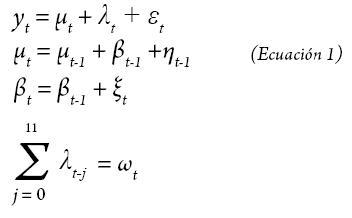Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957On-line version ISSN 2500-7440
Rev Col Gastroenterol vol.25 no.2 Bogotá Apr./June 2010
Epidemiological evolution of acid peptic disease in an Endoscopic Diagnostic Center in Bogotá from 1993 and 2007
Luis Alberto Ángel Arango, MD (1), Diego Gómez Boada (2), María Paula Villalba Cuadrado (2)
(1) Internist, Gastroenterologist, Nutritionist. Professor, Gastroenterology Unit, Department of Medicine, Faculty of Medicine. Universidad Nacional de Colombia. Bogotá, Colombia.
(2) Medical student VIII semester. Universidad Nacional de Colombia, Bogotá, Colombia.
Received: 31-07-09 Accepted: 26-05-10
Abstract
Objective: To describe epidemiological characteristics of individuals suffering from acid peptic disease found by a first level endoscopic diagnostic center from 1993 and 2007.
Methods: A retrospective, descriptive and analytical study of series of patients.
Results: 12,638 registers were included in the study. 62.3% of the patients women, 37.7% were men (mean 44.0 ± 16 years). Main endoscopy findings: peptic esophagitis 14.1%, gastric ulcer 4.1%, duodenal ulcer 7.0%, total acid peptic duodenal lesions 17%. Corporal gastric ulcers were most common (56.4%). Erosive gastric lesions represented 21% of cases. Only the frequency of duodenal acid peptic disease decreased significantly in the period observed. This was associated with increased consumption of proton pump inhibitors. No seasonal variation was observed in any of the lesions.
Conclusions: 1. Acid peptic disease is frequently observed at the first level endoscopic center, most frequently in men. 2. We described a significant reduction in the frequency of duodenal acid peptic disease associated with increased consumption of proton pump inhibitors. 3. It is necessary to realize multi-centric studies to confirm our results.
Key words
Endoscopic diagnostic, acid peptic disease, time series, peptic ulcer, epidemiology.
Introduction
Dyspepsia has a high prevalence in the general population. Studies show prevalences ranging from 8% to 54% depending on the definition and methodology used (1). High rates of dyspepsia have resulted in increased use of health services around the world (2). Although whether early diagnostic endoscopy or empirical therapy in patients without previous study is the most appropriate management and diagnosis strategy remains to be defined (3), in our environment early esophageal-gastro-duodenoscopy has been recommended (EGD) as the method of choice. This is due to the high prevalence of gastric cancer in some regions of Colombia (4) and the moderate prevalences among the general population (5-9). Endoscopy of the upper digestive tract is considered to be the gold standard for the study of dyspepsia (10). It allows macroscopic and histological identification of lesions from biopsies which can guide the proper treatment of the patient.
Acid Peptic disease includes a variety of lesions that can occur from the esophagus to the duodenum and even the jejunum and ileum in some cases. Gastric and duodenal ulcers are the main group and have the highest risk of morbidity and mortality related to complications (bleeding, perforation and obstruction) (11). The literature describes a progressive reduction in the frequency of gastric and duodenal ulcers (12-16) resulting from decreases in the rate of infection by Helicobacter pylori in the general population of developed countries (11, 13, 15-18), and in the middle and upper classes of developing countries (19, 20). Meanwhile, mortality rates from high gastrointestinal bleeding remain between 10% and 15% (12, 21, 22). Although trends in rates of Helicobacter pylori infection do not appear to have changed in our country (23-25), consumption of non steroidal anti-inflammatory drugs (NSAIDs) has increased very significantly throughout the world (13, 16, 18 ) including in Colombia (26). This is also associated with the main complication, bleeding peptic ulcers (11, 17, 21, 22, 26, 27). Some studies have also reported a seasonal variation of peptic ulcer disease and complications such as bleeding (12, 18, 22, 28-32) and perforation (33) during the months of March, April, September and October which may or may not be associated (28, 29, 30, 31) with the use of NSAIDs and seasonal variations in Helicobacter pylori infections. Some authors have affirmed that seasonal variation is particularly noticeable for dyspepsia and duodenal ulcers (32, 34), but other authors have been unable to confirm this, or have suggested that this periodicity has disappeared (35, 36, 37).
Endoscopic studies of large series of dyspepsia patients over time have not been conducted in Colombia, nor has anyone described a tendency toward seasonality. In our experience, observations of lesions related to exposure to acid have been made less and less frequently, but as of this writing no one in Colombia has quantified this decrease in the Colombian population as has been done elsewhere (12-16). By the same token no study has yet been made of the possibility of a tendency towards increased NSAIDS use in our country (28, 29). In Colombia the rate of Helicobacter pylori infection has remained stable and high, between 63% and 98% of patients (38-41) in the various descriptions depending on the pathology studied. Meanwhile consumption of potent inhibitors of gastric acid secretions has apparently been increasing with the emergence in the market of unrestricted sales of many low cost generic potassium pump inhibitors. Their indiscriminate use by the general population to self-treat dyspeptic symptoms could be a contributing factor in lower frequencies and reduced severity of peptic acid lesions observed at the time of diagnostic endoscopy.
The aim of this study, undertaken on a series of patients at a first level endoscopic diagnostic center between 1993 and 2007, is to determine the evolution of peptic acid disease frequency in terms of its trend, seasonality and other associated factors.
General objective
Describe the epidemiological characteristics of individuals with acid-peptic disease who were cared for at a first level diagnostic center between 1993 and 2007.
Specific objectives
1. Establish the frequency of acid peptic disease (esophagitis, gastric ulcer, duodenal ulcer, erosive gastritis and duodenitis) in patients referred for endoscopic evaluation of dyspepsia to a first level diagnostic center.
2. Learn about some of the demographic characteristics of subjects with peptic acid disease.
3. Determine the frequency of acid peptic disease in the period under review and its relationship with the frequency of use of risky drugs and with management of acid peptic disease.
4. Compare the results with a control population with no evidence of acid-peptic disease (chronic superficial gastritis, chronic atrophic gastritis and hiatal hernia) as determined by endoscopy. Compare the results with the frequency of Helicobacter pylori.
Subjects and methods
Type of study: A retrospective, descriptive and analytical study of a series of patients.
Subjects: The sample was obtained from subjects who were referred for diagnostic upper endoscopy after showing symptoms suggestive of acid peptic disease. All procedures were performed by an endoscopist. Patients underwent the examination as ordered by the attending physician and reports of findings were made. Biopsies were taken when directly requested by the attending physician or when deemed necessary by the endoscopist. The archives of all the patients evaluated were used to establish the relevant variables for this study: esophagitis, gastric ulcer, duodenal ulcer, acute and/or chronic gastritis (whether or not it was erosive) , and duodenitis (whether or not it was erosive).
A second group of subjects consisted of those patients who tested negative for acid peptic disease as well as for chronic superficial gastritis (GCS), chronic atrophic gastritis (ACG) and hiatal hernia, the frequency of which are unlikely to change over time
The frequency of Helicobacter pylori was assessed with the rapid urease test described by Arvind (42).
Methods
Each report included the consecutive number of the examination, the date, the patient's age, a description of the procedure, and a description of the lesions found. That description included the size, location and characteristics of the lesions and whether or not there were esophageal varices. Rankings were done according to a combination of the Savary-Miller, classification scheme for reflux esophagitis (43) and the Los Angeles Classification of Esophagitis 1 (starting in 1997) (44) which assigned grades of 1 to 4 for purposes of statistical analysis.
Gastric lesions were described as gastric ulcers when there was a parietal defect in the mucosa with a depth exceeding 1 mm. They were described as acute erosive gastritis when there were flat erosions in the corpus or antrum less than 1 mm. deep). Lesions were classified as chronic erosive gastritis when there were raised and eroding lesions of the mucous membrane with the appearance of acne varioliformis. Descriptions included locations (subcardial, corporal and pyloric antral) and maximum diameter in centimeters, and used the Sydney System for the classification of gastritis (45).
Duodenal lesions are classified as either non-ulcerative or ulcerative duodenitis. Non-ulcerative duodenitis includes non-erosive and erosive duodenitis. Non-erosive duodenitis is considered to be mild when congested areas are present. It is classified as moderate when congested areas are present together with edema which reduces the caliber of the bulb slightly and when the first portion does not fully distend with insufflation. It is considered to be severe when congested areas are present together with edema which reduces the caliber of the bulb so severely that the size and elasticity of the bulb become difficult to determine visually, but there is no presence of ulceration or erosion. Erosive duodenitis is defined by the presence of parietal defect in the mucosa which are less than one millimeter deep and less than one millimeter in diameter, and by variables related to lesion location. A duodenal ulcer is diagnosed when there is a parietal defect of the mucosa exceeding 1 mm in depth. Its stage is determined by the amount of scarring, whether or not there is linear or non-linear ulceration, whether or not the form is regular, oval, or rounded and, for the last two, by its diameter in centimeters (45).
