Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.27 no.3 Bogotá July/Sept. 2012
Primary follicular lymphoma of the duodenum: Case report and literature review
Juan Carlos Hiromi López T., MD (1), María del Pilar Casasbuenas D., MD (2), Jinneth Acosta F., MD (3), Orlando Ricaurte G., MD (3)
(1) Fourth-year Resident in Clinical and Anatomical Pathology.
(2) Gastroenterologist and Endoscopist at the Clínica de Marly, Assistant Professor in the Faculty of Medicine at the Universidad del Bosque in Bogotá, Colombia.
(3) Medical Pathologist, Associate Professor in the Molecular Pathology Group of the Department of Pathology in the Faculty of Medicine at the Universidad Nacional de Colombia in Bogotá, Colombia. Correspondence: oricaurteg@unal.edu.co
Abstract
Patient was an 80 year old woman with dyspepsia. An upper endoscopy showed confluent polypoid lesions in the second portion of the duodenum. A histological study showed a follicular B-cell lymphoma. This is a rare low grade neoplasm of duodenum which must be differentiated from follicular reactive hyperplasia and other lymphomas.
Keywords
Duodenal polypoid lesion, primary B-cell lymphoma of the digestive tract, primary duodenal lymphoma, follicular lymphoma.
INTRODUCTION
Primary lymphomas of the digestive tract are uncommon and constitute a heterogeneous group of neoplasms. Their most frequent histological types are marginal zone lymphomas including mucosa-associated lymphoid tissue (MALT) and diffuse large B-cell lymphomas. Both occur primarily in the stomach, and are the most common extranodal lymphomas. Other lymphomas such as lymphomatous polyposis (mantle cell lymphoma), non- Hodgkin B-cell lymphoma and T-cell lymphoma associated with enteropathy occur in the gastrointestinal tract at significantly lower frequencies (1-5). Primary follicular lymphomas of the gastrointestinal tract are even more unusual, accounting for only 1% to 3.6% of primary non-Hodgkin lymphomas of the digestive tract (6, 7). One of its common locations is the duodenum. Histological differential diagnosis of primary follicular lymphomas of the gastrointestinal tract must consider follicular lymphoid hyperplasia, secondary digestive compromise from follicular lymphomas originating in the lymph nodes, primary marginal zone lymphomas derived from MALT and lymphomatous polyposis. We present the case of a patient with a primary follicular lymphoma of the duodenum.
CASE PRESENTATION
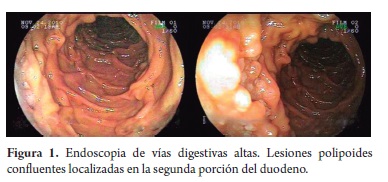
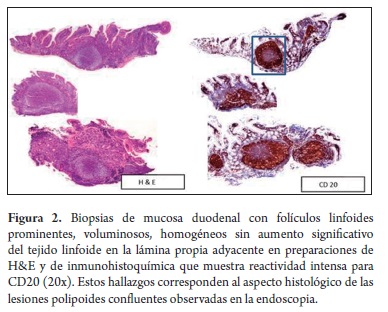
An 80 year old woman who was born and raised in Malaga, Santander came to the towns hospital with dyspepsia and unmeasured weight loss. An upper endoscopy performed there identified an antral polyp. Histopathological study showed a tubular adenoma. A CT scan showed no masses, visceromegaly or lymphadenopathy at any level. Chest x-rays, blood count and glycemia were also within normal limits and no lymphadenopathy was identified. Subsequently the patient was referred for examination in Bogotá because of the same symptoms and unmeasured intermittent fever. Physical examination showed no superficial lymphadenopathy, masses, visceromegaly, or other remarkable findings. Patient underwent endoscopy again. This time it showed confluent polypoid lesions located in the second portion of the duodenum which had displaced the ampulla of Vater (Figure 1). Other findings suggested chronic gastritis. The esophagus and duodenal bulb showed no alterations. After biopsies of the duodenal and gastric lesions were taken, histological study showed atypical nodular lymphoid infiltrate in the duodenum. It consisted entirely of bulky and uniform lymphoid follicles (Figure 2) with non polarized germinal centers which predominantly consisted of small cloven lymphocytes, centrocytes, occasional large cells, centroblasts (less than 5 per 40x field) which lacked stainable body macrophages. There was no significant increase in the lymphoid population in the adjacent lamina propria, and no lymphoepithelial lesions were identified.
