Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
CES Odontología
Print version ISSN 0120-971X
CES odontol. vol.27 no.2 Medellìn July/Dec. 2014
Identificación molecular y genotipificación de Streptococcus mutansde muestras de saliva de niños de Medellín, Colombia
Molecular identification and genotyping of Streptococcus mutans from saliva samples of children in Medellin, Colombia*
Mary L. Arévalo-Ruano,1 Flor Y.Canacuán-Melo,1 Julián Echeverry-Chica,1 Clara L. Salazar-González,1 Cecilia M. Martínez-Delgado,1 María C. Martínez-Pabón,2 Margarita M. Correa,3 Astrld V. Clenfuegos-Gallet3
1Grupo de Investigación en Bacterias Anaerobias y Aerobias de importancia clínica, Escuela de Microbiología, Universidad de Antloqula. Medellín-Colombia. E-mall: malu.ruano@gmail.com, anny563l@gmail.com, julianprotozoo@gmail.com, clarlin27@gmail.com, cmariamar@hotmail.com.
2Grupo POPCAD, Facultad de Odontología, Universidad de Antioquia. Medellín-Colombia. E-mail: macemapa@hotmail.com.
3Grupo de Microbiología Molecular, Escuela de Microbiología, Universidad de Antioquia. Medellín-Colombia. E-mail: mmaria.correa@udea.edu.co, vanessa.cienfuegos@gmail.com
*Trabajo participante por el área de la salud de la Universidad de Antioquia al Concurso Otto de Greiff, 2013
Forma de citar: Arévalo-Ruano ML, Canacuán-Melo MY, Echeverry-Chlca J, Salazar-González CL, Martínez-Delgado CM, Martínez-Pabón MC, Correa MM, Clenfuegos-GalletAV. Identificación molecular y genotipificación de Streptococcus mutans de muestras de saliva de niños de Medellín, Colombia. Rev CES Odont. 2014; 27(2) pág 47-60
Recibido: agosto de 2014. Aprobado: noviembre de 2014
Abstract
Introduction and objective: Tooth decay is one of the most prevalent infectious diseases of the oral cavity. The aim of this study was to determine the prevalence of Streptoccocus mutans and its genotypes in saliva samples from 6 and 7 year old children with and without dental caries.
Materials and methods: Forty seven saliva samples were obtained from 6-7 year-old children, randomly selected from a Public School in Medellin, Colombia. Oral examinations in order to determine the DMF-T Index (Decay, Missing and Filling Teeth) were carried out. Isolates were identified by the Streptoccocus sobrinus and Streptococcus mutans PCR, API 20STREP and 16S rRNA sequence analysis. Genotypes c, e, f and k were detected by PCR.
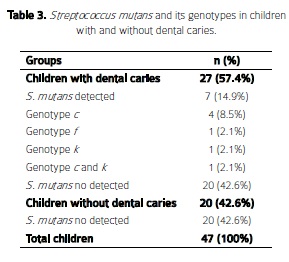
Results: Prevalence of S. mutanswas 14.9%. Of the 47 saliva samples, 57.4% (27) corresponded to children with dental caries, and 8.5% (4) were positive for S. mutans genotype c, 2.1% (1 each) genotype f genotype k, and genotype cand k, respectively.
Conclusion: Prevalence of S. mutanswas lower than in previous studies of Colombian children with similar demographic characteristics. Streptoccocusmutans'genotype c, fand kwere found in children with caries but not in the group without caries. This is the first report of S. mutans genotype kin Colombia; this genotype requires further study to clarify its relation with dental caries and cardiovascular disease in Colombia.
Key words: Streptococcus mutans, Tooth decay, Polymerase Chain Reaction, genotyping, 16S rRNA.
Resumen
Introducción y objetivo: La caries dental es una de las enfermedades infecciosas de la cavidad oral más comunes en el mundo. El objetivo de este estudio fue determinar la prevalencia de Streptococcus mutansy sus genotipos en muestras de saliva de niños de 6 y 7 años, con y sin caries dental.
Materiales y métodos: Cuarenta y siete (47) muestras de saliva fueron obtenidas de niños de 6-7 años, seleccionados aleatoriamente de una escuela pública en Medellín, Colombia. Se realizaron evaluaciones orales para determinar el índice COP-D (número de dientes permanentes cariados, obturados y perdidos). Los aislamientos fueron identificados mediante PCR para diferenciación de S. sobrinusy S. mutans, API-20STREP y análisis de ARNr 16S. Se determinaron mediante PCR los genotipos c e, fy ken las cepas identificadas como S. mutans.
