Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957On-line version ISSN 2500-7440
Rev Col Gastroenterol vol.24 no.3 Bogotá July/Sept. 2009
Metal stent versus plastic stent for malignant distal biliary stricture
Rodrigo Castaño MD.(1), Óscar Álvarez MD. (2), Alvaro García MD. (3), Víctor Quintero MD. (4), Eugenio Sanín MD. (5), Faruk Erebrie MD. (5), Edilberto Núñez MD. (6), Víctor Calvo MD. (7), Luz Helena García (8).
(1) Professor, Gastrohepatology group, Universidad de Antioquia-Hospital Pablo Tobón Uribe
(2) Gastroenterologist. Mac Allen. Texas.
(3) Surgery resident. Universidad Pontificia Bolivariana.
(4) Surgery resident. Universidad de Antioquia
(5) Professor, Gastrohepatology group, Universidad de Antioquia
(6) Medical student. Gastrohepatology group, Universidad de Antioquia
(7) Health information system manager. Facultad Nacional de Salud Pública - Universidad de Antioquia
(8) Lead Nurse, Endoscopy Unit, Hospital Pablo Tobón Uribe.
Received: 05-02-09 Accepted: 12-08-09
Summary
Background: Biliary stent clogging is the principal limitation in the use of palliative treatments for malignant biliary stricture. The patency of metal stents lasts longer than that of plastic stents but metal stents are more expensive. Well-established criteria for choosing metal or plastic stents do not yet exist
Patients and Methods: A retrospective trial was carried out with 80 patients in Pablo Tobón Hospital, between January 2002 and June 2008. 40 patients had plastic stents implanted while the other 40 had metal stents installed. Biliary stricture improvement, stricture recurrence, complications and survival rates for each group were compared.
Results: 48 males (60%) and 32 females (40%) with an overall average age of 66±6 years and ages ranging from 47 years old to 78 years old were diagnosed with inoperable malignant distal biliary stricture. The two groups were comparable in terms of ages, genders, laboratory tests, diagnoses and liver metastases. Clinical success was the same for both treatments (92%). Hospital stays were significantly shorter for patients with metal stents than for patients with plastic stents. (6.1 ± 6.6 days versus 10.6 ± 5.9 days. 95% confidence interval: 1.6 to 7.2, p<0.01). Recurrent biliary stricture was identified in 19 of 40 patients with plastic stents (47.5%) and 13 of 40 patients with metal stents (45%) (95% Confidence Interval: -0.4 to 0.08, p=0.2). Average duration of stent patency was longer in patients with metal stents than in patients with plastic stents (174.2 ± 60.4 days versus 108 ± 50.9 days. 95% confidence interval: 41.1 to 90.8, p<0.01). Early complications occurred in 5% of patients with metal stents and in 12.5% of patients with plastic stents (95% confidence interval: -0.22 to 0.07, p = 0.4). Later complications occurred in 42.5% of patients with plastic stents and 30% of patients with metal stents (95% confidence interval: -0.35 to 0.1, p = 0.3). Patients with metal stents required less intervention for treatment of complications than did patients with plastic stents (6 versus 17. 95% confidence interval: 0.06 to 0.48, p<0.01). Patients with metal stents required endoscopic retrograde cholangiography (ECRP) 11 times, whereas patients with plastic stents required ECRP 18 times (95% Confidence Interval -0.4 to 0.05, p=0.1). Average survival time after stent insertion was 159.4 ± 9.4 days for plastic stents and 189.5 ± 7.6 days for metal stents (p=0,07).
Conclusions: Plastic and metal stents are effective for inoperable distal malignant stricture treatment. However a comparison between the two types of stents reveals that although patient survival rates for the two types of stents are equal, metal stents had better patency, and patients with metal stents had shorter hospital stays, and fewer procedures for complications.
Key words
Malignant distal bile duct stricture, Metal biliary stent, Stent clogging, Plastic biliary stent, Endoscopic drainage, ERCP.
INTRODUCTION
The treatment of malignant bile strictures is a big challenge for specialists. Establishing a surgical treatment which provides better expectations for patient survival is the measure of success for this kind of stenting. However, around 80% of patients can not be drained for purposes of healing due to advanced lesions or metastases (1).
Patients with undrainable lesions survive less than a year on average and are likely to endure a poorer quality of life. To improve quality of life the therapeutic objective is to palliate the symptoms of this stricture such as jaundice, itching, cachexia and vomiting while avoiding complications like cholangitis, sepsis and avoiding hepatic resection (2).
