Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Facultad de Ingeniería Universidad de Antioquia
Print version ISSN 0120-6230
Rev.fac.ing.univ. Antioquia no.79 Medellín Apr./June 2016
https://doi.org/10.17533/udea.redin.n79a17
ARTÍCULO ORIGINAL
DOI: 10.17533/udea.redin.n79a17
Optimization for producing biodiesel from ethanol and waste frying oil with a high concentration of ester
Optimización de la producción de biodiesel con etanol y aceite residual de freír con un alto contenido de éster
Bruna Beatriz Lobo, Adriana Elaine da Costa, Carlos Alberto Klimeck Gouvêa*, Janaina Karine Andreazza, Kassim Shamil Al-Rubaie
Centro Universitário SOCIESC - UNISOCIESC. Campus Boa Vista, Rua Albano Schmidt, 3333. CEP: 89206-001. Joinville, Brazil.
* Corresponding author: Carlos Alberto Klimeck Gouvêa, e-mail: gouvea@sociesc.org.br
DOI: 10.17533/udea.redin.n79a17
ISSN 0120-6230
e-ISSN 2422-2844
(Received July 03, 2015; accepted March 03, 2016)
ABSTRACT
The usual transesterification of waste frying oil with ethanol does not give final concentration of biodiesel above 96.5%. The aim of this paper is to optimize the reaction conditions using ethanedioic acid as an activator of the carbonyl glyceryl ester. With the aid of a factorial design, the effects of the molar ratio of ethanol and oil, as well as the concentration of the catalyst sodium hydroxide were evaluated. The oil used was dehydrated and filtered, and the reactants submitted to mechanical stirring for two hours at 70° C. The data have been modeled by applying the response surface methodology, for the optimal reaction conditions. As results, the presence of ethanedioic acid drives transesterification from waste frying oil giving biodiesel with ester content over 96.5%. The response surface methodology gave the optimum conditions for the production of ethyl esters at molar ratio ethanol:oil of 6:1 and sodium hydroxide content of 1% relative to the mass of oil.
Keywords: Transesterification, recycling waste frying oils, response surface methodology
RESUMEN
Considerando que la transesterificación del aceite residual como resultado de la cocción de alimentos con la presencia de etanol no permite obtener biodiesel con concentración de etil éster superior a 96,5%, esta investigación muestra cómo se optimizaron las condiciones de la reacción con la adición de ácido oxálico. Con la ayuda de un diseño factorial fueron estudiados los efectos de la relación molar de etanol y aceite, así como la concentración del catalizador hidróxido de sodio en la presencia de ácido oxálico como activador del carbonilo en la fase aceite. El aceite usado se deshidrató y se filtró. La reacción fue realizada mediante la agitación mecánica durante un período de dos horas a 70° C. Los datos fueron modelados mediante la aplicación de la metodología de superficie de respuesta, encontrando las condiciones óptimas de la reacción. El camino propuesto demostró ser factible para la producción de biodiesel a partir de etanol y aceite residual con contenido de éster dentro de la concentración especificada por EN 14103 (superior a 96,5%). La metodología de superficie de respuesta mostró las condiciones ideales con la relación molar etanol:aceite de 6:1 y contenido de hidróxido de sodio de 1% respecto a la masa del aceite.
Palabras clave: Transesterificación, reciclaje de aceite residual de alimentos fritos, metodología de superficie de respuesta
1. Introduction
The utilization of waste frying oil (WFO) for producing biodiesel is advantageous from the environmental and economic points of view [1-6], considering that this source of raw material is less expensive, and its final disposition, when improper, constitutes an environmental problem. Regarding different types of alcohol, methanol is widely employed for this purpose, as it is more reactive, but it is toxic. Ethanol displays such advantages as produced from renewable sources and displays a lower degree of toxicity [7], and is a source of income for smallfarmers. However, it needs to be emphasized that ethanol is produced by a fermentation process as it requires the removal of water [8].
