1. Introduction
Industrial effluents commonly contain high levels of organic pollutants such as phenolic compounds, the acute toxicity of these compounds has been extensively studied, and 11 phenolic compounds have been classified as priority pollutants by the US Environmental Protection Agency (EPA). Phenolic compounds are known to be biorecalcitrants and to produce undesired organoleptic properties in water; in addition, these compounds have been associated with different illnesses such as cancer. Different authors have analyzed and employed many alternatives to remove phenolic compounds from water sources [1-6].
The phenolic compounds selected for the present study are widely employed in industry, firstly, Phenol, is used as disinfectant and in the production of polymeric resins such as Baquelite; Phenol is one of the most persistent aromatic compounds frequently found in industrial wastewater, representing a serious problem for the environment [7, 8]. In addition, during the Phenol degradation, it is possible to obtain different intermediates mainly represented by Catechol, Hydroquinone, p-Benzoquinone and short chain organic acids [9]. Hydroquinone (1,4-dihidroxibencene, HQ) and Catechol (1,2-dihidroxibencene, CT) are positional isomers of phenolic compounds, Hydroquinone is used in cosmetics, in the treatment of skin pathologies and in photography [10]. On the other hand, Catechol is an intermediary compound in pharmaceutics industry and in the production of agrochemicals; in addition, Catechol is an important precursor in the production of flavors such as Vanillin and Eugenol, which are widely employed in food industry and in the production of fragrances and cleaning products [11].
The treatment of water sources polluted with phenolic compounds has been a big technology challenge; it is because in many cases the conventional treatments are not totally efficient, for that reason in the last decades the development of alternative environmental remediation processes has fast evolution; thus, heterogeneous photocatalysis, currently represents a suitable alternative in the treatment of phenolic compounds in liquid phase. Photocatalytic treatment involves the irradiation of a solid semiconductor by UV-Visible light, as a result of this irradiation and the presence of an oxidant agent, •OH radicals are generated by redox reactions which occur on the photocatalyst surface [1-8, 12].
Titanium dioxide has been mainly employed as a photocatalyst in the photodegradation of phenolic compounds; however, Zinc oxide also has pecial attention in the last years as an alternative to TiO2, thus, ZnO has been successfully employed in the total mineralization of organic pollutants [13, 14]. The properties of these semiconductors are very similar, thus, both of them are active under UV light, the band gap value corresponds to 3.2 eV and 3.3 eV for TiO2 and ZnO, respectively. However, one of the main disadvantages of TiO2 is that this semiconductor only presents absorption in the UV region, compared with their counterpart the ZnO which presents oxygen vacancies as surface defects, these defects lead to obtain high optic transmission in the visible region of the electromagnetic spectrum [15]. It is important to take into account that ZnO is unsuitable to use as photocatalyst in reaction medium with low pH values, it is because this solid can be soluble at low pH [16].
In the present work, it was evaluated the effectiveness of ZnO photocatalyst modified by gold addition in the photodegradation of Phenol, Catechol and Hydroquinone; samples containing a mix of these compounds were also prepared and the evaluation of the reaction mechanism was also attempted.
2. Experimental section
2.1. Synthesis of Au-ZnO photocatalytic materials
Commercial Zinc oxide provided by Riedel de Haen was used as received, Au-ZnO materials were obtained by photodeposition method using tetrachloroauric acid (III) (HAuCl4) provided by Aldrich with 99.9% of purity, as gold precursor. Two series of samples with different Au content (i.e 0.5 and 2 wt.%) were prepared. Firstly, a suspension containing commercial ZnO, isopropanol (electrons donor) provided by Merck with 99.9% of purity and the metal precursor was prepared. The suspension was illuminated during 120 min under N2 atmosphere with constant stirring. An Osram ultra-vitalux lamp (300 W) was used as the light source, the illumination intensity was measured by using a Delta OHM HD 2102.1 photoradiometer, being 40 W/m2. The obtained powders (0.5 wt.% Au-ZnO and 2 wt.% Au-ZnO) were recovered by filtration, washing and drying at 120 °C for 12 hours. Commercial TiO2 P25 Evonik was used as received and employed as the reference material.