Palliative methods for malignant bile duct obstructions include surgical bilioenteric derivatives (3), intraluminal or external (5) radiotherapy (brachytherapy) (4), chemotherapy (6), photo-dynamic therapy (7), percutaneous therapy (8), and endoscopic palliation (9).
Endoscopic retrograde cholangiopancreatography (ERCP) allows palliation of malignant bile duct strictures in patients with inoperable tumors. In some cases, this procedure also allows histological diagnosis, as we reported previously (10).
Plastic or metal stents can palliate jaundice and its complications, improving patients sensation of well-being. Stent patency is an important factor because it affects patient quality of life and service costs (11). There are two kinds of biliary stents on the market: plastic and metal. Plastic stents are made of polyethylene, polyurethane or tephlon, with sizes that vary between 7 Fr and 11 Fr. Self-expandable metallic stents, covered or uncovered, are made of nitinol with sizes from 24 Fr to 30 Fr.
A smaller rate of occlusion is the most important advantage of metal stents over plastic stents (6-12 months average patency for metal stents versus 3-4 months for plastic stents). Occlusion is frequently caused by proximal or distal growth of tumors into the stent and also, but less frequently, biliary mud (12).
On the other hand, disadvantages of metal stents include high prices and greater difficulties in replacement or removal once they become loose. Occlusions in plastic stents are most commonly caused by the growth of biofilms on biliary mud deposits. Plastic stents frequently need repeated changes to maintain biliary drainage (13).
Worldwide, the number of publications comparing metal stent and plastic stent patency and effectiveness, and which mention palliation in patients with malignant distal biliary stenosis, is limited (14). To date there have been no reports in Colombia.
The present work addresses this issue by comparing the effectiveness of 10 Fr plastic stents with uncovered metal stents in patients with inoperable malignant bile duct distal strictures.
PATIENTS AND METHODS
We carried out a retrospective and descriptive study in Medellin, Colombia in three level three centers: Hospital Pablo Tobón Uribe, Hospital San Vicente de Paúl and Clínica el Rosario. The study was conducted between January 2002 and June 2008.
We selected a data base of patients with plastic stents or metal stents, discarding patients with strictures at the hepatic confluence. We analyzed 40 patients with metal stents and 40 with plastic stents.
Diagnosis
In most cases diagnoses were made through imaging using computerized axial Tomography or abdominal magnetic resonance was used to diagnose the malignancy. Less frequently, we obtained percutaneous biopsies of lesions, endoscopic cytology of brushed lesions (10), or diagnoses were made from clinical sources when histopathological diagnosis was not available.
Procedure
Each endoscopy was done according to the preferences of the endoscopist, using general anesthesia or Midazolam/Fentanyl, Ketamin sedation.
Prophylactic antibiotics were administered intravenously one hour before the proceeding: 3gr of ampicillin sulbactam and 400 mg of ciprofloxacin.
Cholangiopancreatography was done before the proceeding to determine the anatomy of the biliary system. To view the stenting process we cleared the biliary confluence using an absorbent guide.
Stents
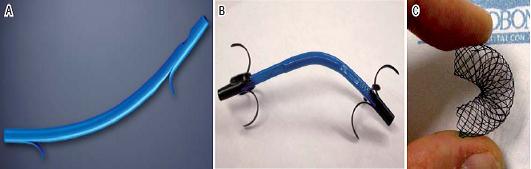
Three plastic prostheses were used in the experiment. They consist of 10 Fr, polyethylene or polyurethane conduits (no tephlon prostheses were used). We modified the end of the prosthesis to adapt them to different sizes of flaps (figure 1).
Figure 1. Stents used. A. 10 Fr Biliary stent, Cotton-Leung variety. Produced by Cook Medical Inc. Winston-Salem, NC. B. 10 Fr Biliary stent, RX Biliary System variety, Boston Scientific Corporation, Natick, Mass. C. Nitinol biliary stent, 10 mm diameter. Produced by Tecnostent. Medellín.
The metal stents used in this study were developed by Tecnostent of Seoul, South Korea and produced at their plant in Medellin, Colombia under the supervision of Drs. Song and Kan. The stents are self expanding and flexible along their longitudinal axis.
9 Fr stents come premounted in 220 cm introductory systems. To reduce friction they must be irrigated externally and internally with saline solution prior to use. Once they are located in the stenosis, progressive retirement of the cover frees the metal stent. It expands up to 10mm (30 Fr) with a shortening of 30% once it is released. The introductory system with in-situ stent is passed over a 4m long, 0.35 mm caliber radio-opaque guide (ideally without papilotomy).