Pathologies considered for use as control variables were those that, once established, are persistent and/or reversible only under special conditions. These included chronic superficial gastritis and hiatus hernia. Chronic superficial gastritis may be characterized only by patchy erythema. Patchy erythema with depressed areas with or without areas of apparent metaplasia in the corpus and antrum, will be called "by definition", chronic atrophic gastritis, despite its poor histological correlation (45). Hiatus hernia location is characterized by the length of the gastric folds above the esophageal hiatus, or the distance from the inferior esophageal sphincter to the esophageal hiatus, of greater than 2 cm.
All variables mentioned were analyzed as functions of time in years from the beginning to the end of the study. Their frequencies were measured monthly, bi-monthly, quarterly, and semi-annually throughout the entire study. Frequencies of acid peptic disease and controls, and the evolution of those frequencies, were compared over time.
A patient survey about overall consumption of medicine was conducted for the initial period of the series and the final period of the series. This information was used to compare frequency of consumption of antisecretory and medications to neutralize gastric acid (aluminum hydroxide, magnesium, H2 receptor antagonists and proton pump inhibitors) with the risk of developing peptic ulcer disease.
Ethical considerations
All subjects were ambulatory and had undergone a low risk diagnostic procedure (<2%) (46). The procedure had been prescribed and justified by either a general practitioner or specialist and then approved by the patient's EPS. In some cases the procedure had been requested from the EPS directly by the patient. Prior to performing the procedure the researchers checked the referral orders, diagnoses, indications and contraindications, and made sure that the patients' conditions were optimal for undergoing the procedure without risk. Then they routinely explained the procedure directly to all subjects before performing the procedure.
All subjects received a written informed consent form as required for such tests.
This research was consistent with the universal ethical principles for research involving human beings (47). It was an investigation without risk in which did not take into consideration the identities of any of the patients. It was based exclusively on the retrospective study of endoscopy reports and was conducted solely for academic purposes.
Statistical analysis
Data are presented as averages or proportions depending on the variable. Studies of the differences among the averages and proportions were conducted using Student's t-test, the Wilcoxon signed-rank test, the chi-square test, Spearman's rank correlation coefficient, and/or Pearson's correlation depending upon whether variables are parametric or nonparametric.
For statistical analysis of time series, the number of patients with the following peptic acid pathologies within the entire series were considered: esophagitis (grades 1-4); gastric ulcers; acute and chronic erosive gastritis; congestive and erosive (grades 1-4); ulcerative duodenitis (grades 1-3); and duodenal peptic acid disease (grades 1-7). In addition the three control variables, chronic superficial gastritis, chronic atrophic gastritis and hiatal hernia, were analyzed.
Statistical methods were used to analyze time series data from November 1993 to January 2007. The analysis took into account the premise that it is more appropriate to use absolute values than relative values with health-related problems, especially as they relate to the number of patients (48).
The analysis was done in two phases. TSW® was used in the first phase of the program. Because of the data configuration an analysis of missing data was performed using Kalman filters, the proposal of Gomez and colleagues (49), and smoothing techniques. For the best imputation of missing data analysis the analysis was first undertaken with a descriptive component for identification of missing data (damage to the original database) and other analytical components (see explanation in the Results section).
These data were entered into a database and analyzed using Excel statistical package (Microsoft Office ® 2003), Simstat for Windows 1.1 ® (Provalis Research Inc.) and SPSS 17 ® (SPSS Inc.) and STAMP 6.0 ®. (49) An alpha error of 5% was considered acceptable.
Results
In the period under study, a total of 15,500 upper endoscopies were preformed, of which 12,638 were included in the usable reports digital database. Lost reports were due to damage or loss of media or destruction or damage of files by computer viruses. The resulting population consisted of 4,765 men (37.7%) with an average age of 44.0 ± 16.1 years and with a standard error of 0.23, and 7,873 women (62.3%) with an average age of 44.6 ± 16.61 years and with a standard error of 0.19 (p <0.03. Patients ages ranged between 3 years old and 97 years old, with 211 minor patients.
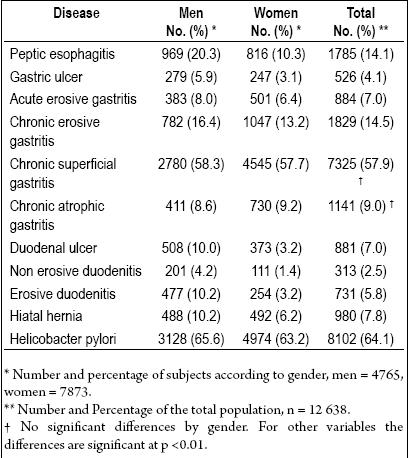
Table 1 shows the number and percentage of subjects with chronic peptic acid disease and the endoscopic diagnoses chosen to be used as controls in this study. The most frequently occurring was chronic erosive gastritis, followed in order of frequency by peptic esophagitis and duodenal ulcer. All conditions included in the study were more frequent among men than among women except for chronic superficial gastritis and chronic atrophic gastritis. The ulcerative lesion size was 1 ± 0.84 cm (median = 0.8 cm) for gastric ulcers and 0.74 ± 0.39 cm (median = 0.7 cm) for duodenal ulcers with no differences according to gender.
Table 1. Global frequency and gender of some endoscopic diagnostic in a first level diagnostic endoscopy center in Bogotá between 1993 and 2007.
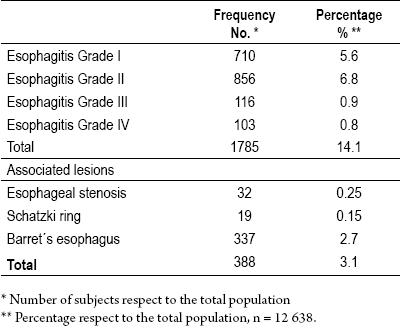
Secondary complications of acute and chronic esophageal lesions resulting from peptic acid action were found in 14% of the patients examined (as shown in Table 2). Most of these cases were not severe. Our findings were suggestive of Barrett's esophagus in 19% of the patients with esophagitis.
Table 2. Lesion frequency of acid peptic esophageal in the endoscopy center, of first level in Bogotá between 1993 and 2007.
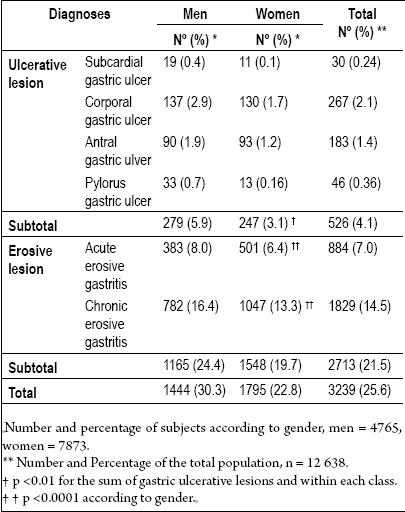
Regarding gastric lesions (Table 3) chronic erosive gastritis is most common, followed by acute erosive gastritis and finally ulcers. Among the ulcers the subcardial ulcers and corporal ulcers, both related primarily to gastric hyposecretion, represent just over half of the ulcerative lesions (56.4%). The remaining ulcers are related to gastric acid hypersecretion (43.6%). The number of chronic erosive lesions is twice the number of acute erosive lesions. These two findings were very common, comprising 21.5% of the examined population. In all cases, all lesions occurred significantly more frequently among men.
Table 3. Stomachal acid peptic frequency in a diagnostic center of first level in Bogotá between 1993 and 2007
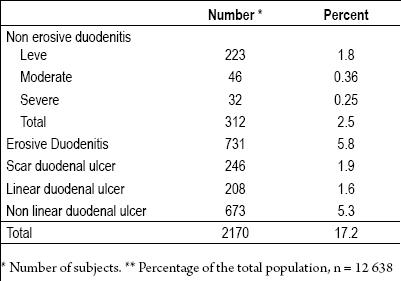
Table 4 shows that for acid peptic duodenal lesions the frequency of linear and nonlinear ulceration taken together slightly exceeded the frequency of erosive duodenal lesions and were three times greater than non-erosive duodenal lesions. In total 17% of all patients evaluated were diagnosed with acid peptic duodenal lesions.
Table 4. Frequency of acid peptic lesion of duodenum in a diagnostic endoscopy center of first level in bogota between 1993 and 2007.
Frequencies of diagnoses designated as controls can be seen in Table 1. There are no differences in frequency between genders for chronic superficial gastritis and chronic atrophic gastritis which together comprise 67% of the diseases observed. The sum of the lesions mentioned is greater than 100% because of overlapping pathologies (particularly esophagitis and hiatal hernia) and because of lower frequency of other diseases. Rapid urease tests for helicobacter pylori showed a frequency of 64% with a small but significant gender difference of 2.4% (p <0.01) (Table 1).
Time series analysis
We first analyzed the major acid peptic disorders: gastric ulcers, peptic esophagitis and duodenal ulcers. The periods of observation are located at the abscissae (x coordinates). The raw proportions of subjects with each lesion, defined as the number of subjects with a lesion divided by the total examined during that month, are located at the ordinates (y coordinates).