2.2 Au-ZnO photocatalysts characterization
All the photocatalytic materials were analyzed by X-ray diffraction (XRD), X-ray fluorescence (XRF), Scanning Electron Microscopy (SEM), N2 Physisorption (SBET), Fourier Transformation Infrared Spectroscopy (FTIR) and UV-Vis Diffuse Reflectance (UV-Vis DRS). The analysis conditions employed are summarized as follows:
The composition of the crystalline phases was determined by XRD, this analysis was carried out by using XPERT Pro Panalytical equipment and Cu Kα radiation, a continuous scanning was carried out with a 2θ angle between 20° and 80°, an 80 seconds step was employed.
Chemical composition determination was carried out by XRF; a Panalytical Minipal 2 equipment was employed, for these analyzes He, 20KV and 180 seconds were used as experimental parameters.
In order to observe the morphology and gold particles dispersion on ZnO surface, SEM analyzes were performed by using an EVO MA10 Zeiss microscope.
Specific surface area measurements were carried out by low-temperature N2 adsorption in a Micromeritics ASAP 2020 instrument. Degasification of the samples was performed at 150 ºC. The functional groups present in the samples were analyzed by FTIR, a Thermo Scientific Nicolet TM iSTM 50 FT-IR spectrophotometer was used, all the samples were analyzed in a wavenumber between 4000 and 1000 cm-1, for these analyzes the samples were dried at 120 °C for 12 h and then prepared in a KBr tablet.
Light absorption properties of the samples were studied by UV-Vis spectrophotometry. The UV-vis DR spectra were recorded on a UV-2600 SHIMADZU spectrophotometer equipped with an ISR-2600 integrating sphere. Band-gaps values were calculated from the corresponding Kubelka-Munk functions, F(R∞), which are proportional to the absorption of radiation by plotting (F(R∞) × hv)1/2 against hv.
2.3 Phenolic compounds photodegradation tests
Photocatalytic tests were performed by using 25 ppm solutions of Phenol, Catechol or Hydroquinone provided by Aldrich and 1 g/L of photocatalyst suspended in the liquid phase; each photoreaction was followed by a total time of 120 min under continuous stirring, 120 W/m2 of light intensity was provided by an UV-Visible Osram Ultravitalux lamp. In order to favor the adsorption-desorption equilibrium, the phenolic compounds solutions were mantained in dark during 10 min before switching on the lamp. Photolysis experiments without photocatalyst were also carried out.
The phenolic compounds identification during the photocatalytic reactions was performed by High Performance Liquid Chromatography (HPLC), a Shimadzu LC - 2030 chromatograph equipped with an UV-Vis detector and a column MACHEREY-NAGEL C18 (150 mm, 5 mm x 5 µm). The experimental conditions employed in these analyzes are described as follows: injection volume 10 µL, the mobile phase was composed by water: methanol (provided by Merck with 99.5% of purity) with a 65:35 ratio, 1.3 mL/min of flux, total run time of 5 min and 40 °C of temperature.
An Analytikjena Multi N/C 2100 equipment was employed for Total Organic Carbon (TOC) analysis, for this analysis 100, (L of filtered samples were measured, by using a furnace temperature of 750 ºC. Three measurements were performed for each sample and the average value was considered.
3. Results and discussion
3.1 Photocatalysts characterization
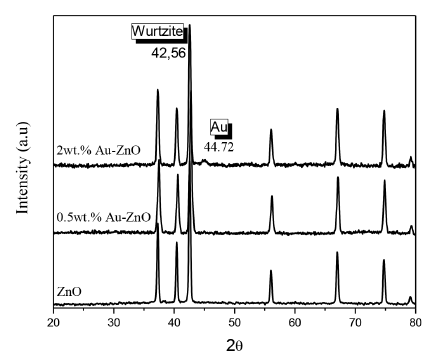
Figure 1 shows the XRD patterns for the Zinc Oxide modified photocatalysts, by this technique it was possible to identify the diffraction peaks (100), (002), (101), (102), (110), (103), (200), (112) and (201) these peaks are located in the 2 θ positions of 37.26°, 40.40°, 42.56°, 56.04°, 67.05°, 74.78° and 79.17°, respectively [17]. These peaks correspond to the hexagonal Wurtzite, the crystalline form characteristic of ZnO. It was also possible to determine that the photodeposition of 0.5 wt.% Au on ZnO surface did not modify the XRD patterns; it can be mainly due to the low content and size of the gold nanoparticles in this material; however, in the photocatalyst prepared with the highest gold content (2 wt.% Au-ZnO), it is evident the presence of a diffraction peak characteristic of the metallic gold, located at 44.72º, as it is indicated in Figure 1.
