Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957On-line version ISSN 2500-7440
Rev Col Gastroenterol vol.27 no.2 Bogotá Apr./June 2012
Friedrich Krukenberg of Krukenberg's Tumor: Report of a series of cases
Martin Gómez Zuleta, MD (1), Luis Fernando Benito, MD. (2), Cristina Almonacid, MD. (3)
(1) Assistant Professor in the Gastroenterology Unit of the Department of Internal Medicine at the Universidad Nacional de Colombia and Hospital El Tunal in Bogotá, Colombia.
(2) Third Year Surgery Resident at the Universidad de San Martin in Bogotá, Colombia.
(3) Pathologist at the Hospital El Tunal and the Clínica de la Policía in Bogotá, Colombia.
Received: 17-01-12 Accepted: 15-05-12
Abstract
Krukenberg's tumor is an ovarian tumor first described by the German physician Friedrich Krukenberg. It is a metastasis of a primary tumor which is usually located in the stomach. This article presents a brief overview of the history of these tumors and a series of 5 cases which were handled in our service. The aim of this article is to demonstrate the complexity of this diagnosis, the therapeutic approach, and the pessimistic prognosis that this condition has.
Key words
Krukenberg's tumor, gastric cancer.
Friedrich Ernst Krukenberg (1871-1946) was a German physician who worked in the city of Marburg under the tutelage of Felix Jacob Marchand, the Chief Medical Officer of the Department of Pathology, during his undergraduate education (1846-1928). Dr. Marchand had had six cases of atypical ovarian tumors which he gave to Krukenberg as material for his doctoral thesis. At the age of 25 years old in 1896, Krukenberg published his thesis. He proposed that, because of the appearance of malignant cells, these tumors were a new type of primary ovarian sarcomas which he named "fibrosarcoma ovarii mucocellulare (carcinomatodes) (1-3)."
In 1902 Schlagenhauffer proposed that these tumors were not primary ovarian entities but rather were metastases of epithelial neoplasia. Decades later it was confirmed that Krukenberg tumor is in fact a metastasis of signet ring cells typically from gastric adenocarcinoma which is associated with disproportionate desmoplastic reactions. This explains the sizes reached by these lesions and the prominent stromal component that puzzled Krukenberg and Marchand. In 1928 Gauthier-Villars' described a series of microscopic tubes and glands in Krukenberg tumors. In 1981, Bouillon described in great detail what he called a tubular Krukenberg tumor (3-5).
The use of the term Krukenberg tumor is based on textbooks published by Scully, Young and Kurman. It is defined as a carcinoma that occurs in the ovary and which has a significant component of stringy mucin and typically has signet ring cells within a proliferative cellular stroma derived from the normal stroma of the ovary. Small glands with a squamous epithelium or large glands with intestinal and mucinous epithelium can be found. In 2006 Kiyokawa, Young and Scully reported their experience with 120 cases of Krukenberg tumors. In the same year Lerwill and Young reported 4 cases of ovarian metastasis of gastric intestinal type carcinoma (4-7).
Nearly all Krukenberg tumors are considered to be metastases. However, there have been rare and isolated cases which have been interpreted as primary tumors. Krukenberg tumors constitute 1% to 1.5% of all ovarian tumors. Although they can occur at any age, most occur between the ages of 30 and 40. They rarely occur during menopause. This tumor's evolution and growth are rapid and are accompanied by significant degeneration.
Clinically, a patient presents gynecological symptoms associated with ascites. The tumor is bilateral in up to 80% of cases, and, because of its external structure and consistency, it does not present adhesions in the peritoneal cavity. Due to its histological composition it is classified as an epithelial tumor with connective tissue. It is transmitted through contiguous tissue, blood, lymphatic cells through the peritoneum. Its mode of transmission has been the subject of ongoing study, and the exact pathway of its transmission has not yet been defined (10-12).
CLINICAL CASES
Case 1
A 48 year old woman came to the emergency department with abdominal pain and distension associated with 10kg weight loss and anorexia. A physical examination showed vital signs within normal limits and a patient who otherwise was not in the best condition. Patient presented major abdominal distention. An abdominal mass and ascites could be felt in the pelvic region, but there were no signs of peritoneal irritation. Patient was hospitalized for study. She tested positive for CA 125 (549), but normal for CA19.9 antigen (7.46). A complete abdominal ultrasound found positive evidence of a 16x3 cm mass in the right ovary and a 13x10 cm mass in the left ovary. An upper endoscopy showed an ulcerated lesion with infiltration extending from the upper third to the middle third of the corpus which was compatible with Borrmann III advanced gastric cancer.
