Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
CES Medicina
Print version ISSN 0120-8705
CES Med. vol.30 no.1 Medellín Jan./June 2016
Reporte de caso
Sedation at the end of life
Sedación al final de la vida
Viviana Calderón Flórez1, Manuel E. Machado-Duque1, Jorge E. Machado-Alba1
1Grupo de investigación en Farmacoepidemiología y Farmacovigilancia. Universidad Tecnológica de Pereira-Audifarma S.A. Pereira, Colombia.
Forma de citar: Rada-Ortega C, Pérez Toro LM, Echeverri Saldarriaga s, Reyes Márquez DC. Sedation at the end of life. Rev CES Med 2016. 30(1): 46-54.
Recibido en:Â mayo 30 de 2015. Revisado en: diciembre 17 de 2015.. Aceptado en: febrero 24 de 2016.
Abstract
Life expectancy has increased; consequently, the incidence of chronic illness has risen. Oncologic patients may present in their final days of life severe symptoms refractory to treatment, such as delirium, dyspnea, psychological stress and existential suffering. Sedation at the end of life is a strategy for the relief of those symptoms, highlighting that the intention is not to shorten life. We describe a pharmacological strategy for end-of-life sedation, along with ethics and legal aspects.
Keywords: Terminal care, Palliative care, Euthanasia, Neoplasm, Deep sedation.
Resumen
La expectativa de vida se ha incrementado, y con ella, la incidencia de enfermedades crónicas terminales. En el grupo de los pacientes oncológicos existe un subgrupo que se encuentra en sus últimos días de vida, cursando con síntomas severos y refractarios a los tratamientos. En esto pacientes la sedación al final de la vida constituye una estrategia para el alivio de sus síntomas, resaltando que en ningún momento se tiene como objetivo acelerar el proceso de muerte. Se presenta una estrategia farmacológica de sedación al final de la vida, junto con algunos aspectos éticos.
Palabras clave: Cuidado terminal, Cuidado paliativo, Eutanasia, Neoplasia.
Introduction
The concept of sedation at the end of life has gained importance during the last years due to the population aging and the increased incidence of terminal chronic diseases (1). This has taken specialized physicians to treat patients who are facing their last days of life; patients who have experienced several physical and psychological suffering which relief becomes impossible with targeted therapy. For this purpose, there is a medical practice involving the patient’s sedation to relieve the symptoms resistant to conventional treatment, but there is not a consensus in relation to definition, indications or treatment, which becomes a difficulty to prepare protocols for palliative care programs (2). The need for implementing a program to regulate sedation at the end of people’s life has gained importance in Colombia since the entry into force of Law 1733 of 2014 that states the following:"…to decree the right of people experimenting terminal, chronic, degenerative, and irreversible diseases to palliative care, as an attempt to improve the standard of living of them and their families through an integral pain therapy and the relief of suffering and other symptoms, taking into consideration relevant psycho-pathological, physical, emotional, social, and spiritual aspects according to the clinical practice guidelines set by the Ministry of Health and Social Protection for each pathology. To further decree their right to voluntarily and in advance reject unnecessary medical care that is not in compliance with the therapeutic proportionality and fails to represent a dignified life for the patients, especially in cases where a terminal chronic, degenerative, and irreversible disease with a significant impact on living standards has been diagnosed" (3). From the entry into force of law above, there is an interest for creating programs and guides to set sedations procedures in patients with terminal diseases who request such procedures; for this reason, professionals and specialists in palliative care and pain management areas must be entirely trained and knowledgeable about this option. This article is a description of the main aspects alleged for sedation at the end of people’s life.
Definition
Concepts have evolved throughout history: The first person to introduce the term was Enck in 1991 (4). By 1998-1999, this number of behaviors was known as “terminal sedation.” The term, however, was seen as improper since it suggested an intervention performed with the purpose of “terminating” the patient’s life (5).
Some other definitions involved life expectancy and treated the patient as a person facing an imminent death (a patient with two or less life expectancy weeks); since 2007, most authors have been using the term “palliative sedation” (6).
According to the WHO, palliative therapy is intended to provide relief of pain and other disturbing symptoms, and not to accelerate or postpone death; palliative therapy integrates psychology and spiritual aspects of care and offers a supporting system to help patients live as actively as possible (7).