Resultados: La prevalencia de S. mutansfue 14,9%. De las 47 muestras de saliva, 57,4% (27) correspondieron a niños con caries dental, y 8,5 % (4), fueron positivas para S. mutans genotipo c, 2,1 % (1 cada una) genotipo f genotipo ky genotipo cy k, respectivamente.
Conclusión: La prevalencia de S. mutansfue menor que la reportada en estudios previos de niños colombianos con características demográficas similares. Se encontró presencia de genotipos c, fy kde S. mutansen niños con caries dental, pero no en el grupo de niños sin caries dental. Este es el primer estudio en reportar la presencia del genotipo ken Colombia. Es necesario realizar más investigaciones en nuestro país para aclarar la relación de S. mutans genotipo k con caries dental y enfermedad cardiovascular.
Palabras clave: Streptococcus mutans, Caries dental, Reacción en Cadena de la Polimerasa, Genotipificación, ARNr 16S.
Introduction
Tooth decay is one of the most common diseases of the oral cavity, affecting especially children between 5 and 12 years, and it is considered the most recurrent infectious disease worldwide (1, 2); it is an infectious pathological process, multifactorial, localized, post-eruptive and transmissible, that finally destroys hard dental tissues (3). The initiation and development of this disorder is linked to the presence of abundant microorganisms which vary depending on the stage of the lesion (4). Oral bacteria colonizing teeth are organized in biofilms which exists in a dynamic balance with the organism immune defenses (5). In addition to the presence of bacterial communities associated with fermentative metabolism, an increase in dietary carbohydrates, particularly sucrose (4,5), result in further acid production that may exceed both the capacity of the saliva to remove acid end-products and the neutralizing power of the salivary/plaque buffer system, producing additional acidification of the biofilm (6).
At each stage of progression of the carious lesion, succession of different bacterial species occurs. At the onset and progression of the lesion, species belonging to the Mutans streptococci group, particularly Streptococcus mutans and Streptococcus sobrinus are the main microorganisms found, while Lactobacillus and Bifidobacterium dominate in the advanced stages of the disease (4,5). Usually, the emergence of S. mutansin the tooth cavities is followed by the presence of caries after the infective window of 6 to 24 months (6).
The important role of S. mutansin caries initiation is due to its acidogenic and aciduric potential (7, 8). In addition, S. mutansgenerate an extracellular matrix polysaccharide in dental biofilms, the exopolysaccharides. These are glucans synthesized by the products of the glucosyltransferases genes gtfB and gtfC , and provide binding sites that promote the establishment of pathogenic biofilms (9,10).
Mutans streptococci are classified serologically on the basis of the chemical composition of its exopolysaccharides, also known as cell surface serotype-specific rhamnose-glucose polymers (RGPs). Six genetic groups and seven serotypes are recognized: S mutans serotypes c, e, f k S. sobrinus serotype g S. downeiserotype g S. rattus'serotype b, S cricetusserotype a and S. macacae serotype c (isolated from monkeys) (11,12). Data regarding the distribution and prevalence of S. mutans serotypes are limited. In Japan, the prevalence of S. mutans serotypes in oral isolates, is primarily serotype c (76 %), followed by serotype e (26 %), serotype f (8 %), and serotype k(2 - 5%) (12,13). Studies on acidogenicity of S. mutans serotypes had shown it is higher in subjects with dental caries and that the cserotypes strains are the most acidogenic (14,15).
Serotype k has been found in patients with infective endocarditis, a complication that can lead to heart valves replacement (16). Importantly, the frequency of serotype k was significantly higher (75%) in samples of dental plaque from patients with infectious endocarditis compared with oral samples from healthy patients (20%), suggesting a possible association with the development of cardiovascular disease (17-20).
In Colombia, some studies have analyzed the relationship between dental caries and counts of S. mutansin children with and without dental caries; however no statistically significant differences in S. mutans counts among these groups were found (21). Thirty years ago, presence of S. mutans serotype cand f, and other serotypes dand g classified as S. sobrinus according to Coykendall were reported in school-age population in the country (22,23). Since then, there are no reports on studies evaluating prevalence of S. mutansand its serotypes in school-age population in Colombia. The aim of the present study was to determine the prevalence of Streptococcus mutans and its genotypes in saliva of children aged 6 and 7 years with and without dental caries in a public school in Medellin.
Materials and methods
Saliva sampling: One saliva sample was obtained from 47 children of ages 6 and 7, randomly selected from a public primary school of Medellin city. Each child received oral examination to determine the Decay, Missing and Filling Teeth (DMF-T index). Saliva was collected in 50 mL sterilized conical tubes, after minimum an hour of food intake for 3 minutes. Evaluated children chewed 1 gr of wax (Ivoclar Vivadent AG FL-9494, Schaan, Liechtenstein). The saliva samples were stored at 4°C and processed within the following two hours.