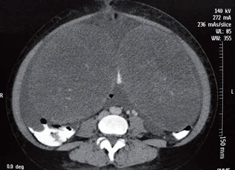
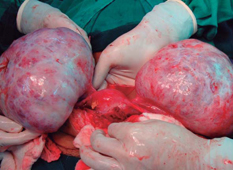
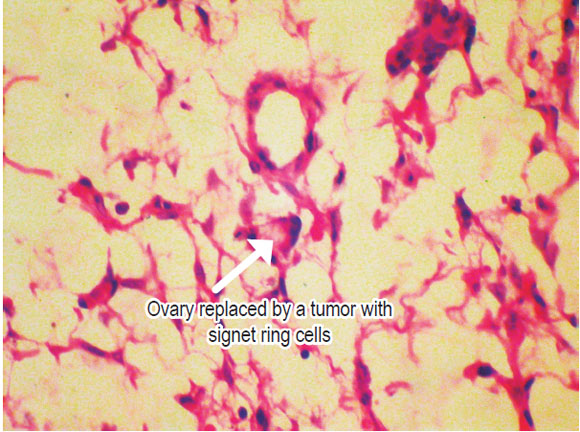
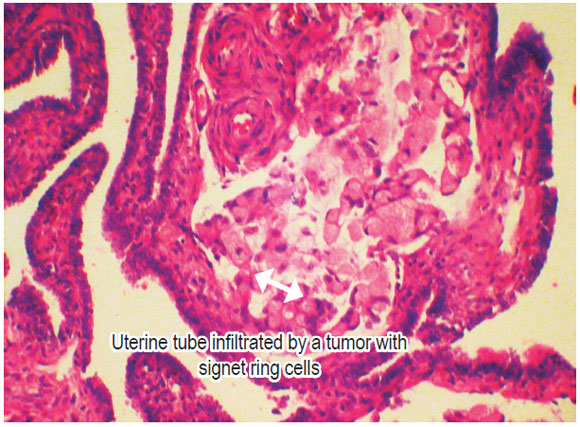
A CT scan showed a 20 cm mass in the right ovary and a 23cm mass in the left ovary (Figure 1). Her pathology revealed adenocarcinoma with diffuse ulcerated signet ring cells. After a week's hospital stay for study, it was decided to initiate palliative treatment because of the prognosis of the patient. Surgery was performed by the gynecology service to resect the ovarian masses and reduce symptoms (Figure 2). Pathology showed signet ring cells not only within the ovarian stroma (Figure 3) but also within the fallopian tube (Figure 4). Patient's postoperative evolution was adequate and was followed up with complementary chemotherapy. The patient died 4 months after diagnosis.
Figure 1. Dependent masses are observed in the ovaries, a 20 cm mass in the right ovary and a 23 cm mass in the left ovary.
Figure 2. Bilateral salpingo-oophorectomy, bilateral masses dependent on left and right ovary are shown.
Figure 3. Signet-ring cells infiltrating the ovary.
Figure 4. Signet-ring cells infiltrating the uterine tube.
