Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Facultad de Ingeniería Universidad de Antioquia
Print version ISSN 0120-6230On-line version ISSN 2422-2844
Rev.fac.ing.univ. Antioquia no.56 Medellín Oct./Dec. 2010
Simulation of two alternatives for SO2 removal from wet cement kiln exhaust gases
Simulación de dos alternativas para la remoción de SO2 de los gases de escape de hornos cementeros
Carlos Duque1, Consuelo Montes1, Felipe Bustamante1, Alejandro Ortiz2
1Grupo Catálisis Ambiental. Departamento de Ingeniería Química. Universidad de Antioquia. Sede de Investigación Universitaria (SIU) - Carrera 53 N.° 61-30. Medellín. Colombia.
2Cementos Argos S.A. Calle 7 D N.° 43A-99. Medellín. Colombia.
Abstract
This work aims at simulating two processes for capturing sulfur dioxide from exhaust gases of wet clinker processes. The goal is to present a guide to cement manufacturers when selecting the most appropriate technology for wet processes in order to comply with environmental regulations. The available commercial technologies chosen for desulfurization process were: wet limestone and wet Cement Kiln Dust (CKD) removal processes. A commercial simulator (Aspen Plus v.2006.5) was used. The absorption tower -considered the core of the process- was simulated with an Aspen RadFrac model combined with Aspen RateSep calculations to provide better and more accurate simulation results than a simulation with the traditional equilibrium approach. This combination also allows better estimates for equipment sizing. The convergence issues, both for the absorption of sulfur dioxide and the overall process, were solved using Aspen Plus tools.
Keywords: Aspen plus, CKD, limestone, radfrac, ratesep, SO2 removal process, simulation, sulfur dioxide.
Resumen
El objetivo de este trabajo es similar dos procesos para capturar el dióxido de azufre liberado en los gases de escape de una cementera que opere con el proceso de clinker húmedo. De esta manera se pretende ayudar a las empresas cementeras en la selección de la tecnología más adecuada para cumplir las regulaciones ambientales. Se seleccionaron dos tecnologías comerciales para la remoción de SO2: wet limestone, y wet Cement Kiln Dust (CKD), que se simularon usando un software comercial (Aspen Plus v.2006,5). La torre de adsorción, que es considerada el punto crítico del proceso, se simuló usando el modelo Aspen RadFrac combinado con los cálculos de Aspen RateSep, de modo que se obtuvieran resultados más exactos que los alcanzados con la alternativa tradicional del diseño basado en el equilibrio. Además, los resultados obtenidos con esta combinación dan mejores estimaciones para el diseño de los equipos. Los aspectos relacionados con la convergencia de la simulación, tanto para la torre de absorción como para el proceso global, fueron resueltos usando las herramientas del software.
Palabras clave: Aspen plus, CKD, limestone, radfrac, ratesep, procesos de remoción de SO2, simulación, dióxido de azufre.
Introduction
Sulfur dioxide (SO2) is an important atmospheric pollutant and acid rain precursor. Several technologies for sulfur dioxide removal have been developed and nowadays it is possible to achieve 99% removal of SO2 [1]. The best alternative for SO2 removal depends on: feedstock availability, final waste handling, and -of course- capital investment. In the case of cement plants both the first and second issues play an important role for reducing operating costs.
Cement production by wet methods include mining to obtain the main feedstock -limestone (CaCO3). After reducing particle size and mixing with water (30-40%) the raw material is introduced in a large rotary kiln. In this kiln water is evaporated and feedstock calcined (1700 K), becoming a black rock called clinker, formed primarily by calcium oxides. Finally, the clinker is milled with gypsum (CaSO4.2H2O) and other additives to obtain different cement types. The particulate material present in the gaseous effluent of the kiln is removed in an electrostatic precipitator, resulting in the so-called Cement Kiln Dust (CKD).
A large variety of fuel sources have been used in cement clinker kilns. Since high fuel consumption is required, coal has become one of the most common fuels [2]. When medium- or high-sulfur coal is used as fuel, the SO2 content of the flue gas may exceed air-quality-emission limits. The SO2 concentration of the flue gas depends on the fuel sulfur content and also on the mineral raw materials, as well as, the design and operating conditions of the cement kiln.