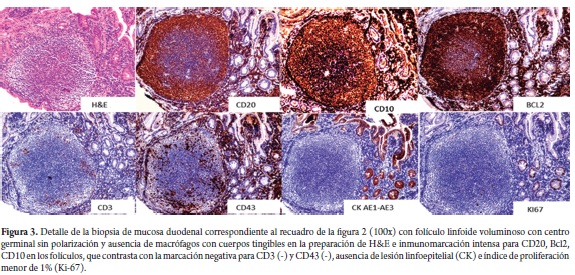
Based on these findings we proposed a differential diagnosis for follicular lymphoma and reactive follicular lymphoid hyperplasia for which a complementary immunohistochemistry study was conducted (Figures 2 and 3). The study showed reactivity for CD20, Bcl 2 and CD10 in the cellular elements of the follicles while markings for CD3 and CD43 were restricted to scattered reactive mature T lymphocytes which were predominantly located in the lamina propria in the vicinity of the follicles. No lymphoepithelial lesions were found with cytokeratin AE1/AE3. Cell proliferation index with Ki-67 was less than 1%. The markings for follicular lymphocytes and the low rate of cell proliferation are characteristics of follicular non-Hodgkin B lymphoma and exclude reactive follicular hyperplasia. The absence of superficial, abdominal and thoracic lymphadenopathy during the physical examination and extension studies (abdominal CT, chest radiography) indicate the primary duodenal character and exclude secondary digestive compromise from a nodal lymphoid neoplasia. The study of gastric biopsies showed non atrophic active chronic antral gastritis associated with Helicobacter pylori infection. This was treated with triple eradication therapy which resulted in improvement of symptoms of dyspepsia. Constitutional symptoms resolved spontaneously.
DISCUSSION
The lymphoid tissue of the gastrointestinal tract (GI tract) is the largest component of the mucosa-associated lymphoid tissue (MALT). Located in the lamina propria and the submucosa, its density varies across segments. Any segment can present primary lymphoid neoplasms or secondary compromise from lymphomas from neighboring nodal groups (1, 2, 4, 5). Several risk factors for the development of gastrointestinal lymphomas have been identified. Among them are Helicobacter pylori infections, immunosuppression after transplantation of solid organs, celiac and inflammatory intestinal disease and infection by human immunodeficiency virus (8).
Primary GI tract lymphomas are rare neoplasms but correspond to the most common extranodal location of non-Hodgkin lymphomas of which they account for about 30%. They often compromise the stomach, although less frequently they can affect other segments from the esophagus to the rectum (1- 5, 8, 9).
Hematolymphoid neoplasias secondary compromise of the GI tract is frequent in advanced states. It is usually multifocal and affects several segments of the GI tract because it metastasizes from mesenteric and retroperitoneal lymph nodes that have been compromised. In contrast, primary lymphomas usually involve a single segment of the GI tract. Criteria for diagnosing primary gastrointestinal lymphomas are absence of palpable superficial adenopathy, radiographic studies of the chest and abdominal tomography showing no lymphadenopathy, normal total and differential white cell count, predominant compromise of the GI tract segment involved. In the case of a compromised lymph node, it should be restricted to that nodes drainage group and not extend to the liver or spleen (8, 10).
The most common histological type of GI tract primary lymphomas is marginal zone lymphoma associated with MALT. This represents up to 10% of B lymphomas and up to 50% of gastric lymphomas. MALT lymphomas are followed by diffuse large B cell lymphomas which account for 60% of gastric lymphomas. These may be secondary to the transformation of a marginal zone lymphoma or they may recur. Mantle cell lymphoma or lymphomatous polyposis accounts for up to 30% of primary GI tract lymphomas. Its presentation is different from the previously described lymphomas since it is usually multifocal. Follicular B lymphomas, Burkitt lymphomas, T cell lymphomas associated with enteropathy, and monomorphic CD56+ intestinal lymphomas are much less frequent. Hodgkins lymphoma in the GI tract is very unusual (1-5).