Case 2
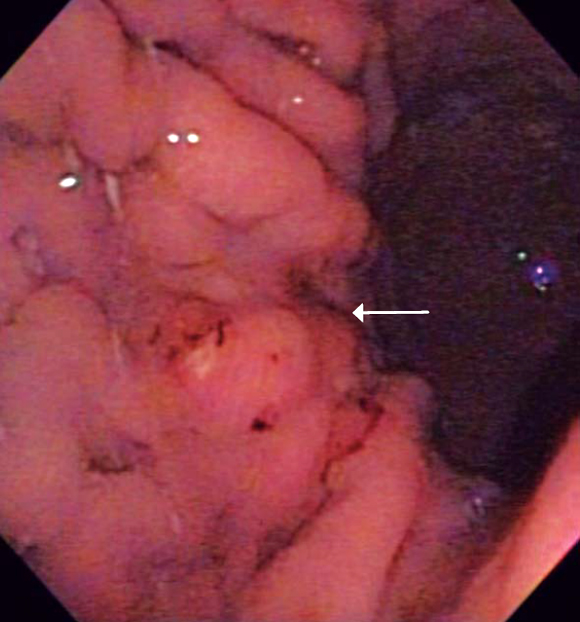
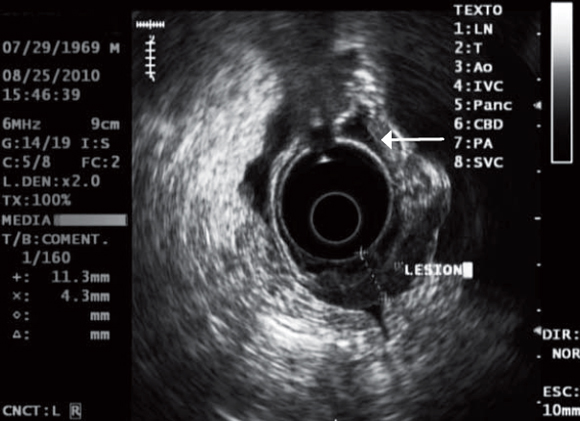
A 41 year old woman who complained of pelvic pain associated with progressive alterations in her menstrual cycle and weight loss for 3 months was examined. Patient's physical condition was acceptable conditions. She was well hydrated, had pale mucous membranes, but had a protruding abdomen and ascites. Bilateral ovarian masses found in the pelvic examination were confirmed by ultrasound and CT scan. Upper endoscopy showed a slight thickening and convergence of folds in an area no bigger than 1cm (Figure 5) near the greater curve of the upper third of the stomach. Biopsies taken from this location showed a diffuse carcinoma. Endoscopic ultrasound showed ascites and a hypoechoic lesion that destroyed all gastric layers in this area. The rest of the stomach was normal (Figure 6). The patient underwent bilateral salpingo-oophorectomy followed by chemotherapy but died 5 months later.
Figure 5. The arrow indicates an approximately 1 cm lesion, the only evidence of which is loss of folds at this level.
Figure 6. A focal lesion has destroyed all layers and thickened the gastric wall to 11.3 mm. Note the normal wall which measures 4.3 mm, indicated by the arrow.
Case 3
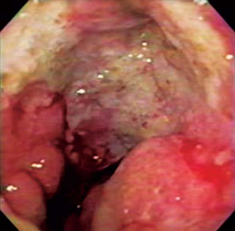
A 48 year old woman with complaints of progressive pelvic pain and an abnormal menstrual cycle was examined. She was found to be in poor condition with anemia, abdominal distension and a positive ascitic wave. Gynecologic evaluation showed bilateral ovarian masses. Patient was hospitalized for testing. An abdominal CT scan showed bilateral ovarian masses. Endoscopy showed an ulcerated and infiltrative lesion compromising the entire gastric corpus which was diagnosed as Borrmann level IV cancer (Figure 7). The pathology report showed a diffuse carcinoma. Due to the patient's poor condition, she and her family chose not to undergo any treatment except for analgesia. She died 2 months after diagnosis.
Figure 7. Ulcerated and infiltrative lesion that compromised the entire corpus and part of the antrum.
Case 4
A 50 year old woman with complaints of progressively increasing abdominal girth, but with no other associated symptoms such as dyspepsia or gastrointestinal bleeding, was examined. Examination showed pink mucous membranes, normal vital signs, ascites and bilateral ovarian masses. A pelvic CT scan showed bilateral ovarian masses. CA 125 tested at 712 (normal value: 33 IU/ml) and an endoscopy showed an ulcerated lesion extending from the cardia to the angular incisure compromising the greater curve. It was reported as a Borrmann level III cancer. Pathology reported diffuse carcinoma.
The patient underwent surgery for removal of ovarian masses followed by chemotherapy. As in the previous three cases gastric surgery was not performed. Development was acceptable, but the patient died 4 months following diagnosis.
Case 5
The patient was a 32 year old woman who was suffering from pelvic and perineal pain associated with progressive abdominal distension, vomiting, anorexia, but no weight loss. Symptoms had been developing for 2 months prior to consultation of evolution. Physical examination showed that the patient was in generally poor condition with a distended abdomen and an ascitic wave. Patient was hospitalized for study by the gynecology department. A CT scan showed bilateral ovarian masses with peritoneal tumor seeding. Endoscopy showed an approximately 4 cm ulcerated lesion with infiltration in the upper third of the posterior wall. It was diagnosed as Borrmann type II cancer. Pathology reported a poorly differentiated ulcerated carcinoma with signet ring cells. The patient underwent surgery for resection of gynecological ovarian masses. The tumor on the right ovary measured 20 cm x 20 cm while that on the left ovary measured 15 cm x 10cm. Patient underwent chemotherapy with mixed results, but died 4 months after diagnosis.