As mentioned above, some of the data were missing (These missing data are clearly visible in Figures 1 and 2. To estimate the missing data we used Program TSW statistical software with the method proposed by Gomez and colleagues (49). This method produced optimal estimates of missing values in autoregressive moving average (ARMA) models through development of a Kalman filter (49). A maximum likelihood estimator (MLE) for the model parameters was then obtained. This was followed by the use of a smoothing algorithm. Interpolation of missing data was performed using both a fixed-point smoother and additive approximation for atypical data. MLEs for ARMA model parameters were calculated using approximation of atypical additive parameters assuming that where these values were absent any value could be used and treated as an atypical additive. As can be seen in Figures 1 and 2, this interpolation of x and y values (as defined above, and after making the corrections outlined above) provides continuity to the series in the whole interval under study. Thus, techniques for handling missing data in these time series conserve the temporary components that describe and characterize the time series.
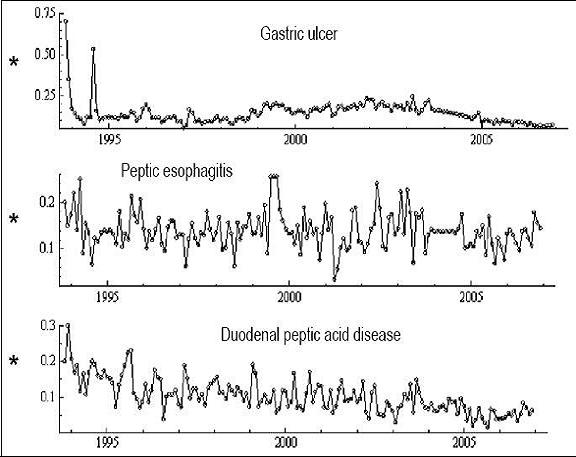
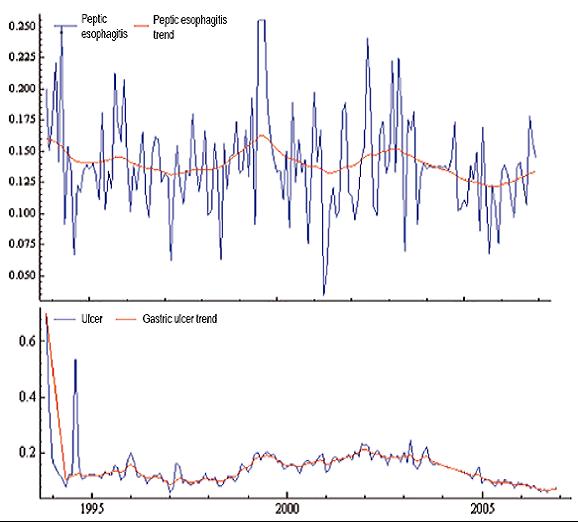
Figure 1. Interpolated series of peptic acid diseases. The Y axis corresponds to the raw proportion of cases of the total population and the X axis the period between 1993 and 2007. The graph shows the interpolated time series for gastric ulcer gastric lesion groups in terms of their location, peptic esophagitis grades 1-4 according to Savary-Miller and Los Angeles and duodenal peptic acid disease in which diagnoses are grouped non-erosive duodenitis, erosive and ulcerative in one variable (see text for explanation).
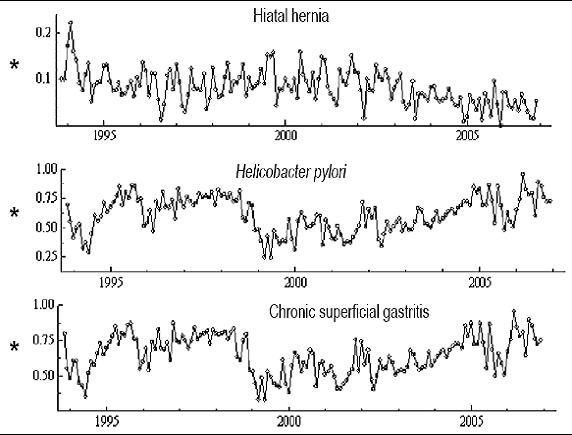
Figure 2. Interpolated series of control variables. The Y axis corresponds to the raw proportion of the total cases and the X axis the period between 1993 and 2007. It shows the interpolated time series for the control entities: hiatal hernia, Helicobacter pylori and chronic superficial gastritis.
As the main interest was to analyze trends of variables over time, we used the STAMP (Structural Time Series Analyser, Modeller and Predictor) 6.0 program (50) to apply structural equation modeling to separate the different components. A structural equation model is determined by: (Equation 1)
With these models for analyzing time series we were able to break down the different components of our time series through the use of Kalman filters (50) to obtain an estimate of the stochastic trend of the observed series (μt), the stochastic seasonality effect (λt) that is described by qualitative variables that met the fourth condition in equation 1 with ωt, a white noise process with zero mean and variances of σ2ω, y, εt, ηtyξt, and also inter-correlated white noise processes with mean 0 and σ2εt , σ2ηt , σ2ξt variance respectively.
These models allow more versatility than the use of the X-11 filter and provide components that are expected to preserve behaviors such as trend, seasonality and the irregular component. In this way once we decompose the time series observed in these components, we can more clearly analyze the trend and seasonality of the series.
At the time of adjustment of each of the models different possible atypical data were considered for use in adjusting each model according to the indications. To evaluate the evolution of the slope and seasonality of curves the vector of the final state was evaluated to determine the significance of each of the components in the temporal series. For the diagnoses of the models nonparametric tests of randomness of the residues of the model were applied. The Box-Pierce test and the Ljung-Box test were used to determine non-correlation of model residues and thus to evaluate each of the proposed models.
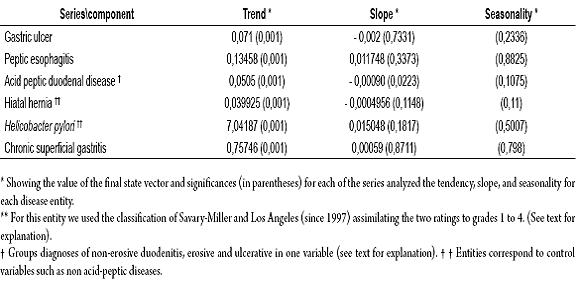
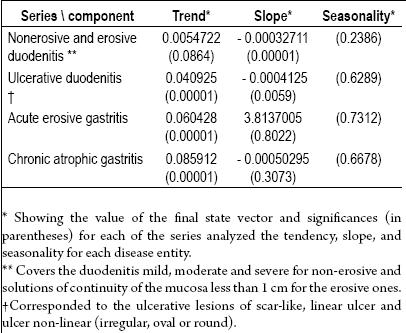
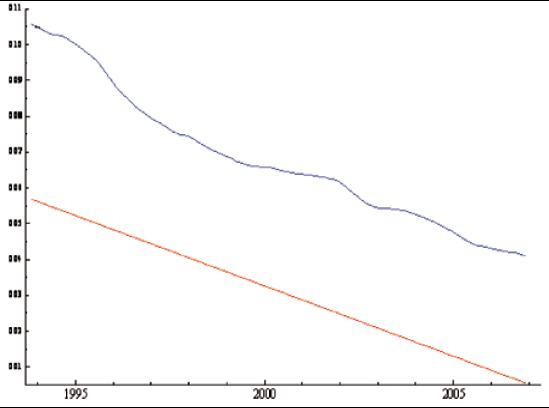
Table 5 shows the significance of the final state vector for the trend, slope and seasonality of all the series analyzed followed by their respective p-values in parentheses. Although the trend components (The overall average of the series which varies over time) for almost all variables tabulated show statistical significance, the model is unstable once the slope is determined for each variable. There is one exception: duodenal peptic acid disease which had a negative significance for its slope of (=βt-0.00090; p = 0.0233). In Figure 3, until the middle of 1994 gastric ulcers occur atypically in a relatively high number of patients, then for the rest of the study period it maintains stable behavior. Frequency of peptic esophagitis decreases near the end of the study, although in the years 1999 and 2003 there are ascending peaks, without significant slopes.
Table 5. Temporary components estimates for peptic acid disease and control variables.
Figure 3. Estimated components for variables peptic esophagitis and gastric ulcer. The Y axis corresponds to the raw proportion of cases of the total population and the X axis the period between 1993 and 2007. The graph shows the estimated trend component for the time series of gastric ulcer.
