Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em
SciELO
Similares em
SciELO -
 Similares em Google
Similares em Google
Compartilhar
Revista colombiana de Gastroenterología
versão impressa ISSN 0120-9957
Rev Col Gastroenterol vol.27 no.3 Bogotá jul./set. 2012
Cystic tumors of the pancreas: Surgical vision and a patient cohort
Manuel Santiago Mosquera, MD. (1), Akram Kadamani A, MD. (1), Carlos Felipe Chaux MD. (2), Humberto Bohórquez, MD. (3), Carlos A. Sánchez Pignalosa, MD. (4), Gabriel Sánchez de Guzmán, MD. (4)
(1) Surgical and Specialty Service at the Fundación Cardioinfantil - Instituto de Cardiología (FCI IC) in Bogotá, Colombia.
(2) General and Laparoscopic Surgeon at the Clínica VIP in Bogotá, Colombia.
(3) Abdominal Organ Transplant Surgeon in the Abdominal Organ Transplant Department at the Ochsner Clinic in New Orleans, LA USA.
(4) Resident IV in General Surgery at the Universidad de la Sábana in Bogotá, Colombia.
Received: 30-08-11 Accepted: 17-08-12
Abstract
We present a retrospective study of a cohort of ten patients with cystic tumors of the pancreas, most of whom underwent surgery. The study emphasizes study, diagnosis, and treatment of the four who underwent laparoscopic surgery. It presents some of the variables analyzed in their management and pays special attention to their surgery treatment. A systematic review of the literature is also included.
Key words
Cystic tumors of the pancreas, serous cystadenoma, mucinous cystadenoma, intraductal papillary tumors, cystic papillary tumors.
INTRODUCTION
Cystic tumors of the pancreas (CTP) have recently gained attention from surgeons and gastroenterologists due to progress in the diagnostic images that frequently detect them. The prevalence of the cystic tumors is uncertain in the general population since only tumors that cause symptoms are usually identified (1). A prevalence of 1.2% was found in series of surgical and radiological records of CTP. Half of these lesions were asymptomatic (2).
When CTP produces symptoms, a patient may manifest recurrent pancreatitis, chronic chest pain or jaundice. These symptoms normally imply that there is compression or obstruction in the pancreatic duct.
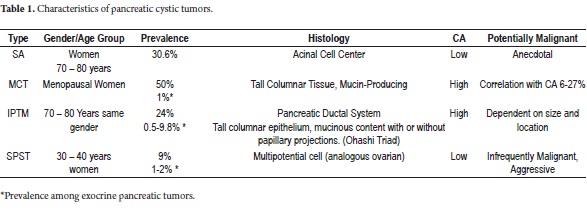
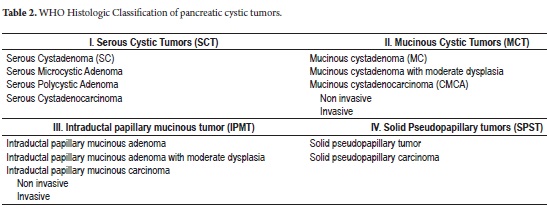
Among the different classifications of CTP we find serous cystadenoma (SC), mucinous cystic neoplasms (MCN), intraductal papillary mucinous tumors (IPMT) and solid pseudopapillary neoplasms (SPN) (3-15). The histology, prevalence, potential for malignancy, and distributions of gender and age at presentation vary according to the neoplasm (Table 1). The World Health Organization (WHO) has described and established their histological classification (Table 2) (16, 17).
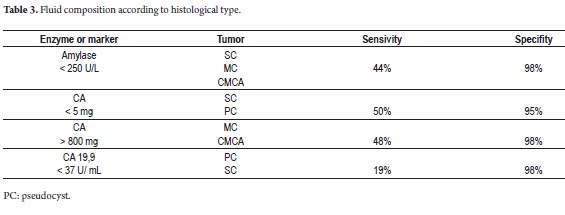
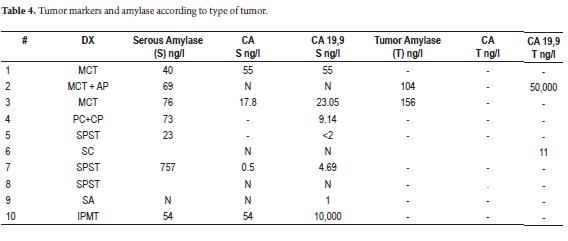
The majority of CTPs are detected by CT scans or MRIs. For doubtful lesions, including those with diameters less than or equal to 3cm, endoscopic ultrasound (EUS) facilitates typing with fine needle aspiration (sensitivity 50-60%) since the study of markers in the liquid is an innocuous procedure (18-20). The level of carcinoembryonic antigen (CA) is the best predictor of mucinous tumors: its accuracy is 79% with levels higher than 192ng/ml (21). This predictor can be complemented with CA 19.9 and amylase measured in the serum in the and tumor which has low sensitivity but high specificity (Table 3) (22).