The transesterification of WFO is more difficult due to the lower degree of reactivity on account of its characteristics of residual acidity, humidity concentration, etc. This is the reason why trials with biodiesel with waste frying oil are normally, conducted by methanol, since ethanol is less reactive to alkali in the formation of alkoxide and also because ethoxide is a Lewis base weaker than metoxide [9]. An excess of alcohol is used to increase the yields of the alkyl ester, and use of ethanol as a raw material complicates the separation of glycerol from the reaction medium as its solubility is greater than methanol [10].
There are very detailed reviews showing the art standard for WFO transesterification, making clear the necessity of a methodology to drive the final methyl or ethyl ester content above 96.5% [11-18] and they confirm ester concentrations lower than 96.5% of biodiesel from waste frying oil with ethanol, the minimum concentration as established by EN 14103 [19].
Thus, it is important to carry out studies on catalysts or reagents for making it possible for WFO transesterification to reach a minimum ethyl ester concentration of 96.5%. It is possible to activate the glyceride carbonyl, in the Fischer Esterification model, through the addition of a strong acid. Considering the corrosiveness of strong mineral acids, it is seen that trials with strong organic acids can form a carbocation in the carbonyl of ester capable of facilitating the reaction of the attack by the ethoxide ion [20].
An earlier study of WFO transesterification applied 1.5 wt% of H2SO4 in a 1st step reaction to increase the ethyl ester content, but the results did not reach the EN 14103 specification [21].
Preliminary trials on the authors laboratory was conducted seeking for strong organic acids soluble in WFO. The addition of ethanedioic acid (oxalic acid) in the oily phase, in a concentration of 2% ratio of the oil mass, before the addition of ethanol and catalyst, helped the transesterification reaction to occur.
The objective of this study is to transesterify waste frying oil with ethanol to obtain an ester concentration above 96.5% and to optimize the mass ratio of sodium hydroxide and ethanol with oil carbonyl activated with 2% of ethanedioic acid. As an experimental basis, the alcohol/referential oil molar ratio will be 6:1 and, for the base, it will be 1% of the oil mass [21-31]. The experiments were performed according to factorial experimental design and the optimization of parameters using the surface response methodology, which is widely used in chemical processes, including biodiesel production [19, 25, 26, 29-31]. The evaluated results are restricted to the ethyl ester concentration. However, it is not the object of this discussion, at the moment, to evaluate other parameters as established by EN 14214.
2. Materials and methods
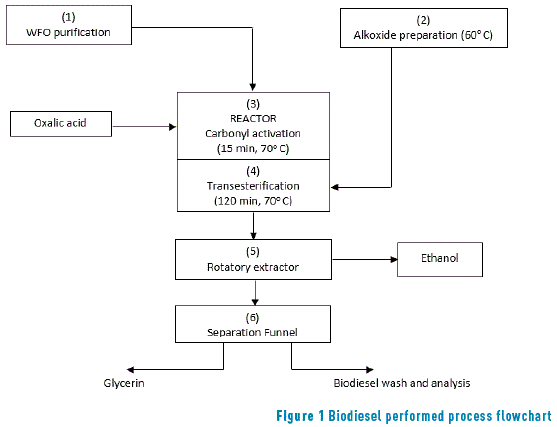
The production of biodiesel was performed in six steps: drying the WFO, production of alkoxide, activation of the used oil by the addition of ethanedioic acid, transesterification, extraction of the non-reacted alcohol, and the separation of glycerin. Figure 1 presents the flowchart of biodiesel performed process.
2.1. Preparation of the used oil
The WFO came from the university restaurant, and it is soybean oil. It was dried with magnesium sulfate anhydrous (+/- 1 g/380g of oil) (PA, VETEC) for all the tests, followed by vacuum filtration, on a paper filter with a porosity of 6.0 m. The residual acidity was equal to 1.98 mg of KOH/g (EN 14104). It was added 2 % (in relation to the oil mass) of ethanedioic acid (7.6 g) to the oil heated at 70° C and agitated for 15 min for a complete dissolution and the expected protonation of the ester carbonyls.
2.2. Alkoxide preparation
For the sodium ethoxide preparation, anhydrous ethanol (commercial grade, VETEC) and NaOH (PA, VETEC) were used. The mas of sodium hydroxide was 1% in relation to the oil mass, with variation according to factorial design. The ethanol mass variations were based on the 6:1 molar ratio, considering the soybean oil molar mass equal to 874.6 g/mol [19, 24, 31], with variations according to factorial design. The oil mass (380 g) employed in the tests was the same for all the experiments.