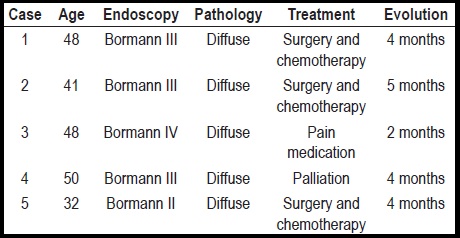
The table 1 shows the main characteristics of the five (5) patients evaluated with Krukenberg tumors.
Table 1. Main characteristics of the 5 patients evaluated with Krukenberg Tumors.
DISCUSSION
Krukenberg tumors account for 1% to 2% of all ovarian neoplasms. In most cases this lesion is simultaneously diagnosed with the primary neoplasm, but 20% to 30% occur following removal of the primary tumor. The prognosis of a patient with Krukenberg tumor is extremely poor with average survival time between 3 and 10 months. Patients in our service survived 3.7 months on average. Only 10% of patients survive more than two years after diagnosis (18-20).
Krukenberg tumors have two important characteristics. The first is ovarian metastasis characterized by the presence of signet ring cells, and second is pseudo sarcoma proliferation of ovarian stroma. By-products of neoplastic epithelial cells induce proliferation of fused ovarian stromal cells and abundant collagen deposits resulting in disproportionate desmoplastic reactions. It is important to note that these tumors do not always have gastric origins: 76% originate in the stomach, 11% in the bowel, 4% come from breast cancer, 3% from the biliary system, 3% from the cecal appendix and the remaining 3% come from the pancreas, cervix, bladder and renal pelvis. About 80% are bilateral. When examined by pathology the ovaries have a smooth, spongy or gelatinous consistency, although they can also be firm. This depends on the degree of edema, on aggregates of mucin within the stroma, on the degree of hypercellularity, and on the degree of fibrous transformation. The ovary generally retains its shape despite the large size that these lesions can reach. They range in size from 5 cm to larger than 20 cm (as in the first case reported where the tumor in the left ovary reached 23 cm in diameter).
Immunological markers for this tumor include MUC 1, CK 7 and estrogen receptors which are of great importance. A good example of their importance is the second case we presented. The gastric tumor was very small (<2cm), but the ovarian masses were very large and had ascites. Only immunohistochemistry allowed us to confirm that the primary tumor was in the stomach, and this completely changed treatment and prognosis of the patient (22-24).
Krukenberg tumor patients usually consult for abdominal and pelvic pain, changes in their menstrual cycles or dyspareunia. In more advanced stages patients present ascites. It is noteworthy that all 5 cases presented had ascites indicating that these patients were in advanced stages. The second case is striking because the patient had a small gastric lesion but already presented ascites and ovarian masses of 12 and 15cm. This gives us an idea of the aggressiveness of these diffuse cancers and raises the question of how these tumors spread. The pathways and mechanisms by which cancer cells reach the ovaries from the stomach, long mystery, still remain only partially understood (10-12).
Three possible pathways for metastasis have been considered: lymphatic, hematic and peritoneal. Since these tumors show no peritoneal involvement on the ovarian surface and rarely involve the peritoneum, the theory of peritoneal dissemination has been discarded. The lymphatic route is most frequently postulated because lymphatic permeation resulting from carcinomatous emboli in adjoining structures (ovarian hilum, mesovarium and mesosalpinx) is commonly found in the histological study of these tumors. There are also many studies of patients with early gastric cancer and Krukenberg tumors which consider that the rich lymphatic plexuses that drain the gastric mucosa and submucosa could be the route by which the tumor spreads to the ovary. It has been found that the risk of ovarian compromise is greater when there are a larger number of lymph nodes affected by gastric cancer (8, 9). Some authors propose that the cancer spreads directly through the circulatory system via the thoracic lymph duct rather than through purely retrograde dissemination via the lymphatic route (22-24).