It was observed that the real gold content increases with the nominal content used in the synthesis of the photocatalysts prepared; low content of chloride remaining from the gold precursor was also detected in the samples by XRF.
In order to analyze the morphology and gold particles size in the photocatalysts prepared, SEM analyzes were carried out and a selected micrographs of 0.5 wt.% and 2 wt.% Au-ZnO photocatalysts are presented in Figure 2. As it can be observed in these images, the Au nanoparticles are heterogeneously distributed on ZnO surface. It has been also observed that when the gold content increases the Au particle size also increases, thus, in the 2 wt.% Au-ZnO photocatalyst, were observed Au particle sizes close to 100 nm.
Specific surface area was found to be 5.66, 2.89 and 3.69 m2/g for ZnO, 0.5 wt.% Au-ZnO and 2 wt.% Au-ZnO photocatalysts, respectively.
As it can be observed, the SBET value decreases after Au photodeposition, it is due to the presence of Au nanoparticles which causes obstruction of the ZnO pores; however, in the photocatalyst modified by the addition of 2 wt.% of gold, it is observed a slight increase in the specific surface area, this is due to the higher size of the Au particles present in this material compared with 0.5 wt.% Au-ZnO sample as it was observed by SEM analyzes [Figure 2], thus, the aggregation of gold particles of high size, leads to obtain the lowest number of Au particles over ZnO surface, thus leading to less surface obstruction.
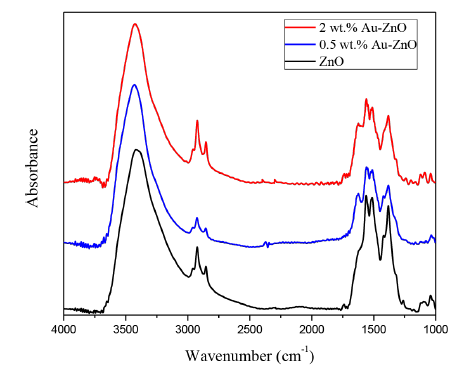
Figure 3 shows the FTIR spectra obtained for the photocatalysts analyzed, in the ZnO spectrum it is possible to identify a wide band located at 3,408 cm-1, which corresponds to hydroxyl groups; the bands observed in the region between 1,558 cm-1 and 1,386 cm-1 correspond to H-O-H flexion vibration signals, these bands indicate the presence of water linked to the ZnO structure [18, 19]. By FTIR, it was also observed that the intensity of the bands located between 2,500 and 3,000 cm-1 and between 1,000 and 2,000 cm-1 slightly decreases after gold photodeposition, it is most evident in the photocatalyst prepared with 0.5 wt.% of Au compared with the material prepared with 2 wt.%, it is mainly due to the higher number of Au nanoparticles present in the catalysts 0.5 wt.% Au-ZnO compared with their counterpart 2 wt.% Au-ZnO, thus, the gold nanoparticles can reduce the starting surface hydroxylation of ZnO, this effect is less evident when the gold content increases, it is because, the photocatalyst prepared with 2 wt.% of gold shows the highest particles size, as it can be observed by the SEM analyzes presented in the previous section [Figure 2].
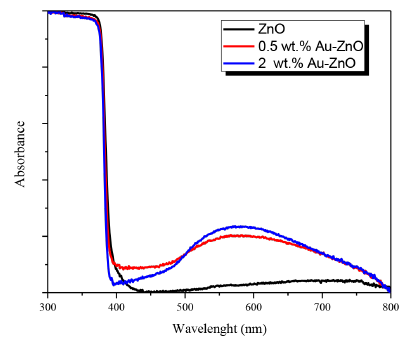
UV-Vis DR spectra for the photocatalysts analyzed are presented in Figure 4.