Recently, Colombian government issued stricter environmental regulations for cement industries, including SO2 emissions [3]. Consequently, Colombian cement manufacturers need to take measures to achieve the required limits. Several Colombian facilities rely on the wet clinker process, which -compared to the dry clinker process- requires a larger amount of coal leading to larger SO2 emissions. Only few sulfur dioxide emission removal technologies are adequate for the wet clinker process (e.g. wet limestone, sodium bicarbonate, wet CKD, dry lime or oxidation). Thus, modeling and simulation are valuable tools for selecting the most appropriate option.
Among the available commercial SO2 removal technologies, the wet limestone (calcium carbonate, CaCO3) process has received much attention. Several mathematical models have been proposed and verified experimentally [4]. However, these models are developed for specific operating conditions and require programming of complex algorithms. On the other hand, several reports suggest that wet Cement Kiln Dust (CKD) process is also a viable gas desulfurization option for wet clinker production [5, 6]. However, published papers on the modeling of this process are scarce.
In contrast with the specific mathematical models discussed above, simulation with commercial software provides a user-friendly, yet rigorous, approach for designing and optimizing process units for different process conditions. This work aims at simulating two processes -wet limestone and wet CKD- for capturing sulfur dioxide in wet cement kilns based on available commercial technologies. Aspen Plus software package [7] was used for the simulation and preliminary sizing of process units. The process simulation model is expected to provide the following information: product stream concentrations, required liquid feed, and a first estimate of equipment size. This information is important when selecting the best alternative for the SO2 removal process.
Detailed process model
Alternatives for desulfurization process
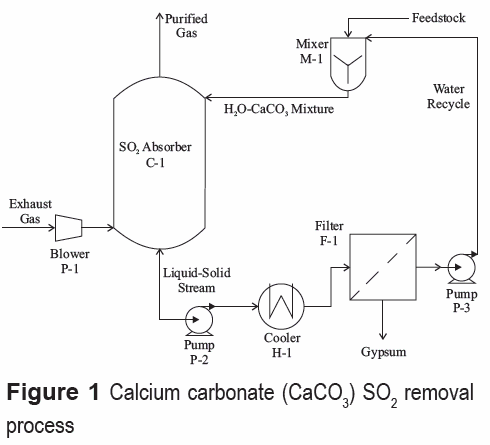
As we mentioned above, an alternative for the desulfurization process should be selected based on different criteria: process efficiency, feedstock availability, final waste handling, and capital investment, among others. Several reasons make the wet limestone removal process an interesting choice: high removal efficiency; required feedstock can be obtained directly from the plant (i.e., limestone is a raw material for clinker production); gypsum (dihydrate calcium sulfate, CaSO4.2H2O) generated as byproduct might be used as a cement additive, thus reducing production costs and avoiding final solid disposal. Figure 1 shows a scheme of the wet limestone process. Flue gas desulfurization occurs in the absorption tower, where a calcium carbonate mixture removes SO2 by chemical absorption. The precipitated gypsum contained in the liquid outlet stream is removed by filtration. Only one filter is needed for solid separation since gypsum is not required to be completely dry for its further use in cement manufacture. Finally, the liquid (without gypsum) is recycled to the absorber.