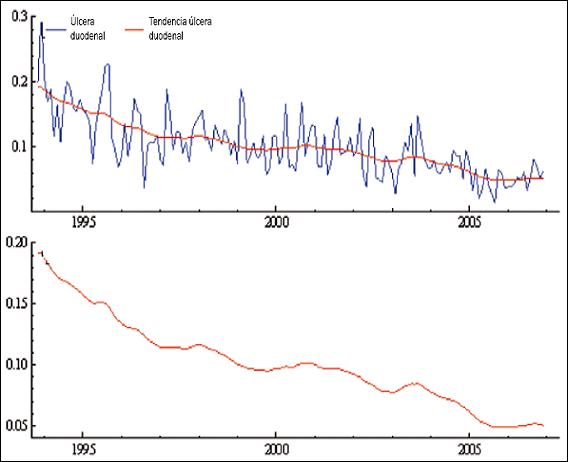
For the various acid peptic duodenal lesions a continuum has been hypothesized. For both non-erosive and erosive non-ulcerative lesions the continuum is based on severity, while for ulcerative lesions it is based on both activity and severity. Mild to severe duodenitis is ranked as grades 1-3 grades (according to reports of endoscopy), and erosive duodenitis is ranked as grade 4. Duodenal ulcers in an advanced state of healing are classified as grade 1, linear ulcers are classified as grade 2, and non-linear ulcers are classified as grade 3 (The three of them were so reported in the endoscopic report.) Finally, the two classifications were combined into grades 1 to 7. Ulcerations are ranked from 5 to 7 according to the severity of scarring and whether or not the lesion is linear or nonlinear. As shown in Table 5 and Figure 4, ulceration was the only variable that showed significance in the slope of the model, demonstrating constantly decreasing behavior throughout the period.
Figure 4. Estimated componentfor the variable duodenal acid peptic disease. The Y axis corresponds to the raw proportion of the total cases and the X axis the period between 1993 and 2007. The graph shows the estimated component of tendency for the time series of duodenal acid peptic disease and their tendency.
The same methodology was used for the control variables. They show decreasing frequency of hiatal hernia between the start of this study and 1997 followed by a slight increase of frequency which persists until 2002, after which frequency again decreases. No slope was identified for the period observed. The frequencies of Helicobacter pylori infections and chronic superficial gastritis behaved in a very similar manner, increasing increase between 1996 and 1998, followed by a decrease in the proportion of diagnosed cases for these two variables up until mid-1999 and a progressive increase of the two entities until 2005, and slightly oscillate until the end of the series, but without showing any definitive slope.
The others entities in the study, acute erosive gastritis, chronic erosive gastritis and chronic atrophic gastritis are analyzed using the same process with similarly plotted time series for each, plus their respective estimates and graphic interpolations of missing data. Trend, slope, and seasonality were studied for each of these (Table 6), as they were for the other entities. Despite the apparent trend for chronic erosive gastritis, there were no significantly declining slopes for any of these three lesions. Similarly, none had significant seasonality.
Table 6. Estimated temporary components for duodenal peptic acid disease, erosive gastritis and chronic atrophic gastritis.
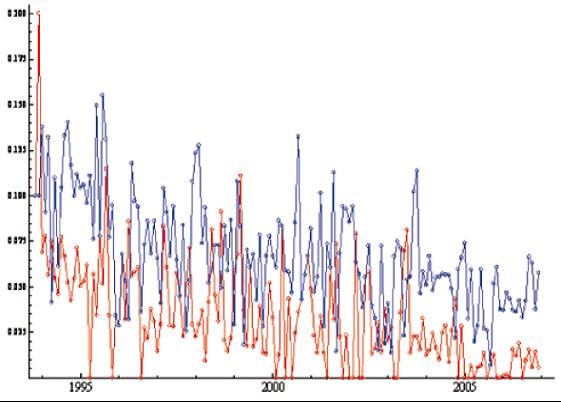
Given that all duodenal lesions were classified as a single variable, we proceeded to analyze them as two variables: ulcerative and non ulcerative (non-erosive and erosive). We identified whether these two were behaved similarly. Figure 5 shows the time series for non-ulcerative and ulcerative lesions. The interpolation of missing data and trends and slopes for these two types of lesions are shown in Table 6 and Figure 6. Significant reductions in time, but without seasonal patterns, were found for both types of lesions. Seasonality was also absent in the analysis of the control variable, chronic atrophic gastritis.
Figure 5. Interpolated time series of the two groups of duodenal peptic acid disease. The Y axis corresponds to the raw proportion of cases of the total population and the X-axis the period between 1993 and 2007. The graph shows the time series interpolated for the two groups of acid peptic disease, duodenitis mild to severe as grades 1-3 and erosive duodenitis as Grade 4 (-o-o-), ulcer in healing process as degree 5, linear ulcer grade 6 and non-linear ulcer to grade 7 (-o-o-).
Figure 6. Estimated tendency components of two groups of duodenal peptic acid disease. The Y axis corresponds to the raw proportion of cases of the total population and the X-axis the period between 1993 and 2007. The graph shows the trend for the two groups of duodenal peptic acid disease: duodenitis mild to severe as grades 1-3, and erosive duodenitis Grade 4 (-o-o-) and the state of healing ulcers as grade 5, linear ulcer and ulcer grade 6 to grade 7 non-linear (-o-o-).
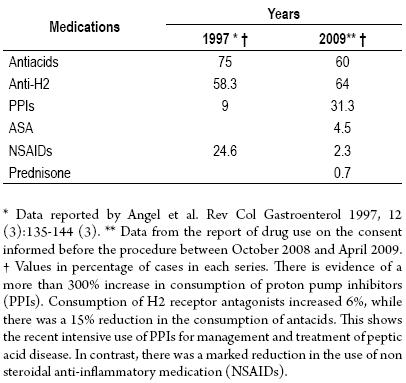
Table 7 shows the frequency of consumption of both medications for protection against peptic acid disease and of medications with risks of causing or aggravating peptic acid by patients who underwent upper gastrointestinal endoscopies in two different periods. Data from the first period, which corresponds to the beginning of our series, were reported by Angel et al in 1997 for patients with gastric and duodenal ulcers (38). Data for the second period is drawn from the medication consumption report in the preliminary informed consent procedure of our service. There is evidence of a more than 300% increase in consumption of proton pump inhibitors (PPIs). Consumption of H2 receptor antagonists increased 6%, while there was a 15% reduction in the consumption of antacids. This shows the recent intensive use of PPIs for management and treatment of peptic acid disease. In contrast, there was a marked reduction in the use of non steroidal anti-inflammatory medication (NSAIDs).
Table 7. Consumption of protective and risk medications for acid peptic disease in patients under dyspepsia study before 1997 and 2008-2009.
Discussion
This study presents the first set of results from patients of an endoscopy service in Colombia spanning a long period of time. It has allowed us to establish the behavior of different pathologies over time, especially peptic acid disease. The only other similar time series have covered short periods of time. These include that by Laverde and Fassler which reported the overall results of a five year period (51) and a study by Sierra et al which reviewed 20,000 reports of endoscopies performed between 1983 and 1994 to diagnose upper GI bleeding (26).
The distribution of patients by sex and age in our study was similar to that in the series by Laverde (51), although that study was restricted to chronic gastritis, gastric ulcers and gastric cancer. Small series of Colombian patients screened for peptic acid diseases have shown frequencies for chronic superficial gastritis similar to those we are reporting, albeit they reported lower frequencies for other diseases (25). This could be explained by tests on asymptomatic individuals.
Comparisons with three foreign series show interesting similarities and differences with our study. The 11.1% frequency of gastric and duodenal peptic ulcers in our series was lower than the 14.9% frequency for these lesions found by Wai et al (52), but our finding of a 14.1% frequency of esophagitis was higher than their report of 5% for this condition. Our findings were similar to those of Voutilainen (53) for gastric ulcers, duodenal ulcers and gastritis. In contrast, Bartels found a frequency of gastric ulcers of 10%: twice that found in our study. That study found similar frequencies of peptic esophagitis and duodenal ulcers (54). The predominance of occurrences among males for some of these lesions was also described in the last mentioned series. It also described increases of diagnoses of chronic superficial and atrophic gastritis, among other lesions, with advancing age (26, 50). Table 1 shows the predominance of the most frequent diagnoses among men with the exceptions of chronic superficial gastritis and chronic atrophic gastritis. In the case of Helicobacter pylori, the difference, although significant, was only 2.4%.
Peptic esophagitis is a common finding, accounting for 14.1% of subjects examined with a 2:1 ratio for males (Table 1). Mild forms of erosive esophagitis predominate, while there are low frequencies of extensive lesions, scarring and the metaplasia of Barrett's esophagus (Table 2) (55). Given that diagnoses of the metaplasia of Barrett's esophagus by endoscopy is confirmed by histology nearly 60% of the time (55), these endoscopic findings are significant even though they are not frequent. They are significant because of their implications for treatment and follow-up although naturally these diagnoses require histological testing for confirmation (55).
Acid peptic disorders include the ulcerative lesions already mentioned, as well as the acute and chronic erosions shown in Table 3. These are more common among males than among females, as described in the literature (56). Gastric ulcers are less common than duodenal ulcers (Table 3), similar to findings reported by other authors (38, 40, 41). There are a few other descriptions of the frequency of erosive lesions in endoscopy series in Colombian literature. Gomez (41) reported a similar frequency of 5.8% for gastric erosions. In bulletins from the National Day of Digestive Endoscopy in 1999-2002 (57, 58) the Colombian Association of Gastrointestinal Endoscopy reported a frequency for gastric erosions of 8.2%. Both are lower than the frequency we report. This can probably be explained by the fact that these were series related to gastric cancer screening in which a large number of participants were asymptomatic or had minor discomfort. In contrast our series included only patients whose dyspeptic discomfort was B enough for their doctors to order a diagnostic study.