From UV-Vis DRS analysis, it was possible to observe that the gold addition on ZnO surface improves the absorption of this semiconductor in the visible region of the electromagnetic spectrum, thus after gold addition a high absorption was observed in the region between 400 and 800 nm, it is mainly due to the purple color of the metallized ZnO; thus in this region the gold plasmon located at 576 nm is clearly observed [Figure 4]. The highest intensity and therefore the highest absorption was observed in the band ascribed to the gold plasmon in the photocatalyst prepared with 2 wt.% of gold as it can be seen in Figure 4. The band gap values of the photocatalysts were also calculated from UV-Vis DRS analyzes, these values were found to be close to 3.3 eV, and any significant modification in the band gap value of ZnO was observed after gold addition.
3.2 Phenolic compounds photodegradation
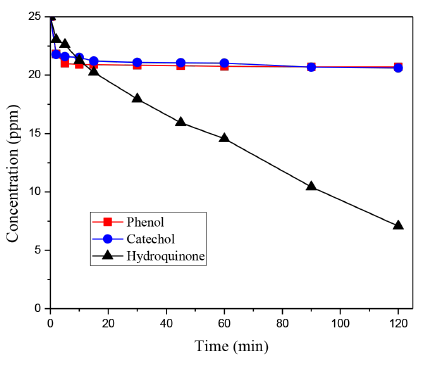
Photolysis experiments for each phenolic compound without catalyst and under UV-Visible light were carried out as the blank test; the results obtained are represented in Figure 5. As it can be seen in this figure, the photolysis phenomenon is negligible in the case of Phenol and Catechol; however, Hydroquinone is very susceptible to be degraded under illumination, thus, the Hydroquinone concentration highly decreases by photolysis from 25 to 7 ppm. Taking into account these results, it is expected that this compound presents the highest degradation rate during the photocatalytic reactions.
After photolysis tests, the effectiveness of the photocatalysts prepared was evaluated in the Phenol, Catechol and Hydroquinone photodegradation, the results obtained for each phenolic compound in these analyzes are described as follows.
Phenol photodegradation
Figure 6 shows the HPLC signals identified after five minutes of Phenol photodegradation by using ZnO as photocatalyst. In this figure, it is possible to clearly identify 4 signals, thus indicating that phenol can be transformed into three new compounds during reaction, these compounds were identified as Catechol (retention time 0.5 min), Hydroquinone (retention time 0.98 min), at the retention time of 1.26 minutes was detected an unidentified intermediate compound, this signal is probably due to the formation of Resorcinol or a short chain organic acid, as it was indicated by different authors [20-23]; finally, the phenol signal is located at 1.77 minutes.
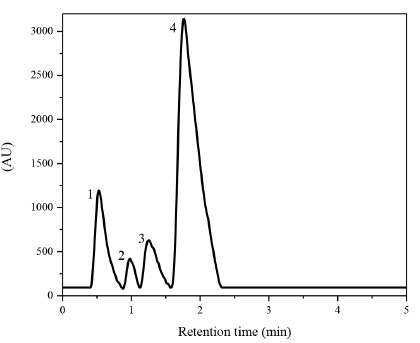
Figure 6 HPLC signals identified for Phenol photodegradation over ZnO. (1) Catechol; (2) Hydroquinone; (3) other intermediate and (4) Phenol
For comparison purposes, the evolution of phenol transformation during photocatalytic reaction over ZnO is represented in Figure 7, as it can be seen according to HPLC results, after 15 minutes of reaction only Catechol and Hydroquinone remained in the reaction medium, then, the phenol transformation continues until 120 minutes of photoreaction when the total phenolic compounds elimination was achieved; however by TOC analyzes, it was possible to determine that in fact, the total organic carbon decreases as the photoreaction time increases, but at the end of 120 min of reaction almost 10 mg/L of TOC was still detected, thus indicating that the total phenolic compounds mineralization was not achieved under the reaction conditions used in the present work. Additional analyzes lead to observe that by increasing the reaction time the TOC values significantly decreases.