Looking directly through the endoscope we performed cutaneous cholangioscopic insertion of the stent. The stent is radio-opaque and has two gold marks on each end for better radiological recognition. The stent can be recovered and remounted immediately if no more than 2/3 of its length have left the introductory system. However, after 2 days have passed from the time of release, recovery becomes very difficult and can have serious complications.
Monitoring
One week after the stent was placed, biliary drainage was considered successful if bilirubin levels had fallen by more than 50%. Stent patency is the time between placement and displacement, or between placement and the appearance of fever and jaundice, an increase in bilirubin or the patients death. Stay in hospital was measured as the number of days the patient stayed in the hospital after the first stent was placed until death (including days spent in the hospital during return stays for treatment of complications). Early complications were defined as those occurring during the first week following stent placement. Late complications were defined as those which occurred after the end of the first week following placement. We evaluated ERCP complications and biliary sphincterotomy according to Freemans guide (15).
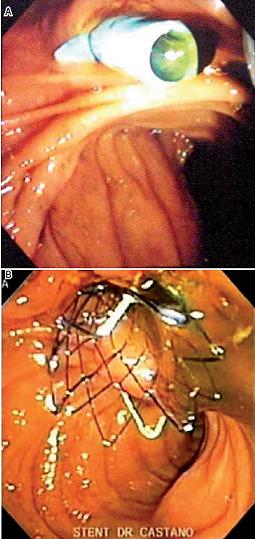
Given that greater patient comfort and improved quality of life are the principal objectives of endoprosthesis, the measure of palliative success is the degree to which biliary symptoms are reduced. If there was no direct access to the patients medical history file, the patient, a family member or the attending physician was contacted by telephone to determine when jaundice or symptoms of cholangitis reappeared after stent placement. In a case of obstruction of a metal stent, a plastic stent is used to open the passage. In the case of obstruction of a plastic stent, the stent must be removed and replaced with another plastic stent (figure 2).
Metal stent obstruction opened with plastic stent. Obstructed plastic stent replaced with another plastic stent (figure 2).
Figure 2. Endoscopic views of stents after placement. A. Transpapillar Plastic Stent. B. Metallic Stent released
Statistical analysis
The SPSS (Statistical Package for the Social Sciences, Version 15.0, Chicago, IL, USA) was used for analysis of data. Patency and survival were estimated according the Kaplan-Meier technique. Stent patency (measured in days) represents the time interval between insertion and time of first intervention (replacement with new stent) or patients death. All results are shown in averages. Statistical analysis was done using nonparametric tests. The Mann-Whitney test was used for quantitative variables and the chi square test for qualitative variables. Statistical significance was established with a p value smaller than 0.05.
Results
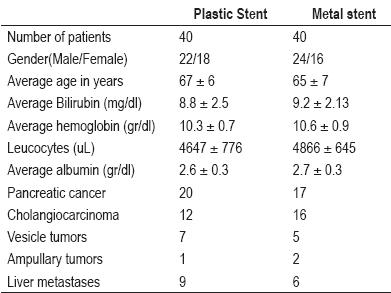
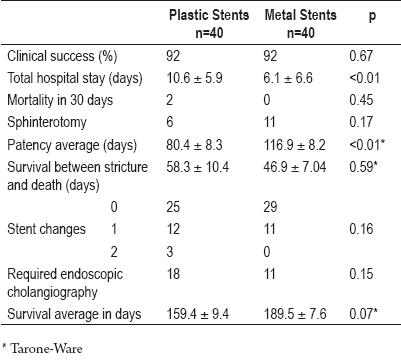
Between January 2002 and June 2008 medical records of 80 patients who fit the evaluation criteria were included in this study. Patients average age was 66.3 years (47 to 78 years, SD ± 6.5). The two groups were similar in terms of gender, malignances, laboratory tests, tumor locations and liver metastases. The basics characteristics of patients who received plastic stents and metal stents are shown in table 1.
Table 1. Basic characteristics of patients.
10 sphincterotomies were performed on patients in the metal stent group while 3 were performed in the plastic stent group. 1 precut was done in the metal stent group and 3 were done in the plastic stent group.
Complications appeared frequently in the plastic stent group, especially late obstructive complications.
Of the three bleeding papillas presented, two required endoscopic intervention (esclerotherapy with adrenalin dilution 1:10.000) but did not require transfusions.