The need for surgical treatment is determined by histology, size, location of neoplasia and the risk to the patient. When an SC is considered to be benign but the diagnosis is questionable and the patient is symptomatic and has a lesion > 4 cm, surgery is required (23, 24). Because of their potential malignancy great when an MCN is 3 cm or larger it must be resected (25). Whether or not there are nodules in the duct wall, an IPMT 2 cm or larger in the main duct or in a secondary duct absolutely requires surgery. When nodules are present, surgery is also required even for smaller lesions. When surgery is not possible, these lesions are managed medically and patient is observed (26) although good results have been described after the use of ablation (21, 27). Regardless of their size or location SPNs are potentially malignant. Surgical treatment is recommended as medical groups with experience have found that it provides benefits in terms of long-term survival (28).
Our objective in this study is to present the approach, surgical treatment and results from the patients we treated.
METHODOLOGY
A retrospective cohort of patients seen at the Fundación Cardioinfantil Instituto de Cardiología between 2006 and 2010 was studied. To date patients have been monitored for between 24 to 78 months following each diagnosis of a cystic neoplasm of the pancreas.
Data was transferred directly from patients clinical histories and stored in a data base for later analysis. Descriptive statistics were used in consideration of the type of variables studied.
RESULTS
A total of 10 patients were included. For each patient clinical presentation, evaluation, treatment and the histologic diagnosis are shown. The average age was 50 years old (Range 29-86). Eight of the patients were women with various types of pancreatic cystic tumors, while another female patient was diagnosed histologically with a pancreatic pseudocyst from the surgically removed piece although she had not previously been diagnosed or suspected of having this cyst. The male patient presented an IPMT.
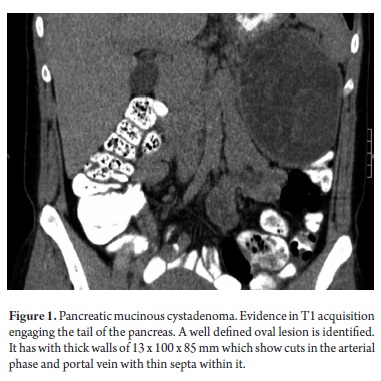
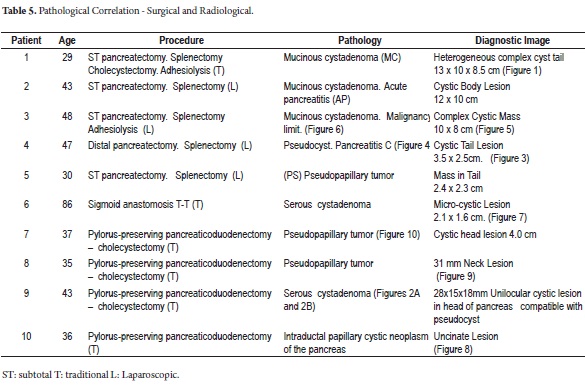
Most of the patients initially manifested a clinical picture of severe chest pain of variable intensity. One patient manifested repeated episodes of pancreatitis while another presented acute abdomen after surgery was performed in another institution. A biopsy was performed and the patient was referred for complete treatment. A cyst was incidentally found at the head of the pancreas (Figure 1) in another female patient who was undergoing extension studies for sigmoid adenocarcinoma. Histological diagnosis of a pancreatic biopsy taken during resection of a colon tumor showed serous cystadenoma. This resection was done two years after resection had removed another metachronous tumor from her right colon. Study results, diagnoses and treatments are presented in Tables 4 and 5.
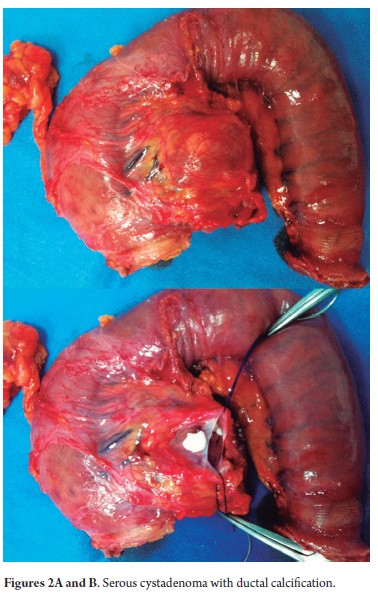
Nine out of the ten patients were treated surgically. Four of the patients with lesions in the body and tail of the pancreas were treated laparoscopically. Time of surgery varied between 165 and 210 minutes with an average of 185 minutes. In contrast, the patient who underwent the conventional laparoscopic reoperation required only 105 minutes. The other five patients with lesions in the body and tail of the pancreas required pancreaticoduodenectomies which preserved their pylori. One patient developed sepsis as the result of a staphylococcus infection from a catheter. Another patient developed nosocomial enterobacter cloacae pneumonia and self-limiting upper digestive tract bleeding. Another patient developed repeated bacterobilia. All of these situations were resolved with medical handling (directed antibiotics). We performed a lateral pancreaticojejunostomy in two planes, a lateral hepatic jejunostomy in a single plane and a lateral duodenojejunostomy in two planes. We did not have any fistulas in any of the anastomosis we performed nor did any intra-abdominal or surgical site infections occur. There were no mortalities. The serum tumor marker of the patient with SC remained normal during more than 60 months of observation and no surgical intervention was required. Between studies of the patient who was treated for SC endoscopic ultrasound reported a 1 cm cystic lesion and an intraductal papillary lesion of 0.5 cm. As a result, surgery was performed. The final pathology report showed a serous cystadenoma with intraductal calcification (Figures 2 and 2A).