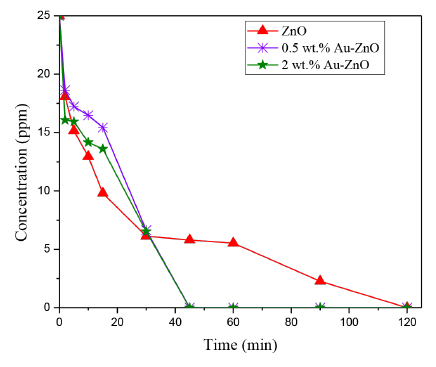
The phenol concentration was calculated in each step of reaction and the results are shown in Figure 8, this figure presents the evolution of the pollutant concentration as a function of photoreaction time over the ZnO modified photocatalysts analyzed.
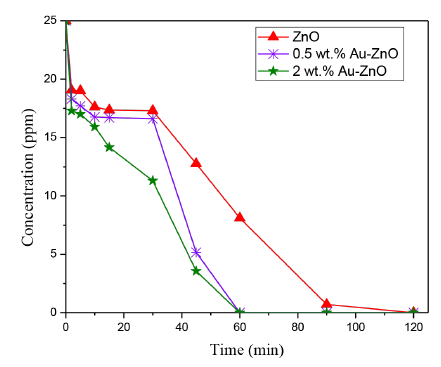
Figure 8 Evolution of the Phenol concentration after photocatalytic reaction by using the materials analyzed
As it can be observed in Figure 8, the Phenol concentration significantly decreases under UV-Visible light and in presence of a photocatalyst compared with the results observed in the photolysis tests previously presented. It is also observed that the Au addition on ZnO surface, significantly improves the photocatalytic efficiency of this semiconductor in the Phenol photodegradation; thus, the total Phenol degradation was achieved in half the time with Au-ZnO compared with bare ZnO. In 60 minutes of photoreaction, no Phenol was detected in the liquid phase by using 0.5 or 2 wt.% Au-ZnO; however, the highest reaction rate in the phenol degradation was achieved by using the photocatalyst prepared with 2 wt.% of gold (i.e 6.9x10-3 mg of Phenol. L-1. s-1).
It is widely known that the noble metal doping of ZnO leads to improve the photocatalytic efficiency of this semiconductor [13, 24], this is because the noble metal nanoparticles act as a sink for the electrons generated during the photocatalytic reaction, thus avoiding the electron - hole recombination, and therefore increasing the half life time of the photogenerated charges. In addition, the gold nanoparticles addition modifies the ZnO color; starting ZnO present a white color and after gold addition a purple color is observed, this color change leads to increase the absorption of this oxide in the visible region of the electromagnetic spectrum as it was observed by UV-Vis DRS analyzes [Figure 4].
The positive effects of the gold nanoparticles in the photocatalytic processes, above described, can explain the results observed in the phenol photodegradation over Au-ZnO materials. On the other hand, the highest photoactivity observed with the 2 wt.% Au-ZnO photocatalyst, may be due to the lower number of gold nanoparticles on ZnO surface compared with their counterpart the photocatalyst prepared with 0.5 wt.% of Au, as it can be observed by SEM analyzes [Figure 2], which led to a lower obstruction on ZnO surface as it was observed by SBET and FTIR results previously discussed.
Catechol photodegradation
By HPLC, it was possible to observe that during the photocatalytic reactions carried out by using Au-ZnO materials, Catechol is transformed only into Hydroquinone (for the sake of brevity the chromatograms obtained are not included). It was also observed that the Catechol photodegradation increases by using the photocatalyst modified by gold addition, similar to the behavior observed in the Phenol degradation tests; however, in the case of the Catechol there is not an important difference in the results obtained with the photocatalyst prepared with 0.5 or 2 wt.%, in this case total Catechol mineralization was achieved after 45 min of photocatalytic reaction as it can be seen in Figure 9.
