Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Acta Neurológica Colombiana
Print version ISSN 0120-8748
Acta Neurol Colomb. vol.31 no.4 Bogotá Oct. 2015
Trabajo original
Cutis verticis gyrata en población masculina institucionalizada con enfermedad mental crónica en colombia. Serie de casos
Cutis verticis gyrata in adult male institutionalized population in colombia. Case series.
José Fernando Hernández (1), Lina María Espejo Jiménez (2), Ruth Liliana Correa Díaz (2), Ricardo de la Espriella (3).
(1) Neurologist, Nuestra Señora de La Paz Clinic, Bogotá, Colombia.
(2) Psychiatrist, Nuestra Señora de La Paz Clinic, Bogotá, Colombia.
(3) Psychiatrist, MSc Clinical Epidemiology, Professor School of Medicine Javeriana University, Bogotá, Colombia.
Recibido: 19/05/15. Aceptado: 13/10/15.
Correspondencia: Ricardo de la Espriella: rde@javeriana.edu.co
Resumen
El artículo describe casos de cutis verticis gyrata (CVG) encontrados institución de protección en Sibaté, Colombia. Se trata de una serie de casos de 65 hombres adultos; se describen las variables sociodemográficas y clínicas. Se encuentra en la institución una prevalencia de 5,9%, mucho más alta que lo esperada en la población general (1:100.000 hombres). El promedio de edad fue de 38,5 años, la mayoría analfabetas (98,46%). El diagnostic más frecuente fue de retard mental profundo en 59 casos (90,77%), con alta comorbiidad multiaxial. La presencia de surcos en cuero cabelludo tuvo un rango de 1 a 16. La edad de inicio y el tipo de CVG no pudieron ser establecidos.
Se trata de la mayor serie de casos reportada en una sola institución. Existe necesidad de estudios posteriores para determinar el tipo, origen y evolución del hallazgo.
Palabras clave: Enfermedad mental, Retardo mental, Malformación cutánea (DECS).
Summary
To describe Cutis Verticis gyrate (CVG) in cases found in an institutionalized people facility in Colombia. Case series with 65 male adult in-patients, describing socio-demographic and clinical variables. Prevalence of 5.9 % was higher than expected in the general population (1:100.000 in males). Mean age: 38.5 years, most illiterate (98.46%). The most frequent diagnosis was profound mental retardation in 59 (90.77 %) cases, with high multi-axis comorbidity. Folds range from 1 to 16. Age of onset and type of CVG couldn't be established.
This is the largest case series reported from a single institution. There is need of further study to determine type, origin, and evolution of the sign.
Key words: Mental illness, Mental retardation, Cutaneous malformation (MeSH).
Introduction
The unusual high presence of folds in scalp observed in male patients from a protection facility in Sibaté (Colombia), led the authors to investigate the diagnosis initially, and then to establish some characteristics of the population. This malformation is very rare and poorly studied worldwide, there are no previous data in Colombia.
Cutis Verticis Gyrata (CVG) was described for the first time in 1843 by Robert (quoted by Akesson and Diven et al), as a physical sign characterized by the presence of scalp circumvolutions resembling cerebral cortex (1, 2). It has been estimated that it has a worldwide prevalence of 1 in 100,000 men and 0.26 in 100,000 women (3). It is considered that the CVG is the manifestation of several comorbid conditions; based on etiology, two groups have been described: primary and secondary causes (3). These clinical signs have been described with different names, including cutis sulcata, bulldog scalp, and cutis capitus strata (4).
While Albert was the first author who mentioned this malformation, Robert described it in 1843, then Auvert and Devergie (1854) extended the description (quoted by Akesson). Poggi (1884) was the first to describe a case in a demented patient; in 1890 it was noted in 3 criminals in Italy; in 1893 were reported cases of CVG in patients with mental retardation, microcephaly and epilepsy; Moller in 1903 described the first CVG case associated with nevus. Only until 1906, CVG called the attention of the German Dermatological Association through Jadassohn who reported 4 cases published in the journal "Transactions of the German Society of Dermatology" (McDowall in 5). Unna introduced the term Gyrata verticis Cutis in 1907 (2, 4); from this time to the present date case reports have been described, such as Adrian Foster's 1919, Desana Peruccio's 1938 38 cases; in 1939 Rudberg described a case with findings in abdominal skin and scalp (5).