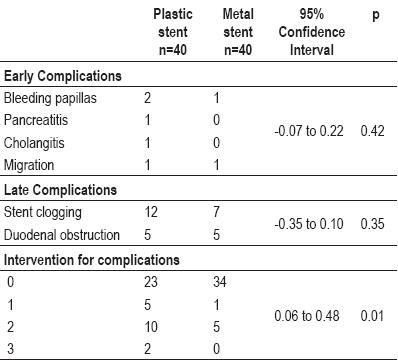
Patients with complications who had plastic stents required a greater number of interventions than patients with complications who had metal stents. Table 2 shows early and late complications for both groups.
Table 2. Early and late complications for both groups.
Biliary drainage occurred in both kinds of patients, documented by decreases of more than 50% of the initial bilirubin value after the first week in 92% of the cases. One week after stent placement, jaundice had not decreased nor did ecography show any evidence of biliary dilation for two patients in the metal stent group, however both patients improved spontaneously thereafter. Another patient had a bleeding papilla which needed endoscopic intervention. This intervention improved the patients biliary stricture. In patients with plastic stents that did not work it was found that the stents had loosened early and new plastic stents were needed. An early obstruction occurred in a patient with hepatic metastases. The obstruction never was resolved despite the placement of two stents. This patient also suffered from a gastroduodenal obstruction that was handled with another stent. The patient survived only 47 days. Another patients stent never functioned. Death occurred after only 23 days.
Total hospital stays were significantly greater for the plastic stent group than for patients with metal stents. (10.6 ± 5.9 versus 6.1 ± 6.6 days, p<0,01). Table 3 shows the results for both groups.
Table 3. Results for different groups.
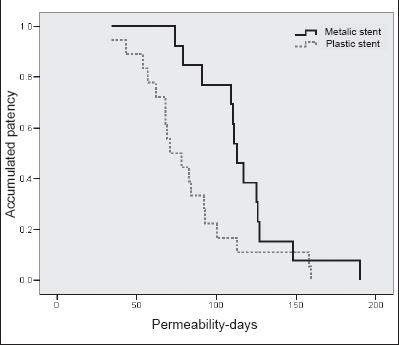
The accumulated patency of the first stent was significantly better for the metal stent group than for the plastic stent group. Average patency of the first stent was 108 ± 50.9 days for the plastic stent group (range: 20 to 201 days) while average patency of the first stent was 174.2 ± 60.4 days for the metal stent group (figure 3).
Figure 3. Accumulated patency for metal and plastic stents.
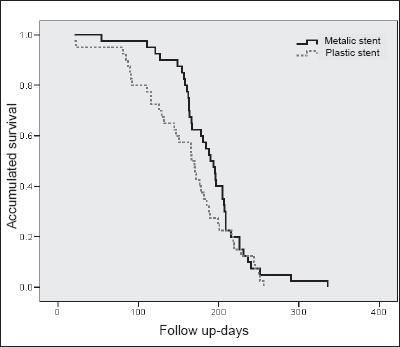
After stent occlusion the survival rate was lower in the metal stent patients because these obstructions appeared later instance than in the plastic stent group and at the stage when there was little that could be offered to these patients. No percutaneous drainage was done in these cases. All patients had died by the final evaluation of the study.
No differences in survival were found between the two groups. Average survival for the metal stent groups was 189.5 ± 7.6 days (range: 54 to 336 days) and 159.4 ± 9.4 days for the plastic stent group (figure 4).
Figure 4. Accumulated survival for plastic and metal stents.
DISCUSSION
Obstructive jaundice caused by an inoperable distal malignant biliary illness is associated with different complications including poor absorption, coagulopathy, hepatocellular and progressive renal dysfunction, cholangitis, and itching. These complications make the quality of life of terminal patients worse than it would be otherwise. Endoscopic stent placement decreases risks of these complications. It is considered to be the best palliative procedure for patients with inoperable pancreatobiliary tumors (16). Plastic stents were introduced 30 years ago, and metal stents were introduced 15 years ago.
A disadvantage of plastic stent use is the high risk of obstruction three to four months after placement. Nevertheless, they relieve jaundice very efficiently once they are placed. Plastic stents showed a greater incidence of repeated obstructions. These obstructions are the result of multiple factors including stent design, constituent materials, proteins, and bile viscosity, but the most important factors are the presence of bacteria with calcium bilirubinate and calcium palmitate crystals (13).