Hydroquinone photodegradation
Hydroquinone is transformed into Catechol during the photocatalytic reactions evaluated, the evolution of this substrate concentration as a function of the reaction time is represented in Figure 10.
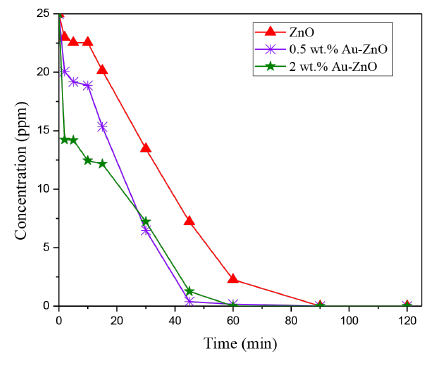
Figure 10 Evolution of the Hydroquinone concentration after photocatalytic reaction by using the materials analyzed
In this case, it is also observed that the gold addition is an important factor influencing the effectiveness of ZnO semiconductor in the photodegradation of this phenolic compound. It was found that the Hydroquinone reaction rate obtained over the 2 wt.% Au-ZnO photocatalyst is slightly higher than the obtained with the material prepared with the lowest gold content, i.e 7.13x10-3 and 7.3x10-3 mg of hydroquinone. L-1. s-1, respectively.
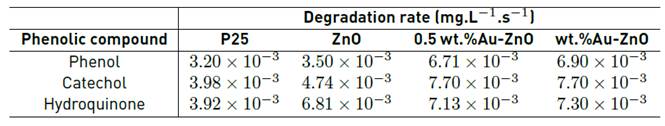
The total reaction rate during the first 60 min of illumination was calculated in each photocatalytic test and the results obtained are presented in Table 1. As it can be seen in this table, the degradation rate for all the phenolic compounds analyzed increases with the addition of gold; thus, the lowest degradation rate was obtained over ZnO and the highest value for this parameter was observed by using 2 wt.% Au-ZnO as photocatalyst. These results are in good agreement, with the results observed when the phenolic compounds concentration was calculated. It was also observed that Catechol and Hydroquinone present higher degradation rate than Phenol, it can be due to the presence of two OH- groups in these compounds, the O-H bond can be effectively broken leading to the transformation of Catechol and Hydroquinone; on the other hand, Phenol can be less reactive containing only one hydroxyl group in their molecular structure.
On the other hand, and taking into account that TiO2 has been considered as the typical reference semiconductor in photocatalysis, in this section, a commercial sample of Evonik P25 was tested in the degradation of the phenolic compounds, and it was observed that the efficiency of TiO2 in the phenolic compounds photodegradation is lower than the obtained with ZnO and Au-ZnO photocatalyst, these degradation rate results have been included in Table 1.
It general, from the results obtained it can be concluded that the mechanism through the Phenol photodegradation carried out is similar to that previously reported [20], these authors have studied the phenol degradation by TiO2, in that work HPLC and GC-MS analyzes were performed and it was found that Phenol is transformed over Titania mainly in three intermediates, i.e Catechol, Hydroquinone and Resorcinol, other compounds also were detected.
In the present work, we employ ZnO as semiconductor, and it was observed that Phenol is transformed firstly in Catechol, then, this one is transformed in Hydroquinone and then Resorcinol is formed, other intermediates were not detected in the liquid phase during 120 min of photoreaction. It is possible that Resorcinol can be finally mineralized leading to CO2 and H2O formation.
It is also possible that similar to the reported by TiO2 also in the presence of ZnO, the mechanism of the Phenol photodegradation takes place by •OH radicals attack to phenyl ring, yielding Catechol, Resorcinol and Hydroquinone.
In the same vein, It is also important to note that Resorcinol was only detected during the Phenol photodegradation, in the case of Catechol or Hydroquinone degradation over ZnO, this intermediate was not detected by HPLC. It was also observed that gold addition did not modify the proposal Phenol degradation route, in the reactions carried out by using Au-ZnO photocatalysts Resorcinol was not detected. On the other hand, during the Hydroquinone degradation over Au-ZnO only Catechol was detected; on the contrary, in the case of Catechol degradation, the presence of gold in the photocatalyst leads to the formation of two other intermediates different from Hydroquinone.