In 1953 Simon Polan and Thomas Buttenworth reviewed 275 cases of patients with cutis verticis gyrata, they only found enough information in 195 patients. They found that 84.8% of cases were men, and 15.2% were women (ratio 5.6:1) (5), they classified this pathology as primary or secondary. 47.7% of patients had primary CVG and 52.6% secondary (5). In most cases they were unaware of the start time. In 65 patients it was determined that the age of onset was in childhood. In 25% of men it existed from birth. In 87.5% of men and 88.2% of women it started before the end of the third decade of life. Its occurrence was not limited by any particular racial or geographic factor. The cases were reported among people of Europe, North America, Central America, South America, China, Korea and Japan. While there were only two cases in black patients, it was concluded that the presence of grooves in scalp of Africans and Melanesians was a common feature. There was an incidence of family involvement in 17 of the 275 cases of which were described 2 brothers "idiots" with microcephaly and two other brothers who previously had scalp eczema (5).
CVG was described for the first time in women by Anderson in 1928, in a 43 year old woman, with no physical abnormalities, with a biopsy that reported nonspecific hypertrophy of the dermis (1, 3). In 1964, Akesson reported that from 38 patients in Sweden he found only one woman, Larsen and Birchal presented 3 cases, with a woman in New Zealand (3, 6).
In world literature there have only been reported a few cases of essential primary CVG adult men without ophthalmological or neurological abnormalities, all possible causes of secondary CVG were discarded (7 ,8 )
Literature Review
The scalp of people with mental retardation has been investigated extensively, but without much success in finding differences compared with no retarded people (9-11). Furthermore, there are a large number of findings on the skin of patients with mental retardation, of which the CVG is only one possibility.
McDowall and Fisher in 1893 were the first authors to notice the relationship between CVG and mental retardation, describing the disorder in two mentally retarded men, with epilepsy and hemiparesis (12). That year Cowan reported two additional cases, which had as common findings strabismus, and microcephaly (5).
In 1913 Fiocco reported a case of CVG with mental retardation and cretinism. Three years later Ganter reported two new cases, both mentally retarded with microcephaly; one with epilepsy had strabismus and the other had rotatory nystagmus (5).
In 1918 Galant reported two cases of CVG in men, one in a 53-year old male with adipose genital dystrophy and acromegaly; the other one in a 65 years old male (5).
In 1929 Truffi reported five cases in patients with mental retardation, one woman with severe mental retardation with microcephaly and gait disturbance and three men with severe mental retardation, two with epilepsy and one with microcephaly. The fifth had microcephaly (5).
In 1933 Hellerstrom CVG described a case of a 20 year old man with mental retardation, history of acanthosis nigra, diabetes; the disorder disappeared after castration (5). Miescher found a similar case in a man with normal intelligence (13).
In 1938 Touraine and Gole described the case of two men with mental retardation, CVG possibly due to infection; there were no associated neurological disorders (5).
In 1940 Radner reported a case of CVG in a 27 years old man with mental retardation, epilepsy, microcephaly, congenital nystagmus, strabismus and bilateral keratoconus (5).
In 1953 Polan and Butterworth made a report of six cases of men with mental retardation and CVG, two of them with a diagnosis of epilepsy, one with spastic dysplasia, one with congenital cataract; imaging reports showed enlargement of the sella. Symptoms appeared after puberty (5).
In 1958 Kratter reported three cases in men with microcephaly, two with spastic diplegia. In 1959 Saetra reported a case of CVG, unspecified endocrinopathy, mental retardation, and schizophrenia, with infantile genitalia (1).
In 1962 Berg and Windrath-Scott described a man with mental retardation, CVG, and quadriplegia with onset at puberty (1).
In 1964 Hans Olof Akesson reported a series of 47 cases of patients with mental retardation and CVG in special institutions in Switzerland, 46 of them were men and only one woman. A genetic study was conducted, finding that chromatin 6 and the genetic study of parents were normal. Parents had similar age at birth of patients. 8% of patients had parents with first-degree consanguinity (cousins). Parents morbidity did not differ with respect to the general population. A genetic condition was suspected, they reviewed various modes of transmission, but they could not determine any genetic etiology, which led them to believe that they were environmental (3).
In 1996 Striano et al reported a case of a male patient with CVG, mental retardation and epilepsy resistant to medication. Genetic studies showed increased number of ruptures in 3p14 and 16q23. They proposed the hypothesis of a critical period in the neurodevelopmental brain abnormality which arises and develops CVG (14).
In 2001 Mégabarné et al reported the case of two brothers with microcephaly, CVG, retinitis pigmentosa, cataracts, sensorineural deafness and mental retardation (13).
In 2001 Filosto et al made a case report of a patient with mental retardation, Lennox-Gastaut syndrome, and CVG. Hormone levels were reported as normal, MRI showed atrophic changes, CVG etiology remained unknown (15).