2.3. Transesterification
The transesterification reaction was conducted by the addition of alcohol heated to 60° C over the oil in a temperature of 70° C, to avoid reducing the temperature. The reaction was agitated by the blade agitator set at a constant speed throughout the experiment (300 rpm) for 120 minutes. In order to guarantee greater thermal balance, the system was mounted in a 1.5 liter container of water bath at 70° C. The reaction was conducted at 70° C because studies have proven that higher temperatures favor saponification [23, 29].
2.4. Extraction of Non-Reacted Ethanol and Separation of Glycerin
After the reaction is finished, the mixture was placed in a rotating evaporator to extract the non-reacted ethanol (excessive stoichiometric) and then, the mixture was placed in a separation funnel for removing the glycerin phase. The collected biodiesel underwent washing in deionized water (1:1), followed by analysis.
2.5. Analysis
The ethyl esters obtained from the transesterification reaction were analyzed by gas chromatography by the EN-14103:2003 method (Brazilian Standard for ethyl ester ABNT NBR 15342), using an equipment Varian 3400 CX instrument equipped with a capillary injection system RTX-5MS, column (15 m × 0.25 mm), coated with a 0.25 μm film, with a split ratio of 100:1 and a sample size of 1 μL. The injector temperature was 230 °C, the oven temperature was programmed from 50 °C to 250 °C, and the detector temperature was 200 °C.
2.6. Data treatment
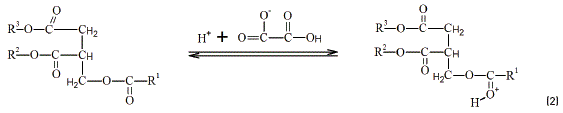
The data have been treated using Statistica software program version 10.0. The obtained data from the reaction yield was submitted to variance analysis (ANOVA) by adjusted model obtained for ethyl esters production. The response variable (%) was fitted with a full quadratic model in order to correlate the % to the operating variables. The form of the full quadratic model as a function of the more significant variables is shown in Eq. (1).
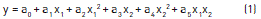
where, y: ester concentration; x1: codified ethanol mass; x2: codified NaOH mass; a0, a1, a2, a3, a4, a5: model coefficients.
In the experimental condition of highest concentration of ethyl ester was obtained a reaction without addition of oxalic acid in the oil phase was performed (experiment 12).
3. Results and discussion
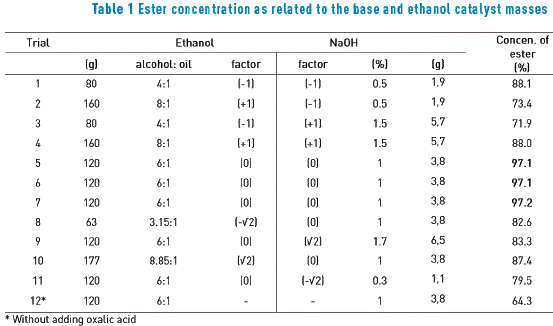
Table 1 shows the results related to the variations of sodium hydroxide (%) and ethanol (molar ratio) informing the concentrations (%) of esters produced (biodiesel).
The results from the trials 5, 6, and 7 (the triplicate point) showed a product with an ethyl ester concentration in conformity to the specification required by EN 14103, which is above 96.5%. Trial 9 showed the formation of emulsion. The ester concentration with the absence of the acidic activator (trial 12) was quite similar to the results found in literature [23].
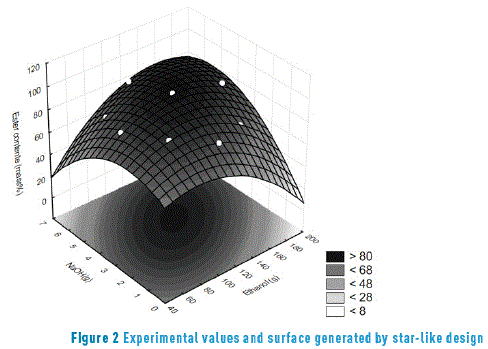
The obtained results generated a quadratic convex surface based on the mass variations of NaOH and ethanol with the localization of points relative to the ester concentrations (Figure 2).