Nodal follicular lymphoma (FL) is one of the most frequent types of non-Hodgkin lymphoma (NHL). Most cases of FL are initially detected in the lymph nodes. Visceral compromise is secondary to dissemination from compromised nodal groups. It has an indolent course and therefore is usually diagnosed in advanced stages (stages III or IV). (3, 6). Primary duodenal follicular lymphoma, first described in 1997 (11), is a very rare neoplasm accounting for only 1% to 3.6% of all primary GI tract lymphomas (4, 6, 7). Until 2010 there were only 249 cases reported in the Anglo-Saxon literature. About 50% of these were in Japan, possibly due higher frequency of endoscopic studies in that country (6, 7). Endoscopic diagnoses must differentiate between Primary duodenal follicular lymphoma and epithelial polyps and eventually lymphomatous polyposis (mantle non Hodgkin B lymphoma) while histological studies include those for reactive follicular lymphoid hyperplasia, secondary compromise from nodal follicular lymphoma and primary gastrointestinal marginal zone lymphoma when accompanied by prominent reactive lymphoid follicles and mantle cell lymphoma (lymphomatous polyposis), although these are also rare in the duodenum.
The average age for patients with primary follicular lymphoma of the GI tract is 56 years with a range between 26 and 81 years with predominance in females. Most patients (43%) are asymptomatic, although up to 10% present vague gastrointestinal symptoms. Their diagnosis is incidental in endoscopies performed for other reasons. Other symptoms reported include abdominal pain (28%), nausea and vomiting (8%) and gastrointestinal bleeding (6%). 66% of cases are Stage I at the time of diagnosis and 27% are Stage II. This is similar to other extranodal follicular lymphomas such as primary skin follicular lymphoma (4, 6, 7, 8, 12).
Although primary follicular lymphoma of the GI TRACT is usually unifocal, the increasing use of capsule video enteroscopy has led to some reports of multifocal compromise of distal segments of the small intestine. The most frequent location involved is the second portion of the duodenum in the vicinity of the ampulla of Vater, as was this case with the patient in this report. This location is followed by the terminal ileum, the stomach, and more rarely the colon and rectum. The endoscopic appearance of primary follicular lymphoma of the GI tract is characterized by dispersed confluent or whitish polypoid lesions up to 2 mm in diameter. They are usually covered by normal appearing mucosa when their location is in the submucosa or is subepithelial. Due to their slow growth they are not usually associated with ulceration (4, 6, 7, 8).
Histopathologic findings of primary follicular lymphoma of the GI tract are similar to those for follicular lymphoma originating in the lymph nodes. They are characterized by a nodular growth pattern or less frequently by a nodular and diffuse pattern consisting of bulky homogeneous follicles with non polarized germinal centers composed of two kinds of cells. For the most part they have small and irregular nuclear contours and inconspicuous cytoplasm (centrocytes), but to a lesser extent are composed of large cells with vesicular nuclei with loose chromatin, prominent nucleoli and visible cytoplasm (centroblasts). Their size in a 40x field determines the histological grade of the tumor (grade 1: 0-5, Level 2: 6-15, grade 3: more than 15). They proliferate slowly because they are neoplasms with an indolent course. Typically they lack stainable body macrophages. This confers a starry sky pattern in the germinal centers of reactive follicles which is a characteristic of follicular hyperplasia. In some cases they are accompanied by marginal zone lymphoma for which a differential diagnosis must be established. In most cases of primary follicular lymphoma of the GI tract, no tumor cells infiltrate any glandular structures. As a result there are usually no lymphoepithelial lesions as there characteristically are in marginal zone lymphomas (4, 6, 7, 8).
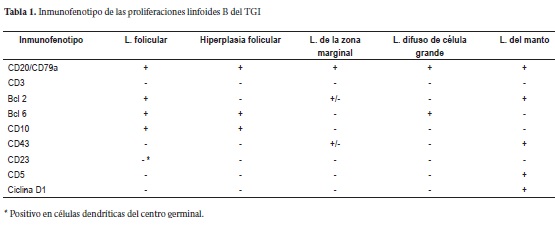
The immunophenotype of tumor cells in primary follicular lymphoma of the GI tract is characterized by the expression of pan-B markers (CD19, CD20, CD22 and CD 79a among others). It tests negative for CD43, CD5, D1 and cyclin. CD43 is a significant marker expressed in marginal zone lymphomas while CD5, D1 and cyclin characterize mantle lymphomas. 90% of primary follicular lymphomas of the GI tract present markers for CD10 and Bcl-2 in the germinal centers. These are useful markers for differentiation of reactive follicular hyperplasia, mantle lymphomas and marginal zone lymphomas which are all MALT derivatives. Table 1 compares primary follicular lymphoma of the GI TRACT immunophenotypes with those of reactive follicular hyperplasia and other non-Hodgkin B lymphomas of the digestive tract (4, 6, 7, 8).