In 2002 Akai et al reported a CVG case of genetic origin, with gene fibroblast receptor 2 mutation, expressed at birth as Beare-Stevenson Syndrome. CVG was confirmed by DNA analysis. At birth, patients have ptosis and ocular protrusion, forehead nevus, and presence of umbilical stump. This patient developed craniosynostosis, facial dysmorphism and hydrocephalus. There are few cases reported of Beare-Stevenson Syndrome, four with genetic mutation (16, 17).
A review of 1100 institutionalized patients in La Colonia, Sibaté (Colombia) in a protection facility for men with chronic mental illness, actively seeking the presence of cutaneous malformation, 65 of them presented CVG. The demographic, sociocultural, and clinical characteristics, and pharmacological treatment and psychiatric diagnosis are described, using multiaxial classification (DSM-IV TR). Description of features of the folds is done, looking for etiology and comorbidities.
Type of study: Case series
Study Population: Male institutionalized patients in special center La Colonia - Sibaté, with CVG.
Study variables: variables studied were age, gender, age of onset, number of folds, psychiatric diagnosis, medications received, clinical progress over time, associated diseases.
Data collection: Permission was obtained from the Beneficencia of Cundinamarca, organization responsible for the protection of the patients who were studied, review of medical records, family interviews were conducted when possible. We included men with chronic mental disorder with physical signs present. Analysis: The data were organized into frequency distributions, including absolute and relative frequencies to be presented in tables and graphs. Central tendency and dispersion measures of quantitative variables were obtained.
Ethical Aspects: The protocol was approved by the Bioethics Committee of Nuestra Señora de La Paz Clinic, as well as access to medical records, photographies, publication of material was approved by the organization and the staff responsible for the protection and care of them. Identity of the participants is kept in reserve by assigning a random number to data analysis. Under Colombian research norm (Resolution 1993 008 430 Article 11) investigation is considered with no risk.
Results
From 1100 institutionalized patients in the facility, were obtained 65 men cases, mean age 38.5 years, range 22 to 72,64 (98.46%), reported no schooling and only one referred schooling. 86% of patients were from Bogota and Cundinamarca zone (Central Colombia area).
The most frequent diagnosis in the medical record was behavioral disorder in 30 cases (46.15%), followed by impulse control disorder and schizophrenia; Axis I diagnosis was not stated in 26 cases (40%); there were a total of 54 different diagnoses, some patients having one or two diagnoses.
On axis II only one patient (1.5%) had no diagnosis at the time of data collection, appearing in the category of deferred diagnosis, 64 patients were diagnosed with unspecified Mental Retardation, in 1 case (1.5%), moderate mental retardation in 4 cases (6.15%), and severe-profound in 59 cases (90.77%)
Axis III had 55 different diagnoses, and 1 patient (1.5%) had no diagnosis, a case was deferred, the range in this category was between 0 and 5 diagnostics.
The most frequent diagnosis was symptomatic epilepsy in 28 cases (43%), together with 5 cases (7.69%) in total, 33 patients were diagnosed with epilepsy of diverse origin.
Language disorder was the next category with 18 cases (27.7%), chronic gastritis in 10 cases (15.38%), spastic quadriplegia in 8 cases (12.3%) and dyslipidemia in 8 cases (12.3%)
In the medical record family abandonment was stated in 19 cases (29.23%), partial abandonment in 1 case (1.5%), without a social support network 7 cases (10.77%), low support network in 20 cases (30,8%), proper network in 16 cases (24.61%), not recorded in two cases (3%).
Medication: Failed to establish a history of exposure to various substances, there was only information of the present medication. Range: 0-6 medications. As for medication received at time of consignment data shows that only 2 patients had no medication at all (3%), 7 cases were not receiving psychiatric medication (10.8%)
The most prescribed drug in these patients, carbamazepine, was followed by haloperidol and clonazepam.
As anticonvulsants, the more frequent was carbamazepine in 28 cases (43.1%), followed by valproic acid in 15 cases (23.1%), clonazepam in 14 cases (21.54%), phenobarbital in 12 cases (18 , 46%) and phenytoin in 6 cases (9.23%).
Antipsychotics, the most used was haloperidol in 24 cases (36.92%), followed by levomepromazine in 19 cases (29.23%), clozapine in 17 cases (26.15%), pipotiazine in 7 cases (10.77 %) some patients received two antipsychotics.
Antidepressants: Fluoxetine 7 patients (10.77%), trazodone 2 (3%) and amitriptyline 1 (1.53%).