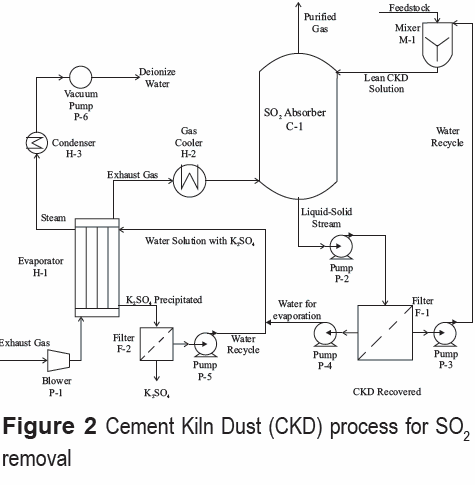
Although limestone appears to be a good alternative for flue gas desulfurization, several reports suggest that wet CKD process is also a viable gas desulfurization option for clinker production [5, 6]. There are several reasons for using CKD: it is a byproduct from clinker process with no commercial value; potassium sulfate (K2SO4) -one of the byproducts of the removal process- has applications as a fertilizer and could be sold. Besides, a solid compound -primarily composed of calcium carbonate, and free of alkaline ions- is also obtained in the process; this "recovered CKD" has similar characteristics to the feedstock of the clinker process (e.g., high CaCO3 content) and, therefore, could be reused as raw material. The flowsheet of the wet CKD process is shown in figure 2. The need to recover potassium sulfate by evaporation makes the wet CKD process more complex than the calcium carbonate process. The main difference with the limestone process is that part of the recycling liquid is sent to an evaporator for K2SO4 recovery (see figure 2). In order to get an objective criterion for comparing and selecting the most appropriate technology, both processes, limestone and CKD, were simulated under the same removal efficiency and inlet conditions, namely, sulfur content, flowrate, temperature and pressure. Aspen Plus version 2006.5 [7] was used as the commercial process simulator due to its wide modeling capability.
Simulation
A detailed understanding of the underlying phase, chemical, and combined phase and chemical equilibria, accounting for the presence of electrolytes and solids -which is required for an accurate simulation of the process- is a challenging task. The framework used in Aspen Plus for modeling a three phase system is based on liquid-solid and vapor-liquid equilibria. The reactions considered in this study occur in the liquid phase, involving electrolytes in solution and precipitation of solids. Consequently, the Electrolyte-NRTL model [8] for the liquid and solid phases, and the Redlich-Kwong equation for the gas phase [9, 10] were used. The ENRTL system is capable of modeling solid precipitation from aqueous mixtures. Moreover, ENRTL implicitly includes calculation models of heat and mass transfer parameters and properties [10].
The absorption tower, which can be regarded as the core of the process, was simulated with the RadFrac module; RadFrac is a rigorous model for simulating all types of multistage vapor- liquid fractioning operations [11]. Furthermore, in addition to the default equilibrium stage mode (hereinafter referred as RF-equil), RadFrac was combined with RateSep calculation (hereinafter referred as RF-RS) for a better estimation based on mass and heat transfer parameters; RateSep is a rigorous rate-based approach which considers mass and heat transfer limitations, liquid and vapor film diffusion, equipment hydrodynamics and chemical reaction mechanisms [12]. RateSep is, therefore, a great complement to RadFrac due to its realistic -although more complex- calculations. Moreover, RF-RS calculations for the reactive absorption considered in this study use the electrolyte system and equilibrium reactions to determine the number of stages based on transport phenomena. This approach provides better estimates of the number of stages without the need of an initial guess of plate efficiencies. Thence, as it will be illustrated below, RF-RS calculations provide better and more accurate simulation results, allowing better estimates for equipment sizing.
Reaction sets
The simulation of the absorption tower is complex because of the multiple reaction equilibria present. However, it is possible to simultaneously simulate all the involved reactions in the system using the RadFrac tower. The set of equations shown in table 1 was used for the limestone process [13].
The same reaction set, plus the alkali reactions [6], were used in the simulation of the wet CKD process (see table 2); it is necessary to consider the alkaline reactions due to the significant amount of alkaline oxides present, which might prevent the recirculation of CKD.
Although there are other minor reactive species present in the CKD feedstock, they were neglected in the calculations. In a similar manner, calcium carbonate feedstock is approximately 75% pure.
Equilibrium constants for each reaction are either included in the Aspen database or have been calculated by Gibbs free energy minimization [7]. Some of the equilibrium constants were compared to values reported by Kohl [13] obtaining similar results; therefore, it was not necessary to introduce corrections in the Aspen database.
Results
Process model and convergence
As all reactions occur in the absorption tower, the overall convergence of the simulation for both SO2 removal processes strongly depends on the convergence of this unit. Several options for process convergence, including those suggested in the help files, were tried for the RadFrac tower with the default equilibrium stage mode, RF-equil [7]. It was observed that in addition to the standard convergence (Inside-Out algorithm), the use of temperature estimates and activation of the option for calculating solids by stages was required. Initial equilibrium convergence is needed because RF-RS mode uses the results of the equilibrium stage mode as input for solving its algorithms.
