Introduction
Soybean is one of the most economically important crops in the USA whilst in Europe it is very limited, due to climate and soil conditions (A. Geoffrey Norman, 2006). Neither all tank reactors are perfectly mixed, nor do all tanks have their composition and temperature independent from the length of the reactor (Plug-flow behavior - Ideal Reactor) (H. Scott Fogler, 2006). For Soymilk production, this is the case. The latter is an inexpensive source of protein, minerals, carbohydrates, oil (blends), vitamins and calories for human consumption. All its compounds formula have not been described completely due to the different species and varieties in the world and their chemical variation such as the well-known health benefits: Proteins, Isoflavones, and dietary fibre (glucose, uronic acids, galactose, arabinose and xylose) - (Ren, Liu, Endo, Takagi & Hayashi, 2006). Moreover, Soymilk is a low-cost substitute for dairy milk for the poor in the developing countries. Being free of cholesterol, gluten and lactose, soymilk is also a suitable food for lactose-intolerant consumers, vegetarians and milk-allergy patients. Today, consumer demands are more and more directed toward high-quality, additive free, minimally processed, nutritious, and deteriorative organisms as well as inactivate undesirable enzymes (Seibel & Beleia, 2009). Nevertheless, high pasteurization temperatures impact negatively on the nutritional quality and taste of milk (Chou & Hou, 2010). Soybean seeds are known to contain different anti-nutritive factors, such as: trypsin inhibitors, phytic acid, raffinose and stachyose many of which lose their effects after processing. Previous research (Becker-Ritt, Mulinari, Vasconcelos, & Carlini, 2004; Kumar, Rani, Solanki, & Hussain, 2006) shows that there is an increasing interest from scientists in soybean which is focused on the characterization of its components. The aim of this paper is attain new data and information about the Soymilk composition which may be relevant to the scientific and industrial - health community, using a Simulation Aspen Hysys software which has customer testimonials from: LG Chemistry, Chevron, Arithmetek, Siam, Petro-bras, Campbell, ConocoPhillips, Fluor, Hitachi Zosen, Genesis Consulting, INEOS and others (Fusco, 2013). The macromolecules described here are the onset for new specific soymilk compounds.
Materials and methods
Soybean samples and water extract of soybeans (soymilk)
0,5 Kg/h were feedstock for the process resembling the one undergone in markets in Brazil and Canada, taking care to ensure that good quality of soybeans (Chemical composition). Whole soybeans were first washed and soaked with hot water and undergo pre-heating, to inactivate the Lipoxigenase to foresee bad properties in the final flavor, until the water boiling point (at 89,326 KPa) -(CDMB 2013) for 5 min. The soybeans were blended with 2 times of volume of soybean in water, then the soybeans were taken in a grinding unit for 3 minutes. Then the mixture of soybean was filtered in a 1,2 μιτι filter. The solid residue was used to measure the PH between 8 and 8,5 in two measures. The final liquid filtered was heated (138 °C) before entry to the pasteurization process 600 MPA and (148 °C) streamed to a container at -2 °C and 89.326 KPa in 3 seconds (Figure 1).

Source: Aspen Tech Hysys v 7.1 - Flow sheet panel
Figure 1 Soymilk Production, Plant Simulation by Aspen Hysys
Soybean composition and Soybean composition prediction.
Table 1 Chemical composition prediction through the software for soybeans