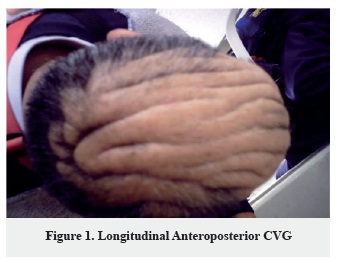
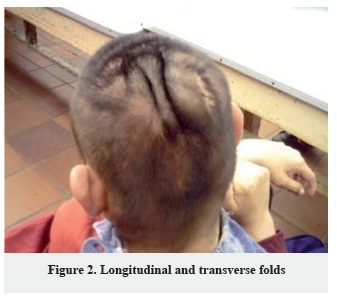
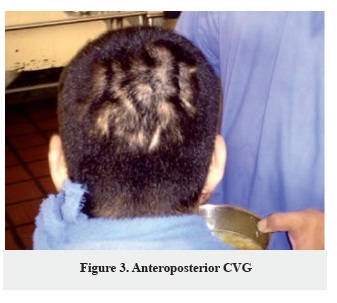
Number of folds: The number of folds has wide distribution of 1-16 scalp folds, only in one patient the number of folds was not established (Figure 1, 2, 3).
Discussion
CVG etiological classification has been described as primary and secondary, but because it is a description, this division has been deemed unnecessary and confusing by some authors (2).
The primary or ‘true' CVG is essentially characterized by normal histopathology (6). The term primary essential CVG has been proposed for those cases where there are no other abnormalities and nonessential primary cases associated with mental retardation, cerebral palsy, epilepsy, schizophrenia, cranial abnormalities (microcephaly), deafness, eye abnormalities (cataracts, strabismus, blindness, retinitis pigmentosa) or the combination of these (4, 18). CVG is considered to affect 0.2% - 5% of patients with mental retardation (1, 3) and patients with cerebral palsy, eye abnormalities, seizures, microcephaly and schizophrenia (3).
The secondary etiology is due to underlying diseases, with no mental deficiency. Causes are: Inflammation (Eczema, psoriasis, folliculitis, impetigo, erysipelas, pemphigus), Nevus, hamartomas (fibroids, neurofibroma, can be present at birth, and can be seen in women), tumors (advanced stages of lung cancer) Acromegaly (19, 20), myxedema, posttraumatic, idiopathic hypertrophic osteopathy (pachydermoperiostosis), amyloidosis, syphilis, leukemia, tuberous Sclerosis, catatonic Schizophrenia, and Ehlers Danlos (3, 9, 10, 21, 22). It has been reported a case of pseudoacromegalia by the use of minoxidil, with scalp and facial alterations CVG type (23), breast cancer (24) and foreign body granulomata (19). The histopathology is altered by neoplastic or inflammatory basis (6).
The term ‘latent' or ‘potential' CVG has been proposed to describe scalp folds by lateral pressure exerted inwardly, in previous normal scalp (Merenlender quoted by 2).
Pathophysiology
Primary Form: Folds are symmetrical, with normal histology or hypertrophy and hyperplasia of connective tissue or epidermal appendages (25); it develops mainly in puberty.
In the primary essential form the cause is unknown, although it has been seen in sporadic cases, autosomal recessive and dominant factors involved, usually occurs after puberty, in men only (3).
In the primary form could be involved nonessential endocrine disorders, it occurs mainly in men, appearing before puberty. It has been reported as due to the increasing use of peripheral testosterone, by measuring the level of free testosterone, which is found lower in patients with CVG compared with controls, possibly linked to X chromosome anomalies, fragility sites in chromosomes 9, 10 and 12 and the rupture of 3p14 and 16q23 bands (3).
Secondary form: scalp folds are predominantly asymmetric, with abnormal histology, and develop at any age (except associated with nevus, which are usually congenital) (3). The pathology of the process depends on the basic cause (e.g. , inflammatory, neoplastic). Lymphedema has been postulated as the cause of CVG in patients with Turner syndrome (4, 26, 27) There have been reported cases of Klinefelter syndrome with CVG, the suggested mechanism does not include lymphedema (28).
In children CVG cases are rare; however, primary non-essential form is associated with genetic syndromes and familial cases (29).
CVG familial form usually occurs in patients with normal intelligence, associated with idiopathic hypertrophic osteoarthropathy and pachydermoperiostosis with autosomal dominant variable expression (3, 22). This disease usually occurs in men after puberty. In complete form it can be found as follows: fingers in hands and feet widened and elongated, arms and legs with cylindrical appearance, gross facial traits, skin wrinkles and hypertrophy causing facial expression of anxiety. In the extremities can be found ossification of long bones periosteum and to a lesser extent in the phalanges, metacarpals and metatarsals, these changes are not seen in the sella (21).