The Sphincter of Oddi acts as barrier between a colonized duodenum and a sterile biliary tract, a kind of mechanical defense against microbial colonization. A disruption of this barrier through the placement of a prostheses through the sphincter in the duodenal lumen allows bacteria to reach the biliary tract. This event allows biofilms and bile mud to form in the endoprosthesis.
The process of stent occlusion begins with the fixation of bacterial pili to the stent surface.
Bilirubin diglucuronide conjugation and biliary lecithin formation by bacterial beta-glucoronidase and phospholipase lead to the precipitation of calcium bilirubinate, calcium salts and fatty acids. Over time, biofilm progression and progressive agglomeration of biliary sediment cause the formation of biliary mud which in turn leads to stent occlusion.
Our patients received a single dose of prophylactic antibiotic: ciprofloxacine or ampicillin sulbactam according to the endocopists preference. Prolonged antibiotic use has not demonstrated decreased incidence of stent occlusion. In the presence of cholangitis the recommendation is to use ciproflaxicine at least for 10 days because this agent effectively penetrates to the obstructed biliary tract and can be orally-administrated (17, 18).
In our study implanted metal stent patency lasted longer than plastic stent patency (116.9 ± 8.2 days versus 80.4 ± 8.3 days, p<0,01). Patients with metal stents have less symptomatic biliary complications, longer periods without symptoms, less need to repeat ERCP, plus and in other studies they have had shorter hospital stays (2). This superiority of metal stents is probably due to their greater diameter (around 10 mm, 30 Fr) and to how the occlusions are produced.
Strictures in metal stents develop more slowly and are due to internal growth or growth at the end of the stents rather than not to biofilm formation with biliary mud formation as in plastic stents.
Studies have demonstrated that tumor growth frequently exceeds stent ends which is crucial for selecting stent length. Nevertheless, the use of metal stents is limited by their high cost compared with plastic stents, a difference of almost 10 times in our environment.
Universal specifications for choosing between metal stents and plastic stents for malignant biliary stricture do not yet exist. However, most studies suggest choosing metal prostheses for patients who are likely to survive more than four months (19-21).
Usage of plastic prostheses have been related to higher costs because of the need for more ERCPs and renewed hospital cholangitis management. The studies of Prat (19) and Kaasis (20) evaluate different variables for developing prognoses to guide prosthesis election. The best forecast indicator in Prats multivariable analyses was the presence of a tumor larger than 3cm, while the best indicator in the study done by Kaasiss was the presence of hepatic metastases.
Recently, Moss and colleagues (22) systematically reviewed the use of different stents and made a global cost benefit analysis of the use of the two different kinds of stents. They reviewed 7 randomized and controlled studies, with 724 patients. They compared metal stents and plastic stents for malignant biliary strictures. Parameters considered included: technical excellence, quality of life, survival, and cost-benefit trade-offs. While they did not find any significant differences in technical or therapeutic excellence, complications, mortality or survival between plastic stents and metal stents, they did find that plastic stent patency (62 to 165 days) was significantly less than metal stent patency (111-273 days).
Metal stents have cost benefits for palliation in patients with extrahepatic biliary stricture who survive more than six months (19, 23-25).
Other studies, using other kinds of measurements, have demonstrated improvements in quality of life thanks to stents due to control of many secondary symptoms in patients with extrahepatic biliary stricture (26-28).
It is not clear whether or not stent changes should be programmed. Although some people suggest programmed stent changes (19), others suggest that changes only be undertaken in dysfunctional situations in order to avoid cholangitis (20, 29).
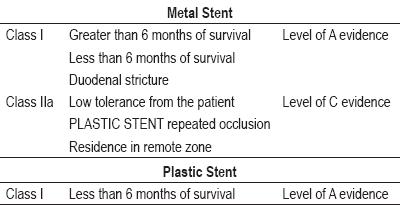
One proposal for stent selection based on the medical evidence is reported in table 4 (13).
Table 4. Recommendations for the selecting plastic stents or metal stents.
The evidence-based classification system seen in the table has the following form: Class I: Evidence and agreement exist regarding utility and effectiveness of a certain procedure. Class II: Conflicting evidence and/or opinion exist regarding a certain procedure. Class III: Existing evidence and opinion indicates that a procedure is useless or ineffective.
The present study together with the literature on this subject show both the reliability and effectiveness of plastic stents and metal stents for management of malignant distal biliary stricture. Metal stents resulted in shorter hospital stays and greater patency rates, although survival time was not significantly different.
As more studies of stents are done, stents need to be developed with the capacity to avoid bacterial or tumor colonization or which have anti-bacterial and/or chemo-radio therapeutic elements. The stents of the future should have longer patency, less need of reinterventions, better cost-benefit relationships and longer patient survival.