Bacterial cultures: Serial dilutions of the saliva samples in sterilized PBS (10-1 to 10-6) were seeded in Mitis Salivarius Agar medium (Difco Laboratorios, Detroit MI, USA), added with 0.2 U/ mL bacitracin (Sigma, St Louis, MO, USA), and subsequently incubated at 37°C, 5% CO2 for 48-72 hours. One hundred and sixteen isolates of grampositive cocci with colony morphology, positive mannitol and sorbitol fermentation compatible with S. mutans were stored at -80°C in 20% glycerol brain-heart infusion broth (Difco, Detroit, MI, USA).
PCR for differentiation of Streptoococcass sobrrinus-Streptococcus mutans: Bacterial DNA for PCR amplification was extracted using DNeasy® Blood 8 Tissue kit following the manufacturer's instructions (DNeasy® Blood 8 Tissue Kit, Qiagen, Germantown, USA). Differentiation between S. sobrinus and S. mutans was performed using a multiplex PCR (24). Two primer pairs amplified gtf l and gttB genes for S. sobrnnus and S. mutans, respectively (Table 1). Each PCR mixture contained 16 mM (NH4)2SO4, 67 mM Tris-HCl (pH 8.8), 0.01% Tween-20, 1.5 mM MgCl2, 0.5 uM of each oligonucleotide primer, 0.2 mM of each dNTP (Bioline, London, UK), 0.65U Taq DNA polymerase (Bioline, London, UK) and 50-100 ng DNA. Samples were amplified in an iCycler thermocycler (BioRad Laboratories, Hercules, CA, USA) under the following PCR conditions, initial denaturation at 96°C for 2 min, followed by 30 cycles of denaturation at 95°C for 30 sec, annealing at 59°C for 30 sec and extension at 72°C for 1 min.
In silico primer analysis: Specificity of S. mutans specific primers (24) were evaluated using in silico analyses because PCR amplification of a fragment similar to the size expected for S. mutanswas found in other species of oral streptococci. Analyses were done using Primer-BLAST (25) and sequences from the Streptococcus genus in GenBank, including complete genomes of S. mttis strain B6 (GI: 288906474), S. salivariusstrain 57.I (GI: 339291081) and S. parasangunnssFW213 strain (GI: 387878539) and ATCC 15912 (GI:335369081).
Biochemical identification: To corroborate the PCR results particularly on isolates not identified by the PCR, 39 out of the 116 isolates were identified using biochemical tests. These strains corresponded to isolates identified as S. mutans and strains not identified by the S. sobrinus - S. mutansPCR, randomly selected using the program EPIDAT (SERGAS, Xunta of Galicia, PAHO, WHO). The isolates stored at -80°C were recovered on 5% blood agar and incubated at 37°C, 5% CO2 for 4872 hours. Negative catalase test and gram-positive cocci in the Gram staining helped to define the Streptococcus genus. Biochemical identification was performed using API-20STREP (bioMerieux SA. Inc., Durham, USA) and readings were performed at 4 and 24 hours using the software api-WebTM (bioMerieux SA. Inc., Durham, USA).
Amplification and sequencing of the 16S rRNA: Because the biochemical identification of several isolates was not accurate, amplification and sequencing of the 16S rRNA for 29 and a control strain was performed to confirm bacterial identification. Universal primers for 16S rRNA Eubac 27F and 1492R were used (Table 1) (2628). Each reaction mixture contained 16 mM (NH4)2SO4, 67 mM Tris-HCl (pH 8.8), 0.01% Tween-20, 1.5 mM MgCl2, 0.2 mM each dNTPs (Bioline), 0.5 uM of each oligonucleotide primer, 0.65U of Taq Polymerase (Bioline) and 50-100ng DNA. Samples were amplified in a thermocycler iCycler (BioRad) by PCR conditions described previously (28).
These products were purified using the QIAquick Spin Kit (QIAGEN, Hilden, Germany) and subsequently sequenced in the forward and reverse directions. Sequences were edited using the software Geneious (Geneious Pro 5.5.6 created by Biomatters. (Available from http://www.geneious.com. Sequences with a Q value > 20 were assembled and compared to the sequences reported in GeneBank (National Center for Biotechnology Information NCBI) (29).
Genotype analysis: Genotypes c, e and f of isolates identified as S. mutanswere detected using the multiplex PCR (30). Primers used in the PCR assay were SC-F, SC-R, SE-F, SE-R, SF-F, SF-R (Table 1).
