Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.27 no.3 Bogotá July/Sept. 2012
The experience of a Venezuelan medical center in diagnosis and treatment of nonpolypoid neoplastic colorectal lesions with high resolution equipment without magnification
Yolette Martínez, MD. (1), María Alexandra Guzmán, MD. (2), Raúl Monserrat, MD. (3), Sandra Romero, MD. (4)
(1) Gastroenterologist at the Policlínica Santiago de León.
(2) Gastroenterology Fellow at the Hospital de Clínicas in Caracas, Venezuela.
(3) Gastroenterologist at the Centro Médico in Caracas, Venezuela.
Abstract
Rationale. It is well known that adenomas are the morphological precursors of the vast majority of colorectal cancers. Nevertheless, nonpolypoid lesions follow pathways which differ from the known sequence from adenoma to carcinoma. Since these pathways appear to be more aggressive, early detection is crucial for definition of the natural history of these cancers.
Objective. In our study we used both conventional chromoendoscopy with blue methylene and indigo carmine and electronic chromoscopy with the NBI (Olympus) system to identify nonpolypoid neoplastic colorectal lesions without magnification. All lesions were treated by endoscopic mucosal resection since this technique is safe and effective and allows the complete specimen to be saved for histopathological study.
Design. We found 155 (12.6%) cases of nonpolypoid neoplastic colorectal lesions as defined by the Paris classification in a total of 1,234 colonoscopies performed between 2006 and 2010. All were removed through EMR in single sessions without complications.
Results. Nonpolypoid neoplastic lesions were found in 155 patients (from 1.234 colonoscopies, 12,6%). The lesions were classified as follows: 125 were IIa (80,65%), 24 were lateral spreading tumors (15,48%), 4 were IIb (2,58%), and 2 were IIc (1,29%). 70 were in the right colon (45%), and 85 were in the left colon (55%). Average patient age was 61 years and the age range was from 42 to 81 years old. 87 patients were male, and 68 were female. The sizes of the lesions ranged from 5mm to 50 mm. 16 (10,32%) nonpolypoid lesions were diagnosed as normal and hyperplastic. The other 139 (89,68%) were neoplastic lesions: 10 were diagnosed as serrated adenomas, 119 were tubular adenomas (18 with high-grade dysplasia), 9 were tubulovillous adenomas with high degrees of dysplasia, and 1 was a well differentiated adenocarcinoma. Sensitivity was 100% and specificity was 98,5%.
Limitations. There were two limitations: the study was carried out in only one medical center and the sample size was limited.
Findings. The results suggest that endoscopy without magnification was useful and sensitive for identification of nonpolypoid lesions in the colon. It is important to note that a significant number of lesions were found in the right colon. Endoscopic mucosal resection is a safe, minimally invasive procedure that can be performed widely. In addition it allows the physician to obtain the complete specimen for histological study.
Key words
Nonpolypoid neoplastic lesions, high resolution, magnification, the Paris classification.
INTRODUCTION
Nonpolypoid neoplastic lesions of the colon represent a variety of premalignant colon pathology that sometimes leads to greater risk for cancer when than do classic polypoid lesions with their well-known adenoma-carcinoma sequence. Although they were described in 1985 by Japanese authors (1) there are few studies in the Western Hemisphere (2, 3). This verified the small importance initially given to identification of these small lesions which now are known to vitally require early identification. For these reasons we consider it useful to present our experience in the diagnosis and treatment of Nonpolypoid neoplastic lesions of the colon.
MATERIALS AND METHODS
The study population consisted of all adult patients who underwent colonoscopies in Caracas Medical Center between January 2006 and December 2010. Patients were included if they met the following criteria:
1. Patients were asymptomatic but had intermediate risk of colorectal cancer.
2. Patients were symptomatic and had anemia, were positive for occult blood, had changes in bowel habits, and exhibited rectal bleeding, weight loss and abdominal pain.