70-95% of nodal follicular lymphomas present translocation t(14;18)(q32,q21) that causes the fusion of the Bcl-2 gene with the gene for IgH (immunoglobulin heavy chain). This translocation has also has been described in primary follicular lymphoma of the GI tract but is absent in the primary skin follicular lymphoma. This suggests a closer relation between primary follicular lymphoma of the GI tract and nodal follicular lymphoma than between primary follicular lymphoma of the GI tract and skin follicular lymphoma (4). However, follicular lymphoma originating in the duodenum lacks activity of cytidine deaminase, the enzyme involved in induction of recombination and somatic hypermutation. This is the cause of network patterns of dendritic cells, a characteristic of follicular nodal lymphomas (14). Some cases of primary follicular lymphoma of the GI tract share genotypical and molecular features with those of marginal zone lymphoma. These shared features include restriction of the immunoglobulin heavy chain gene to VH4 and VH5 regions. This is related the possibility that they develop from B lymphocytes subject to persistent antigenic stimulation. This would be similar to the process that occurs with marginal zone gastric lymphoma and its association with persistent antigenic stimulation due to H pylori infection (14). This pathogenic association has also been corroborated by regression analyses of cases of primary follicular lymphoma of the GI tract following H pylori antibiotic eradication therapy (15). All of these features suggest that primary follicular lymphoma of the GI tract is a specific nosological entity which is different from the other lymphomas mentioned.
There are different types of treatment for non-Hodgkin lymphomas. Treatment selection depends on the type, histological grade and clinical condition diagnosed. For primary GI tract follicular lymphoma which has survival times ranging from 10 to 16 years, treatment regimes must be customized according to the characteristics of each patient.
Below we list the treatment options for primary follicular lymphoma of the GI tract (12) according to the recommendations of the British National Lymphoma Investigation in 2003 (16).
Watch and wait: Observation without starting treatment for patients without evidence of local spread, itching, weight loss, fever higher than 38.5 ° C, night sweats, vital organ and bone marrow involvement, bone lesions, renal infiltration, macroscopic compromise of the liver or rapid progression of the disease.
Specific treatment for patients with evidence of disease progression. The following treatment options are available for patients who have the symptoms listed above:
Radiation therapy for local control. Primary GI tract follicular lymphoma is highly sensitive to radiation.
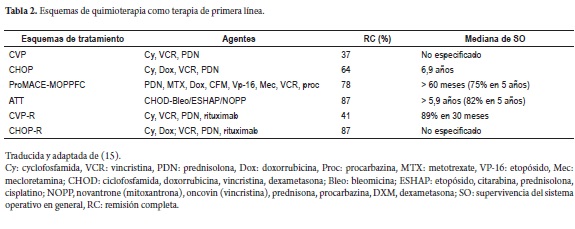
Chemotherapy: CVP and CHOP schemes among others have proven to be highly effective (Table 2).
Interferon and Rituximab- humanized monoclonal antibody anti-CD20.
Radioimmunotherapy (RIT): Systemic administration of monoclonal antibodies (anti-CD20) along with a radioisotope that allows radiation therapy specifically against tumor cells.
CONCLUSION
We presented the case of an elderly female patient with primary follicular lymphoma of the duodenum. This is an uncommon low grade neoplasia which is usually diagnosed incidentally at early stages and consequently patients have good prognoses. Its endoscopic polypoid appearance suggests a differential diagnosis of epithelial lesions and lymphomatous polyposis, the most frequent such entities. Its specific diagnosis is established by histological and immunohistochemical studies which exclude reactive follicular lymphoid hyperplasia and B lymphomas. Among these secondary gastrointestinal compromise due to nodal follicular lymphoma stands out out because it occurs in advanced stages. Advanced states of the disease are also characterized by superficial thoracic and abdominal lymphadenopathy and marginal zone and mantle lymphoma (lymphomatous polyposis). These are principally in the digestive tract, but are unusual in the duodenum where their biological behavior and treatment differ.