Each 50 uL reaction mixture contained 16 mM (NH4)2SO4, 67 mM Tris-HCl (pH 8.8), 0.01% Tween-20, 2 mM de MgCl2, 0.2 mM of each dNTP, 0.5 uM of each oligonucleotide primer, 1U Taq DNA polymerase and 50-100ng DNA. Samples were amplified in a thermocycler iCycler (Bio-Rad). PCR conditions were described previously (30). S. mutans genotype kwas identified by conventional PCR using a set of specific primers CEFK-F and KR based on the sequence of genes rgpF and rgpE (Table 1) and the PCR conditions previously described.
Statistical analysis
Data analyses were performed using the Statistical Software SPSS® (Statistical Package for Social Sciences) for Windows, Version 19 (SPSS Inc., Chicago, IL). Quantitative variables were analyzed using measures of central tendency (mean and standard deviation) and qualitative variables in percentages. The Kolmogorov-Smirnoff test was used as normality test. Because the variables assumed a non-normal distribution, non-parametric statistical tests were performed with a significance of p<0.05.
Results
Demographic and oral characteristics of patients: From the 47 children evaluated, 38% (18) were six and 62% (29) were seven years old, 62% (29) were male. The overall pattern of dental caries experience and history was of 8% and 13%, respectively. The average DMF-T was of 2.7 (SD ± 3.2), suggesting very low levels of caries prevalence and severity; similar to the results obtained during the sixth oral health monitoring in Medellin (31).
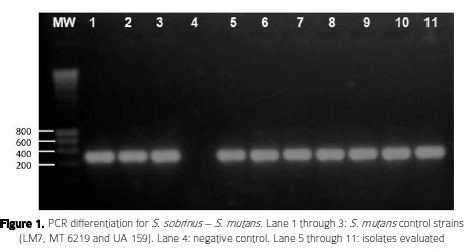
Streptococcus mutans identification and genotyping: S. sobrinus - S. mutans PCR differentiated 16 isolates as S. mutans and four isolates as S. sobrinus (Figure 1). Although, differences in species identification of the isolates by the S. sobrnnus- S. mutans PCR and 16S RNA analysis were found, in silico analyses of S. mutans specific primers against Streptococcus sequences in GenBank revealed the putative amplification of a 517 bp DNA fragment of the glucosyltransferase gene exclusively in five S. mutans strains. These results showed the expected size of this gene in S. mutans and hence support the species specificity of the primers used. Likewise, nn silico analyses of S. sobrinus specific primers revealed the putative amplification of 712 bp in S. sobrinus, S. downei, S. dentirousetti, S. dent/'su/'s, S. orisuis and S. cricetti. These results indicate the need of laboratory confirmed strains to check the specificity of these primers.
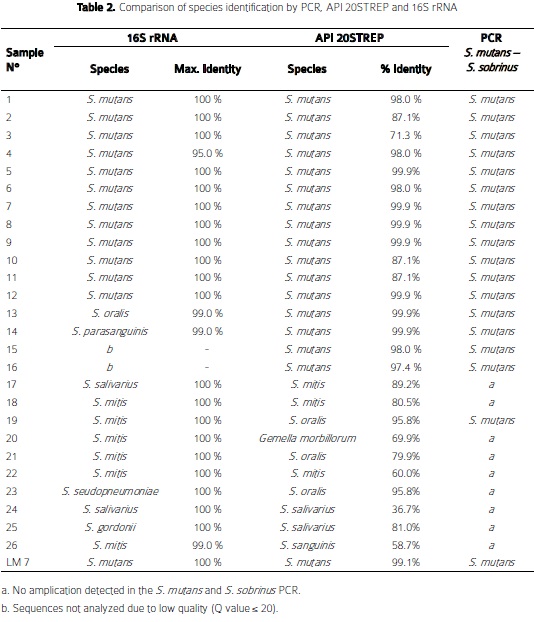
By API-20STREP 16 isolates and by 16S rRNA sequencing 12 isolates were identified as S. mutans (Table 2). The Kappa concordance test value between API-20STREP and 16S rRNA species identification was of 0.73. Overall, agreement between biochemical and molecular identification (16S rRNA) was observed for S. mutans isolates; however, contradictory results of species identification were found in 9 strains by API- 20STREP and 16S rRNA sequencing. Seven out of nine strains showed a lower percentage of identification by API (58.7% - 95.8%) than 16S rRNA sequencing (99% - 100%), suggesting higher accuracy with the last method. 2 out of 7 strains that showed discrepancies were identified by 16S rRNA sequence analysis with 100% similarity as Streptococcus gordonii and Streptococcus pseudoneumoniae ( species not included in the API-20STREP database), while the API system identified those isolates as Streptococcus oralis (95.8%) and Streptococcus salivarius (81%). Likewise, 2 other strains were identified by 16S rRNA sequence analysis with 99% similarity as Streptococcus oralis and Streptococcus parasanguinis (the latter not included in API-20STREP database), while those isolates were identified by the API system as S. mutanswith 99.9% similarity.