The results for RF-equil model showed that the removal could be achieved in just two stages -the minimum number allowed by the model-. This is a consequence of the reaction products being favored by the equilibrium constant regardless of equipment dimensions; therefore, the RF-equil does not provide the necessary information for equipment sizing. One option to circumvent this problem would be to use corrections for plate efficiency. This option was not used in this work because calculations were conducted with RF- RS, which correlates geometry and residence time of either phase to the conversion of the electrolyte reaction in each stage, i.e. RF-RS is a more accurate approach and provides a better estimation of the number of stages than RF-equil. As a consequence, the simulation and comparison of removal alternatives was conducted with the Rate-Based approach (i.e. RF-RS).
However, the option of calculating precipitation of solids is not included in the RateSep model. Nevertheless, according to our results, the model does handle the precipitation of solids indirectly. Indeed, when the simulation converges the Aspen control panel displays a warning indicating that the system saturation point is greater than 1 and, therefore, precipitation might exist on some stages (the electrolyte model "forces" all ions to be in solution by somewhat inhibiting the precipitation reactions: this may result in saturation point greater than 1). Moreover, the calculations are not affected, as can be inferred by noting that the liquid outlet stream properties are similar to those obtained with RF-equil -which does explicitly include precipitation-. Then, it is possible to conclude that the RF-RS results in the tower are correct, despite its apparent lack of solid precipitation in the simulation tower. Subsequently, the RF-RS module was combined with an additional unit (e.g., a heat exchanger) to ensure the formation of the solid precipitate.
Processes were simulated as a closed flowsheet with ions diluted in the water recycle stream from the filter. This recycle makes the simulation convergence more challenging, because the system required two convergences: RadFrac convergence per iteration and process convergence. For process convergence, adjustment of the feedstock (makeup water stream and solid stream) in either iteration is needed. The makeup water stream depends on water lost by evaporation in the outlet stream gas, and water consumed in the reaction; solid stream amount depends on the quantity of ions present on the recycle water. Then, implementation of a calculator block was necessary to correct per iteration the values of makeup water and solid stream. Other important factors for the process convergence were the choice and the initial estimation of the tear streams (selected in order to help reaching process convergence with recycle streams) and the calculation sequence for the blocks. Finally, as the Rate-Based approach (RF- RS) depends on the tower geometry for mass and heat transfer calculations, the equipment design is also an iterative process. Therefore, it was necessary to guess design values, such as plate height and diameter, and correct them with the results. In addition, it was required to make an initial guess of the number of stages for obtaining the required SO2 removal.
Simulation results
Stream Inlets
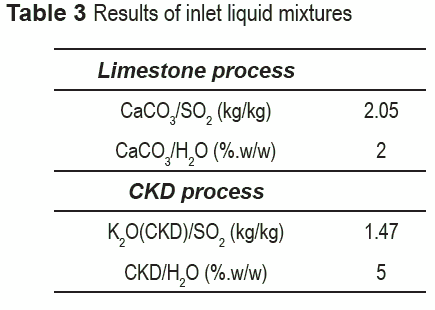
he amount of solid calcium carbonate in the liquid stream entering the absorption tower was fixed as the stoichiometric amount necessary to achieve 99% removal of sulfur dioxide. The amount of water in the inlet stream was fixed according to the flow parameter factor [14]: there exists a rule-of-thumb applicability range of flow parameter -the ratio of liquid to vapor kinetic energy- in equipment design. The specific value within the range was selected considering that due to the low solubility of calcium carbonate, a larger amount of water (i.e., a dilute solution) favors the presence of ions in solution speeding up the reaction. The inlet stream for the process using CKD was also fixed using that rule of thumb. Considering that the goal is to recycle all the CKD as feedstock for clinker production, it is required to remove all alkaline ions present. As a result of the low concentration of K2O in the CKD, and despite its high solubility, a larger amount of water was required. Notwithstanding, the higher concentration facilitates recovery of solids. CKD is expressed in terms of potassium oxide (K2O) in table 3 because this is the primary component for SO2 removal reaction, and its concentration varies from plant to plant. K2O and SO2 concentrations are not reported due to a privacy agreement between the University and the Company.