REFERENCES
1. Isaacson PG, Wright DH. Malignant lymphoma of mucosa associated lymphoid tissue. A distinctive type of B-cell lymphoma. Cancer 1983; 52: 1410-1416.
2. Wotherspoon AC, Ortiz-Hidalgo C, Falzon M, et al. Helicobacter pyloriassociated gastritis and primary B-cell gastric lymphoma. Lancet 1991; 338: 1175-1176.
3. Harris NL, Swerdlow SH, Jaffe ES, Olt G, Nathwani BN, de Jong D, Yoshino T, Spagnolo D. Follicular lymphoma in: Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pireli S, Stein H, Thiele J, Vardiman J. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. 4th Edition. International Agency for Research on Cancer: Lyon-France; 2008. p. 220-228.
4. Nakamura S, Müller- Hermelink HK, Delabie J, Ko YH, Jaffe ES. B cell lymphoma of the small intestine in: Bosman FT, Carneiro F, Hruban RH, Theise DT. WHO Classification of Tumours of the digestive system. 4th Edition. International Agency for Research on Cancer: Lyon-France; 2010. p. 69-73.
5. Nakamura S, Müller- Hermelink HK, Delabie J, Ko YH, van Krieken JH, Jaffe ES. Lymphoma of the stomach in: Bosman FT, Carneiro F, Hruban RH, Theise DT. WHO Classification of Tumours of the digestive system 4th Edition, 2010. International Agency for Research on Cancer: Lyon-France; 2010. p. 108-111.
6. Yamamoto S, Nakase H, Yamashita K, Matsuura M, Takada M, Kawanami C, Chiba T. Gastrointestinal follicular lymphoma: review of the literature. J Gastroenterol 2010; 45: 370-388.
7. Yoshino T, Miyake K, Ichimura K, Mannami T, Ohara N, Hamazaki S, et al. Increased incidence of follicular lymphoma in the duodenum. Am J Surg Pathol 2000; 24: 688-93.
8. Ghai S, Pattison J, Ghai S, OMalley ME, Khalili K, Stephens M. Primary Gastrointestinal Lymphoma: Spectrum of Imaging Findings with Pathologic Correlation. RadioGraphics 2007; 27: 1371-1388.
9. López R, Andrade R. Tejido linfoide y linfomas gástricos. Rev Col Gastroenterol 2010; 4: 409-421.
10. Dawson IM, Cornes JS, Morson BC. Primary malignant tumors of the intestinal tract. Br J Surg 1961; 49: 80-89.
11. Misdraji J, Fernandez del Castillo C, Ferry JA. Follicle center lymphoma of the ampulla of Vater presenting with jaundice: report of a case. Am J Surg Pathol 1997; 21: 484-8.
12. Sentani K, Maeshima AM, Nomoto J, Maruyama D, Kim S-M, Watanabe T, Kobayashi Y, Tobinai K, Matsuno Y. Follicular Lymphoma of the Duodenum: A Clinicopathologic Analysis of 26 Cases. Jpn J Clin Oncol 2008; 38(8): 547-552.
13. Goodlad JR, MacPherson S, Jackson R, Batstone P, White J. Extranodal follicular lymphoma: a clinicopathological and genetic analysis of 15 cases arising at non-cutaneous extranodal sites. Histopathology 2004; 44: 268-76.
14. Takata K, Sato Y, Nakamura N, Kikuti YY, Ichimura K, Tanaka T, Morito T, Tamura M, Oka T, Kondo E, Okada H, Tari A, Yoshino T. Duodenal and nodal follicular lymphomas are distinct: the former lacks activation-induced cytidine deaminase and follicular dendritic cells despite ongoing somatic hypermutations. Modern Pathology 2009; 22: 940-949.
15. Toyoda H, Yamaguchi M, Nakamura S, Nakamura T, Kimura M, Suzuki H, et al. Regression of primary lymphoma of the ampulla of Vater after eradication of Helicobacter pylori. Gastrointest Endosc 2001; 54: 92-6.
16. Rohatiner AZ, Davies A, Montoto S, Lister TA. Follicular Lymphoma (Chapter 19) in: The lymphomas. Second edition. Elsevier Inc; 2006. p. 348-365.