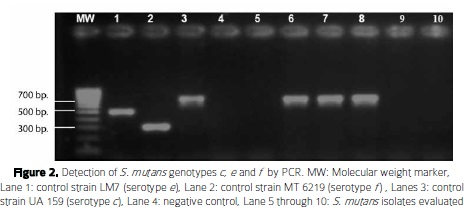
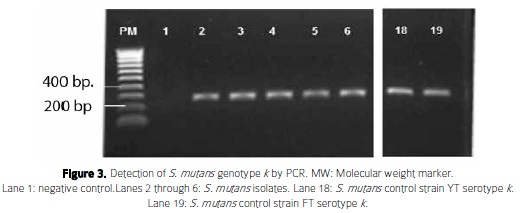
Regarding the genotype analysis, the most frequent S. mutans genotype detected was genotype c (n= 4, 8.5%) (Figure 2, Table 3). Notably, genotype k ( n=2, 4.2%) was detected for the first time in Colombia in this work (Figure 3).
Prevalence of Streptococcus mutans and its genotypes in children with and without caries: Overall, prevalence of S. mutans based on 16S rRNA species identification was 14.9 % (Table 3). In the group without dental caries S. mutans was not detected. There was no significant statistical relationship (p> 0.05) in the presence of S. mutans between the groups with and without dental caries. The subjects harboring S. mutans genotype c, had an average DMF-T of 6.7 (SD ± 1.6) and the subjects with genotypes f kand co-infection of c and k had a DMF-T of 6. Oral examination of one patient with genotype cand kshowed the presence of dental caries in permanent teeth.
Discussion
Tooth decay is a multifactorial disease strongly associated with socio-cultural, economic, environmental and behavioral factors; its distribution and severity varies from region to region (32). Although several works have studied the relationship between dental caries and the presence of S. mutans (33-35); in Colombia, studies on S. mutans prevalence and circulating genotypes comparing children with and without dental caries are scarce.
An approximation to the complete history and experience of dental caries in the population is achieved by the measure of the DMF-T index. Oral examination in this study detected a low dental caries index (DMF-T 0.04 ± 0.3), indicating low dental caries prevalence. However, is also frequent to find a DMF-T low in younger children because change to permanent teeth occurs between six and seven years old (36), in older children this probability increases because of the higher number of permanent teeth exposed to disease.
In general, the DMF-T index and prevalence of dental caries in this study (57.4 %) was lower than previously reported in Medellin for children of 2.5 -4 years with a prevalence of 74.9 % (37). Similarly, a Monitoring Oral Health Survey of 2006 found a lower prevalence of dental caries (24%) in children aged 6 and 7 (31). Contrary, higher prevalence of dental caries (77,8%) has been reported in children 1-5 years old (38). It is important to mention that in the former study, an inclusion criteria was of children with a low socioeconomic status, aspect associated with higher prevalence and severity of dental caries (39); this variable was not consider in the present study.
Considering the possible role of S. mutansin the initiation and progression of dental caries (4), it is important not only to described its prevalence, but also characterize the strains present in children with dental caries. Therefore, it is important to guaranty the proper identification of S. mutans (40). This study evidenced the problems occurring during biochemichal identification of the viridans group streptococci; for example, the identification of some S. mutans isolates showed incongruent results between biochemical and genetic tests (Table 2). Furthermore, the in silicoanalyses with the S. mutans-S. sobrinus PCR, several isolates were identified as S. mutans; however, by 16S rRNA comparisons they showed a high sequence identity with S. mitis, S. oralis and S. parasanguinis (Table 2), which indicated that this PCR is not as specific as anticipated.
In general, In the present study, high concordance (0.61 - 0.80) in species identification was found between API-20STREP and 16S rRNA sequencing (k=0.73, p< 0.001); indicating that both methods appropriately identify these oral bacteria, especially S. mutans. However, caution should be taken when using API-20STREP, because in some occasions, isolates identified as S. mutansby this system were identified as other streptococci by 16S rRNA; this occurred more frequently with species not included in the API-20STREP database. Given that the 16S rRNA is highly conserved within bacterial species and had proved to be highly accurate for bacteria identification and especially useful for discriminating among isolates difficult to distinguish (41,42), in the present study, species were assigned according to the 16S rRNA sequencing results. This is supported by the consideration that the 16S rRNA is the suggested barcode for identification of both culture and non-culture bacterial species (43).