The frequency of duodenal ulcers that we found (Table 4) is higher than the one described by Gomez (41), but lower than that described by Wong. This included gastric ulcers (19) and variables according to those described by Sung (17). There are no series comparable to ours about congestive and erosive lesions. Their frequency was described as being 4.2% in the 2002 bulletin of the Colombian Association of Gastrointestinal Endoscopy. This was slightly lower than the frequency we reported, probably for the same reasons mentioned above in the case of gastric erosions.
The entities used as controls for comparison have frequencies that match those reported in the bulletins of the Colombian Association of Gastrointestinal Endoscopy mentioned above and which were within the ranges reported in the literature (23, 41, 51, 55, 59, 60).
For the study of epidemiology in the time period of the study, time series studies were conducted to test the hypothesis of declining frequencies of acid peptic diseases and to compare them with esophageal and gastro-duodenal lesions, pathologies and diseases unrelated to acid secretion. These should not differ if acid secretion levels have changed positively or negatively in the population because of the use of mucous protectors or mucous aggressors. The study also attempted to use the same methodology to evaluate the seasonality hypothesis for peptic ulcers advanced by some authors (12, 18, 22, 28-33).
Figures 1 and 2 graph the evolution over time of major peptic acid lesions. They have been corrected by the addition missing data to allow the analyses to be made. The resulting vectors are shown in Table 5, Figure 3 and Figure 4. They show that frequency of duodenal peptic acid disease has a significantly decreasing slope for this time series (Figure 3 and 4). This is the first time this has been demonstrated to be a fact here in Colombia, as had already been done in descriptions in international literature (12-20). Although in some of those cases this decline was associated with reductions in the prevalence of Helicobacter pylori, this was not the case in our series. It demonstrated no significant variation over time for H. pylori prevalence. In those other tests the frequencies of positive urease tests is similar to the average of ours at all times (38-41, 59). The two series reported by the Colombian Association of Gastrointestinal Endoscopy in bulletins of the National Day of Digestive Endoscopy (57, 58) also have similar frequencies for positive urease test results. For the other acute and chronic erosive gastric lesions, as well as for control entities (Table 6), there were also no significant changes over time. Seasonality was ruled out for all lesions studied (Tables 5 and 6), consistent with descriptions by some other authors (35-37).
To establish whether or not that hypothesis that clustering of duodenal peptic acid diseases occurs is incorrect, non-ulcerative lesions and ulcers were analyzed separately. We found the same downward trend for ulcers and non-ulcerative lesions (Figures 5 and 6), corroborating the hypothesis that duodenal acid peptic disease is decreasing. This reduction could be due to increasing consumption of PPIs, including without prescriptions (13, 61, 62). Since PPIs are capable of reducing and curing acid peptic lesions (63, 64), this could account for the observation shown in Table 7. This table shows significantly increased PPI consumption and reduced consumption of NSAIDs. This is at variance with other findings described in literature, however it should be noted that elevated consumption is very common among patients with increased risk of development of complicated peptic ulcers, especially gastrointestinal bleeding (17, 21, 24, 26, 38), but not for the general population who undergo endoscopies from which our series was drawn.
These two factors combined, PPIs and NSAID, could explain how and why endoscopic manifestations of duodenal acid peptic disorders have decreased over the course of the 14 years of this series. Proton pump inhibitors and H2 receptor antagonists have gained an important place as the first choices for management and treatment of peptic acid disease and self-medication of symptoms. This is partly because of their unrestricted sale, but also because of the exuberant presence of very low cost generic medications.
The variability observed for duodenal peptic acid disease differs from those observed for gastric and esophageal peptic acid diseases. This is probably due to the pathophysiologies associated with these lesions in which the role of the acid is less important and is combined with other factors. This is the case for gastric ulcers. More than half were located in the proximal stomach where they can be affected by gastric hyposecretion, unlike antral ulcers and pyloric ulcers (56). They would require a greater number of lesions to establish such an association if it existed. Similarly, gastroesophageal reflux disease is characterized by recurrent periods of time in which severe lesions are stable (55, 65). These are associated with mechanical, anatomical and motility factors.
A weakness of this study is the lack of histological findings when it came time to take samples and establish a true diagnosis. This is true even though endoscopic and histopathological diagnoses do not always match precisely. Sydney system diagnoses (45) for peptic ulcer and erosions visible in endoscopy do not correlate very precisely with histopathological diagnoses in relation to gastritis, although the match is quite good for diagnoses of ulcerative lesions (66). On the other hand, consumption was not systematically assessed for each patient. They are referred to a series contemporary to the start of this study. Specifically for patients with peptic ulcers (38), we know that they have a history of increased consumption of drugs (NSAIDs) which favor this. Also, information about consumption at the end of the series is from informed consent forms rather than from a specific survey. However, these two factors favor a larger gap after the initial period given that the bias is against our hypothesis. This would suggest higher consumptions of NSAIDs and PPIs than in the general population, whereas the spontaneous, unguided and undirected reports of patients on the informed consent forms support favors lower consumption rates. This does not prevent demonstration of a significant difference.
Prospective multicenter studies are needed since they would allow concentration of a large number of patients for assessment for each period, and would thus allow better analyses of trends and the possible seasonality of the lesions under study.
Conclusions
1. Acid peptic disease is a common finding in first level medical diagnostic centers. The frequency is higher among men than among women.
2. The frequency of duodenal peptic acid disease has significantly declined in the last 14 years, probably associated with increased consumption of protective medications.
3. No seasonality was observable for any of the peptic acid lesions and other lesions evaluated.
4. Multicenter studies are needed to corroborate our findings.
Acknowledgements
The authors thank Mr. Ronald Fernery Herrera Calvino, who did most of the statistical analysis, and without whose intervention this manuscript would have not been possible.
Conflicts of interest
None. The costs of this study were born in full by the researchers.
References
1. Heading RC. Prevalence of upper gastrointestinal symptoms in the general population: a systematic review. Scand J Gastroenterol Suppl 1999; 231: 3-8.
2. Schwartz MD. Dyspepsia, peptic ulcer disease, and esophageal reflux disease. West J Med 2002; 176: 98-103.
3. Hammer J, Eslick GD, Howell SC, Altiparmak E, Talley NJ. Diagnostic yield of alarm features in irritable bowel syndrome and functional dyspepsia. Gut 2004; 53: 666-672.
4. Correa P, Cubillos C, Duque E, Barbosa O, Erazo JH. Patología geográfica del carcinoma y de la metaplasia intestinal del estómago en Colombia. Antioquia Médica 1969; 19(5): 41-56.
5. Ángel LA, Giraldo A, Pardo CE. Mortalidad por cánceres del aparato digestivo en Colombia entre 1980 y 1998. Análisis de tendencias y comparación regional. Rev Fac Med Univ Nac Colomb 2004; 52(1): 19-37.
6. Ángel LA, Giraldo A, Pardo CE. Tasas de mortalidad por canceres del tubo digestivo según género y grupos de edad en Colombia entre 1980 y 1998. Rev Colomb Gastroenterol 2007; 23(2): 124-35.
7. Medina MR. Atlas de mortalidad por cáncer. Colombia 1990. 1ª edición. Instituto Nacional de Cancerología. Talleres de Nuevas Ediciones, Santafé de Bogotá, Colombia 1994.
8. Instituto Nacional de Cancerología, E.S.E., Instituto Geográfico Agustín Codazzi IGAC. Atlas de mortalidad por cáncer en Colombia. Imprenta Nacional de Colombia, Bogotá D.C. 2003.
9. Piñeros M, Ferlay J, Murillo R. Cancer incidence estimates at the national and district levels in Colombia. Salud Pública Mex 2006; 48: 455-465.
10. Asociación Española de Gastroenterología, Sociedad Española de Medicina de Familia y Comunitaria y Centro Cochrane Iberoamericano. Grupo de trabajo de la guía de práctica clínica sobre dispepsia. Manejo del paciente con dispepsia. Guía de práctica clínica. Barcelona 2003.
11. Soll AH, Graham DY. Peptic ulcer disease. En: Yamada T. Textbook of Gastroenterology. Fifth edition. Blackwell Publishing Ltd. Oxford, USA 2009. p. 955-981.
12. Bobadilla J, Vargas-Vorackova F, Gómez A, Jesus Villalobos J. Incidence of peptic ulcer at the Instituto Nacional de la Nutrición "Salvador Zubirán": study of localization, associated factors and temporal trends. Rev Gastroenterol Mex 1996; 61(1): 31-5.
13. Perez-Aisa MA, Del Pino D, Siles M, Lanas A. Clinical trends in ulcer diagnosis in a population with high prevalence of Helicobacter pylori infection. Aliment Pharmacol Ther 2005; 21(1): 65-72.
14. de Jesus Villalobos Perez J, Quinones Menendez NF, Cruz Lomeli G, Vargas Vorackova F. Epidemiology of peptic ulcer at the "Salvador Zubirán" National Institute of Nutrition. Rev Gastroenterol Mex 1990; 55(2): 55-60.
15. Lam SK. Aetiological factors of peptic ulcer: perspectives of epidemiological observations this century. J Gastroenterol Hepatol 1994; 9(Suppl 1): S93-8.