It could be affirmed then that this is the medical procedure most frequently used to relieve severe refractory symptoms of patients with terminal diseases whose symptoms cannot be treated with targeted therapy (8); this is then a final measure to be used at the end of people’s life.
The methodology for this kind of procedures involves administration of sedatives that decrease consequences or even take patients to an unconscious state (9,10). This will depend upon the patient’s needs, and the procedure can be performed from a slight sedation to a moderate-severe sedation, also known as deep terminal sedation (a continued and sustained sedation until death), which is the most controversial procedure when trying to make it different from euthanasia (6,11).
Epidemiology
In Colombia, no data related to this topic is available. In a survey administered to 89 health practitioners from several countries in South America and the Caribbean, it was found that 71% was properly trained in palliative therapy and 94% agreed to use sedation; 53% agreed to use sedation regardless of the patient’s life expectancy; and only 1.2% affirmed that they would do it only if the patient’s life expectancy was above two weeks. Finally, 68% of respondents affirmed agreed to use sedation based on the opinion of a multidisciplinary team (12).
In a systematic revision, it was first found that in patients with advanced cancer a delirium condition was the most common indication of sedation (30%); psychological stress and suffering was the second indication (19%), followed by dyspnea (14%), vomiting (7%), and pain (3%) (13).
Indications
Sedation at the end of life has been indicated for patients with advanced or incurable severe diseases, intended to relieve symptoms refractory to other ways of treatment (14). A symptom is deemed as “refractory” when it cannot be relieved and the option is to reduce consciousness to avoid the patient’s perception of the symptom.
The symptoms that more frequently become refractory are: delirium, dyspnea, psychological stress, and existential suffering; this latter symptom requires an exhaustive evaluation of the patient before any pharmacological management attempt, not only at a physical level but also at an emotional level so that the patient can be given some other choices such as spiritual accompaniment and psychological support (8).
Worthy of attention is the fact that the option of sedation at the end of a person’s life due to spiritual symptoms such as loss of interest for life, fear to death or wish to control the moment of the disease has been taken into consideration. It has been found that this kind of symptoms are sometimes more relevant than physical manifestations and account for 13% of patients facing the last stage of their lives (12).
Duration
Sedation at the end of life is administered during the last days of life. When the beginning of the sedation process is determined, there are reported durations from sedation to death ranging from 0.8 to 2.7 days (6,13). In Colombia, a series reported a range from 1 to 35 days (15).
It is very important to note that this treatment is not intended to reduce the patient’s life expectancy and that onset of treatment does not change the time the person would stay alive, since the treatment is only intended to relieve symptoms (16).
Prior considerations
Before starting the sedation process, the informed consent process should be completed with both patient and family; they should be explained risks and benefits and the patient’s diagnosis should be evaluated. An evaluation should also be performed to different treatments administered in the past and the patient’s response to such procedures; prognosis, survival forecast, intended sedation depth, and the possibility of discontinuing sedation are also aspects to be taken into consideration.
The patient should be clearly explained the alterations he/she may experience concerning consciousness, mental activity, communication, and the potential risk of avoiding food consumption. The patient should also be explained the adverse effects of some drugs, such as paradoxical reaction, late or improper relief or the possibility that death process can be accelerated for aspiration or over-sedation (17).
Drugs
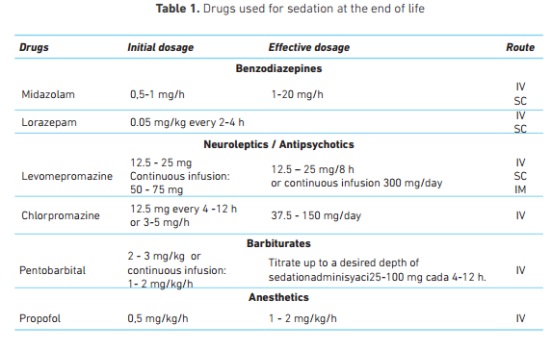
The most frequently used drug is Midazolam, with a dosage ranging from 30 to 120 mg/day. This is the drug of choice among benzodiazepines since it exhibits a short half-life and a rapid onset of action (6).
Other choices such as Levomepromazine and Chlorpromazine are specifically useful when patients show delirium symptoms, since the use of benzodiazepines could result in worse symptoms. Phenobarbital and Propofol could also be administered since they are useful drugs with a patient suffers seizures not responding to treatment (15).