Source: Software Aspen Tech Hysys v 7.1
In this Stage of the process, 3 units were included (Heating, Pasteurization and Cooling) to guarantee the quality of the food by the microorganism inactivation. The fluid packages fo this stage were the Generic COMthermo Pkg. (Van Laar - Virial for Liquid blends) and extended NRTL- Virial (For Soybean Solid properties and Pasteurization Final Product). The heating unit was included to prevent damage due to the high increase of the temperature at Pressure values of 600 MPa. Since an increase (DT = 123 °C and DP = 498675 Pa) is furnish for the entire process, the latter was caused by the collisions of the particles. Besides, the Heat supply from the heat source makes necessary a rigorous and strict control for the temperature, the Heating unit at 101,325 KPa is a cheaper alternative. In this stage, the condition for the inlet and outlet streams can be appreciated including the duty (Heat Flux) 3,766 KJ/h. The unit of cooling is included to guarantee the inactivation of pathogenic microorganism by the collision of hot particles (148 °C) with a container with fluid at -2 °C value.
Table 2 Thermodynamics properties for the soybeans after PreHeating Stage (Inactive Lipoxygenase)

Source: Software Aspen Tech Hysys v 7.1
Stage1, 2 and 3 are continuously linked in a unique flow sheet diagram (Figure adapted for the journal template)
Results and discussion
Element composition and soymilk compounds
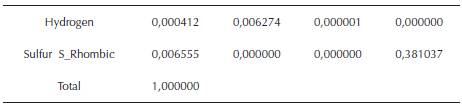
The final soymilk was formed by 16 amino-acids, the Polysaccharides stachyose, Rafinosse, Sucrose, Daidzein as the total isoflavones, and thiamin and niacin as Vitamin B content. The final mass fractions of Carbon wC = 0.002455, Nitrogen wN = 0.005726, and Oxygen wO = 0.006541, Copper wCu = 0,012988, Calcium wCa = 0,008193, Sodium wNa = 0,004699, Magnesium wMg = 0,004968, Phosphorus wP = 0,006632, iron wFe = 0,011416, Potassium wK = 0,007992, Hydrogen wH = 0,000412, Sulfur wS = 0,006555 indicate a presence of macromolecules besides the cited above, which chemical Structures can be derived from the empirical formula and subsequently by the Global molecular formula.
Table 3 Pasteurization Thermodynamics conditions for UHT and UHPH units

Source: AspenTech Hysys - Flowsheet Conditions
Global molecular formula assessment (For t = 1 hour) Source data: Table 4
mi = mt * wi
Equation (1)- Mass calculation for each component
mi = mass of each element
mt= total mass
wi = mass fractio
Equation (2)- Mole calculation for each component
For Carbon replacing (1) e.g.
mc = mt * wc
Equations and calculation procedure
After dividing all moles in 0,001703 k-mole to find the mole empirical ratio we obtain:
We must find a ratio of integers according to the law of mass action.
Conclusions
Based in the composition predicted by the software, we can make a classification of the specie for the soybean (Glycine Max. L.). It resulted in the presence of C20 H40 S20 N40 O40 that indicates the presence of Protein (the presence of an essential amino acid with sulfur). Multiple Biomolecule options can be derived from this analysis such as: Organometallic Compounds, Organo Phosphorus Compounds, Proteins, Carbohydrates, Fat, Vitamins. One option for this could be large linear chains with methionine or cysteine both considered as unique source. 1,1111 mole of C8H18N2O4S2 or a 1 mole of (CH2(NO)2)20 (Fulminic Acid Family compound) according to (H Yang & L. Zhang, 2007), and an Organ phosphorus compound (Phospholipids). Although this last one is present mostly in pesticides, two hypothesis can be considered: The presence of them due to residuals in soybean after chemical process in a non-mortal level, or their presence as any of these compounds: P ( = O)(OR)3; RP ( = O) (OR')2; R3P = O;. According to (Seibel & Beleia 2009), one kind is C6H18O24P6 written as (C2H6)3(O4P)6 (phytic acid) where the remaining 15 atoms of P would be inorganic Phosphorus. The remaining proportions of mole for protein analysis may correspond to new proteins which are not well-known such as those found in studies by the Hospital de Rhode Island (Jonathan Kurtis, 2014 PfSEA-1 2014 Journal: Science) for the presence of trace elements (Micronutrients - trace elements Cu and Fe) (Organometallic compounds).