16. Sadic J, Borgström A, Manjer J, Toth E, Lindell G. Bleeding peptic ulcer - time trends in incidence, treatment and mortality in Sweden. Aliment Pharmacol Ther 2009; 30: 392-398.
17. Sung JJY, Kuipers EJ, El-Serag HB. Systematic review: the global incidence and prevalence of peptic ulcer disease. Aliment Pharmacol Ther 2009; 29: 938-946.
18. Arents NL, Thijs JC, van Zwet AA, Kleibeuker JH. Does the declining prevalence of Helicobacter pylori unmask patients with idiopathic peptic ulcer disease? Trends over an 8 year period. Eur J Gastroenterol Hepatol 2004; 16(8): 779-83.
19. Wong SN, Sollano JD, Chan MM et al. Changing trends in peptic ulcer prevalence in a tertiary care setting in the Philippines: a seven-year study. J Gastroenterol Hepatol 2005; 20(4): 628-32.
20. Ramirez Ramos A, Chinga Alayo E, Mendoza Requena D, Leey Casella J, Segovia Castro MC, Otoya C. Changes in the prevalence of H. pylori in Peru; during the 1985-2002 period in medium and upper socio-economic strata. Rev Gastroenterol Peru 2003; 23(2): 92-8.
21. Van Leerdam ME. Epidemiology of acute upper gastrointestinal bleeding. Best Pract Res Clin Gastroenterol. 2008; 22(2): 209-24.
22. Bjorkman DJ. Endoscopic diagnosis and treatment of nonvariceal upper gastrointestinal hemorrhage. En: Yamada T. Textbook of Gastroenterology. Fifth edition. Blackwell Publishing Ltd. Oxford, USA 2009. p. 3018-3031.
23. García del Risco F, Caraballo JM, Urbina D. Helicobacter pylori en sujetos asintomáticos, con úlcera duodenal, úlcera gástrica y dispepsia no ulcerosa en Cartagena. Rev Col Gastroenterol 1994; 9(4): 188-93.
24. Bejarano M, Méndez F. Interacción de los factores asociados a enfermedad úlcero-péptica. Rev Col Gastroenterol 2006; 21(1): 25-35.
25. Gómez MA, Ricaurte O, Gutiérrez O. Costo efectividad de la endoscopia digestiva alta como prueba diagnóstica en una campaña para detección de cáncer gástrico. Rev Col Gastroenterol 2009; 24(1): 34-50.
26. Sierra F, Hernández BE, Botero RC, De Molano B. Hemorragia de vías digestivas altas, incidencia en 20.000 endoscopias. Rev Col Gastroenterol 1994; 9(4): 175-85.
27. Higham J, Kang J-Y, Majeed A. Recent trends in admissions and mortality due to peptic ulcer in England: increasing frequency of haemorrhage among older subjects. Gut 2002; 50: 460-464.
28. Yen FS, Wu JC, Wang LM, Kuo BI, Hu SC, Lee SD. Seasonal variation in the incidence of peptic ulcer and esophageal variceal bleeding in Taiwan. Zhonghua Yi Xue Za Zhi (Taipei) 1996; 57(1): 22-7.
29. Sezgin O, Altintaş E, Tombak A. Effects of seasonal variations on acute upper gastrointestinal bleeding and its etiology. Turk J Gastroenterol 2007; 18(3): 172-6.
30. Thomopoulos KC, Katsakoulis EC, Margaritis VG, Mimidis KP, Vagianos CE, Nikolopoulou VN. Seasonality in the prevalence of acute upper gastrointestinal bleeding. J Clin Gastroenterol 1997;25(4):576-9.
31. Zimmerman J, Arnon R, Beeri R et al. Seasonal fluctuations in acute upper gastrointestinal bleeding: lack of effect of nonsteroidal anti-inflammatory drugs. Am J Gastroenterol 1992; 87(11): 1587-90.
32. Moshkowitz M, Konikoff FM, Arber N. Seasonal variation in the frequency of Helicobacter pylori infection: a possible cause of the seasonal occurrence of peptic ulcer disease. Am J Gastroenterol 1994; 89(5): 731-3.
33. Wysocki A, Kaminski W, Krzywon J. Seasonal periodicity of perforated peptic ulcers. Przegl Lek 1999; 56(3): 189-91.
34. Xirasagar S, Lin H-C, Chen C-S. Role of Meteorological Factors in Duodenal Ulcer Seasonality. A Nation-wide, Population-Based Study. J Gen Intern Med 2007; 22(10): 1439-46.
35. Tulassay Z, Papp J, Lengyel G, Szathmari M. Seasonal disposition of gastroduodenal ulcer--legend or reality? Wien Med Wochenschr 1987; 137(4): 76, 78.
36. López-Cepero JM, López-Silva ME, Amaya-Vidal A et al. Influencia de los factores climáticos en la incidencia de hemorragia digestiva alta. Gastroenterol Hepatol 2005; 28: 540-5.
37. Soszka A, Kusz-Rynkun A. Seasonal fluctuation in peptic ulcer occurrence: a disappearing phenomenon? Pol Merkuriusz Lek 1998; 4(22): 208-9.
38. Angel LA, Gutiérrez O, Rincón D. Epidemiología de la enfermedad ácido péptica. Rev Col Gastroenterol 1997; 12(3): 135-144.
39. Gutiérrez O. Aspecto endoscópico en empedrado de la mucosa gástrica (Gastritis folicular): relación con el Helicobacter pylori. En: Sociedad Colombiana de Gastroenterología (editores). Temas escogidos de Gastroenterología. Tomo XXVII. Gráficas Aguilera Ltda., Santafé de Bogotá 1993. p. 75-94.
40. Martínez JD, Henao SC. Hiperplasia linfoide folicular gástrica e infección por Helicobacter pylori en adultos colombianos. Rev Col Gastroenterol 2009; 24: 148-156.
41. Gómez MA, Ricaurte O, Gutiérrez O. Costo efectividad de la endoscopia digestiva alta como prueba diagnóstica en una campaña para detección del cáncer gástrico. Rev Col Gastroenterol 2009; 24(1): 34-50.
42. Arvind AS, Cook RS, Tabaqchali S, et al. One minute endoscopy room test for Campylobacter pylori. Lancet 1988: 1: 704.
43. Ollyo JB, Lang F, olliet C, Monnier P. Savary-Miller's new endoscopic grading of reflux-oesophagitis: a simple, reproducible, logical, complete and useful classification. Gastroenterology 1990; 98: A100.
44. ArmB D, Bennett JR, Blum AL, et al. The endoscopic assessment of esophagitis: a progress report of observer agreement. Gastroenterology 1996; 111: 85.
45. Tygat NJ. The Sydney System: endoscopic division. Endoscopio appearances in gastritis/duodenitis. J Gastroenterol Hepatol 1991; 6: 223-234.
46. Santacoloma M. Indicaciones y complicaciones. En: Roldan LF, Hani AC, Aponte DM et al (editores). Técnicas en endoscopia digestive. ACED Asociación Colombiana de Endoscopia Digestiva, Bogotá D.C., Colombia, 2007.
47. Ministerio de Salud Nacional, República de Colombia. Resolución No. 008430. Por la cual se establecen las normas científicas, técnicas y administrativas para la investigación en salud. Rev Col Psiquiatría 1996; 25(1): 38-59.
48. Sanchez. R, Tejada P. Martinez J. Comportamiento de las muertes violentas en Bogotá, 1997-2003. Revista de Salud Pública 2005; 7(3): 254-267.
49. Gómez V., Maravall A., Peña D. Missing Observation in ARIMA models: Skipping approach versus additive outlier approach. Journal of Econometrics 1999; 88: 341-363.
50. Koopman S.J, A.C. Harvey, J.A Doornik and N. Shephard, STAMP 6.0: Structural Time Series Analyser, Modeller and Predictor, London: Timberlake Consultants Press. 2000.
51. Laverde M, Fassler S. Gastritis crónica: factores de riesgo, correlación endoscópica-histológica. En: Sociedad Colombiana de Gastroenterología (editores). Temas escogidos de Gastroenterología. Tomo XXV. Bogotá 1989. p. 93-108.
52. Wai CT, Yeoh KG, Ho KY, Kang JY, Lim SG. Diagnostic yield of upper endoscopy in Asian patients presenting with dyspepsia. Gastrointest Endosc 2002; 56(4): 548-51.
53. Voutilainen M, Mantynen T, Kunnamo I, Juhola M, Mecklin JP, Farkkila M. Impact of clinical symptoms and referral volume on endoscopy for detecting peptic ulcer and gastric neoplasms. Scand J Gastroenterol 2003; 38(1): 109-13.
54. Bartels F, Hahn HJ, Stolte M, Schmidt-Wilcke HA. Quality of diagnostic procedures and frequency of endoscopically defined diseases of the upper gastrointestinal tract. Z Gastroenterol 2003; 41(4): 311-8.
55. Vakil N, Zanten SV van, Kahrilas P, Dent J, Jones R, Global Consensus Group. The Montreal Definition and Classification of Gastroesophageal Reflux Disease: A Global Evidence-Based Consensus. Am J Gastroenterol 2006; 101: 1900-1920.