The 7 day hospital stay for the patient who underwent conventional distal pancreatectomy was shorter than average 9 day stay for minimally invasive surgery although they varied from 6 to 12 days.
In most cases complications were considered to be minor. Postoperative pain was the most frequent. In the two worst cases, patients suffered intestinal obstructions. One was initially treated with laparoscopic surgery but postoperative percutaneous drainage was required. The other underwent conventional left colectomy because of a stenosis caused by ischemia in the splenic flexure of the colon. Patient’s postoperative evolution was appropriate. Neither case resulted in death.
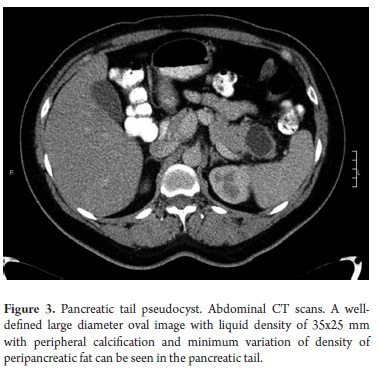
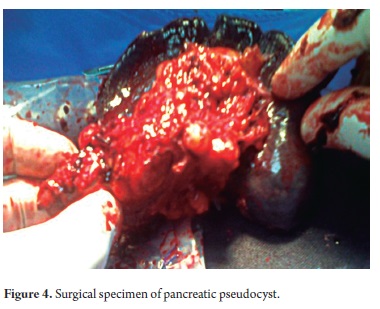
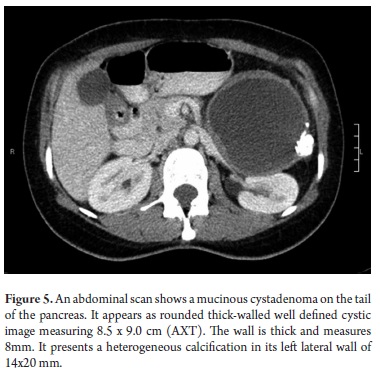
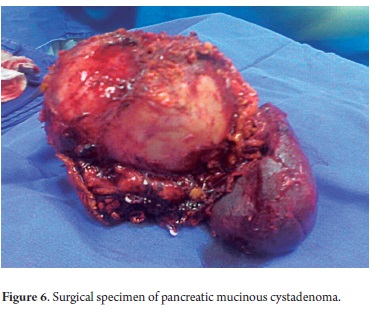
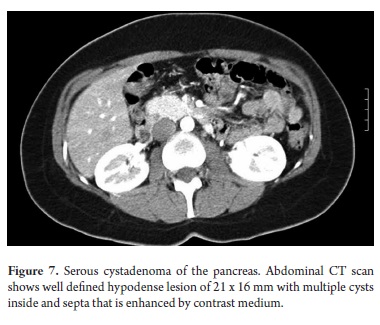
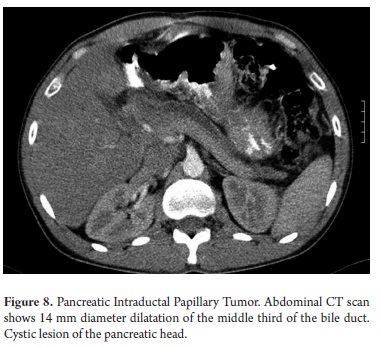
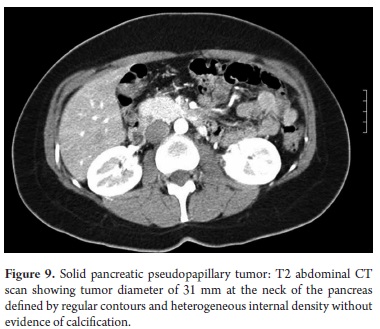
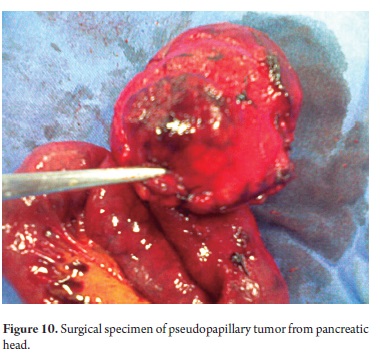
The pathological study reported mucinous cystadenoma in 3 cases, one of which presented pancreatic inflammation. Three cases reported solid pseudopapillary cystic tumors, two reported serous cystadenomas, one was diagnosed as the result of a biopsy performed for another reasons, and one had a questionable diagnosis which could have been either an intrapapillary mucinous tumor or a pancreatic pseudocyst. One had an intraductal papilloma and another had a pancreatic pseudocyst (Table 5). All patients in the series underwent complete resections and were negative for tumor margins. No malignant or infiltrating lesions were documented. (Figures 3, 4, 5, 6, 7, 8, 9,10)
As part of the preoperative study, punctures were performed on 3 patients. Serum tumor markers and serum amylase were studied. CA 19.9 with a value of 50,000 was reported in just one patient (Table 5).