The best experimental condition was confirmed when using 120g of ethanol and 3.8 g of NaOH, resulting in an ester concentration of 97.2%. This was in the alcohol/oil molar ratio of 6:1 as well as the base/oil percentage of 1% in relation to the oil, equivalent to the transesterification constants mentioned in literature for virgin oil [21, 23, 25, 26].
Comparing the results to the similar studies [1, 2], even in methyl transesterification, the same mass relation of NaOH and oil, as well as the molar ratio alcohol: oil were found to be ideal.
The reaction in the tested concentration, temperature, and pressure conditions, but in the absence of oxalic acid, generates only 64.3% of biodiesel. Therefore, the new fact is the use of oxalic acid as the carbolic activator, proving that its absence for used oil transesterification, in the stoichiometric ratio employed, did not produce adequate amount of ethyl ester content.
Sodium or potassium hydroxides are traditionally used as reaction catalysts, producing methoxide or ethoxide, and as they are catalysts, the base is always regenerated at the end of the reaction. Therefore, in the reaction conducted here, the generation of anion oxalate occurs, which could react with the ionized metal, forming corresponding salts. Thus, due to the consumption of the base catalyst, sodium hydroxide and oxalic acid must be considered as reagents.
To understand the role of ethanedioic acid in the reactions, some hypotheses are presented. The hydrogen ion derived from the strong acid makes a link with the carbonyl oxygen Eq. (2) in a Fischer Esterification model [20]. The electrons permits the positive charge drives to the carbonyl carbon Eq. (3). The alkoxide attacks the carbonyl carbon easily Eq. (4), followed by the formation of the respective ester Eq. (5).

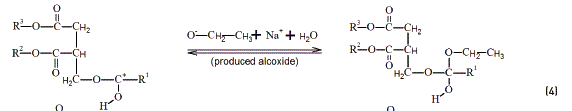
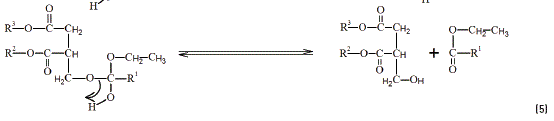
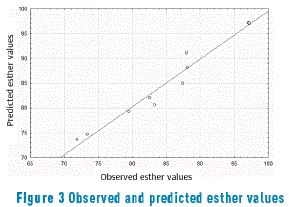
In Figure 3, a comparison between the observed and predicted values from the ANOVA model is presented, whereas it was possible to verify if there is an adjustment between the predicted model and the obtained results, as being small variations.
Figure 3 confirms the predicted model, similar to the results found in literature for oil esterification of coconut oil with methanol [29]. Applying the variance analysis (ANOVA) in star-like design through equations (2) and (3), it is possible to find the ''p'' significance levels for 95% confidence (Table 2).
The Eq. (6) shows adjusted model obtained for ethyl esters production, as a function of the most significant variables.
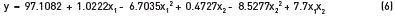
Based on the data in Table 3, the quadratic sum of the wastes is small when compared to the regression on the percentage of explained variance as 96.49%, which is high and sufficiently close to the maximum explained variance as 99.98%. The results of this study explained the variance of 96.4% between the models, as quite satisfactory for variances from the empirical equation [29-31].
The obtained statistical indexes through star-like design, applied to equation (1), displayed optimal conditions on the evaluated parameters for the production of biodiesel, as presented in Table 3.
Based on Table 3, the refinement of the quantities of ethanol and sodium hydroxide vary very little as compared to the concentrations employed in the experiment, which are compliant to what has been defined by EN 14103.