Since Krukenberg's tumor is a metastatic tumor, it has been proposed that the primary cancer must be found and treated first. Nevertheless, most cases have such poor prognoses that radical surgery such as oophorectomies improve survival only in cases of solitary ovarian metastases or local extensions of the disease only located within the pelvis. Chemotherapy and radiation therapy are indicated as adjuvant therapy for cytoreduction and gastric cancer control which is no longer susceptible to surgical treatment. Even though we were already aware of the bad prognoses, in 4 of the cases in this series bilateral salpingo-oophorectomies were performed. Since these tumors had apparently not disseminated elsewhere, we performed surgery primarily to reduce tumoral burdens before chemotherapy. Probably, to control Krukenberg's tumor and avoid its bad prognosis we must prevent gastric carcinoma since, as is well known, it has a B genetic component identified as inherited familial cancer associated with the CDH1 gene. If we identify a family that meets the Amsterdam criteria for this tumor genetic testing should be performed to identify mutations in the CDH-1 gene in asymptomatic relatives. If a patient tests positive, he or she should have a gastrectomy to prevent the appearance of the tumor (23-25).
CONCLUSIONS
Krukenberg's tumor should not be diagnosed in all patients who have ovarian masses because there are many primary tumors of this organ. Krukenberg's tumor of the ovary is a metastasis that has the histological patterns that we have discussed in this article. It is important to communicate with the medical team to identify the primary tumor before surgery. Primary tumors are commonly located in the stomach but may be located in other organs including the colon, rectum, gallbladder, and pancreas. The prognoses for patients with this type of metastatic tumor are poor: most die within the first year of evolution. There are rare cases in which patients survive several years (26).
REFERENCES
1. Kim SH, Kim WH, Park KJ, Lee JK, Kim JS. CT and MR findings of Krukenberg tumors: comparison with primary ovarian tumors. J Comput Assist Tomogr 1996; 20(3): 393-8.
2. Tavassoli FA, Devilee P (editors). WHO Classification Tumors of the Breast and Female Genital Organs. Lyon: IARC Press; 2003.
3. Irving JA, Lerwill MF, Young RH. Gastrointestinal stromal tumors metastatic to the ovary: a report of five cases. Am J Surg Pathol 2005; 29(7): 920-6.
4. Lash RH, Hart WR. Intestinal adenocarcinomas metastatic to the ovaries: a clinicopathologic evaluation of 22 cases. Am J Surg Pathol 1987; 11(2): 114-21.
5. Mccluggage W, Wilkinson N. Metastatic neoplasms involving the ovary: a review with an emphasis on morphological and immunohistochemical features. Histopathology 2005; 47(3): 231-47.
6. Al-Agha OM, Nicastri AD. An in-depth look at Krukenberg tumor: an overview. Arch Pathol Lab Med 2006; 130(11): 1725-30.
7. Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin 2005; 55(2): 74-108.
8. Krukenberg F. Ueber das fibrosarcoma ovarii mucocellulare (carcinomatodes). Arch Gynaecol Obstet 1896; 50(2): 287-321.
9. Woodruff JD, Novak ER. The Krukenberg tumor: study of 48 cases from the ovarian tumor registry. Obstet Gynecol 1960; 15: 351-60.
10. Yakushiji M, Tazaki T, Nishimura H, Kato T. Krukenberg tumors of the ovary: a clinicopathologic analysis of 112 cases. Acta Obstet Gynaecol Jpn 1987; 39(3): 479-85.
11. Hale RW. Krukenberg tumor of the ovaries. A review of 81 records. Obstet Gynecol 1968; 32(2): 221-5.
12. Israel SL, Helsel EV Jr, Hausman DH. The challenge of metastatic ovarian carcinoma. Am J Obstet Gynecol 1965; 93(8): 1094-101.
13. Kakushima N, Kamoshida T, Hirai S, Hotta S, Hirayama T, Yamada J, et al. Early gastric cancer with Krukenberg tumor and review of cases of intramucosal gastric cancers with Krukenberg tumor. J Gastroenterol 2003; 38(12): 1176-80.
14. Kim NK, Kim HK, Park BJ, Kim MS, Kim YI, Heo DS, et al. Risk factors for ovarian metastasis following curative resection of gastric adenocarcinoma. Cancer 1999; 85(7): 1490-9.
15. de Palma P, Wronski M, Bifernino V, Bovani I. Krukenberg tumor in pregnancy with virilization. A case report. Eur J Gynaecol Oncol 1995; 16(1): 59-64.