DISCUSSION
In our experience with tumors, a CAT scan is the first step in evaluating the body which allows the study of the characteristics of the tumor and thus diagnosis. Percutaneous puncture and endosonography are alternatives for characterizing a mass in questionable cases or when surgery is being considered. Either transpapillary or transgastric techniques guided by endosonography can be used (29, 30). One of the two patients with a lesion smaller than 3cm underwent laparoscopic distal pancreatectomy without echo-endoscopy while the other underwent pancreaticoduodenectomy using echo-endoscopy. In this case the question arose with a magnetic resonance cholangiopancreatography (MRCP) for an IPMT. In neither case were puncture cytology or tumor markers studied. Perhaps if these tests had been used we would have changed our behavior in favor of observation thereby avoiding the potential for morbidity and mortality in a major procedure such as a pancreatoduodenectomy. The foundation does not have EUS equipment which is why the number of patients treated is low. This is especially so when all the difficulties argued by the EPSs and the prolonged time required for performance of high cost diagnostic procedures which are not included in the list of approved POS services. (Translators note: EPS stands for Entidades Promotoras de Salud which are institutions analogous to HMOs in the USA, POSs are Planes Obligatorios de Salud which are compulsory health insurance plans connected to the EPSs.)
In this cohort, study of liquids did not provide much information that was useful for diagnosis. CA and amylase values did not correlate with the tumor type as described in the literature. Nevertheless, we consider these studies to be helpful for diagnosis so that they should not be ruled out for specific cases, as we mentioned earlier.
Standard surgical treatment for confirmed neoplastic cystic lesions and for those which are highly suspicious or have high potentials for malignancy is not up for debate although the type of cystic tumor, location and risk to the patient must all be considered (31). Minimally invasive treatment has gained strength as a means of treatment while distal and/or partial pancreatectomies have also been reported to produce good results in terms of low rates of recurrence and morbidity, and in terms of preserving the spleen in patients with tumors as large as 5cm (30).
Not counting the patient who had recently been operated on with conventional surgery, the four patients who underwent laparoscopies are part of the beginning of an experience in our institution. Although we did not find any reductions in lengths of hospital stays or surgery time, it was evident that postoperative pain was reduced and patients were able to begin oral intake sooner. Also, there was no need for blood transfusions during or after the procedures. As shown in the meta-analysis published in Annals of Surgery of 2012, there is clear evidence that laparoscopic procedures decrease blood loss during surgery, decrease length of hospital stays, reduce total complications including infection at the site of surgery, oncology intention (positive margin in cases of malign neoplasms), incidence of the pancreatic fistula and mortality (32). All of this is accomplished without loss of operating time. Although in the literature there are reports of conversion rates of between 0% and 30%, there were no patient conversions even though some lesions were larger than 10cm. We believe that this affected the obstructive complications of the colon resulting from intimate adherence of the mass to the mesocolon that was probably traumatized by release and subsequent ischemia and stenosis (32). We had no pancreatic fistulas in any of these patients for which we consider stapling of polypropylene reinforcement of the posterior pancreatic stump to be relevant. In the same meta-analysis fistulas rates, according the International Study Group of Pancreatic Fistula (ISGPF) classification by severity, ranged from 12.5% to 19.1% (33). We did not have any intra-abdominal or surgical site infections, a result which matches those reported in the literature.
We performed a total of 4 pancreatoduodenectomies all of which preserved the pylorus. Morbidity related to nosocomial infections developed in three of these patients. Two of these had origins unrelated to surgery (pneumonia and bacteremia from a catheter). The one infection which was due to bacterobilia had no documentable bilioenteric anastomotic stenosis, so a second intervention was not required and the patient improved with medical treatment. As we notice, this procedure presupposes risks which vary between 30% and 40% in published series (34, 35). Even when there is zero mortality among patients, associated with a group of young patients without comorbidities and for which appropriate surgical techniques were used, risks are not negligible as has been shown in other studies (36, 37). Hence the importance of an accurate diagnostic and surgical indication. There are reports in the literature that show the feasibility of performing this procedure in a total laparoscopic way or video assisted with good results regarding mortality, surgery time, transoperative blood loss, quick recovery and oncological safety (38).