3. Patients were being monitored because of histories of colon polyps and/or colon cancer surgery.
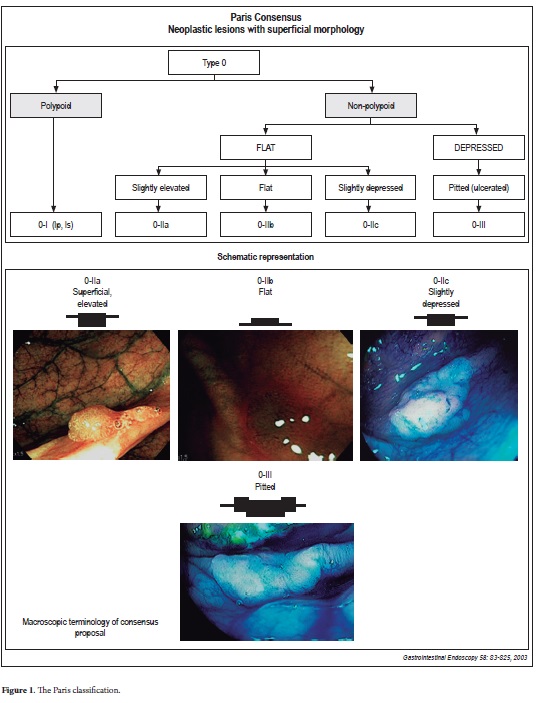
Exclusion criteria were incomplete colonoscopies and unprepared colons. The endoscope used was an Olympus colonoscope F-H180 A1 series. Endoscopies were performed in the morning after colon cleansing with divided doses combining phosphosoda (90 cc) the evening before and Polyethylene glycol (2 liters) before the endoscopy. Sedation with and IV Hyoscine Bromide 5 mg and either Midazolam 7.5 mgr or Propofiol was administered by the gastroenterologist. Endoscope withdrawal time was at least 6 minutes. Methods of enhancement used included acetic acid, indigo carmine, and electronic Chromoscopy (NBI). The Paris classification was used to classify all lesions (Figure 1) (4).
Figure 1. The Paris classification.
All lesions were resected endoscopically except when contraindicated and when the endoscopist considered that lesions were advanced cancer without adequate indication for endoscopic mucosal resection. Cases of laterally spreading tumors larger than 3 cms were partially resected. 30% hypertonic glucose was used sued for endoscopic mucosal resections. All specimens were examined by a pathologist who is an expert in the gastrointestinal area.
RESULTS
Of a total of 1,234 colonoscopies performed, 155 patients presented nonpolypoid tumors (12.6%). Lesion sizes ranged between 5 and 50 mm. Lesions were identified as follows: 125 (80%) type IIa: 24 (15.48 %) lateral spreading tumors, 4 (2.58 %) type IIb, 2 (1.29%) types IIa and IIc (Table 1). The only lesions larger than 2 cm were found in cases of lateral spreading tumors. 139 lesions (89.6%) were histologically diagnosed as neoplasias, but the remaining cases were histologically normal or hyperplastic. Benign neoplastic lesions were distributed as follows: 119 tubular adenomas, 9 tubulovillous adenomas, 10 serrated adenomas and 1 well-differentiated adenocarcinoma (Table 2). It should be noted that 18 of the lesions presented high-grade dysplasia. In two cases delayed bleeding occurred and was controlled endoscopically. There were no perforations.
Table 1. Type of lesion found.
Table 2. Histological diagnosis.
DISCUSSION
Although non polypoid neoplastic lesions were first described more than twenty years in Japan (1), they have received little attention by Western endoscopists. Only recently have studies been reported in the U.S. and Europe which shows that Western endoscopists may be waking up to this pathology (2, 3). The literature shows that the prevalence of these lesions ranges from 4 to 25%, (13) but there is still uncertainty about the clinical relevance of these findings. The prevalence of these lesions in our study (12.6%) compares favorably with those found in series in the USA, Italy and China (2, 5, 6). Also, as in these other series, we found a predominance of these lesions in the right colon. The clinical significance of this fact most likely lies in the high incidence of right colon cancer that has gone undetected during early diagnostic colonoscopies (7). This emphasizes the need for better and more detailed exploration of this segment of the colon in light of recent progress in understanding the events that lead to polypoid and nonpolypoid colorectal cancer.