56. Rengifo A, De Lima E. Úlcera péptica. En: Alvarado J, Otero W, Archila PE et al (editores). Gastroenterología y Hepatología. Segunda edición. Editorial Médica Celsus, Bogotá, Colombia 2006. p. 529-547.
57. Asociación Colombiana de Endoscopia Digestiva. Día Nacional de la Endoscopia. Santafé de Bogotá, D.C. 1999.
58. Asociación Colombiana de Endoscopia Digestiva. Boletín Informativo: Informe Especial: Día Nacional de la Endoscopia Digestiva. Cartagena, Colombia 2002.
59. Otero W, Hani A, Arbeláez V. Enfermedad por reflujo gastroesofágico. En: Alvarado J, Otero W, Archila PE et al (editores). Gastroenterología y Hepatología. Segunda edición. Editorial Médica Celsus, Bogotá, Colombia 2006. p. 461-480.
60. Martínez JD, Henao SC. Consumo crónico de medicamentos inhibidores de la bomba de protones (IBP) y el desarrollo de gastritis atrófica corporal. Rev Col Gastroenterol 2007; 22(4): 302-307.
61. Jacobson BC, Ferris TG, Shea TL, Mahlis EM, Lee TH, Wang TC. Who is using chronic acid suppression therapy and why? Am J Gastroenterol 2003; 98(1): 51-8.
62. Lassen A, Hallas J, Schaffalitzky De Muckadell OB. Use of anti-secretory medication: a population-based cohort study. Aliment Pharmacol Ther 2004; 20(5): 577-83.
63. Smith T, Verzola E, Mertz H. Low yield of endoscopy in patients with persistent dyspepsia taking proton pump inhibitors. Gastrointest Endosc 2003; 58(1): 9-13.
64. Talley NJ. Yield of endoscopy in dyspepsia and concurrent treatment with proton pump inhibitors: the blind leading the blind? Gastrointest Endosc 2003; 58(1): 89-92.
65. AGA Institute. American gastroenterological association medical position statement on the management of gastroesophageal reflux disease. Gastroenterology 2008; 135: 1383-1391.
66. Ricaurte O, Gutiérrez O. Gastritis crônica. En: Alvarado J, Otero W, Archila PE et al (editores). Gastroenterología y Hepatología. Segunda edición. Editorial Médica Celsus, Bogotá, Colombia 2006. p. 549-563.
1. Heading RC. Prevalence of upper gastrointestinal symptoms in the general population: a systematic review. Scand J Gastroenterol Suppl 1999; 231: 3-8. [ Links ]
2. Schwartz MD. Dyspepsia, peptic ulcer disease, and esophageal reflux disease. West J Med 2002; 176: 98-103. [ Links ]
3. Hammer J, Eslick GD, Howell SC, Altiparmak E, Talley NJ. Diagnostic yield of alarm features in irritable bowel syndrome and functional dyspepsia. Gut 2004; 53: 666-672. [ Links ]
4. Correa P, Cubillos C, Duque E, Barbosa O, Erazo JH. Patología geográfica del carcinoma y de la metaplasia intestinal del estómago en Colombia. Antioquia Médica 1969; 19(5): 41-56. [ Links ]
5. Ángel LA, Giraldo A, Pardo CE. Mortalidad por cánceres del aparato digestivo en Colombia entre 1980 y 1998. Análisis de tendencias y comparación regional. Rev Fac Med Univ Nac Colomb 2004; 52(1): 19-37. [ Links ]
6. Ángel LA, Giraldo A, Pardo CE. Tasas de mortalidad por canceres del tubo digestivo según género y grupos de edad en Colombia entre 1980 y 1998. Rev Colomb Gastroenterol 2007; 23(2): 124-35. [ Links ]
7. Medina MR. Atlas de mortalidad por cáncer. Colombia 1990. 1ª edición. Instituto Nacional de Cancerología. Talleres de Nuevas Ediciones, Santafé de Bogotá, Colombia 1994. [ Links ]
8. Instituto Nacional de Cancerología, E.S.E., Instituto Geográfico Agustín Codazzi – IGAC. Atlas de mortalidad por cáncer en Colombia. Imprenta Nacional de Colombia, Bogotá D.C. 2003. [ Links ]
9. Piñeros M, Ferlay J, Murillo R. Cancer incidence estimates at the national and district levels in Colombia. Salud Pública Mex 2006; 48: 455-465. [ Links ]
10. Asociación Española de Gastroenterología, Sociedad Española de Medicina de Familia y Comunitaria y Centro Cochrane Iberoamericano. Grupo de trabajo de la guía de práctica clínica sobre dispepsia. Manejo del paciente con dispepsia. Guía de práctica clínica. Barcelona 2003. [ Links ]
11. Soll AH, Graham DY. Peptic ulcer disease. En: Yamada T. Textbook of Gastroenterology. Fifth edition. Blackwell Publishing Ltd. Oxford, USA 2009. p. 955-981. [ Links ]
12. Bobadilla J, Vargas-Vorackova F, Gómez A, Jesus Villalobos J. Incidence of peptic ulcer at the Instituto Nacional de la Nutrición "Salvador Zubirán": study of localization, associated factors and temporal trends. Rev Gastroenterol Mex 1996; 61(1): 31-5. [ Links ]
13. Perez-Aisa MA, Del Pino D, Siles M, Lanas A. Clinical trends in ulcer diagnosis in a population with high prevalence of Helicobacter pylori infection. Aliment Pharmacol Ther 2005; 21(1): 65-72. [ Links ]
14. de Jesus Villalobos Perez J, Quinones Menendez NF, Cruz Lomeli G, Vargas Vorackova F. Epidemiology of peptic ulcer at the "Salvador Zubirán" National Institute of Nutrition. Rev Gastroenterol Mex 1990; 55(2): 55-60. [ Links ]
15. Lam SK. Aetiological factors of peptic ulcer: perspectives of epidemiological observations this century. J Gastroenterol Hepatol 1994; 9(Suppl 1): S93-8. [ Links ]
16. Sadic J, Borgström A, Manjer J, Toth E, Lindell G. Bleeding peptic ulcer - time trends in incidence, treatment and mortality in Sweden. Aliment Pharmacol Ther 2009; 30: 392-398. [ Links ]
17. Sung JJY, Kuipers EJ, El-Serag HB. Systematic review: the global incidence and prevalence of peptic ulcer disease. Aliment Pharmacol Ther 2009; 29: 938-946. [ Links ]
18. Arents NL, Thijs JC, van Zwet AA, Kleibeuker JH. Does the declining prevalence of Helicobacter pylori unmask patients with idiopathic peptic ulcer disease? Trends over an 8 year period. Eur J Gastroenterol Hepatol 2004; 16(8): 779-83. [ Links ]
19. Wong SN, Sollano JD, Chan MM et al. Changing trends in peptic ulcer prevalence in a tertiary care setting in the Philippines: a seven-year study. J Gastroenterol Hepatol 2005; 20(4): 628-32. [ Links ]
20. Ramirez Ramos A, Chinga Alayo E, Mendoza Requena D, Leey Casella J, Segovia Castro MC, Otoya C. Changes in the prevalence of H. pylori in Peru; during the 1985-2002 period in medium and upper socio-economic strata. Rev Gastroenterol Peru 2003; 23(2): 92-8. [ Links ]
21. Van Leerdam ME. Epidemiology of acute upper gastrointestinal bleeding. Best Pract Res Clin Gastroenterol. 2008; 22(2): 209-24. [ Links ]
22. Bjorkman DJ. Endoscopic diagnosis and treatment of nonvariceal upper gastrointestinal hemorrhage. En: Yamada T. Textbook of Gastroenterology. Fifth edition. Blackwell Publishing Ltd. Oxford, USA 2009. p. 3018-3031. [ Links ]
23. García del Risco F, Caraballo JM, Urbina D. Helicobacter pylori en sujetos asintomáticos, con úlcera duodenal, úlcera gástrica y dispepsia no ulcerosa en Cartagena. Rev Col Gastroenterol 1994; 9(4): 188-93. [ Links ]
24. Bejarano M, Méndez F. Interacción de los factores asociados a enfermedad úlcero-péptica. Rev Col Gastroenterol 2006; 21(1): 25-35. [ Links ]
25. Gómez MA, Ricaurte O, Gutiérrez O. Costo efectividad de la endoscopia digestiva alta como prueba diagnóstica en una campaña para detección de cáncer gástrico. Rev Col Gastroenterol 2009; 24(1): 34-50. [ Links ]
26. Sierra F, Hernández BE, Botero RC, De Molano B. Hemorragia de vías digestivas altas, incidencia en 20.000 endoscopias. Rev Col Gastroenterol 1994; 9(4): 175-85. [ Links ]
27. Higham J, Kang J-Y, Majeed A. Recent trends in admissions and mortality due to peptic ulcer in England: increasing frequency of haemorrhage among older subjects. Gut 2002; 50: 460-464. [ Links ]
28. Yen FS, Wu JC, Wang LM, Kuo BI, Hu SC, Lee SD. Seasonal variation in the incidence of peptic ulcer and esophageal variceal bleeding in Taiwan. Zhonghua Yi Xue Za Zhi (Taipei) 1996; 57(1): 22-7. [ Links ]