CONCLUSIONS
Cystic tumors of the pancreas remain a rare pathology within our population, although this diagnosis is made more frequently every day due to the evolution and resolution of diagnostic methods. In our series, the abdominal CAT scans with contrast were sufficient to achieve diagnosis and adequate management of lesions larger than 3cm. In regards to smaller lesions, we consider relevant the use of endoscopy combined with histological study and tests for tumor markers relevant for establishing an accurate diagnostic. However, this is not always easy.
Laparoscopic treatment continues to evolve. As experience is gained, more complex procedures such as pancreatectomies become possible to perform in minimally invasive ways without affecting oncological principles. The four cases of patients who underwent laparoscopy showed complete resections with minimal complications plus some benefits from minimizing invasiveness. In any case this procedure requires experience and advanced laparoscopy skills. At the beginning of this experience we did not reduce times of surgery of patients lengths of hospital stays.
Even when the preservation of the spleen in distal pancreatectomy is described, this procedure is not recommended for lesions greater than 5cm. It would be even more difficult to try this procedure in the initial phase of the learning curve.
This initial experience encourages us to continue performing minimally invasive treatments for these neoplasms regardless of their size. Our idea is to evaluate in specific and prospective ways the different variables to be considered and the results of these procedures over the medium and long term.
Laparoscopic pancreaticoduodenectomies have been performed in increasing numbers by American, French and Indian groups. As volume and experience in advanced laparoscopic pancreatic surgery and edge technology increase, results become comparable to those achieved in centers of excellence in standard surgery. Thus we are faced with the question of whether these favorable outcomes can be achieved under different conditions.
REFERENCES
1. Fasanella KE, McGrath K. Cystic lesions and intraductal neoplasms of the pancreas. Best Pract Res Clin Gastroenterol 2009; 23(1): 35-48.
2. Spinelli KS, Fromwiller TE, Daniel RA, Kiely JM, Nakeeb A, Komorowski RA, Wilson SD, Pitt HA. Cystic pancreatic neoplasms: observe or operate. Ann Surg 2004; 239(5): 651-7.
3. Zinner MJ. Solid and papillary neoplasms of the pancreas. Surg Clin North Am 1995; 75(5): 1017-24.
4. Aaloten LA, et al. World Health Organization Classification of Tumours. Pathology and Genetics Tumour of the Digestive System. Lyon: IARC Press; 2000.
5. Kloppel G, et al. Histological typing of tumours of the exocrine pancreas. World Health Organization International Histological Classification of Tumours. Berlin: Springer-Verlag; 1996.
6. Khalid A, Brugge W. ACG practice guidelines for the diagnosis and management of neoplastic pancreatic cysts. Am J Gastroenterol 2007t; 102(10): 2339-49.
7. Centeno BA, Lewandrowski KB, Warshaw AL, Compton CC, Southern JF. Cyst fluid cytologic analysis in the differential diagnosis of pancreatic cystic lesions. Am J Clin Pathol 1994; 101(4): 483-7.
8. Frossard JL, Amouyal P, Amouyal G, Palazzo L, Amaris J, Soldan M, Giostra E, Spahr L, Hadengue A, Fabre M. Performance of endosonography-guided fine needle aspiration and biopsy in the diagnosis of pancreatic cystic lesions. Am J Gastroenterol 2003; 98(7): 1516-24.
9. Zamboni G, et al. Mucinous cystic neoplasms of the pancreas. En Aaolten LA, Hamilton SR, eds. World Health Organization Classification of Tumours. Pathology and Genetics Tumour of the Digestive System. Lyon:IARC Press; 2000.
10. Tanaka M, Chari S, Adsay V, Fernandez-del Castillo C, Falconi M, Shimizu M, Yamaguchi K, Yamao K, Matsuno S; International Association of Pancreatology. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas. Pancreatology 2006; 6(1-2): 17-32.
11. Reddy RP, Smyrk TC, Zapiach M, Levy MJ, Pearson RK, Clain JE, Farnell MB, Sarr MG, Chari ST. Pancreatic mucinous cystic neoplasm defined by ovarian stroma: demographics, clinical features, and prevalence of cancer. Clin Gastroenterol Hepatol 2004; 2(11): 1026-31.
12. Zamboni G, Scarpa A, Bogina G, Iacono C, Bassi C, Talamini G, Sessa F, Capella C, Solcia E, Rickaert F, Mariuzzi GM, Klöppel G. Mucinous cystic tumors of the pancreas: clinicopathological features, prognosis, and relationship to other mucinous cystic tumors. Am J Surg Pathol 1999; 23(4): 410-22.
13. Kosmahl M, Pauser U, Peters K, Sipos B, Lüttges J, Kremer B, Klöppel G. Cystic neoplasms of the pancreas and tumor-like lesions with cystic features: a review of 418 cases and a classification proposal. Virchows Arch 2004; 445(2): 168-78.