Table 3 shows the amount of CaCO3, K2O (CKD), and water necessary for sulfur dioxide removal using both alternatives; the presence of other species in CKD that may react with sulfur dioxide (e.g., MgO, Na2O), can be neglected since they are normally in lower proportions than K2O. The stream conditions of exhaust gas to be treated are listed in table 4.
The CKD/water ratio depends on several factors: equipment size (which in turn depends on the flow parameter), and energy consumption (a function of the flowrate) being the more important ones. All these parameters affect investment and process costs.
Absorber
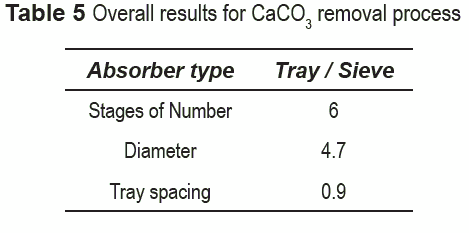
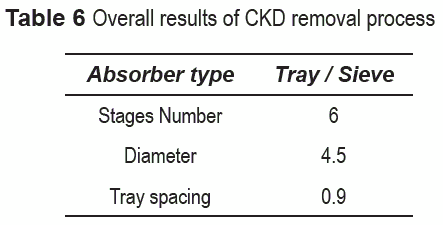
The main characteristics of the absorber, once the convergence was reached, are shown in tables 5 and 6 for limestone and CKD, respectively. The diameter depends primarily on the gas flowrate because this is the determining factor in the flow parameter, i.e. the liquid flow rate does not notably affect the diameter. Therefore, the diameter in both processes is quite similar.
The number of stages in both processes is the same which may be an indication that the process is controlled by gas-liquid mass transfer. In fact, Aspen Plus does not account for solid-liquid mass transfer limitations. On the other hand, in actual operation, particle size distribution may play a significant role in the solid-liquid diffusion rate; however, this kind of calculation would make the model much more complex that it currently is. The above results show that the specifications of the absorption tower are not a determinant factor in selecting the best alternative.
Purified gas
Tables 7 and 8 show the conditions of the purified gas for each removal process. Due to different treatment of the inlet gas streams, outlet streams differ in temperature and, consequently, water concentration. Both processes almost completely removed sulfur dioxide.
Selected alternative
The CKD process has been successfully used in other cement plants [6], nevertheless, the results suggest that it is not applicable for the conditions considered in this study. The most important reason is that sulfur dioxide emissions were too high, so there was not enough K2O available in the CKD to remove all sulfur dioxide without producing gypsum (with the amount of CKD available is possible to remove only 50% of SO2). However, the CKD alternative was still considered because the solid product could be used in other part of the cement process. As we mentioned above the clinker is mixed with gypsum and other process additives. One of these additives would be the solid CKD.
The inversion costs for CKD process would be higher due to the evaporation step needed for potassium sulfate recovery. Furthermore, the flowrate of lean CKD solution is higher than in the limestone process, resulting in larger equipment and higher operating costs (pumping). Moreover, the marketing of the potassium sulfate would pose new problems of packing, storage and transport, among others. Also, as a result of the variable composition of CKD byproduct the purity of the potassium sulfate would vary. However, if the potassium sulfate could be sold and the CKD could be recoverable, the incomes and cost savings can make the process profitable. Therefore, though the limestone process would be less profitable than the CKD -under the optimum conditions of the latter-, it is simpler, widely known and produces gypsum which also can be used directly in cement manufacture.
Conclusions
SO2 removal by limestone and CKD in a cement plant was satisfactorily simulated under the same inlet conditions of the sulfur-laden exhaust gas. A preliminary equipment sizing was carried out based on the gas and liquid flowrate to achieve a 99% removal of sulfur dioxide. The necessary amount of inlet liquid mixture for both processes was established.
The convergence issues, both for the desulfurization process in the absorption tower and the overall process, were solved using Aspen Plus tools. The system was simulated including all the possible reactions involved and the presence of the solid phase as well. It was found that the combination of the absorption module (RadFrac module) with a model accounting for mass and/ or heat transfer limitations (Aspen RateSep) proved to be an excellent option for the reactive absorption process. In fact, the conventional stage equilibrium approach led to a significant underestimation of the absorber.