Disclosure
The authors of this study declare their absolute independence from stent producers and producers of drugs used in patient management.
REFERENCES
1. Stern N, Sturgess R. Endoscopic therapy in the management of malignant biliary obstruction. Eur J Surg Oncol 2008; 34: 313-7.
2. Weber A, Mittermeyer T, Wagenpfeil S, Schmid RM, Prinz C. Self-expanding metal stents versus polyethylene stents for palliative treatment in patients with advanced pancreatic cancer. Pancreas 2009; 38: e7-e12.
3. Lesurtel M, Dehni N, Tiret E, Parc R, Paye F. Palliative surgery for unresectable pancreatic and periampullary cancer: a reappraisal. J Gastrointest Surg 2006; 10: 286-91.
4. Singh V, Kapoor R, Solanki KK, Singh G, Verma GR, Sharma SC. Endoscopic intraluminal brachytherapy and metal stent in malignant hilar biliary obstruction: a pilot study. Liver Int 2007; 27: 347-52.
5. Saito H, Takada T, Miyazaki M, et al. Radiation therapy and photodynamic therapy for biliary tract and ampullary carcinomas. J Hepatobiliary Pancreat Surg 2008; 15: 63-8.
6. Giuliani F, Gebbia V, Maiello E, Borsellino N, Bajardi E, Colucci G. Gemcitabine and cisplatin for inoperable and/or metastatic biliary tree carcinomas: a multicenter phase II study of the Gruppo Oncologico dellItalia Meridionale (GOIM). Ann Oncol 2006; 17 Suppl 7: vii73-7.
7. Zoepf T, Jakobs R, Arnold JC, Apel D, Riemann JF. Palliation of nonresectable bile duct cancer: improved survival after photodynamic therapy. Am J Gastroenterol 2005; 100: 2426-30.
8. Van Delden OM, Lameris JS. Percutaneous drainage and stenting for palliation of malignant bile duct obstruction. Eur Radiol 2008; 18: 448-56.
9. Baron TH. Palliation of malignant obstructive jaundice. Gastroenterol Clin North Am 2006; 35: 101-12.
10. Ruiz MH, Castaño R, Alvarez O, et al. Evaluación de la citología por cepillado en las estenosis biliares y pancreáticas durante la colangiografía endoscópica retrógrada. Revista Col de Gastroenterol 2002; 17: 222-5.
11. Hammarstrom LE. Role of palliative endoscopic drainage in patients with malignant biliary obstruction. Dig Surg 2005; 22: 295-304; discussion 5.
12. Moss AC, Morris E, Leyden J, MacMathuna P. Malignant distal biliary obstruction: a systematic review and meta-analysis of endoscopic and surgical bypass results. Cancer Treat Rev 2007; 33: 213-21.
13. Levy MJ, Baron TH, Gostout CJ, Petersen BT, Farnell MB. Palliation of malignant extrahepatic biliary obstruction with plastic versus expandable metal stents: An evidence-based approach. Clin Gastroenterol Hepatol 2004; 2: 273-85.
14. Moparty B, Carr-Locke DL. Metal or plastic stent for malignant biliary obstruction: whats got the most bang for your buck? Eur J Gastroenterol Hepatol 2007; 19: 1041-2.
15. Freeman ML. Adverse outcomes of endoscopic retrograde cholangiopancreatography. Rev Gastroenterol Disord 2002; 2: 147-68.
16. Katsinelos P, Paikos D, Kountouras J, et al. Tannenbaum and metal stents in the palliative treatment of malignant distal bile duct obstruction: a comparative study of patency and cost effectiveness. Surg Endosc 2006; 20: 1587-93.
17. Leung JW, Libby ED, Morck DW, et al. Is prophylactic ciprofloxacin effective in delaying biliary stent blockage? Gastrointest Endosc 2000; 52: 175-82.
18. Leung JW, Liu YL, Chan RC, Ling TK, Cheng AF. Effects of adherence factors and human bile on bacterial attachment and biliary stent blockage: an in vitro study. Gastrointest Endosc 2002; 56: 72-7.
19. Prat F, Chapat O, Ducot B, et al. A randomized trial of endoscopic drainage methods for inoperable malignant strictures of the common bile duct. Gastrointest Endosc 1998; 47: 1-7.
20. Kaassis M, Boyer J, Dumas R, et al. Plastic or metal stents for malignant stricture of the common bile duct? Results of a randomized prospective study. Gastrointest Endosc 2003; 57: 178-82.