In fact, today we require better detection and characterization of neoplastic lesions thanks to the increasingly frequent use of chromoendoscopy, the use of high resolution and high definition endoscopes, and a better understanding of the molecular biology of polypoid lesions (12). These developments have led us to clearly question the concept of a single pathway in the adenoma-carcinoma sequence (12). Currently, three types of lesions are recognized: polypoid, non polypoid and depressed. Usually, polypoid lesions grow over the surface and polypoid volume correlates with histological stage (13). Nonpolypoid lesions usually are flat or slightly depressed and may eventually progress to lateral spreading tumors or polypoid lesions. Depressed lesions appear to constitute a separate group because of their greater tendency to develop into cancer and lymphatic spread (14). For proper identification of these lesions optimal cleaning of the colon and a high percentage of cecal intubation are essential because many of the lesions are located in the right colon. This last point is of fundamental importance because of the high percentage of incomplete colonoscopies performed. Frequently this is due to inadequate preparation (15).
In our series we highlight the percentage of complete colonoscopies and optimal preparation (99%).
It is important to remark that in this series we only show our findings with endoscopic equipment that allows for characterization of lesions by narrow band imaging (NBI) without magnification. This, in our opinion, should be very useful, because endoscopic magnification equipment is not available to most endoscopists. By using our methodology, we obtained a percentage of 12.4% which relates favorably to those of other studies (2, 5, 6, 10).
The number of injuries with high-grade dysplasia even in small lesions it is of particularly relevance in regard to histological study. This is consistent with the views expressed by other authors as to the higher propensity of nonpolypoid tumor lesions to develop into cancer (14). Also, in this series we highlight the finding of serrated polyps because it suggests that there currently is a need to search for this morphology and have histological studies performed by pathologists who are experts in the gastrointestinal area as we did in this series.
CONCLUSSIONS
In our environment nonpolypoid neoplastic lesions appear at rates similar to those in series published in the literature. This motivates us to take extreme measures to recognize them. The emphasis should be on complete colonoscopies and ensuring optimal colon preparation. Furthermore, we assert that the treatment of these injuries by endoscopic mucosal resection is safe and effective for the eradication of these lesions.
REFERENCES
1. Muto T, Kamiya J, Sawada T, et al. Small flat Adenoma of the large bowel with special reference to its clinicopathological features. Diseases of Colon and Rectum 1985; 28: 887-51.
2. Ress AS, Waxman F. Flat and depressed neoplasms of the Colon in Western Population. Am J Gastroenterology 2006; 101: 172-180.
3. Soetinkno RM, Kattenbach, Rense R, et al. Prevalence of non polipoid (flat and depressed) colorectal neoplasms in asymptomatic adults. JAMA 2008; 299: 1027-35.
4. The Paris Endoscopic Classification of superficial neoplasms lesions: Esophagus, Stomach and Colon. Gastrointestinal Endoscopy. 2003; 58(suppl.): 53.
5. Neugut Al, Jacobson JS, Rella VA. Prevalence and incidence of colorectal adenomas and cancer in asymptomatic persons. Gastrointestinal Endosc Clin N Am 1977; 378-99.
6. Rex DK. Maximizing detection of adenoma and cancer during colonoscopy. Am J Gastroenterology 2006; 101: 2866-77.
7. Harminder Singh, Donna Turner, Lin Xue, et al. Risk of developing of colorectal cancer following a negative colonoscopy examination: evidence for 10 years interval between colonoscopy. JAMA 2006; 295: 2366-2373.
8. Brooker Jr, Sauridus BP, Shohga, et al. Total colonic dye-spray increases the detection of diminute adenomas during routine colonoscopy; a randomized control trial. GIE 2002; 56: 333-8.
9. Jogahus MJ, Helbat T, Napoleon R, et al. Does Chromoendoscopy with structure enhancement increase colonoscopy adenoma detection note. Endoscopy 2006; 38: 444.