1. Isaacson PG, Wright DH. Malignant lymphoma of mucosa associated lymphoid tissue. A distinctive type of B-cell lymphoma. Cancer 1983; 52: 1410-1416. [ Links ]
2. Wotherspoon AC, Ortiz-Hidalgo C, Falzon M, et al. Helicobacter pylori–associated gastritis and primary B-cell gastric lymphoma. Lancet 1991; 338: 1175-1176. [ Links ]
3. Harris NL, Swerdlow SH, Jaffe ES, Olt G, Nathwani BN, de Jong D, Yoshino T, Spagnolo D. Follicular lymphoma in: Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pireli S, Stein H, Thiele J, Vardiman J. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. 4th Edition. International Agency for Research on Cancer: Lyon-France; 2008. p. 220-228. [ Links ]
4. Nakamura S, Müller- Hermelink HK, Delabie J, Ko YH, Jaffe ES. B cell lymphoma of the small intestine in: Bosman FT, Carneiro F, Hruban RH, Theise DT. WHO Classification of Tumours of the digestive system. 4th Edition. International Agency for Research on Cancer: Lyon-France; 2010. p. 69-73. [ Links ]
5. Nakamura S, Müller- Hermelink HK, Delabie J, Ko YH, van Krieken JH, Jaffe ES. Lymphoma of the stomach in: Bosman FT, Carneiro F, Hruban RH, Theise DT. WHO Classification of Tumours of the digestive system 4th Edition, 2010. International Agency for Research on Cancer: Lyon-France; 2010. p. 108-111. [ Links ]
6. Yamamoto S, Nakase H, Yamashita K, Matsuura M, Takada M, Kawanami C, Chiba T. Gastrointestinal follicular lymphoma: review of the literature. J Gastroenterol 2010; 45: 370-388. [ Links ]
7. Yoshino T, Miyake K, Ichimura K, Mannami T, Ohara N, Hamazaki S, et al. Increased incidence of follicular lymphoma in the duodenum. Am J Surg Pathol 2000; 24: 688-93. [ Links ]
8. Ghai S, Pattison J, Ghai S, O'Malley ME, Khalili K, Stephens M. Primary Gastrointestinal Lymphoma: Spectrum of Imaging Findings with Pathologic Correlation. RadioGraphics 2007; 27: 1371-1388. [ Links ]
9. López R, Andrade R. Tejido linfoide y linfomas gástricos. Rev Col Gastroenterol 2010; 4: 409-421. [ Links ]
10. Dawson IM, Cornes JS, Morson BC. Primary malignant tumors of the intestinal tract. Br J Surg 1961; 49: 80-89. [ Links ]
11. Misdraji J, Fernandez del Castillo C, Ferry JA. Follicle center lymphoma of the ampulla of Vater presenting with jaundice: report of a case. Am J Surg Pathol 1997; 21: 484-8. [ Links ]
12. Sentani K, Maeshima AM, Nomoto J, Maruyama D, Kim S-M, Watanabe T, Kobayashi Y, Tobinai K, Matsuno Y. Follicular Lymphoma of the Duodenum: A Clinicopathologic Analysis of 26 Cases. Jpn J Clin Oncol 2008; 38(8): 547-552. [ Links ]
13. Goodlad JR, MacPherson S, Jackson R, Batstone P, White J. Extranodal follicular lymphoma: a clinicopathological and genetic analysis of 15 cases arising at non-cutaneous extranodal sites. Histopathology 2004; 44: 268-76. [ Links ]
14. Takata K, Sato Y, Nakamura N, Kikuti YY, Ichimura K, Tanaka T, Morito T, Tamura M, Oka T, Kondo E, Okada H, Tari A, Yoshino T. Duodenal and nodal follicular lymphomas are distinct: the former lacks activation-induced cytidine deaminase and follicular dendritic cells despite ongoing somatic hypermutations. Modern Pathology 2009; 22: 940-949. [ Links ]
15. Toyoda H, Yamaguchi M, Nakamura S, Nakamura T, Kimura M, Suzuki H, et al. Regression of primary lymphoma of the ampulla of Vater after eradication of Helicobacter pylori. Gastrointest Endosc 2001; 54: 92-6. [ Links ]
16. Rohatiner AZ, Davies A, Montoto S, Lister TA. Follicular Lymphoma (Chapter 19) in: The lymphomas. Second edition. Elsevier Inc; 2006. p. 348-365. [ Links ]











 text in
text in