29. Sezgin O, Altintas E, Tombak A. Effects of seasonal variations on acute upper gastrointestinal bleeding and its etiology. Turk J Gastroenterol 2007; 18(3): 172-6. [ Links ]
30. Thomopoulos KC, Katsakoulis EC, Margaritis VG, Mimidis KP, Vagianos CE, Nikolopoulou VN. Seasonality in the prevalence of acute upper gastrointestinal bleeding. J Clin Gastroenterol 1997;25(4):576-9. [ Links ]
31. Zimmerman J, Arnon R, Beeri R et al. Seasonal fluctuations in acute upper gastrointestinal bleeding: lack of effect of nonsteroidal anti-inflammatory drugs. Am J Gastroenterol 1992; 87(11): 1587-90. [ Links ]
32. Moshkowitz M, Konikoff FM, Arber N. Seasonal variation in the frequency of Helicobacter pylori infection: a possible cause of the seasonal occurrence of peptic ulcer disease. Am J Gastroenterol 1994; 89(5): 731-3. [ Links ]
33. Wysocki A, Kaminski W, Krzywon J. Seasonal periodicity of perforated peptic ulcers. Przegl Lek 1999; 56(3): 189-91. [ Links ]
34. Xirasagar S, Lin H-C, Chen C-S. Role of Meteorological Factors in Duodenal Ulcer Seasonality. A Nation-wide, Population-Based Study. J Gen Intern Med 2007; 22(10): 1439-46. [ Links ]
35. Tulassay Z, Papp J, Lengyel G, Szathmari M. Seasonal disposition of gastroduodenal ulcer--legend or reality? Wien Med Wochenschr 1987; 137(4): 76, 78. [ Links ]
36. López-Cepero JM, López-Silva ME, Amaya-Vidal A et al. Influencia de los factores climáticos en la incidencia de hemorragia digestiva alta. Gastroenterol Hepatol 2005; 28: 540-5. [ Links ]
37. Soszka A, Kusz-Rynkun A. Seasonal fluctuation in peptic ulcer occurrence: a disappearing phenomenon? Pol Merkuriusz Lek 1998; 4(22): 208-9. [ Links ]
38. Angel LA, Gutiérrez O, Rincón D. Epidemiología de la enfermedad ácido péptica. Rev Col Gastroenterol 1997; 12(3): 135-144. [ Links ]
39. Gutiérrez O. Aspecto endoscópico en empedrado de la mucosa gástrica (Gastritis folicular): relación con el Helicobacter pylori. En: Sociedad Colombiana de Gastroenterología (editores). Temas escogidos de Gastroenterología. Tomo XXVII. Gráficas Aguilera Ltda., Santafé de Bogotá 1993. p. 75-94. [ Links ]
40. Martínez JD, Henao SC. Hiperplasia linfoide folicular gástrica e infección por Helicobacter pylori en adultos colombianos. Rev Col Gastroenterol 2009; 24: 148-156. [ Links ]
41. Gómez MA, Ricaurte O, Gutiérrez O. Costo efectividad de la endoscopia digestiva alta como prueba diagnóstica en una campaña para detección del cáncer gástrico. Rev Col Gastroenterol 2009; 24(1): 34-50. [ Links ]
42. Arvind AS, Cook RS, Tabaqchali S, et al. One minute endoscopy room test for Campylobacter pylori. Lancet 1988: 1: 704. [ Links ]
43. Ollyo JB, Lang F, Fontolliet C, Monnier P. Savary-Miller's new endoscopic grading of reflux-oesophagitis: a simple, reproducible, logical, complete and useful classification. Gastroenterology 1990; 98: A100. [ Links ]
44. Armstrong D, Bennett JR, Blum AL, et al. The endoscopic assessment of esophagitis: a progress report of observer agreement. Gastroenterology 1996; 111: 85. [ Links ]
45. Tygat NJ. The Sydney System: endoscopic division. Endoscopio appearances in gastritis/duodenitis. J Gastroenterol Hepatol 1991; 6: 223-234. [ Links ]
46. Santacoloma M. Indicaciones y complicaciones. En: Roldan LF, Hani AC, Aponte DM et al (editores). Técnicas en endoscopia digestive. ACED Asociación Colombiana de Endoscopia Digestiva, Bogotá D.C., Colombia, 2007. [ Links ]
47. Ministerio de Salud Nacional, República de Colombia. Resolución No. 008430. Por la cual se establecen las normas científicas, técnicas y administrativas para la investigación en salud. Rev Col Psiquiatría 1996; 25(1): 38-59. [ Links ]
48. Sanchez. R, Tejada P. Martinez J. Comportamiento de las muertes violentas en Bogotá, 1997-2003. Revista de Salud Pública 2005; 7(3): 254-267. [ Links ]
49. Gómez V., Maravall A., Peña D. Missing Observation in ARIMA models: Skipping approach versus additive outlier approach. Journal of Econometrics 1999; 88: 341-363. [ Links ]
50. Koopman S.J, A.C. Harvey, J.A Doornik and N. Shephard, STAMP 6.0: Structural Time Series Analyser, Modeller and Predictor, London: Timberlake Consultants Press. 2000. [ Links ]
51. Laverde M, Fassler S. Gastritis crónica: factores de riesgo, correlación endoscópica-histológica. En: Sociedad Colombiana de Gastroenterología (editores). Temas escogidos de Gastroenterología. Tomo XXV. Bogotá 1989. p. 93-108. [ Links ]
52. Wai CT, Yeoh KG, Ho KY, Kang JY, Lim SG. Diagnostic yield of upper endoscopy in Asian patients presenting with dyspepsia. Gastrointest Endosc 2002; 56(4): 548-51. [ Links ]
53. Voutilainen M, Mantynen T, Kunnamo I, Juhola M, Mecklin JP, Farkkila M. Impact of clinical symptoms and referral volume on endoscopy for detecting peptic ulcer and gastric neoplasms. Scand J Gastroenterol 2003; 38(1): 109-13. [ Links ]
54. Bartels F, Hahn HJ, Stolte M, Schmidt-Wilcke HA. Quality of diagnostic procedures and frequency of endoscopically defined diseases of the upper gastrointestinal tract. Z Gastroenterol 2003; 41(4): 311-8. [ Links ]
55. Vakil N, Zanten SV van, Kahrilas P, Dent J, Jones R, Global Consensus Group. The Montreal Definition and Classification of Gastroesophageal Reflux Disease: A Global Evidence-Based Consensus. Am J Gastroenterol 2006; 101: 1900-1920. [ Links ]
56. Rengifo A, De Lima E. Úlcera péptica. En: Alvarado J, Otero W, Archila PE et al (editores). Gastroenterología y Hepatología. Segunda edición. Editorial Médica Celsus, Bogotá, Colombia 2006. p. 529-547. [ Links ]
57. Asociación Colombiana de Endoscopia Digestiva. Día Nacional de la Endoscopia. Santafé de Bogotá, D.C. 1999. [ Links ]
58. Asociación Colombiana de Endoscopia Digestiva. Boletín Informativo: Informe Especial: Día Nacional de la Endoscopia Digestiva. Cartagena, Colombia 2002. [ Links ]
59. Otero W, Hani A, Arbeláez V. Enfermedad por reflujo gastroesofágico. En: Alvarado J, Otero W, Archila PE et al (editores). Gastroenterología y Hepatología. Segunda edición. Editorial Médica Celsus, Bogotá, Colombia 2006. p. 461-480. [ Links ]
60. Martínez JD, Henao SC. Consumo crónico de medicamentos inhibidores de la bomba de protones (IBP) y el desarrollo de gastritis atrófica corporal. Rev Col Gastroenterol 2007; 22(4): 302-307. [ Links ]
61. Jacobson BC, Ferris TG, Shea TL, Mahlis EM, Lee TH, Wang TC. Who is using chronic acid suppression therapy and why? Am J Gastroenterol 2003; 98(1): 51-8. [ Links ]
62. Lassen A, Hallas J, Schaffalitzky De Muckadell OB. Use of anti-secretory medication: a population-based cohort study. Aliment Pharmacol Ther 2004; 20(5): 577-83. [ Links ]
63. Smith T, Verzola E, Mertz H. Low yield of endoscopy in patients with persistent dyspepsia taking proton pump inhibitors. Gastrointest Endosc 2003; 58(1): 9-13. [ Links ]
64. Talley NJ. Yield of endoscopy in dyspepsia and concurrent treatment with proton pump inhibitors: the blind leading the blind? Gastrointest Endosc 2003; 58(1): 89-92. [ Links ]
65. AGA Institute. American gastroenterological association medical position statement on the management of gastroesophageal reflux disease. Gastroenterology 2008; 135: 1383-1391. [ Links ]
66. Ricaurte O, Gutiérrez O. Gastritis crônica. En: Alvarado J, Otero W, Archila PE et al (editores). Gastroenterología y Hepatología. Segunda edición. Editorial Médica Celsus, Bogotá, Colombia 2006. p. 549-563. [ Links ]











 text in
text in