On the other hand, when Titania was employed as photocatalyst, the formation of five intermediates during the Phenol photodegradation was detected. Thus, in this case, it is possible that after phenolic compounds formation, the phenyl rings in these compounds break up to give short chain organic acids such as malonic acid, maleic, oxalic, acetic, formic, and finally CO2, as it was previously reported [20, 22, 23].
Phenolic compounds mix
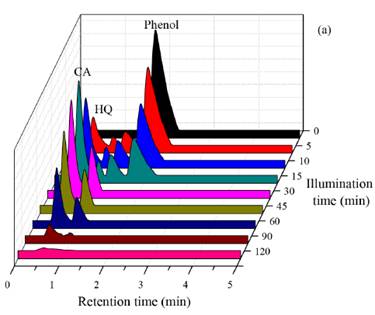
In order to simulate a real industrial effluent, a 25 ppm solution containing the three phenolic compounds (Phenol, Catechol and Hydroquinone) was prepared. Taking into account that the best photocatalytic performance in the individual phenolic compound degradation was observed by using 2 wt.% Au-ZnO as photocatalyst, this material was selected to test its effectiveness in the treatment of this solution. The results obtained are represented in Figure 11.
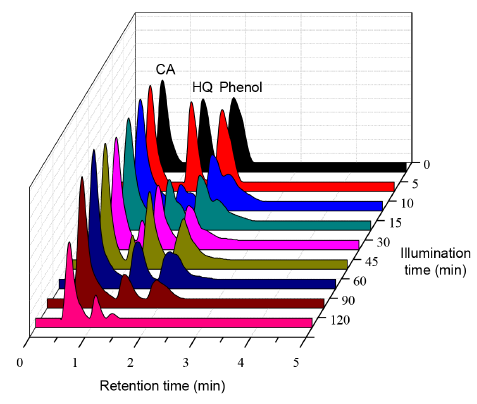
Figure 11 HPLC chromatograms for phenolic compounds mix photodegradation by using 2 wt.% Au-ZnO as photocatalyst
As it can be seen in Figure 11, after 10 minutes of illumination time, both, Phenol and hydroquinone start to be transformed in other compounds, which are evident due to the formation of two new signals in the chromatogram. In the case of the mix phenolic compounds photodegradation, it is observed that the effectiveness of the photocatalytic process is lower than the obtained in the tests performed with the phenolic substrates individually, thus, the total mineralization of these compounds was not achieved even after 120 min of photocatalytic treatment. This behavior is due to the simultaneous presence of three pollutants which can be transformed into others at the same time; in addition, these phenolic compounds can compete with each other by the hydroxyl radicals during the photocatalytic reaction, as it can be also explained by other authors [25]; this is one of the main challenge to deal when the photocatalytic processes are employed at big scale in the treatment of real wastewater polluted simultaneously with different organic and inorganic compounds.
4. Conclusions
ZnO semiconductor is an efficient material in the Phenol, Catechol and Hydroquinone photodegradation, this material has demonstrated to be more effective than commercial TiO2 P25 in the treatment of these compounds in liquid phase.
Gold photodeposition is a suitable method to improve the ZnO photoactivity in the phenolic compounds degradation. The addition of this metal modifies the optical properties of this semiconductor mainly due to the color changes; it was also observed that metal loading is an important factor influencing the effectiveness of ZnO, thus, the highest degradation rate of phenolic compounds was achieved by using the Au-ZnO photocatalyst prepared with 2 wt.% of gold content.
Phenol photodegradation mechanism depends on the catalyst employed in the photocatalytic reaction. Thus, over ZnO Phenol is mainly transformed in Catechol, Hydroquinone and Resorcinol, then the total mineralization of this compound is achieved. The Phenol degradation over TiO2 takes place in a longer reaction route by the formation of five compounds intermediaries before total mineralization.
Gold addition on ZnO did not modify the Phenol or Hydroquinone degradation mechanism observed with bare ZnO. In the case of Catechol degradation over Au-ZnO, the presence of gold leads to the formation of two intermediates different from Hydroquinone.