CKD process advantages has been claimed in the literature [6] -e.g., CKD recovery and potassium sulfate production-, but under the conditions of this study, the wet limestone is considered to be the best option because there are technical hurdles to implement CKD recovery, which would diminish its cost/benefit ratio. However, CKD process is still an interesting option that should be considered for others cement plants.
Acknowledgments
The authors wish to thank the Universidad de Antioquia, Servicio Nacional de Aprendizaje SENA, and Cementos Argos S.A. for their financial support for this study.
References
1. G. T. Joseph, D. S. Beachler. "Scrubber Systems Operation Review". Lesson 9: Flue GasDesulfurization (Acid Gas Removal) Systems. U.S. Environmental Protection Agency (EPA) APTI Course SI: 412C. 2da. ed. 1998. Available from: http://yosemite.epa. gov/oaqps/E0Gtrain.nsf/DisplayView/SI_412C_0- 5?OpenDocument. Consultada en Abril de 2008. [ Links ]
2. K. Schumacher, J. Sathaye. "India's cement industry: Productivity, energy efficiency and carbon emissions. LBNL-41842." Lawrence Berkley National Laboratory, USA. 2007. Available from: http://eetd. lbl.gov/ea/ies/suni6/industry41842.pdf. Consultada en Marzo de 2008. [ Links ]
3. Ministerio de Ambiente, Vivienda y Desarrollo Territorial. República de Colombia. Resolución No 909 de junio 5 de 2008. pp. 15-17. [ Links ]
4. A. Gómez, N. Fueyo, A. Tomás. "Detailed modeling of a flue-gas desulphurization plant." Comput. Chem. Eng. Vol. 31. 2007. pp. 1419-1431. [ Links ]
5. U.S. Environmental Protection Agency (EPA). Report to Congress 1993-Cement Kiln Dust Waste. Available from: http://www.epa.gov/osw/nonhaz/industrial/ special/ckd/cement2.htm. Consultada en Abril de 2008. [ Links ]
6. L. P. Passamaquoddy Technology. Passamaquoddy Innovative Clean Coal Technology Program. Final Report. U.S. Department of Energy. Feb. 1994. [ Links ]
7. Aspen Plus. 2007(A). Aspen Plus version 2006.5. Aspen Technology Inc. Cambridge. MA. USA. [ Links ]
8. C. C. Chen, H. I. Britt, J. F. Boston, L. B. Evans. "Local Composition Model for Excess Gibbs Energy of Electrolyte Systems, Part I: Single Solvent, Single Completely Dissociated Electrolyte Systems". AIChE J. Vol. 28. 1982. pp. 588-596. [ Links ]
9. O. Redlich, J. N. S. Kwong. "On the Thermodynamics of Solutions V. An Equation-of-state. Fugacities of Gaseous Solutions". Chem. Rev. Vol. 44. 1979. pp. 223-244. [ Links ]
10. Aspen Properties. Aspen Physical Property System: Physical Property Methods. Electrolyte NRTL Activity Coefficient Model. Version number 2006.5. Aspen Technology Inc. Cambridge. M. A. USA. 2007. [ Links ]
11. Aspen Plus. Aspen RadFrac Module. offline help. Version 2006.5. Aspen Technology Inc. Cambridge. M. A. USA. 2007. [ Links ]
12. Aspen RateSep. Aspen RateSep Calculations. Online help. Complement of RadFrac module. Aspen Technology Inc. Cambridge. M. A. USA. 2007. [ Links ]
13. A. Kohl, R. Nielsen. Alkaline Earth Processes: Limestone/Lime Process. Gas Purification. Ed. Gulf Publishing Company. Oxford. UK. 1997. pp. 496-528. [ Links ]
14. H. Kister. Distillation Design. Ed. McGraw Hill Company. 1992. pp. 278-279. [ Links ]
(Recibido el 19 de Octubre de 2009. Aceptado el 14 de octubre del 2010)
*Autor de correspondencia: teléfono: + 57 + 4 + 219 85 35, 219 66 09, fax: + 57 + 4 + 219 10 47, correo electrónico: fbustama@udea.edu.CO. (F. Bustamante)