14. Vignesh S, Brugge WR. Endoscopic diagnosis and treatment of pancreatic cysts. J Clin Gastroenterol 2008; 42(5): 493-506.
15. Matsumoto T, Aramaki M, Yada K, Hirano S, Himeno Y, Shibata K, Kawano K, Kitano S. Optimal management of the branch duct type intraductal papillary mucinous neoplasms of the pancreas. J Clin Gastroenterol 2003; 36(3): 261-5.
16. Sugiyama M, et al. Predictive factors for malignancy in intraductal papillary mucinous tumours of the pancreas. Br J Surg 2003; 90(10): 1244-9.
17. Buetow PC, Buck JL, Pantongrag-Brown L, Beck KG, Ros PR, Adair CF. Solid and papillary epithelial neoplasm of the pancreas: imaging-pathologic correlation on 56 cases. Radiology 1996; 199(3): 707-11.
18. Brugge WR. Management and outcomes of pancreatic cystic lesions. Dig Liver Dis 2008; 40(11): 854-9.
19. Brugge WR. Evaluation of pancreatic cystic lesions with EUS. Gastrointest Endosc 2004; 59(6): 698-707.
20. Brugge WR, Lewandrowski K, Lee-Lewandrowski E, Centeno BA, Szydlo T, Regan S, del Castillo CF, Warshaw AL. Diagnosis of pancreatic cystic neoplasms: a report of the cooperative pancreatic cyst study. Gastroenterology 2004; 126(5): 1330-6.
21. Shah JN, Muthusamy VR. Minimizing complications of endoscopic ultrasound and EUS-guided fine needle aspiration. Gastrointest Endosc Clin N Am 2007; 17(1): 129-43.
22. Van der Waaij LA, van Dullemen HM, Porte RJ. Cyst fluid analysis in the differential diagnosis of pancreatic cystic lesions: a pooled analysis. Gastrointest Endosc 2005; 62(3): 383-9.
23. Tseng JF. Management of serous cystadenoma of the pancreas. J Gastrointest Surg 2008; 12(3): 408-10.
24. Harper AE, Eckhauser FE, Mulholland MW. Resectional therapy for cystic neoplasms of the pancreas. Am Surg 2002; 68(4): 353-7.
25. Balcom IV JH, Fernandez-Del Castillo C, Warshaw AL. Cystic lesions in the pancreas: when to watch, when to resect. CurrGastroenterol Rep 2000; 2(2): 152-8.
26. Kanazumi N, Nakao A, Kaneko T, Takeda S, Harada A, Inoue S, Nagasaka T, Nakashima N. Surgical treatment of intraductal papillary-mucinous tumors of the pancreas. Hepatogastroenterology 2001; 48(40): 967-71.
27. Palanivelu C, Shetty R, Jani K, Sendhilkumar K, Rajan PS, Maheshkumar GS. Laparoscopic distal pancreatectomy: results of a prospective non-randomized study from a tertiary center. SurgEndosc 2007; 21(3): 373-7.
28. Jang JY, Kim SW, Lee SE, Yang SH, Lee KU, et al. Treatment guidelines for branch duct type intraductal papillary mucinous neoplasms of the pancreas: when can we operate or observe? Ann SurgOncol 2008; 15(1): 199-205.
29. Roedor B, et al. Endocopic pancreatic pseudocyst drainage. Tech Gastrointest Endosc 2005; 7: 211-18.
30. Connell DB, Gregory JR, Sasaki TM, Vetto RM. Pancreatic pseudocyst. Am J Surg 1982; 143(5): 599-601.
31. Brugge WR. Should all pancreatic cystic lesions be resected? Cyst-fluid analysis in the differential diagnosis of pancreatic cystic lesions: a meta-analysis. Gastrointest Endosc 2005; 62(3): 390-1.
32. Venkat R, Edil BH, Schulick RD, Lidor AO, Makary MA, Wolfgang CL. Laparoscopic Distal Pancreatectomy is Associated with Significantly Less Overall Morbidity Compared to the Open Technique. A Systematic Review and Meta-Analysis. Ann Surg 2012; 00: 1-12.
33. Bassi C, Dervenis C, Butturini G, et al. Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery 2005; 138: 8-13.
34. Cameron J, Riall TS, Coleman J, Belcher KA. One thousand consecutive pancreatoduodenectomies. Ann Surg 2006; 244: 10-5.
35. Buchler M, Wagner M, Schmied BM, Uhl W, Friess H, ZGraggen K. Changes en morbidity afcter pancreatic resección; Toward the end of completion pancreatectomy. Arch Surg 2003; 138: 1310-4.
36. Hoyos S, Duarte A, Franco G, Chavez J, Gomez S, Sánchez JA. Evaluación y seguimiento de los pacientes sometidos a operación de Wipple o duodenopancreatectomía cefálica en un hospital de IV nivel de Medellín. Rev Colomb Cir 2012; 27: 114-120.
