained at room temperature using arsenic concentration of 6.2 mg/L, adsorbent concentration of 0.25-3 g/L, pH of 6.4-6.9, grain size of adsorbent particles 100% < 74 micrometer.
The Freundlich model is valid for a multilayer adsorption model on a surface containing a finite number of sites with mutual interactions between adsorbed ions. It is described by the following equation (Malkoc, 2006):

where, Ce is the mass concentration of arsenic in the synthetic effluent after adsorption (mg/L), qe is the mass of arsenic (mg) adsorbed per mass of adsorbent particles (g) in equilibrium, KF is the Freundlich constant (mg/g)(L/mg)1/n and 1/n is a measure of the adsorption intensity.
The Langmuir model refers to homogeneous adsorption, where all adsorption sites are identical and energetically equivalent (all sites in the adsorbent possess equal affinity for the arsenic). It is described by the following equation (El-Khaiary, 2008):

where, Ce is the equilibrium concentration (mg/L), qe the amount of adsorbent (mg/g), qm is qe for complete monolayer adsorption capacity (mg/g), and KA is the equilibrium adsorption constant (L/mg).
Langmuir and Freundlich isotherms do not give any ideas about adsorption mechanism (Maiti et al., 2007). In order to distinguish the mechanism of adsorption the Dubinin-Astakhov isotherm was used which can be represented by the following equation (Dron and Dodi, 2011):
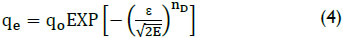
where, E is the adsorption energy (J mol-1), nD is the heterogeneity factor (dimensionless), qo is the maximum adsorption capacity (mg/g), and ε is the adsorption potential (J mol-1) expressed as follows:
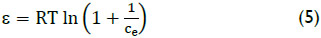
where, R, T and Ce represent the gas constant (8.314 J mol-1 K-1), (K) is the absolute temperature and arsenic equilibrium concentration(mg/L) respectively.
The values of isotherm parameters were obtained by orthogonal distance regression using the statistical software DATAPLOT (El-Khaiary, 2008 Boggs et al., 1987; Boggs et al., 1989; Filliben, 1981) and are presented together with 95% confidence interval and total sum of squared residuals (SSTOTAL) in Table 1. It may be observed from Table 1 that the Langmuir isotherm exhibits the best fit over the range of experimental data. As listed in Table 1, the SSTOTAL value for Langmuir isotherm is the lower.
To ratify if adsorption of arsenic on adsorbent particles takes place in a monolayer the experiment results were also analyzed using the multilayer adsorption (MLA) model described by the equation (6) (El-Khaiary, 2008).
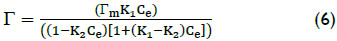
where Γm is the monolayer adsorption capacity (mg/g), Ce the equilibrium concentration (mg/L), and K1 and K2 are the equilibrium adsorption constants of the first and second layers (L/mg). It is noted that in the case of the monolayer adsorption K2 will have a value of zero (El-Khaiary, 2008).
The MLA models parameters, showed in Table 1, confirm the monolayer adsorption because the value of the first layer adsorption capacity Γm (4.57 mg/g) is almost the same as qm of Langmuir models (4.96 mg/g), and also because the value of adsorption constant of the second layer, K2 (0.01 L/mg), is closed to zero and its 95% confidence interval contains the zero, whereas the value of adsorption constant of the first layer K1 (2.67 L/mg) is proximate to KA of Langmuir models (2,14 L/mg).
As shown in Table 1, the E value is 2.63 kJ/mol for arsenic on the adsorbent particles. The value of E is very useful in predicting the type of adsorption and if the value is less than 8 kJ/mol, then the adsorption is physical in nature and if it is between 8 kJ/mol and 16 kJ/mol, then the adsorption is due to exchange of ions. In the present study, as it is evident from the result, the E value is <8 kJ/mol, so the adsorption was physical in nature.
Adsorption Kinetics: The pseudo-first-order equation and the pseudo-second-order equation (Wong et al., 2004, El-Khaiary & Malash, 2011) were used to express the adsorption process of the arsenic ions onto adsorbent particles, and they can be expressed by equation (7) and (8), respectively:
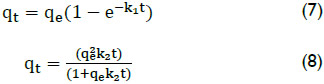
Where qe is the amount of arsenic adsorbed at equilibrium per unit weight of adsorbent, mg/g, qt is the amount of arsenic adsorbed at t time, k1(min-1) and k2 (g/mg min) are the rate constants of the pseudo-first-order and pseudo-second-order adsorption.