Prevalence: In the United States the estimated prevalence of CVG is 0.5% in patients with mental retardation. In Scotland and Switzerland is found a prevalence of 0.71 - 3.4%. In Italy male population prevalence is higher than 14, 3% (21).
The only work that has given the global prevalence was conducted by Akesson in 1964 and was 1 per 100,000 men and 0.26 per 100,000 women (1, 21) The prevalence in male patients with mental retardation is higher in institutionalized population from 0.21 to 2.2% (1, 19, 21, 30). In Colombia has not been reported prevalence of CVG, only case reports (31).
Course: is chronic and progressive.
Mortality: mortality rate is related to the development of melanoma associated with congenital melanocytic nevi (3). Akesson described in 1964 that mortality in people with mental retardation in Switzerland is 30% compared with the general population, and those with skin verticis gyrata mortality is 10% higher than the control group. Life expectancy was 38.1 and death is usually due to infectious diseases, and pneumonia (1, 21).
Race: there are no conclusive studies, there has been reported an increased ratio in Italian patients, possibly attributed to high prevalence of Sicilian descent, and it has been reported also in black people (21).
Gender: male to female prevalence is 5-6:1. In women are reported fewer cases, possibly due to difficulties in detention for long hair (21, 32, 33)
Age: in most cases the onset of the CVG is before puberty, 90% of cases develop before age 30, in the case of intracerebral cerebriform nevus it can be seen at birth (21).
In people with mental retardation and CVG determining the age of onset is virtually impossible, the onset is usually insidious and observed by others. CVG syndrome and mental retardation occurs in patients with IQ below 35 (12, 21).
The secondary CVG can occur at any age depending on the underlying disease, even from birth, in the case of cerebriform intradermal nevus (21).
Genetics: CVG is familiar only when part of an apparent dominant disorder linked to sex, primary paquidermoperiosotosis (4, 34).
Clinical Presentation: usually the scalp surface is not smooth and regular, high folds are observed among which there are folds and wrinkles. The skin turns thick gradually, bending and forming grooves (35).
Primary form only compromises the scalp, the circumvolutions are generally symmetrical, fluffy, and not corrected by pressure or traction, they are usually asymmetric in the secondary forms (4).
The involved area can vary from a small limited location to the entire scalp. The areas most commonly affected are the vertex and occiput, but not uncommon containment to one of the temples, occasionally located in the frontal region. Pleats vary from 2 to 12 or 13, including a thickening of the scalp from 0.75 to 4 cm. Folds are arranged generally in an anterior-posterior and parallel form, and sometimes can be present a union between them or radiate a central common point (4).
In severe cases there have been counted 15 or more convolutions, with 10 -15 cm length and gap between them of from 0.5 to 1 cm. (21).
In most cases hair is described as dark. Usually texture, distribution and amount of hair is not affected, although in some patients hair is thick and dry. Hair tends to disappear in the gyrus, but remains in the folds, infection and maceration of the grooves can occur. In occasional cases, especially in patients with acromegaly, they are accompanied by fold prominences and sometimes vertical transverse forehead folds which exaggerate facial features (5). The occurrence of lesions in the case in primary CVG occurs in puberty, they are located at the vertex and occipital region. In the secondary, the development of the convolutions can be present from birth (3).
There are usually no symptoms, although there may be pain in the case of secondary CVG nevus and intracranial traction alopecia (11).
Primary form only compromises the scalp, the convolutions are generally symmetrical, fluffy, and not corrected by pressure or traction, are usually asymmetric secondary. In cases of CVG nevus or tumors due to their distribution on the scalp, is not usually bilateral and symmetrical. The proportion affected is well demarcated by the surrounding scalp and furrows and folds have cerebriform appearance. The progression of changes is visible (3, 5).
Complementary studies: in cases of primary CVG, laboratories are not required; some patients may find low levels of free testosterone. In secondary cases laboratories depend on the presentation and associated underlying disease (21).
Imaging: MRI or CT in cases of CVG in the newborn or associated with MRI, or ophthalmological or neurological abnormalities shown:
- CAT Scan: Thickening of the skin and subcutaneous tissue distribution and irregular skin furrows, changes like atrophy cortico - subcortical and ventricular dilatation, abnormal calcifications in the brain, bone abnormalities or intracranial tumors, thickening and widening celar intracelar mass may be present in patients with CVG (2, 14, 32).
- In the NMR: abnormalities in the occipital lobe, bilateral polimicrography, small frontal and temporal lobes, atrophy of the frontal and parietal cortex, colpocephaly, hypoplasia of the splenium at corpus callosum, atrophy of the cerebral cortex (2).