37. Qiao QL, Zhao YG, Ye ML, Yang YM, Zhao JX, Huang YT, et al. Carcinoma of de ampulla of Vater: Factors influencing long- term survival of 127 patients with resection. World J Surg 2007; 31: 137-43.
38. Dulucq JL, Wintringer P, Mahajna A. Laparoscopy pancreatoduodenectomy for benign and malignant diseases. Surg Endosc 2006; 20: 1045-1050.
1. Fasanella KE, McGrath K. Cystic lesions and intraductal neoplasms of the pancreas. Best Pract Res Clin Gastroenterol 2009; 23(1): 35-48. [ Links ]
2. Spinelli KS, Fromwiller TE, Daniel RA, Kiely JM, Nakeeb A, Komorowski RA, Wilson SD, Pitt HA. Cystic pancreatic neoplasms: observe or operate. Ann Surg 2004; 239(5): 651-7. [ Links ]
3. Zinner MJ. Solid and papillary neoplasms of the pancreas. Surg Clin North Am 1995; 75(5): 1017-24. [ Links ]
4. Aaloten LA, et al. World Health Organization Classification of Tumours. Pathology and Genetics Tumour of the Digestive System. Lyon: IARC Press; 2000. [ Links ]
5. Kloppel G, et al. Histological typing of tumours of the exocrine pancreas. World Health Organization International Histological Classification of Tumours. Berlin: Springer-Verlag; 1996. [ Links ]
6. Khalid A, Brugge W. ACG practice guidelines for the diagnosis and management of neoplastic pancreatic cysts. Am J Gastroenterol 2007t; 102(10): 2339-49. [ Links ]
7. Centeno BA, Lewandrowski KB, Warshaw AL, Compton CC, Southern JF. Cyst fluid cytologic analysis in the differential diagnosis of pancreatic cystic lesions. Am J Clin Pathol 1994; 101(4): 483-7. [ Links ]
8. Frossard JL, Amouyal P, Amouyal G, Palazzo L, Amaris J, Soldan M, Giostra E, Spahr L, Hadengue A, Fabre M. Performance of endosonography-guided fine needle aspiration and biopsy in the diagnosis of pancreatic cystic lesions. Am J Gastroenterol 2003; 98(7): 1516-24. [ Links ]
9. Zamboni G, et al. Mucinous cystic neoplasms of the pancreas. En Aaolten LA, Hamilton SR, eds. World Health Organization Classification of Tumours. Pathology and Genetics Tumour of the Digestive System. Lyon:IARC Press; 2000. [ Links ]
10. Tanaka M, Chari S, Adsay V, Fernandez-del Castillo C, Falconi M, Shimizu M, Yamaguchi K, Yamao K, Matsuno S; International Association of Pancreatology. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas. Pancreatology 2006; 6(1-2): 17-32. [ Links ]
11. Reddy RP, Smyrk TC, Zapiach M, Levy MJ, Pearson RK, Clain JE, Farnell MB, Sarr MG, Chari ST. Pancreatic mucinous cystic neoplasm defined by ovarian stroma: demographics, clinical features, and prevalence of cancer. Clin Gastroenterol Hepatol 2004; 2(11): 1026-31. [ Links ]
12. Zamboni G, Scarpa A, Bogina G, Iacono C, Bassi C, Talamini G, Sessa F, Capella C, Solcia E, Rickaert F, Mariuzzi GM, Klöppel G. Mucinous cystic tumors of the pancreas: clinicopathological features, prognosis, and relationship to other mucinous cystic tumors. Am J Surg Pathol 1999; 23(4): 410-22. [ Links ]
13. Kosmahl M, Pauser U, Peters K, Sipos B, Lüttges J, Kremer B, Klöppel G. Cystic neoplasms of the pancreas and tumor-like lesions with cystic features: a review of 418 cases and a classification proposal. Virchows Arch 2004; 445(2): 168-78. [ Links ]
14. Vignesh S, Brugge WR. Endoscopic diagnosis and treatment of pancreatic cysts. J Clin Gastroenterol 2008; 42(5): 493-506. [ Links ]
15. Matsumoto T, Aramaki M, Yada K, Hirano S, Himeno Y, Shibata K, Kawano K, Kitano S. Optimal management of the branch duct type intraductal papillary mucinous neoplasms of the pancreas. J Clin Gastroenterol 2003; 36(3): 261-5. [ Links ]
16. Sugiyama M, et al. Predictive factors for malignancy in intraductal papillary – mucinous tumours of the pancreas. Br J Surg 2003; 90(10): 1244-9. [ Links ]
17. Buetow PC, Buck JL, Pantongrag-Brown L, Beck KG, Ros PR, Adair CF. Solid and papillary epithelial neoplasm of the pancreas: imaging-pathologic correlation on 56 cases. Radiology 1996; 199(3): 707-11. [ Links ]