4. Conclusions
Waste frying oil (WFO) reacts with sodium etoxide, in the condition of a 1% base and the molar ratio 6:1 of ethanol and oil at 70° C for two hours, but it does not give ethyl ester content above 96,5%. However, WFO with the addition of oxalic acid (2%) reacts with sodium etoxide forming biodiesel above 96,5%, minimum specified by EN 14103. It is possible to atribute this to the ester carbonil activation by hydrogen ions, according to the Fischer Esterification model. The obtained results show that the significance level is 10-3 and the explained variance percentage is 96.49%, very near to 99.98%, which is the maximum explained variance. As these results are close to the predicted values, it is possible to confirm that the model was compliant to the concentrations values defined as ideal. The obtained statistical levels by equation (2) present values for the mass ratio that proposes to raise values that are extremely close to the experimental values. Based on the performed trials, it has been confirmed that it is possible to produce biodiesel from ethanol and WFO complying with the EN 14103, when 2% of oxalic acid is added, for reaction is conducted at 70° C for 2 hrs and 1% of sodium hydroxide.
5. References
1. Y. Zhang, M. Dubé, D. Mclean and M. Kates, ''Biodiesel production from waste cooking oil: 2. Economic assessment and sensitivity analysis'', Bioresource Technology, vol. 90, no. 3, pp. 229-240, 2003. [ Links ]
2. W. Tsai, C. Lin and C. Yeh, ''An analysis of biodiesel fuel from waste edible oil in Taiwan'', Renewable and Sustainable Energy Reviews, vol. 11, no. 5, pp. 838-857, 2007. [ Links ]
3. X. Meng, G. Chen and Y. Wang, ''Biodiesel production from waste cooking oil via alkali catalyst and its engine test'', Fuel Processing Technology, vol. 89, no. 9, pp. 851-857, 2008. [ Links ]
4. M. Gui, K. Lee and S. Bhatia, ''Feasibility of edible oil vs. non- edible oil vs. waste edible oil as biodiesel feedstock'', Energy, vol. 33, no. 11, pp. 1646-1653, 2008. [ Links ]
5. J. Marchetti, V. Miguel and A. Errazu, ''Possible methods for biodiesel production'', Renewable and Sustainable Energy Reviews, vol. 11, no. 6, pp. 1300-1311, 2007. [ Links ]
6. E. Viola et al., ''Biodiesel from fried vegetable oils via transesterification by heterogeneous Catalysis'', Catalysis Today, vol. 179, no. 1, pp. 185-190, 2012. [ Links ]
7. G. Kohlhepp, ''Análise da situação da produção de etanol e biodiesel no Brasil'', Estudos Avançados, vol. 24, no. 68, pp. 223-253, 2010. [ Links ]
8. M. Canakci, ''The potential of restaurant waste lipids as biodiesel feedstocks'', Bioresource Technology, vol. 98, no. 1, pp. 183-190, 2007. [ Links ]
9. B. Freedman, E. Pryde and T. Mounts, ''Variables affecting the yields of fatty esters from transesterified vegetable oils'', Journal of the American Oil Chemists Society, vol. 61, no. 10, pp. 1638-1643, 1984. [ Links ]
10. A. Phan and T. Phan, ''Biodiesel production from waste cooking oils'', Fuel, vol. 87, no. 17-18, pp. 3490-3496, 2008. [ Links ]
11. A. Refaat, ''Different techniques for the production of biodiesel from waste vegetable oil'', J. Environ. Sci. Tech., vol. 7, no. 1, pp. 183-213, 2010. [ Links ]
12. Z. Yaakob, M. Mohammad, M. Alherbawi, Z. Alam and K. Sopian, ''Overview of the production of biodiesel from Waste cooking oil'', Renewable and Sustainable Energy Reviews, vol. 18, pp. 184-193, 2013. [ Links ]
13. Z. Hamamre and J. Yamin, ''Parametric study of the alkali catalyzed transesterification of waste frying oil for Biodiesel production'', Energy Conversion and Management, vol. 79, pp. 246-254, 2014. [ Links ]