16. Cetin B, Aslan S, Akinci M, Atalay C, Cetin A. A long surviving case of Pseudomeigs' syndrome caused by Krukenberg tumor of the stomach. Jpn J Clin Oncol 2005; 35(4): 221-3.
17. Sugimoto Y, Endo K, Sakahara H, Nakajima K, Abe M, Torizuka K, et al. Sequential measurement of serum CA 125 levels in Krukenberg's tumor. Gan No Rinsho 1985; 31(15): 1893-7.
18. Kikkawa F, Shibata K, Ino K, Nomura S, Kajiyama H, Suzuki T, et al. Preoperative findings in non-gynecologic carcinomas metastasizing to the ovaries. Gynecol Obstet Invest 2002; 54(4): 221-7.
19. Park SY, Kim HS, Hong EK, Kim WH. Expression of cytokeratins 7 and 20 in primary carcinomas of the stomach and colorectum and their value in the differential diagnosis of metastatic carcinomas to the ovary. Hum Pathol 2002; 33(11): 1078-85.
20. Yook JH, Oh ST, Kim BS. Clinical prognostic factors for ovarian metastasis in women with gastric cancer. Hepatogastroenterology 2007; 54(75): 955-9.
21. Cheong JH, Hyung WJ, Chen J, Kim J, Choi SH, Noh SH. Survival benefit of metastasectomy for Krukenberg tumors from gastric cancer. Gynecol Oncol 2004; 94(2): 477-82.
22. Taylor AE, Nicolson VM, Cunningham D. Ovarian metastases from primary gastrointestinal malignancies: the Royal Marsden Hospital experience and implications for adjuvant treatment. Br J Cancer 1995; 71(1): 92-6.
23. Mori T, Fujiwara Y, Yano M, Tamura S, Yasuda T, Takiguchi S, et al. Prevention of peritoneal metastasis of human gastric cancer cells in nude mice by S-1, a novel oral derivative of 5-Fluorouracil. Oncology 2003; 64(2): 176-82.
24. Gretschel S, Siegel R, Estévez-Schwarz L, Hünerbein M, Schneider U, Schlag PM. Surgical strategies for gastric cancer with synchronous peritoneal carcinomatosis. Br J Surg 2006; 93(12): 1530-5.
25. Yonemura Y, Bando E, Kawamura T, Ito H, Endo Y, Miura M, et al. Cytoreduction and intraperitoneal chemotherapy for carcinomatosis from gastric cancer. Cancer Treat Res 2007; 134: 357-73.
26. Xu DZ, Zhan YQ, Sun XW, Cao SM, Geng QR. Meta-analysis of intraperitoneal chemotherapy for gastric cancer. World J Gastroenterol 2004; 10(18): 2727-30.
1. Kim SH, Kim WH, Park KJ, Lee JK, Kim JS. CT and MR findings of Krukenberg tumors: comparison with primary ovarian tumors. J Comput Assist Tomogr 1996; 20(3): 393-8. [ Links ]
2. Tavassoli FA, Devilee P (editors). WHO Classification Tumors of the Breast and Female Genital Organs. Lyon: IARC Press; 2003. [ Links ]
3. Irving JA, Lerwill MF, Young RH. Gastrointestinal stromal tumors metastatic to the ovary: a report of five cases. Am J Surg Pathol 2005; 29(7): 920-6. [ Links ]
4. Lash RH, Hart WR. Intestinal adenocarcinomas metastatic to the ovaries: a clinicopathologic evaluation of 22 cases. Am J Surg Pathol 1987; 11(2): 114-21. [ Links ]
5. Mccluggage W, Wilkinson N. Metastatic neoplasms involving the ovary: a review with an emphasis on morphological and immunohistochemical features. Histopathology 2005; 47(3): 231-47. [ Links ]
6. Al-Agha OM, Nicastri AD. An in-depth look at Krukenberg tumor: an overview. Arch Pathol Lab Med 2006; 130(11): 1725-30. [ Links ]
7. Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin 2005; 55(2): 74-108. [ Links ]
8. Krukenberg F. Ueber das fibrosarcoma ovarii mucocellulare (carcinomatodes). Arch Gynaecol Obstet 1896; 50(2): 287-321. [ Links ]