21. Rey JF, Dumas R, Canard JM, et al. Guidelines of the French Society of Digestive Endoscopy: biliary stenting. Endoscopy 2002; 34: 169-73, 81-5.
22. Moss AC, Morris E, Leyden J, MacMathuna P. Do the benefits of metal stents justify the costs? A systematic review and meta-analysis of trials comparing endoscopic stents for malignant biliary obstruction. Eur J Gastroenterol Hepatol 2007; 19: 1119-24.
23. Schmassmann A, von Gunten E, Knuchel J, Scheurer U, Fehr HF, Halter F. Wallstents versus plastic stents in malignant biliary obstruction: effects of stent patency of the first and second stent on patient compliance and survival. Am J Gastroenterol 1996; 91: 654-9.
24. Arguedas MR, Heudebert GH, Stinnett AA, Wilcox CM. Biliary stents in malignant obstructive jaundice due to pancreatic carcinoma: a cost-effectiveness analysis. Am J Gastroenterol 2002; 97: 898-904.
25. Yeoh KG, Zimmerman MJ, Cunningham JT, Cotton PB. Comparative costs of metal versus plastic biliary stent strategies for malignant obstructive jaundice by decision analysis. Gastrointest Endosc 1999; 49: 466-71.
26. Ballinger AB, McHugh M, Catnach SM, Alstead EM, Clark ML. Symptom relief and quality of life after stenting for malignant bile duct obstruction. Gut 1994; 35: 467-70.
27. Luman W, Cull A, Palmer KR. Quality of life in patients stented for malignant biliary obstructions. Eur J Gastroenterol Hepatol 1997; 9: 481-4.
28. Abraham NS, Barkun JS, Barkun AN. Palliation of malignant biliary obstruction: a prospective trial examining impact on quality of life. Gastrointest Endosc 2002; 56: 835-41.
29. Knyrim K, Wagner HJ, Pausch J, Vakil N. A prospective, randomized, controlled trial of metal stents for malignant obstruction of the common bile duct. Endoscopy 1993; 25: 207-12.
1. Stern N, Sturgess R. Endoscopic therapy in the management of malignant biliary obstruction. Eur J Surg Oncol 2008; 34: 313-7. [ Links ]
2. Weber A, Mittermeyer T, Wagenpfeil S, Schmid RM, Prinz C. Self-expanding metal stents versus polyethylene stents for palliative treatment in patients with advanced pancreatic cancer. Pancreas 2009; 38: e7-e12. [ Links ]
3. Lesurtel M, Dehni N, Tiret E, Parc R, Paye F. Palliative surgery for unresectable pancreatic and periampullary cancer: a reappraisal. J Gastrointest Surg 2006; 10: 286-91. [ Links ]
4. Singh V, Kapoor R, Solanki KK, Singh G, Verma GR, Sharma SC. Endoscopic intraluminal brachytherapy and metal stent in malignant hilar biliary obstruction: a pilot study. Liver Int 2007; 27: 347-52. [ Links ]
5. Saito H, Takada T, Miyazaki M, et al. Radiation therapy and photodynamic therapy for biliary tract and ampullary carcinomas. J Hepatobiliary Pancreat Surg 2008; 15: 63-8. [ Links ]
6. Giuliani F, Gebbia V, Maiello E, Borsellino N, Bajardi E, Colucci G. Gemcitabine and cisplatin for inoperable and/or metastatic biliary tree carcinomas: a multicenter phase II study of the Gruppo Oncologico dellItalia Meridionale (GOIM). Ann Oncol 2006; 17 Suppl 7: vii73-7. [ Links ]
7. Zoepf T, Jakobs R, Arnold JC, Apel D, Riemann JF. Palliation of nonresectable bile duct cancer: improved survival after photodynamic therapy. Am J Gastroenterol 2005; 100: 2426-30. [ Links ]
8. Van Delden OM, Lameris JS. Percutaneous drainage and stenting for palliation of malignant bile duct obstruction. Eur Radiol 2008; 18: 448-56. [ Links ]
9. Baron TH. Palliation of malignant obstructive jaundice. Gastroenterol Clin North Am 2006; 35: 101-12. [ Links ]
10. Ruiz MH, Castaño R, Alvarez O, et al. Evaluación de la citología por cepillado en las estenosis biliares y pancreáticas durante la colangiografía endoscópica retrógrada. Revista Col de Gastroenterol 2002; 17: 222-5. [ Links ]