In the primary essential CVG, MRI may show increased slight thickening of the dermis and subcutaneous tissue (3, 21).
Diagnostic imaging helps classify the CVG and ensure the absence of different associated conditions (36).
Other studies: always applying biopsy is suggested in patients with CVG in order to determine the etiology, in women with or without lymphedema CVG peripheral, perform genetic testing to rule out Turner syndrome; EEG is recommended in patients with mental retardation and CVG, some show slow waves, with high points in the occipital region, and spike-waves complexes in occipital region (21).
Histological changes: there is normal histology in most cases of patients with primary CVG, in some cases thickening collagen, hypertrophy of the pilosebaceous structures, multiple sweat glands and ducts can be found. In the secondary form histopathology findings depend on the underlying disease (21).
Complications: is estimated that 4% of patients with CVG and cerebriform intradermal melanocytic nevi CVG have malignant transformation. In the secondary form outcome depends on the underlying disease (2)
Treatment: There are several treatment modalities that have been investigated for the management of primary and secondary forms of CVG, including observation combined with local skin care, antihistamines, topical antiseptics, steroids, thyroid removal, radiation therapy, sleep, psychotherapy; to date none of them has been proved successful (3). Surgical removal of the skin has been used successfully (4).
In cases of secondary CVG, treating the underlying condition can help correct deformities of the scalp (4).
Hellestrom described a case of CVG, Acanthosis Nigricans and diabetes mellitus, where the finding (CVG) disappeared after castration, the same happened in the case presented by Yamagata where a in a man of 47 years with normal intelligence who had CGV from age 20, a week after castration it was shown that wrinkles began to spread and six months later disappeared (1, 21).
Akesson tried to test the effect of estrogen orally in two patients giving 1 mg of ethinyl estradiol per day: one patient received the medication for two weeks, then with a monthly interval for 4 months and then biweekly for 1 month, the second patient received medication daily for 2 weeks and then weekly for 5 months, none of them reported improvement (21).
Serfling described a case of acromegaly and CVG, wherein findings disappeared when eosinophilia was extracted in the pituitary (3).
Foersters and Winders treated a CVG case with pituitary irradiation, without any effect (3).
Gronberg tried CVG with thyroid medication, without any improvement (21).
CVG surgery depends on the size, location, underlying disease, and patient's preferences (1, 37). This study is the first report of case series of CVG in Colombia, we described cases of male patients with chronic mental illness in one protection facility, describing demographic characteristics of the folds and data from medical records. Genetic studies or complementary examinations were not performed.
It was difficult to establish the age of onset of the scalp folds, as well as previous medications, due to patients characteristics and lack of a family support network. No cases had been detected previously by medical records.
The origin of the patients is mainly in the area of Bogota and Cundinamarca, with few cases in other parts of Colombia.
Patients in the sample had no education (only reported case studies), the average age was 38.5 years, with a diagnosis of mental retardation in 98% of them (axis II) with multiaxial comorbidity: on axis I disorder behavior in 30%, schizophrenia in 11% and impulse control disorder in 11% of cases, on axis III neurological diseases, 47% with epilepsy, and language disorders in 27 % of cases.
On axis IV were documented social problems, as expected for protected population.
Regarding axis V (GAF measuring overall operating range) 100% of the patients were under 30, showing great interference of symptoms.
Regarding drug therapy, we found that 2 patients received no medication, and 7 did not receive any psychiatric medication. The most commonly used drug was carbamazepine in 43.1% of patients, followed by valproic acid in 23.1% and 18.4% phenobarbital, in the group of the most commonly used benzodiazepine clonazepam was at 21.5%. These medications are used both for their anticonvulsant and anxiolytic modulation or affection.
In typical antipsychotics, haloperidol was the most frequently used (36.9%), followed by levomepromazine (29.2%) and within the atypical antipsychotics, clozapine (26.1%) of the depot antipsychotic pipotiazine in 10.7%.
In the group, the most commonly used antidepressant was fluoxetine (10.8%).
It was noted that there are patients with more than one drug in the same group (antipsychotics, benzodiazepines) anticonvulsant medication combinations and frequent use of phenobarbital which should also be included in future studies to expand possible etiologic factors. It should be noted that medication is a possible confounder. In the study it could not be found if there is greater exposure to drugs association, more folds, or some specific distribution.
This is the only study that attempts to link neuropsychiatric drug taking and the appearance of the sign, without concluding that this is a key factor in its appearance.
As in many other studies, we could not determine the age of onset of the sign, the presence of folds in scalp don´t appear in any of the medical records studied.