14. A. Kiakalaieh, N. Amin and M. Hossein, ''A review on novel processes of biodiesel production from waste cooking oil'', Applied Energy, vol. 104, pp. 683-710, 2013. [ Links ]
15. D. Leung, X. Wu and M. Leung, ''A review on biodiesel production using catalyzed transesterification'', Applied Energy, vol. 87, no. 4, pp. 1083-1095, 2010. [ Links ]
16. N. Said, F. Ani and M. Said, ''Review of the Production of Biodiesel from Waste Cooking Oil Using Solid Catalysts'', J. Mech. Engineering and Sciences, vol. 8, pp. 1302-1311, 2015. [ Links ]
17. A. Banerjee and R. Chakraborty, ''Parametric sensitivity in transesterification of waste cooking oil for biodiesel production-a review'', Resources, Conservation and Recycling, vol. 53, no. 9, pp. 490-497, 2009. [ Links ]
18. A. Abbaszaadeh, B. Ghobadian, M. Omidkhah and G. Najafi, ''Current biodiesel production technologies: a comparative review'', Energy Conversion and Management, vol. 63, pp. 138-148, 2012. [ Links ]
19. Agência Nacional do Petróleo, Gás Natural e Biocombustíveis (ANP), RESOLUÇÃO ANP N° 45, 2014. [Online]. Available: http://nxt.anp.gov.br/NXT/gateway.dll/leg/resolucoes_anp/2014/agosto/ranp%2045%20-%202014.xml?fn=document-frameset.htm$f=templates$3.0. Accessed on: Jun. 02, 2015. [ Links ]
20. N. Allinger et al., Química Orgânica, 2nd ed. Rio de Janeiro, Brazil: LTC, 1976. [ Links ]
21. M. Charoenchaitrakool and J. Thienmethangkoon, ''Statistical optimization for biodiesel production from waste frying oil through two-step catalyzed process'', Fuel Processing Technology, vol. 92, no. 1, pp. 112-118, 2011. [ Links ]
22. P. Felizardo, M. Correia, I. Raposo, J. Mendes, R. Berkemeier and J. Bordado, ''Production of biodiesel from waste frying oils'', Waste Management, vol. 26, no. 5, pp. 478-494, 2006. [ Links ]
23. J. Encinar, J. González and A. Reinares, ''Ethanolysis of used frying oil. Biodiesel preparation and characterization'', Fuel Processing Technology, vol. 88, no. 5, pp. 513-522, 2007. [ Links ]
24. S. Froehner, J. Leithold and L. Lima, ''Transesterificação de óleos vegetais: caracterização por cromatografia em camada delgada e densidade'', Quím. Nova, vol. 30, no. 8, pp. 2016-2019, 2007. [ Links ]
25. K. Tan, M. Gui, K. Lee and A. Mohamed, ''An optimized study of methanol and ethanol in supercritical alcohol technology for biodiesel production'', The Journal of Supercritical Fluids, vol. 53, no. 1-3, pp. 82-87, 2010. [ Links ]
26. M. Dorado, E. Ballesteros, M. Mittelbach and F. López, ''Kinetic parameters affecting the alkali-catalyzed transesterification process of used olive oil'', Energy Fuels, vol. 18, no. 5, pp. 1457-1462, 2004. [ Links ]
27. R. Ferrari, V. Oliveira and A. Scabio, ''Biodiesel de soja — Taxa de conversão em ésteres etílicos, caracterização físico-química e consumo em gerador de energia'', Quím. Nova, vol. 28, no. 1, pp. 19-23, 2005. [ Links ]
28. W. Clark, N. Medeiros, D. Boyd and J. Snell, ''Biodiesel transesterification kinetics monitored by pH measurement'', Bioresour Technol., vol. 136, pp. 771-774, 2013. [ Links ]
29. U. Nascimento, A. Vasconcelos, E. Azevedo and F. Silva, ''Otimização da produção de biodiesel a partir de óleo de coco babaçu com aquecimento por microondas'', Eclet. Quím., vol. 34, no. 4, pp. 37-48, 2009. [ Links ]
30. J. Leite, L. Guaraldo, R. Grimaldi, M. Soares and A. Ribeiro, ''Otimização das condições de produção de ésteres etílicos a partir de óleo de peixe com elevado teor de ácidos graxos ω-3'', Quím. Nova, vol. 29, no. 5, pp. 956-959, 2006. [ Links ]
31. G. Silva, F. Camargo and A. Ferreira, ''Application of response surface methodology for optimization of biodiesel production by transesterification of soybean oil with ethanol'', Fuel Processing Technology, vol. 92, no. 3, pp. 407-413, 2011. [ Links ]