Also, the adsorption kinetic parameters were obtained by orthogonal distance regression using the statistical software DATAPLOT and are presented together with 95% confidence interval and total sum of squared residuals (SSTOTAL) in Table 2
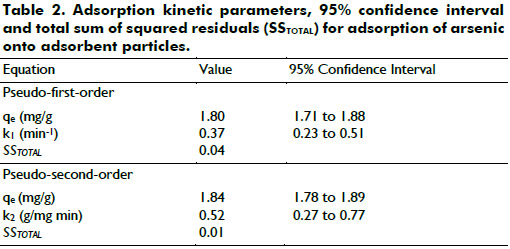
As seen from Table 2, the obtained SSTOTAL value of the pseudo-second-order model (0.01) was lower than that of pseudo-first-order model (0.04), suggesting that the pseudo-second-order model was more suitable to describe the adsorption kinetics of arsenic ions onto adsorbent particles. Moreover, the calculated qe value with the pseudo-second-order model was much closer to the experimental qe value (1.85 mg/g).
Flotation study
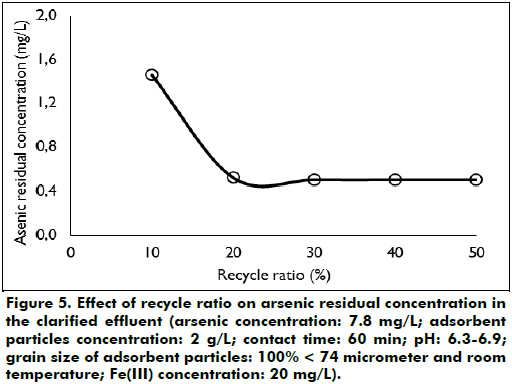
Effect of recycle ratio: Different recycle ratios ranging from 20 to 50% were tested to find out the recycle ratio value that maximizes the removal efficiency. Figure 5 shows that when recycling ratio is equal to 20%, the arsenic residual concentration in the clarified synthetic effluent is 0.50 mg/L; this value of concentration is greater than the optimum reached in the adsorption study. Greater values of recycle ratio increase mildly the arsenic residual concentration in the clarified synthetic effluent. It was observed by visual inspection that the clarified synthetic effluent contained small amounts of adsorbent particles in suspension loaded with arsenic; this probably increased its arsenic residual concentration. The residual turbidity in the clarified synthetic effluent was of the order of 24.7 NTU.
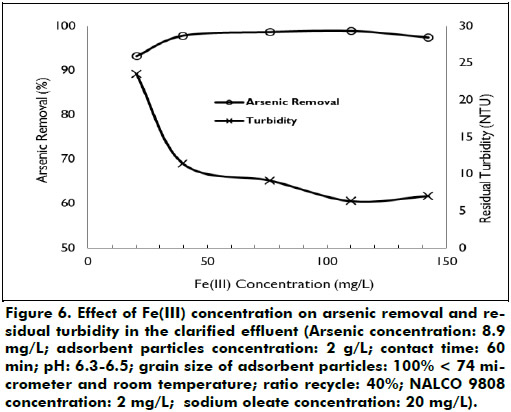
Effect of Fe(III) ions concentration: In order to reduce turbidity in the clarified synthetic effluent, tests increasing the doses of Fe(III) ions were performed with the addition of flocculant and collector. Figure 6 shows the effect of Fe(III) ions concentration on removal of arsenic and residual turbidity in the clarified synthetic effluent. An increase in the concentration of Fe(III) ions increases the removal of arsenic and reduces the residual turbidity in the clarified synthetic effluent. When the Fe(III) ions concentration was increased from 20 to 140 mg/L, the removal of arsenic rose from 93 to 98.9% and the arsenic residual concentration and residual turbidity in the clarified synthetic effluent decreased from 0.6 to 0.12 mg/L and from 23.5 to 6.3 NTU, respectively. An increase in the doses of Fe(III) ions from 10.3 to 30.9 mg/L increases the adsorbent particles removal efficiency of copper ions from 51.72 to 98.26% and reduces residual turbidity in the clarified solution from 36 to 1 NTU (Rubio and Tessele, 1997).