Patients can be receiving opioids as an analgesic treatment or as a therapy for dyspnea; these drugs can produce sedation as a side effect but they should never be used for this only purpose (18). Table 1 below shows the drugs with their dosages and routes of administration.
Monitoring
Monitoring should be intended to achieve the patient’s comfort and as a tool for having a proper control of symptoms until eliminating suffering, avoiding the emergence of side effects (5). Depth of sedation will depend on clinical condition, refractory symptoms, and individual response. Communication allows evaluating the patient since it is the only method to determine whether or not the symptoms have actually improved.
There are several scales to determine the patients’ level of sedation in different scenarios. Maybe the most practical scales (for their easy use for the evaluator and their validity) are RASS (Richmond Agitation-Sedation Scale) and KNMG (Royal Dutch Medical Association Sedation Score) (19).
The objectives of sedation are different for each individual. Hence, initial titration is required with incremental dosages until reaching a suitable level of symptom relief; then, dosage should be adjusted to keep the effect. The route of administration may vary from a continuous infusion to intermittent bolus. The possibility of using rescue bolus in case of worsening of symptoms should be made available (15).
The dosage can be adjusted to reduce the sedation degree, either for unwanted side effects or as a way to reevaluate the patient and allow an interaction with his family, but only if previously agreed in the informed consent (6).
Those who support deep sedation have affirmed that relief of suffering since the very beginning and on a continuous basis should be assured (6). Symptoms such as dyspnea, seizures, and delirium have been described as deep sedation criteria and sometimes the patient’s awakening can become an agitated event and family can be affected (20).
Once dosage has been titrated until reaching the clinical objective, monitoring will depend on the patient’s condition; those with imminence of death do not require routine monitoring of vital signs. Deterioration of breathing does not necessarily result in decreased sedation dosage; it can be part of the natural death process (14).
Impact on survival
One of the most important and controversial points in sedation at the end of life in terms of ethics is the impact it has on survival. The opponents have affirmed that this is a form of euthanasia, especially when nutrition is suspended and hydration is an artificial process. However, those who defend this practice have affirmed that patients with terminal diseases stop eating and drinking during the last days of their life as part of this natural process (4).
For this reason, some authors have affirmed that these two topics should be treated independently and make a joint decision with patient and family. These authors have also stated that starting artificial nutrition and hydration in a patient who is dying (whose natural process makes him stop eating and drinking) would not be an ethical action since this would only extend the patient’s suffering (21).
Survival is not affected when sedation is performed in a safe manner; then, it is of great importance to implement strategies and protocols to provide relief to patients and avoid risks. Morita et al. have studied sedation safety at the end of life and have found a high percentage of complications; they have concluded that sedation should be managed in an organized and safe manner. They have also reported a survival below three weeks in 94% of patients subject to sedation, with an average survival of six days (22).
Another study has not found any differences in the survival of patients who were subject to sedation at the end of their life at home, compared to those who were not subject to sedation (23). Vitetta et al. have not found any differences in survival of sedated and non-sedated patients; they have not found any association between survival and the therapeutic plan chosen by the caregiving physicians (24).
In some countries where euthanasia has been legally authorized, the thin line between sedation at the end of life and the acceleration of death has been found. In a study performed in Flanders (Belgium) at a nursing home where this kind of sedation is a normal practice, a survey was administered to physicians who participated in this type of procedures and it has been found that a third of them affirmed that they partially and expressly intend to shorten life (25).
The Dutch National Guidelines has reported that sedation at the end of life, when administered in a suitable manner and according to set protocols, does not accelerate death process (26). Therefore, evidences suggest that sedation at the end of life, in most cases, does not reduce survival and that the underlying illness is the key factor taking to death.
Sedation and euthanasia: ethical aspects
When physicians face the clinical decision of starting sedation at the end of life, they are actually challenged by ethical conflicts. There are still some debates about the sedation practice at the end of life and how it is seen as a way of euthanasia; the poor clarity on this topic is clearly seen. Sedation is then not a very frequently offered option since it is very frequently seen as a way of euthanasia (27).
This ethical tension emerges from the thin line between both procedures. It is very important then to give clear definitions, so that both procedures can be clearly explained to patients and their families. A clear example of the importance of giving a definition is the name coined by Billing and Block (“slow euthanasia”), since this term caused confusion and fear; the term was finally excluded from literature (28).