In our study, all children with S. mutans had dental caries; similarly, 66.7% of the children with S. sobrnnus (identified by the species PCR), had caries. Prevalence of S. mutans was lower (14.9%) than in a previous study of Colombian children with similar demographic characteristics (51.4%) (22). Multiple could be the reasons for the difference in prevalence; among them, is the nature of the techniques used that could vary in sensitivity and specificity; specifically, Thompson et al. (22), used a fluorescent antibody based methodology, in which the antisera exhibited a strong cross reaction with other oral bacteria (22). In general, in this study the prevalence of S. mutansand S. sobrinuswas lower than in previous surveys in school-age children with dental caries (35,44). These results suggest the importance of evaluating the role of S. sobrinus in dental caries.
Regarding the distribution of serotypes, it is known that serotype cstrains predominate in the human oral cavity (45). Our results showed that the prevalence of 5. mutans genotype c (8.5%) and genotype f(2.1%) was lower than reports in Japan, Finland and Thailand (12, 17, 33, 46). In Colombia, higher prevalence of 5. mutans serotype c(65.1%), serotype e (14.1%) and serotype f (25.4%) was reported in plaque specimens of children with ages ranging from 8 to 14 years, of the Heliconia and Don Matias municipalites in Antioquia Department (22). Remarkably, this study reports for the first time the presence of genotype k i n Colombia. Prevalence of genotype kwas 4.2%, similar to reports from Japan and Thailand (1-5%) (12, 13, 20, 33, 46), but lower than reported in México (16.9%) (47). 5treptococcus mutans serotype k has been found in saliva and blood isolates (12, 19). Study on properties of k strains support their lower cariogenicity (19) and its ability to survive for longer periods in blood, due to resistance to phagocytosis (48). Also, this new serotype has been related with bacteremia, systemic inflammation and was described as one of the risk factors of infective endocarditis and hemorrhagic stroke (49). Thus, future studies in Colombia are required for understanding the role of 5. mutans genotype k in the development of cardiovascular infective disease.
Conflict of interest
The authors declare that they have no competing interests.
Acknowledgements
This work received funding from Estrategia para la Sostenibilidad de Grupos 2013-2014, Universidad de Antioquia (Acta No. E01719), to Grupo de Microbiología Molecular. We express our gratitude to Grupo de Investigación en Bacterias Anaerobias y Aerobias de importancia clínica, Escuela de Microbiología; Grupo POPCAD, Facultad de Odontología, Universidad de Antioquia; BioMerieux Lab (Dr. Carlos Márquez), who provided the API 20STREP; M.M. Chica and L. Gómez for sample collection and technical support.
References
1. Linossier A, Valenzuela C, Soler E, Contreras E. Colonización de la cavidad oral por Streptococcus grupo mutans, según edad, evaluado en saliva por un método semi-cuantitativo. Rev Chil Infect. 2011;28(3):230-237. [ Links ]
2. Gutiérrez S. Microorganismos cariogénicos. En: Fundamentos de ciencias básicas aplicadas a la odontología. 1 ed. Bogotá: Pontificia Universidad Javeriana; 2006. p. 16 - 21. [ Links ]
3. Gamboa F, Herazo B, Martínez M. Control microbiológico sobre Streptococcus mutansy su acción acidogénica. Universitas Scientiarum 2004;9:45-55. [ Links ]
4. Figueroa M, Alonso G, Acevedo A. Microorganismos presentes en las diferentes etapas de la progresión de la lesión de caries dental. Acta Odontol Venez. 2009;47(1):1-13. [ Links ]
5. Reyes B, Martín J, Yevenes I, Neira M, Palma P, Gordan V, et al. Activity and efects of urease and argnine deaminase in saliva and human oral biofilm. Rev Fac Odontol Univ Antioq. 2012;23(2):343- 352. [ Links ]
6. Forssten S, Bjorklund M, Ouwehand A. Streptococcus mutans, caries and simulation models. Nutrients. 2010;2(3):290-298. [ Links ]
7. Ikeda T, Ochiai K, Shiota T. Taxonomy of the oral Streptococcusmutansbased on colonial characteristics and serological, biochemical and genetic features. Arch Oral Biol. 1979;24(10-11):863-867. [ Links ]