The results demonstrated that coagulants, flocculant and collector were necessary to achieve a good removal of adsorbent particles. In the absence of these reagents, an incomplete removal was observed because all the adsorbent particles do not form aggregates with microbubbles.
Conclusions
The result of the studies confirmed that:
The use of the adsorbent particles flotation technique (APF) employing a mineral that contains iron oxide, silicon oxide and amorphous hydrous ferric oxide and dissolved air flotation (DAF) demonstrated to be efficient for arsenic removal.
The adsorption studies demonstrated that it was possible to adsorb above 95% of arsenic in the pH interval from 2 to 7.
Kinetics revealed that adsorption of arsenic on adsorbent particles increased rapidly with contact time, 60 minutes were sufficient to achieve the equilibrium conditions.
Langmuir, Freundlich and Dubinin-Astakhov (D-A) models were used to describe the adsorption isotherms. The results demonstrated that Langmuir isotherm exhibits the best fit over the range of experimental data. The maximum adsorption capacity of adsorbent particles for arsenic determined from Langmuir isotherm was 4.96 mg/g. The mean free energy of adsorption (E) calculated from the D-A isotherm was found to be 2.63 kJ/mol which suggests that the adsorption mechanism is physical.
The pseudo-second-order model was more suitable than the pseudo-first-order model to describe the adsorption kinetics of arsenic ions onto adsorbent particles.
The results of the flotation studies demonstrated that when 120 mg/L of Fe(III), 2 mg/L of Nalco 9808, 20 mg/L of sodium oleate, and 40% of recycle ratio were used in the FAD, it was possible to reach 98% of arsenic removal and 6.3 NTU of residual turbidity in the clarified synthetic effluent.
Acknowledgements
The authors acknowledge financial support from the University of Atacama (Dirección de Investigaciones de la Universidad de Atacama, Proyecto DIUDA 221167) and appreciate the collaboration of Miss Priscilla Bravo and Mr. Bruno Zazzali in the experimental development, and Miss Evelyn Cárdenas in the writing of this article.
References
Baradel, A., Guerriero, R., Meregalli, L., & Vittadini, I. (1986). Extraction of As from Copper Refining Electrolyte. Journal of Metals, 32-37. [ Links ]
Bhakat, P. B., Gupta, A. K., & Ayoob, S. (2007). Feasibility analysis of As (III) removal in a continuous flow fixed bed system by modified calcined bauxite (MCB). Journal of Hazardous Materials, 139, 286-292. [ Links ]
Boggs, P. T., Byrd, R. H., Donaldson, J. R., & Schnabel, R. B. (1987). A stable and efficient algorithm for nonlinear orthogonal distance regression. SIAM Journal Scientific and Statistical Computing, 8, 1052-1078. [ Links ]
Boggs, P. T., Byrd, R. H., Donaldson, J. R., & Schnabel, R. B. (1989). Algorithm 676-ODRPACK: software for weighted orthogonal distance regression. ACM Transactions on Mathematical Software, 15(4), 348-364. [ Links ]
Brunori, C., Cremisini, C. Massanisso, P., Pinto, V., & Torricelli, L. (2005). Reuse of a treated red mud bauxite waste: Studies on environmental compatibility. Journal of Hazardous Materials, 117(1), 55-63. [ Links ]
Cui, J., Du, Y., Xiao, H., Yi, H., & Du, D. (2014). A new process of continuous three-stage co-precipitation of arsenic with ferrous iron and lime. Hydrometallurgy, 146, 169-174. [ Links ]
D. Mohan, D., & S. Chander, S. (2006). Single, binary, and multicomponent sorption of iron and manganese on lignite. Journal Colloid Interface Science, 299(1), 76-87. [ Links ]
Dron, J., & Dodi, A. (2011). Thermodynamic modelling of cl-, no3- and so42- removal by an anion exchange resin and comparison with Dubinin-Astakhov isotherms. Langmuir, 27, 2625-2633. [ Links ]