10. Chiu HM, Chang CY, Chen CC, et al. A prospective comparative study of Narrow Band Imaging, Chromoendoscopy in the diagnosis of colorectal neoplasia. Gut 2007; 56: 373-9.
11. Arain MA, Sauhney S, et al. Status of interval Colon Cancer: Another piece of the puzzle. Am J Gastroenterology 2009; 209: 10-1038.
12. Jas JR, Whitehall VLJ, Yang J, et al. Emerging Concepts in Colorectal neoplasia. Gastroenterology 2002; 123: 862-76.
13. Hanawood GC, Sharma VK, et al. Impact of Colonoscopy preparation quality on detection of colonic neoplasia. Gastrointestinal Endoscopy 2003; 58: 76-79.
14. Kudo S, Tamaura S, Nakajima T, et al. Depressed type of colorectal Cancer. Endoscopy 1995; 27: 54-7.
15. Rex DK, et al. Quality indicator for colonoscopy. Am J Gastroenterology 2006; 101: 873-85.
1. Muto T, Kamiya J, Sawada T, et al. Small flat Adenoma of the large bowel with special reference to its clinicopathological features. Diseases of Colon and Rectum 1985; 28: 887-51. [ Links ]
2. Ress AS, Waxman F. Flat and depressed neoplasms of the Colon in Western Population. Am J Gastroenterology 2006; 101: 172-180. [ Links ]
3. Soetinkno RM, Kattenbach, Rense R, et al. Prevalence of non polipoid (flat and depressed) colorectal neoplasms in asymptomatic adults. JAMA 2008; 299: 1027-35. [ Links ]
4. The Paris Endoscopic Classification of superficial neoplasms lesions: Esophagus, Stomach and Colon. Gastrointestinal Endoscopy. 2003; 58(suppl.): 53. [ Links ]
5. Neugut Al, Jacobson JS, Rella VA. Prevalence and incidence of colorectal adenomas and cancer in asymptomatic persons. Gastrointestinal Endosc Clin N Am 1977; 378-99. [ Links ]
6. Rex DK. Maximizing detection of adenoma and cancer during colonoscopy. Am J Gastroenterology 2006; 101: 2866-77. [ Links ]
7. Harminder Singh, Donna Turner, Lin Xue, et al. Risk of developing of colorectal cancer following a negative colonoscopy examination: evidence for 10 years interval between colonoscopy. JAMA 2006; 295: 2366-2373. [ Links ]
8. Brooker Jr, Sauridus BP, Shohga, et al. Total colonic dye-spray increases the detection of diminute adenomas during routine colonoscopy; a randomized control trial. GIE 2002; 56: 333-8. [ Links ]
9. Jogahus MJ, Helbat T, Napoleon R, et al. Does Chromoendoscopy with structure enhancement increase colonoscopy adenoma detection note. Endoscopy 2006; 38: 444. [ Links ]
10. Chiu HM, Chang CY, Chen CC, et al. A prospective comparative study of Narrow Band Imaging, Chromoendoscopy in the diagnosis of colorectal neoplasia. Gut 2007; 56: 373-9. [ Links ]
11. Arain MA, Sauhney S, et al. Status of interval Colon Cancer: Another piece of the puzzle. Am J Gastroenterology 2009; 209: 10-1038. [ Links ]
12. Jas JR, Whitehall VLJ, Yang J, et al. Emerging Concepts in Colorectal neoplasia. Gastroenterology 2002; 123: 862-76. [ Links ]
13. Hanawood GC, Sharma VK, et al. Impact of Colonoscopy preparation quality on detection of colonic neoplasia. Gastrointestinal Endoscopy 2003; 58: 76-79. [ Links ]
14. Kudo S, Tamaura S, Nakajima T, et al. Depressed type of colorectal Cancer. Endoscopy 1995; 27: 54-7. [ Links ]
15. Rex DK, et al. Quality indicator for colonoscopy. Am J Gastroenterology 2006; 101: 873-85. [ Links ]











 text in
text in