11. Hammarstrom LE. Role of palliative endoscopic drainage in patients with malignant biliary obstruction. Dig Surg 2005; 22: 295-304; discussion 5. [ Links ]
12. Moss AC, Morris E, Leyden J, MacMathuna P. Malignant distal biliary obstruction: a systematic review and meta-analysis of endoscopic and surgical bypass results. Cancer Treat Rev 2007; 33: 213-21. [ Links ]
13. Levy MJ, Baron TH, Gostout CJ, Petersen BT, Farnell MB. Palliation of malignant extrahepatic biliary obstruction with plastic versus expandable metal stents: An evidence-based approach. Clin Gastroenterol Hepatol 2004; 2: 273-85. [ Links ]
14. Moparty B, Carr-Locke DL. Metal or plastic stent for malignant biliary obstruction: whats got the most bang for your buck? Eur J Gastroenterol Hepatol 2007; 19: 1041-2. [ Links ]
15. Freeman ML. Adverse outcomes of endoscopic retrograde cholangiopancreatography. Rev Gastroenterol Disord 2002; 2: 147-68. [ Links ]
16. Katsinelos P, Paikos D, Kountouras J, et al. Tannenbaum and metal stents in the palliative treatment of malignant distal bile duct obstruction: a comparative study of patency and cost effectiveness. Surg Endosc 2006; 20: 1587-93. [ Links ]
17. Leung JW, Libby ED, Morck DW, et al. Is prophylactic ciprofloxacin effective in delaying biliary stent blockage? Gastrointest Endosc 2000; 52: 175-82. [ Links ]
18. Leung JW, Liu YL, Chan RC, Ling TK, Cheng AF. Effects of adherence factors and human bile on bacterial attachment and biliary stent blockage: an in vitro study. Gastrointest Endosc 2002; 56: 72-7. [ Links ]
19. Prat F, Chapat O, Ducot B, et al. A randomized trial of endoscopic drainage methods for inoperable malignant strictures of the common bile duct. Gastrointest Endosc 1998; 47: 1-7. [ Links ]
20. Kaassis M, Boyer J, Dumas R, et al. Plastic or metal stents for malignant stricture of the common bile duct? Results of a randomized prospective study. Gastrointest Endosc 2003; 57: 178-82. [ Links ]
21. Rey JF, Dumas R, Canard JM, et al. Guidelines of the French Society of Digestive Endoscopy: biliary stenting. Endoscopy 2002; 34: 169-73, 81-5. [ Links ]
22. Moss AC, Morris E, Leyden J, MacMathuna P. Do the benefits of metal stents justify the costs? A systematic review and meta-analysis of trials comparing endoscopic stents for malignant biliary obstruction. Eur J Gastroenterol Hepatol 2007; 19: 1119-24. [ Links ]
23. Schmassmann A, von Gunten E, Knuchel J, Scheurer U, Fehr HF, Halter F. Wallstents versus plastic stents in malignant biliary obstruction: effects of stent patency of the first and second stent on patient compliance and survival. Am J Gastroenterol 1996; 91: 654-9. [ Links ]
24. Arguedas MR, Heudebert GH, Stinnett AA, Wilcox CM. Biliary stents in malignant obstructive jaundice due to pancreatic carcinoma: a cost-effectiveness analysis. Am J Gastroenterol 2002; 97: 898-904. [ Links ]
25. Yeoh KG, Zimmerman MJ, Cunningham JT, Cotton PB. Comparative costs of metal versus plastic biliary stent strategies for malignant obstructive jaundice by decision analysis. Gastrointest Endosc 1999; 49: 466-71. [ Links ]
26. Ballinger AB, McHugh M, Catnach SM, Alstead EM, Clark ML. Symptom relief and quality of life after stenting for malignant bile duct obstruction. Gut 1994; 35: 467-70. [ Links ]
27. Luman W, Cull A, Palmer KR. Quality of life in patients stented for malignant biliary obstructions. Eur J Gastroenterol Hepatol 1997; 9: 481-4. [ Links ]
28. Abraham NS, Barkun JS, Barkun AN. Palliation of malignant biliary obstruction: a prospective trial examining impact on quality of life. Gastrointest Endosc 2002; 56: 835-41. [ Links ]
29. Knyrim K, Wagner HJ, Pausch J, Vakil N. A prospective, randomized, controlled trial of metal stents for malignant obstruction of the common bile duct. Endoscopy 1993; 25: 207-12. [ Links ]











 text in
text in