In the sample studied were present from 1 to 16 folds, more than reported in the literature (1-14) (4), there was no uniform pattern of distribution of the folds
Many cases suggest the possibility of secondary CVG, supplementary examinations should be required in order to classify properly the cases.
We were unable to verify cases of consanguinity in patients in the sample.
None of the cases presented were associated with pain.
Conclusions and Recommendations
This is the first case series of CVG in Colombia, which reported the second highest number in the world after that of Polan and Buttenworth and more from a single institution.
In a population sample of 1100 male institutionalized patients, were detected 65 cases of CVG, showing a clear increase in the expected prevalence. The prevalence of CVG for The Colony Sibaté institution was 5.91%, higher than that reported in other series.
The cases presented correspond to 29.55% of that expected for men in the whole country, calculated on the basis of 1 per 100,000 inhabitants and a population base of 44 million people in the country.
It would be interesting to study the evolution of the cases, with additional studies that include genetics and pathology in order to diagnose the specific type of CVG.
Additionally there is the possibility of a multicenter study and search for cases in other regions of Colombia in similar institutions, trying to determine if there are possible environmental factors, comorbidity, exposure to drugs, as well as the prevalence in women.
Acknowledgements:
Saint John of God Order, Colombia.
La Colonia Center, Sibaté, Colombia.
Declaration of Conflicts of Interest: The study did not have any grant; authors have no conflicts of interest with the pharmaceutical industry.
Reference list
1. Akesson HO. Cutis verticis gyrata and mental deficiency in Sweden. I Acta Med. Scand. 1964:115-128. 175, 1. [ Links ]
2. Diven D, Tanus T, Raimer SS. Cutis Verticis Gyrata. International Journal of Dermatology, October 1991, 30, No 10 710-12. [ Links ]
3. Akesson HO. Cutis verticis gyrata and mental deficiency in Sweden. II. Genetic aspects. Acta Med. Scand. 1965:459-464. [ Links ]
4. Garden J, Robinson JK. Essential primary Cutis Verticis Gyrata. Treatment with the scalp reduction procedure. Arch Dermatol. 120 Nov 1984. [ Links ]
5. Polan S, Buttenwort T, 1953 Am J Ment Defic. Cutis verticis gyrata; a review with report of seven new cases. 1953 Apr; 57(4):613-31. [ Links ]
6. Rosso Schons KR, Costa Beber AA. Cutis Verticis Gyrata. N Engl J Med 2012; 367:e23 October 18, 2012. [ Links ]
7. Cribier B, LipsKer D, Mutter D, Grosshands E. Cutis verticis gyrata: reduction surgical treatment. Ann Dermal Verereol 1993; 120:542-5. [ Links ]
8. Schepis C, Romano C. Cutaneous findings in the mentally retarded. Review. International Journal of Dermatology, 35:5, may 1996. [ Links ]
9. Schepis C. Siragusa M. Primary Cutis verticis gyrata or Pachydermia verticis gyrata: A Peculiar Scalp Disorder of Mentally Retarded Adult Males. Dermatology. 191. 4, 1995. [ Links ]
10. David TJ, Osborne CM, Research Article. Scalp hair patterns in mental subnormality. J Med Genet 1976; 13:123-126. [ Links ]
11. Tani, T; Miyamoto, Y; and Kurozumi, N. Surgical treatment of cutis verticis gyrata: A report of two cases. Br. J. Plas. Surg. 30:235, 1997. [ Links ]
12. Mégarbané A, Waked N, Chouery E, Moglabey YB, Saliba N, Mornet E, Serre JL, Slim R. Mycrocefaly, CVG of the scalp, retinitis pigmentosa, cataracts, sensorioneural deafness, mental retardation in 2 brothers. Am J Med Genet 2001:98:244-9. [ Links ]
13. Striano S, Ruosi P, Guzzeta V, Perone L, Manto A, Cirillo S. Mental Deficiency Syndrome: A Patient with Drug-Resistant Epilepsy and Polymicrogyria Epilepsia, 37(3):284-286, 1996. [ Links ]
14. Filosto M, Tonin P, Vattemi G, Bongiovanni LG, Rizzuto, N, Tomelleri, G. Cutis verticis gyrata, mental retardation and Lennox-Gastaut síndrome. Neurol Sci (2001) 22: 253-256. [ Links ]