El-Khaiary, M. I., & Malash, G. F. (2011). Common data analysis errors in batch adsorption studies. Hydrometallurgy, 105, 314-320. [ Links ]
El-Khaiary, M. I. (2008). Least-squares regression of adsorption equilibrium data: Comparing the options. Journal of Hazardous Materials, 158, 73-87. [ Links ]
Féris, L. A., De León, A. T., Santander, M., Rubio, J. (2004). Advances in the adsorptive particulate flotation process. International Journal Mineral Processing, 74, 101-106. [ Links ]
Filliben, J. (1981). DATAPLOT-an interactive high-level language for graphics, non-linear fitting, data analysis and mathematics. Computer Graphics, 15,199-213. [ Links ]
Ghimire, K. N., Inoue, K., Makino, K., & Miyajima, T. (2002). Adsorption removal of arsenic using orange juice residue. Separation Science and Technology, 37(12), 2785-2799. [ Links ]
Gunther, K., & Wanner, D. (1991). Removal of arsenic from washing acid by Sachtleben-Lurgi process, Pa.: Minerals, Metals & Materials Society. IN: Residues and effluents: Processing and environmental considerations, 833-837. [ Links ]
Guo, H., Stüben, D., & Berner, Z. (2007). Adsorption of arsenic(III) and arsenic(V) from groundwater using natural siderite as the adsorbent. Journal of Colloid and Interface Science, 315, 47-53. [ Links ]
Jain, A., & Loeppert, R. H. (2000). Effect of competing anions on the adsorption of arsenate and arsenite by ferrihydrite. Journal of Environmental Quality, 29, 1422-1430. [ Links ]
Kadukova, J., & Vircikova, E. (2005). Comparison of differences between copper bioaccumulation and biosorption. Environment-International, 31(2), 227-232. [ Links ]
Kerfoot, D. (1978, September 19). Method for removing arsenic from copper and/or nickel bearing aqueous acidic solutions by solvent extraction (US Patent N° 4115512 A). [ Links ]
Klamp, G., & Wanner, D. (1991). Removal of arsenic from washing acid by the Sachtleben Lurgi Process. In: R. G. Reddy, W. P. Imrie, P. B. Queneau (Eds.), Residues and Effluents-Processing and Environmental Considerations (pp. 833-837). San Diego, Ca.: TMS. [ Links ]
Krause, E., & Ettel, V. (1989). Solubilities and stabilities ferric arsenate compounds. Hydrometallurgy, 22, 311-337. [ Links ]
Kundu, S., & Gupta, A. (2007). Adsorption characteristics of As(III) from aqueous solution on iron oxide coated cement (IOCC). Journal of Hazardous Materials, 142, 97-10. [ Links ]
Leist, M., Casey, R. J., & Caridi, D. (2000). The management of arsenic wastes: Problems and prospects. Journal of Hazardous Materials, B76, 125-138. [ Links ]
Maiti, A., DasGupta, S., Basu, J. K., & De, S. (2007). Adsorption of arsenite using natural laterite as adsorbent. Separation and Purification Technology, 55, 350-359. [ Links ]
Malkoc, M. (2006). Ni(II) removal from aqueous solutions using cone biomass of Thuja orientalis. Journal of Hazardous Materials, B137, 899-908. [ Links ]
Matis, K. A., Zouboulis, A. I., Malamas, F. B., Ramos Afonso M. D., & Hudson, M. J. (1997). Flotation removal of As(v) onto goethite. Environmental Pollution, 97(3), 239-245. [ Links ]
Mohan, D., & Pittman, C. (2007). Arsenic removal from water/wastewater using adsorbents-A critical review. Journal of Hazardous Materials, 142, 1-53. [ Links ]
Nagarnaik, P. B., Bhole, A. G., & Natarajan, G. S. (2003). Arsenic (III) removal by adsorption on sawdust carbon. International Journal Environmental Pollution, 19(2), 177-187. [ Links ]
Nishimura T., Tosawa K., Itho, T., & Robins, R. (1998). Stabilities and solubilities of metal arsenites and arsenates in water and effect of sulfate and carbonate ions on their solubilities. Arsenic Metallurgy Fundamentals and Applications (pp. 77-98). The Metallurgical Society of AIME. [ Links ]
Nishimura, T., & Tozawa, K. (1988). Kinetics of Oxidation of Arsenious Acid with Aqueous Ozone. J. Min. Met. Inst. Japan, 44(1), 549-153. [ Links ]