Euthanasia is the end of life intentionally and expressly requested by own patients, while sedation is an alternative to relieve the patient’s refractory symptoms; the objective of sedation is not death (29). It should be noted that in Colombia (see Judgment C239 of 1993 pronounced by the Constitutional Court), euthanasia practice has been decriminalized, and the new coined term is “mercy homicide” (30).
When speaking about differences between both procedures, the following should be taken into consideration: intention, action and results.
Intention of euthanasia implies the elimination of the patient’s suffering through death; the intention of sedation implies relief of the patient’s refractory symptoms by decreasing the patient’s consciousness; decease is the result of the illness and not the main intention (31).
WWith respect to euthanasia, the physician, by request of the patient, chooses medication and lethal dosages that drive to a cardiopulmonary arrest and the intervention is then declared as a successful procedure. Concerning sedation at the end of life, the physician is intended to relieve refractory symptoms by choosing medication on an individual basis and under supervision. This kind of sedation does not shorten survival and if the patient dies as a result of the sedation, the case is deemed as an adverse event (4,6).
Very frequently during this kind of discussions the concept of “natural death” emerges, and this has resulted in different ethical debates. Opponents have affirmed that there is a direct intervention with natural death. However, Raus et al. have stated that the concern of physicians, families, and patients should be based on the search for a “good death,” regardless of its natural or artificial cause, and that this concept may vary in a significant manner among different individuals; for this reason, patients and their families should always be involved in the discussion (32).
Conclusion
Sedation at the end of life, administered to a terminal patient, becomes a strategy to relieve symptoms which have refractoriness to the standar medical management.
It is important to implement and standardize some guidelines for the management of patients with terminal diseases. The development of programs concerning sedation at the end of life in our country should be a priority, taking into account the increase of terminal chronic diseases and the regulations governing the palliative care services.
References
1. Otero A. Departmental differences in the causes of death in Colombia. Cartagena; Publishing of the Bank of the Republic, may 30 of 2013. Document 187. [ Links ]
2. Ted C. Braun, Neil A. Hagen, and Trish Clark. Development of a clinical practice guideline for palliative sedation. J Palliat Med. June 2003, 6(3): 345-350. link [ Links ]
3. Law 1733 of September 8 of 2014, Palliative care. Colombian Official Diary, September 8 of 2014. [ Links ]
4. Ten Have H, Welie JVM. Palliative Sedation Versus Euthanasia: An Ethical Assessment. J Pain Symptom Manage. 2014 Jan;47 (1):123-36. link [ Links ]
5. Hasselaar JG, Verhagen SC, Vissers KC. When cancer symptoms cannot be controlled: the role of palliative sedation: Curr Opin Support Palliat Care. 2009 Mar;3(1):14-23. link [ Links ]
6. Maltoni M, Scarpi E, Nanni O. Palliative sedation in end-of-life care: Curr Opin Oncol. 2013 Jul; 25(4):360-7. link [ Links ]
7. Cancer: Palliative care. World Health Organization (WHO), 2015.http://www.who.int/cancer/palliative/es/ (Last access February 15 of 2015) [ Links ]
8. Maltoni M, Scarpi E, Nanni O. Palliative sedation for intolerable suffering: Curr Opin Oncol. 2014 Jul;26(4):389-94. link [ Links ]
9. Cherny NI, Portenoy RK. Sedation in the management of refractory symptoms: guidelines for evaluation and treatment. J Palliat Care. 1994;10(2):31-8. link [ Links ]
10. Graeff AD, Dean M. Palliative Sedation Therapy in the last weeks of life: A literature review and recommendations for standards. J Palliat Med. 2007 Feb;10(1):67-85. link [ Links ]
11. Van Norman GA. Physician aid-in-dying: cautionary words. Curr Opin Anaesthesiol. 2014 Apr;27(2):177-82. link [ Links ]