8. Burne R. Oral streptococci Products of their environment. J Dent Res. 1998;77(3):445-452. [ Links ]
9. Koo H, Xiao J, Klein M, Jeon J. Exopolysaccharides produced by Streptococcus mutans glucosyltransferases modulate the establishment of microcolonies within multispecies biofilms. J Bacteriol. 2010;192(12):3024-3032. [ Links ]
10. Busuioc M, Mackiewicz K, Buttaro B, Piggot P. Role of intracellular polysaccharide in persistence of Streptococcus mutans. J Bacteriol. 2009;191(23):7315-7322. [ Links ]
11. Sánchez L, Acosta E. Estreptococos cariogénicos predominantes, niveles de infección e incidencia de caries en un grupo de escolares. Estudio exploratorio. Rev ADM 2007;LXIV(2):45 - 51. [ Links ]
12. Nakano K, Nomura R, Nakagawa I, Hamada S, Ooshima T. Demonstration of Streptococcus mutans with a cell wall polysaccharide specific to a new serotype k, in the human oral cavity. J Clin Microbiol. 2004 Jan;42(1):198-202. [ Links ]
13. Nakano K, Nomura R, Shimizu N, Nakagawa I, Hamada S, Ooshima T. Development of a PCR method for rapid identification of new Streptococcus mutans serotype kstrains. J Clin Microbiol. 2004;42(11):4925- 4930. [ Links ]
14. Huang X, Liu T, Cai Z, Chen Z, Yang J, Liu J. [Evaluation of the in vitro cariogenic potential of Streptococcus mutans (serotype c) strains isolated from caries-free and -active people: the ability of acidogenicity]. Sichuan Da Xue Xue Bao Yi Xue Ban. 2004;35(4):520-521. [ Links ]
15. Hui-qing F, Bu-ling W, Ling-yun S, et al. Evaluation of the acidogenicity of Streptococcus Mutans (serotype c) isolates of the elderly people with caries. Chinese journal of conservative dentistry. 2006(1):21-23. [ Links ]
16. Martin M, Longman L, Forde M, Butterworth M. Infective endocarditis and dentistry: the legal basis for an association. Br Dent J. 2007;203(1):38-39. [ Links ]
17. Nakano K, Nemoto H, Nomura R, Homma H, Yoshioka H, Shudo Y, et al. Serotype distribution of Streptococcus mutans a pathogen of dental caries in cardiovascular specimens from Japanese patients. J Med Microbiol. 2007;56:551-556. [ Links ]
18. Nakano K, Nomura R, Ooshima T. Streptococcus mutans and cardiovascular diseases. Japanese Dental Science Review. 2008;44:29-37. [ Links ]
19. Nakano K, Nomura R, Matsumoto M, Ooshima T. Roles of oral bacteria in cardiovascular diseases-from molecular mechanisms to clinical cases: Cell-surface structures of novel serotype k Streptococcus mutans strains and their correlation to virulence. J Pharmacol Sci. 2010;113(2):120-125. [ Links ]
20. Nakano K, Nomura R, Nemoto H, Mukai T, Yoshioka H, Shudo Y, et al. Detection of novel serotype k Streptococcusmutansin infective endocarditis patients. J Med Microbiol. 2007;56(Pt 10):1413-1415. [ Links ]
21. Gamboa F, Galindo A, Estupinan M. Presence of Streptococcus mutansin saliva and its relationship with dental caries: antimicrobial susceptibility of the isolates. Universitas Scientiarum. 2004;9:23-27. [ Links ]
22. Thomson L, Little W, Bowen W, Sierra L, Aguirrer M, Gillespie G. Prevalence of Streptococcus mutans Serotypes, Actinomyces, and Other Bacteria in the Plaque of Children. J Dent Res 1980;59(10):1581 - 89. [ Links ]
23. Coykendall A. Proposal to elevate the subspecies of Streptococcus mutansto species status, based on their molecular composition. Int J Sys Bacteriol. 1977;27(1):26 -30. [ Links ]
24. Oho T, Yamashita Y, Shimazaki Y, Kushiyama M, Koga T. Simple and rapid detection of Streptococcus mutans and Streptococcus sobrinus in human saliva by polymerase chain reaction. Oral Microbiol Immunol. 2000;15:258-262. [ Links ]
25. Ye J, Coulouris G, Zaretskaya I, Cutcutache I, Rozen S, Madden T. Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics. 2012;13:134. [ Links ]
26. Spradbery P. Restriction fragment length polymorphisms of mutans streptococci in forensic odontological analysis. Biosciencehorizons. 2010;3(2):166 - 178. [ Links ]
27. Matsuyama J, Sato T, Washio J, Mayanagi G, Ito Y, Abiko Y, et al. PCR for detection of mutans streptococci in human dental plaque. International Congress Series. 2005;1284:158-162. [ Links ]