9. Woodruff JD, Novak ER. The Krukenberg tumor: study of 48 cases from the ovarian tumor registry. Obstet Gynecol 1960; 15: 351-60. [ Links ]
10. Yakushiji M, Tazaki T, Nishimura H, Kato T. Krukenberg tumors of the ovary: a clinicopathologic analysis of 112 cases. Acta Obstet Gynaecol Jpn 1987; 39(3): 479-85. [ Links ]
11. Hale RW. Krukenberg tumor of the ovaries. A review of 81 records. Obstet Gynecol 1968; 32(2): 221-5. [ Links ]
12. Israel SL, Helsel EV Jr, Hausman DH. The challenge of metastatic ovarian carcinoma. Am J Obstet Gynecol 1965; 93(8): 1094-101. [ Links ]
13. Kakushima N, Kamoshida T, Hirai S, Hotta S, Hirayama T, Yamada J, et al. Early gastric cancer with Krukenberg tumor and review of cases of intramucosal gastric cancers with Krukenberg tumor. J Gastroenterol 2003; 38(12): 1176-80. [ Links ]
14. Kim NK, Kim HK, Park BJ, Kim MS, Kim YI, Heo DS, et al. Risk factors for ovarian metastasis following curative resection of gastric adenocarcinoma. Cancer 1999; 85(7): 1490-9. [ Links ]
15. de Palma P, Wronski M, Bifernino V, Bovani I. Krukenberg tumor in pregnancy with virilization. A case report. Eur J Gynaecol Oncol 1995; 16(1): 59-64. [ Links ]
16. Cetin B, Aslan S, Akinci M, Atalay C, Cetin A. A long surviving case of Pseudomeigs' syndrome caused by Krukenberg tumor of the stomach. Jpn J Clin Oncol 2005; 35(4): 221-3. [ Links ]
17. Sugimoto Y, Endo K, Sakahara H, Nakajima K, Abe M, Torizuka K, et al. Sequential measurement of serum CA 125 levels in Krukenberg's tumor. Gan No Rinsho 1985; 31(15): 1893-7. [ Links ]
18. Kikkawa F, Shibata K, Ino K, Nomura S, Kajiyama H, Suzuki T, et al. Preoperative findings in non-gynecologic carcinomas metastasizing to the ovaries. Gynecol Obstet Invest 2002; 54(4): 221-7. [ Links ]
19. Park SY, Kim HS, Hong EK, Kim WH. Expression of cytokeratins 7 and 20 in primary carcinomas of the stomach and colorectum and their value in the differential diagnosis of metastatic carcinomas to the ovary. Hum Pathol 2002; 33(11): 1078-85. [ Links ]
20. Yook JH, Oh ST, Kim BS. Clinical prognostic factors for ovarian metastasis in women with gastric cancer. Hepatogastroenterology 2007; 54(75): 955-9. [ Links ]
21. Cheong JH, Hyung WJ, Chen J, Kim J, Choi SH, Noh SH. Survival benefit of metastasectomy for Krukenberg tumors from gastric cancer. Gynecol Oncol 2004; 94(2): 477-82. [ Links ]
22. Taylor AE, Nicolson VM, Cunningham D. Ovarian metastases from primary gastrointestinal malignancies: the Royal Marsden Hospital experience and implications for adjuvant treatment. Br J Cancer 1995; 71(1): 92-6. [ Links ]
23. Mori T, Fujiwara Y, Yano M, Tamura S, Yasuda T, Takiguchi S, et al. Prevention of peritoneal metastasis of human gastric cancer cells in nude mice by S-1, a novel oral derivative of 5-Fluorouracil. Oncology 2003; 64(2): 176-82. [ Links ]
24. Gretschel S, Siegel R, Estévez-Schwarz L, Hünerbein M, Schneider U, Schlag PM. Surgical strategies for gastric cancer with synchronous peritoneal carcinomatosis. Br J Surg 2006; 93(12): 1530-5. [ Links ]
25. Yonemura Y, Bando E, Kawamura T, Ito H, Endo Y, Miura M, et al. Cytoreduction and intraperitoneal chemotherapy for carcinomatosis from gastric cancer. Cancer Treat Res 2007; 134: 357-73. [ Links ]
26. Xu DZ, Zhan YQ, Sun XW, Cao SM, Geng QR. Meta-analysis of intraperitoneal chemotherapy for gastric cancer. World J Gastroenterol 2004; 10(18): 2727-30. [ Links ]











 text in
text in