12. Moyano J, Zambrano S, Ceballos C, Santacruz CM, Guerrero C. Palliative sedation in Latin America: survey on practices and attitudes. Support Care Cancer. 2008 May;16(5):431-5. link [ Links ]
13. Maltoni M, Scarpi E, Rosati M, Derni S, Fabbri L, Martini F, et al. Palliative sedation in end-of-life care and survival: A systematic review. J Clin Oncol. 2012 Apr 20;30(12):1378-83. link [ Links ]
14. Cherny NI, on behalf of the ESMO Guidelines Working Group. ESMO Clinical Practice Guidelines for the management of refractory symptoms at the end of life and the use of palliative sedation. Ann Oncol. 2014 Sep 1;25(suppl 3):143-52. link [ Links ]
15. Támara LM .Descriptive cohort trial of terminal patients in medical home care in Bogotá - 2008-2012. Rev Col Anest.2014;42:100-6. link [ Links ]
16. Jansen LA. Disambiguating clinical Intentions: The ethics of palliative sedation. J Med Philos. 2010 Feb 1;35(1):19-31. link [ Links ]
17. Cherny NI, on behalf of The Board of the European Association for Palliative Care. European Association for Palliative Care recommended framework for the use of sedation in palliative care. Palliative Medicine 23(7) 581-593. link [ Links ]
18. Mercadante S, Giarratano A. The anesthesiologist and end-of-life care: Curr Opin Anaesthesiol. 2012 Jun;25(3):371-5. link [ Links ]
19. Arevalo JJ, Brinkkemper T, van der Heide A, Rietjens JA, Ribbe M, Deliens L, et al. Palliative Sedation: Reliability and Validity of Sedation Scales. J Pain Symptom Manage. 2012 Nov;44(5):704-14. link [ Links ]
20. Swart SJ, van der Heide A, van Zuylen L, Perez RSGM, Zuurmond WWA, van der Maas PJ, et al. Considerations of physicians about the depth of palliative sedation at the end of life. Can Med Assoc J. 2012 Apr 17;184(7):E360-6. link [ Links ]
21. Schildmann J, Schildmann E. Clinical and ethical challenges of palliative sedation therapy. The need for clear guidance and professional competencies. Int J Clin Pract. 2013 Nov;67(11):1086-8. link [ Links ]
22. Claessens P, Menten J, Schotsmans P, Broeckaert B. Palliative Sedation: A Review of the Research Literature. J Pain Symptom Manage. 2008 Sep;36(3):310-33. link [ Links ]
23. Alonso-Babarro A, Varela-Cerdeira M, Torres-Vigil I, Rodriguez-Barrientos R, Bruera E. At-home palliative sedation for end-of-life cancer patients. Palliat Med. 2010 Jul 1;24(5):486-92. link [ Links ]
24. Vitetta L, Kenner D, Sali A. Sedation and analgesia- prescribing patterns in terminally ill patients at the end of life. Am J Hosp Pall Care 2005;22: 465 - 473. link [ Links ]
25. Rys S, Deschepper R, Mortier F, Deliens L, Bilsen J. Continuous sedation until death with or without the intention to hasten death-A Nationwide Study in Nursing Homes in Flanders, Belgium. J Am Med Dir Assoc. 2014 Aug;15(8):570-5. link [ Links ]
26. Swart SJ, Rietjens JAC, van Zuylen L, Zuurmond WWA, Perez RSGM, van der Maas PJ, et al. Continuous palliative sedation for cancer and noncancer patients. J Pain Symptom Manage. 2012 Feb;43(2):172-81. link [ Links ]
27. Papavasiliou E, Payne S, Brearley S. Current debates on end-of-life sedation: an international expert elicitation study. Support Care Cancer. 2014 Aug;22(8):2141-9.link [ Links ]
28. Papavasiliou ES, Brearley SG, Seymour JE, Brown J, Payne SA. From sedation to continuous sedation until death: how has the conceptual basis of sedation in end-of-Life care changed over time? J Pain Symptom Manage. 2013 Nov;46(5):691-706. link [ Links ]
29. Manzini JL. Palliative sedation: ethical perspectives from Latin America in comparison with European recommendations. Curr Opin Support Palliat Care. 2011 Sep;5(3):279-84. link [ Links ]
30. Judgement C239 of 1997. Colombian Constitutional Court. Magistrate Carlos Gaviria Diaz. link [ Links ]
31. Papavasiliou E, Chambaere K, Deliens L, Brearley S, Payne S, Rietjens J, et al.Physician-reported practices on continuous deep sedation until death: A descriptive and comparative study. Palliat Med. 2014 Jun 1;28(6):491-500. link [ Links ]
32. Ruas, K., Sterckx, S. and Mortier, F. Continuous deep sedation at the end of the life and the "Natural death" hypothesis. Bioethics, 26: 329-336. link [ Links ]