18. Brugge WR. Management and outcomes of pancreatic cystic lesions. Dig Liver Dis 2008; 40(11): 854-9. [ Links ]
19. Brugge WR. Evaluation of pancreatic cystic lesions with EUS. Gastrointest Endosc 2004; 59(6): 698-707. [ Links ]
20. Brugge WR, Lewandrowski K, Lee-Lewandrowski E, Centeno BA, Szydlo T, Regan S, del Castillo CF, Warshaw AL. Diagnosis of pancreatic cystic neoplasms: a report of the cooperative pancreatic cyst study. Gastroenterology 2004; 126(5): 1330-6. [ Links ]
21. Shah JN, Muthusamy VR. Minimizing complications of endoscopic ultrasound and EUS-guided fine needle aspiration. Gastrointest Endosc Clin N Am 2007; 17(1): 129-43. [ Links ]
22. Van der Waaij LA, van Dullemen HM, Porte RJ. Cyst fluid analysis in the differential diagnosis of pancreatic cystic lesions: a pooled analysis. Gastrointest Endosc 2005; 62(3): 383-9. [ Links ]
23. Tseng JF. Management of serous cystadenoma of the pancreas. J Gastrointest Surg 2008; 12(3): 408-10. [ Links ]
24. Harper AE, Eckhauser FE, Mulholland MW. Resectional therapy for cystic neoplasms of the pancreas. Am Surg 2002; 68(4): 353-7. [ Links ]
25. Balcom IV JH, Fernandez-Del Castillo C, Warshaw AL. Cystic lesions in the pancreas: when to watch, when to resect. CurrGastroenterol Rep 2000; 2(2): 152-8. [ Links ]
26. Kanazumi N, Nakao A, Kaneko T, Takeda S, Harada A, Inoue S, Nagasaka T, Nakashima N. Surgical treatment of intraductal papillary-mucinous tumors of the pancreas. Hepatogastroenterology 2001; 48(40): 967-71. [ Links ]
27. Palanivelu C, Shetty R, Jani K, Sendhilkumar K, Rajan PS, Maheshkumar GS. Laparoscopic distal pancreatectomy: results of a prospective non-randomized study from a tertiary center. SurgEndosc 2007; 21(3): 373-7. [ Links ]
28. Jang JY, Kim SW, Lee SE, Yang SH, Lee KU, et al. Treatment guidelines for branch duct type intraductal papillary mucinous neoplasms of the pancreas: when can we operate or observe? Ann SurgOncol 2008; 15(1): 199-205. [ Links ]
29. Roedor B, et al. Endocopic pancreatic pseudocyst drainage. Tech Gastrointest Endosc 2005; 7: 211-18. [ Links ]
30. Connell DB, Gregory JR, Sasaki TM, Vetto RM. Pancreatic pseudocyst. Am J Surg 1982; 143(5): 599-601. [ Links ]
31. Brugge WR. Should all pancreatic cystic lesions be resected? Cyst-fluid analysis in the differential diagnosis of pancreatic cystic lesions: a meta-analysis. Gastrointest Endosc 2005; 62(3): 390-1. [ Links ]
32. Venkat R, Edil BH, Schulick RD, Lidor AO, Makary MA, Wolfgang CL. Laparoscopic Distal Pancreatectomy is Associated with Significantly Less Overall Morbidity Compared to the Open Technique. A Systematic Review and Meta-Analysis. Ann Surg 2012; 00: 1-12. [ Links ]
33. Bassi C, Dervenis C, Butturini G, et al. Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery 2005; 138: 8-13. [ Links ]
34. Cameron J, Riall TS, Coleman J, Belcher KA. One thousand consecutive pancreatoduodenectomies. Ann Surg 2006; 244: 10-5. [ Links ]
35. Buchler M, Wagner M, Schmied BM, Uhl W, Friess H, Z'Graggen K. Changes en morbidity afcter pancreatic resección; Toward the end of completion pancreatectomy. Arch Surg 2003; 138: 1310-4. [ Links ]
36. Hoyos S, Duarte A, Franco G, Chavez J, Gomez S, Sánchez JA. Evaluación y seguimiento de los pacientes sometidos a operación de Wipple o duodenopancreatectomía cefálica en un hospital de IV nivel de Medellín. Rev Colomb Cir 2012; 27: 114-120. [ Links ]
37. Qiao QL, Zhao YG, Ye ML, Yang YM, Zhao JX, Huang YT, et al. Carcinoma of de ampulla of Vater: Factors influencing long- term survival of 127 patients with resection. World J Surg 2007; 31: 137-43. [ Links ]
38. Dulucq JL, Wintringer P, Mahajna A. Laparoscopy pancreatoduodenectomy for benign and malignant diseases. Surg Endosc 2006; 20: 1045-1050. [ Links ]











 texto em
texto em