15. Bryan D. Hall, Ronald G. Cadle, Mahin Golabi, Colleen A. Morris, Dr. M. Michael Cohen Jr. Beare-Stevenson cutis gyrata syndrome. American Journal of Medical Genetics. 44:1 (82-89), 1 September 1992. [ Links ]
16. Akai T, Lizuka H, Kishibe M, Kawakami S, Kobayashi A, Ozawa T; A case of Beare-Stevenson Cutis gyrata syndrome confirmed by mutation analysis of the fibroblast growth factor receptor 2 gene. Pediatric Neurosurgery; aug 2002. [ Links ]
17. Larsen F, Birchall N; Cutis verticis gyrata: three cases with different etiologies that demonstrate the classification system. Australasian Journal of Dermatology (2007) 48, 91-94. [ Links ]
18. Skibinska http://temedicine.medscape.com/article/1113735-treatment#a1128. [ Links ]
19. Thomson J, MacGillivray RC. Cutis Verticis Gyrata with foreign body granulomata. British Journal of Dermatology. 81: 10 (766-768), October 1969. [ Links ]
20. Al-Bedais M, Sultan AK. Acromegaly presenting as cutis verticis gyrata. International journal of Dermatology 2008, 47, 164. [ Links ]
21. Malgorzata D Skibinska M., Janniger C., Cutis Verticis gyrata. Emedicine from Wed. MED, Jan 23 2007. [ Links ]
22. Rimoin DL. Pachydermoperiostosis (idiopathic clubbing and periosis): Genetic and physiologic considerations. N. Engl. J. Med. 272; 923, 1965. [ Links ]
23. Nguyen, KH, Marks JG. Pseudoacromegaly induced by the long-term use of minoxidil. J Am Acad Dermatol 2003; 48: 692-5. [ Links ]
24. Rahman A, Mahmood A. Cutis Verticis Gyrata Secondary to Infiltrating Ductal Carcinoma Breast. Journal of the College of Physicians and Surgeons Pakistan 2012, 22 (2): 120-122. [ Links ]
25. Holbrook, Karen A, Wolff, K. Estructura y desarrollo de la piel en Fitzpatrick, T. Dermatología en medicina general, 3a ed. Editorial Panamericana, Bogotá, 1988. [ Links ]
26. Larralde M, Gardner SS, Torrado M, Fernhoff P, Santos Muñoz A, Spraker M, Sybert V. Lymphedema as a postulated cause of Cutis Verticis Gyrata in Turner Syndrome. Pediatric Dermatology. 15, 1 18-22 1998. [ Links ]
27. Parolin Marinoni L, Taniguchi K, Giraldi S, Carlvalho VO, Furucho M, Bertogna J. Cutis verticis gyrate in a child with Turner Syndrome. Pediatr Dermatol 16: 242-243, 1999. [ Links ]
28. Keller K, Williams C, Seagle B. Klinefelter syndrome and Cutis Verticis Gyrata. American Journal of medical genetics 103:249-251 (2001). [ Links ]
29. Hsieh Hl, Fisher DE, Bronson DM, Fretzin DF. Cutis verticis gyrata en neonatos. Pediatr Dermatol 1983; 1983:153-6. [ Links ]
30. Palo J, Iivanainen M, Blomqvist K, Pesonen S. Etiological aspects of the Cutis Verticis Gyrata and Mental Retardation Syndrome. Journal of Intellectual Disability Research. 14, 1: (33-43), March 1970. [ Links ]
31. Hurtado PM, Pachajoa H. Imágenes en Medicina. Cutis verticis gyrata. MEDICINA (Buenos Aires) 2012; 72: 132. [ Links ]
32. Tan O, Eegen, D. Primary essential Cutis Verticis Gyrata in an adult female patient: a case report. The journal of dermatology, 33, 7: (492-495), july, 2006. [ Links ]
33. Tani, T; Miyamoto, Y; and Kurozumi, N. Surgical treatment of cutis verticis gyrata: A report of two cases. Br. J. Plas. Surg. 30:235, 1997. [ Links ]
34. Casals J., Chinelli P., Piquero V. Pereira G. Fortes A. Cutis vértices gyrata como manifestación clínica de paquidermoperiostosis. Localizador Web 03- 046. [ Links ]
35. Masson P, Fayton M, Lamireau T, Lacombre D, Taieb A, et al, Unusual form of Noonan syndrome: neonatal muti-organ involvement with chilothorax and nevoid CVG. Pediatrics 1993: 48: 59-62. [ Links ]
36. Alorainy, IA. Magnetic Resonance Imaging of Cutis Verticis Gyrata. J Comput Assist Tomogr. 32, 1, January/February 2008. [ Links ]
37. Synder, M. Jhonson P. Hollins R. Congenital primary cutis verticis gyrata. Case report. Plastic & Reconstructive Surgery: September 1, 2002 - 110, 3: (818-821). [ Links ]