Onyango, M. S., Matsuda, H., & Ogada, T. (2003). Sorption kinetics of arsenic onto iron-conditioned zeolite. Journal Chemical Engineering of Japan, 36(4), 477-485. [ Links ]
Papassiopi, N., Virèiková, E., Nenov, V., Kontopoulos, A., & Molnár, L. (1996) Removal and fixation of arsenic in the form of ferric arsenates. Three parallel experimental studies. Hydrometallurgy, 41, 243-253. [ Links ]
Robins, R.G. (1987). Solubility and stability of scorodite, FeAsO4.2H2O: Discussion. American Mineralogist, 72(8), 842-844. [ Links ]
Robins, R. G. (1990). The Stability and Solubility of Ferric Arsenate: An Update. In: D. R. Gaskell (Ed.), EPD Congress '90 (pp. 93-104). Warrendale, PA. [ Links ]
Rubio, J., Souza, M. L., & Smith, R. W. (2002). Overview of flotation as a wastewater treatment technique. Minerals Engineering, 15,139-155. [ Links ]
Rubio, J., & Tessele, F. (1997). Removal of heavy metal ions by adsorptive particulate flotation. Minerals Engineering, 10(7), 671-679. [ Links ]
Rubio, J. (1998). Effluent treatment in the mining industry, University of Concepción, Chile. In: S. H. Castro, F. Vergara, M. Sanchez (Eds.), Environmental applications of the flotation process (pp. 333-364) (Chapter 8). [ Links ]
Saha, B., Bains, R., & Greenwood, F. (2005). Physicochemical characterization of granular ferric hydroxide (GFH) for arsenic (V) sorption from water. Separation Science Technology, 40(14), 2909-2932. [ Links ]
Santander, M., Tapia, P., Pávez, O., Valderrama, L., & Guzmán, D. (2009). Remoción de partículas adsorbentes de iones cobre por flotación Jet. Revista de Metalurgia, 45(5), 365-374. [ Links ]
Santander, M., Valderrama, L., Guevara, M., & Rubio, J. (2011). Adsorbing colloidal flotation removing metals ions in a modified jet cell. Minerals Engineering, 24, 1010-1015. [ Links ]
Sharma , V. K., & Sohn, M. (2009). Aquatic arsenic: Toxicity, speciation, transformations, and remediation. Environment International, 35(4), 743-759. [ Links ]
Tessele, F., Misra, M., & Rubio, J. (1998). Removal of Hg, As and Se ions from gold cyanide leach solutions by dissolved air flotation. Minerals Engineering, 11(6), 535-543. [ Links ]
Tikhomolova, K., & Urakova, I. (2002). Interaction of Co(II), Ni(II) and Cu(II) with quartz surface in aqueous solutions at varied pH. Russian Journal of Applied Chemistry, 75(6), 894-899. [ Links ]
Valenzuela, A., & Sánchez, M. (2001). Manejo de Arsénico en la industria Metalúrgica: Recuperación y neutralización. In: J. P. Ibáñez, E. Patiño, & X. Veloso (Eds.), II Encuentro Minero del Cono Sur de América, VII Encuentro Minero de Tarapacá, Iquique, Chile, II-25-I-35. [ Links ]
Wilkie, J. A., & Hering, J. G. (1996). Adsorption of arsenic onto ferric hydroxide: effects of adsorbate/adsorbent ratios and co-occurring solutes. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 107, 97-110. [ Links ]
Wong, Y. C., Szeto, Y. S., Cheung, W. H., & McKay, G. (2004). Pseudo-First-Order Kinetic Studies of the Sorption of Acid Dyes onto Chitosan. Journal of Applied Polymer Science, 92, 1633-1645. [ Links ]
Zhang, N., Blowers, P., & Farrell, J. (2005). Evaluation of density functional theory methods for studying chemisorption of arsenite on ferric hydroxides. Environmental Science Technology, 39(13), 4816-4822. [ Links ]
Zouboulis, A. I. (1995). Silver recovery from aqueous streams using ion flotation. Minerals Engineering, 8(12), 1477-1488. [ Links ]
Zouboulis, A. I., Kydros, K. A., & Matis, K. A. (1992). Adsorbing flotation of copper hydroxo precipitates by pyrite fines. Separation Science and Technology, 27(15), 2143-2155. [ Links ]
Zouboulis, A. I., Matis, K. A., Lazaridis, N. K., & Golyshin, P. N. (2003). The use biosurfactants in flotation: Application for the removal of metal ions. Minerals Engineering, 16, 1231-1236. [ Links ]