28. Dunbar J, Ticknor L, Kuske C. Phylogenetic specificity and reproducibility and new method for analysis of terminal restriction fragment profiles of 16S rRNA genes from bacterial communities. Appl Environ Microbiol. 2001;67(1):190-197. [ Links ]
29. Altschul S, Gish W, Miller W, Myers E, Lipman D. Basic local alignment search tool. J Mol Biol. 1990;215(3):403-410. [ Links ]
30. Shibata Y, Ozaki K, Seki M, Kawato T, Tanaka H, Nakano Y, et al. Analysis of loci required for determination of serotype antigenicity in Streptococcusmutansand its clinical utilization. J Clin Microbiol. 2003; 41(9):4107-4112. [ Links ]
31. Franco A, Ochoa E, Ramírez B, Segura A, Tamayo A, García C. Situación de salud bucal de los escolares de Medellín. VI monitoreo. Año 2006. 2007;2(1):57-69. [ Links ]
32. Petersen P. The World Oral Health Report 2003: continuous improvement of oral health in the 21st century the approach of the WHO Global Oral Health Programme. Community Dent Oral Epidemiol. 2003;31(Suppl 1):3-23. [ Links ]
33. Taku Y, Kasuko T. Distribution and Characterization of Serotype k Streptococcus mutans. Int J Oral-Med Sci. 2011;10(2):89-95. [ Links ]
34. Kanasi E, Johansson I, Lu S, Kressin N, Nunn M, Kent R, et al. Microbial risk markers for childhood caries in pediatricians' offices. J Dent Res. 2010;89(4):378-383. [ Links ]
35. Okada M, Soda Y, Hayashi F, Doi T, Suzuki J, Miura K, et al. Longitudinal study of dental caries incidence associated with Streptococcus mutansand Streptococcus sobrnnus in pre-school children. J Med Microbiol. 2005;54:661-665. [ Links ]
36. Klein H, Palmer C., Knutson J. Studies on Dental Caries, I, Dental status and Dental Needs of elementary school. Pub Health Rep. 1938;53:751-765. [ Links ]
37. Saldarriaga A, Arango C, Cossio M. Dental caries in the primary dentition of a Colombian population according to the ICDAS criteria. Braz Oral Res. 2010;24(2):211-216. [ Links ]
38. Escobar G, Ramírez B, Franco A, Tamayo A, Castro J. Experiencia de caries dental en niños de 1- 5 años de bajos ingresos. Medellín, Colombia. Rev CES Odont. 2009;22(1):21-28. [ Links ]
39. Frazáo P. Epidemiology of dental caries: when structure and context matter. Braz Oral Res. 2012;26(Spec Iss 1):108-114. [ Links ]
40. Truong T, Menard C, Mouton C, Trahan L. Identification of mutans and other oral streptococci by random amplified polymorphic DNA analysis. J Med Microbiol. 2000;49(1):63-71. [ Links ]
41. Lal D, Verma M., Lal R. Exploring internal features of 16S rRNA gene for identification of clinically relevant species of the genus Streptococcus. Ann Clin Microbiol Antimicrob. 2011;10:28. [ Links ]
42. Bosshard P, Abels S, Altwegg M, Bottger E, Zbinden R. Comparison of conventional and molecular methods for identification of aerobic catalase-negative gram-positive cocci in the clinical laboratory. J Clin Microbiol. 2004;42(5):2065-2073. [ Links ]
43. Ahn J, Yang L, Paster B, Ganly I, Morris L, Pei Z, et al. Oral microbiome profiles: 16S rRNA pyrosequencing and microarray assay comparison. PLoS One. 2011;6(7):e22788. [ Links ]
44. Zhou Q, Qin X, Qin M, GE L. Genotypic diversity of Streptococcusmutansand Streptococcussobrinus in 3-4-year-old children with severe caries or without caries. Int J Paediatr Dent. 2011;21(6):422-431. [ Links ]
45. Biswas S, Biswas I. Complete genome sequence of StreptococcusmutansGS-5, a serotype c strain. J Bacteriol. 2012 Sep;194(17):4787-4788. [ Links ]
46. Lapirattanakul J, Nakano K, Nomura R, Nemoto H, Kojima A, Senawongse P, et al. Detection of serotype k Streptococcusmutansin Thai subjects. Oral Microbiol Immunol. 2009;24(5):431-433. [ Links ]
47. Espinosa L, Martínez G, Martínez R, Loyola J, Patiño N, Reyes J, et al. Antimicrobial sensibility of Streptococcus mutansserotypes to silver nanoparticles. Mater Sci Eng C Mater Biol Appl. 2012;32:896-901. [ Links ]
48. Tsuda H, Yamashita Y, Toyoshima K, Yamaguchi N, Oho T, Nakano Y, et al. Role of serotype-specific polysaccharide in the resistance of Streptococcusmutansto phagocytosis by human polymorphonuclear leukocytes. Infect Immun. 2000;68(2):644-650. [ Links ]
49. Nakano K, Hokamura K, Taniguchi N, Wada K, Kudo C, Nomura R, et al. The collagen-binding protein of Streptococcusmutansis involved in haemorrhagic stroke. Nat Commun. 2011;2(485). [ Links ]