Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.29 no.1 Bogotá Jan./Mar. 2014
First-degree relatives of patients with gastric cancer have high frequencies of achlorhydria and premalignant gastric lesions
Martín Gómez Zuleta MD. (1), Diana Garzón Nossa MD. (2), William Otero R. MD. (3)
(1) Assistant Professor of Gastroenterology at the Universidad Nacional de Colombia, Gastroenterologist at the Hospital El Tunal in Bogotá, Colombia.
(2) Internist and Gastroenterologist at the Universidad Nacional de Colombia in Bogotá, Colombia.
(3) Professor of Gastroenterology at the Universidad Nacional de Colombia, Gastroenterologist at the Clínica Fundadores in Bogotá, Colombia.
Received: 17-01-13 Accepted: 19-12-13
Abstract
Objective: The objective of this study was to establish the frequency of preconditions for gastric cancer including atrophy, metaplasia, dysplasia and achlorhydria (pH > 5) in first degree relatives of patients with gastric cancer (FDR-GC). Design: This is a prospective case-control study with incidental cases. Patients: One hundred twelve first degree relatives of gastric cancer patients (case group) were paired by age and gender to 117 patients with functional dyspepsia but without GC family history (control group). Study location: This study was conducted in the gastroenterology service of a level three hospital in Bogotá, from March 1st, 2011 to March 31st, 2012. Procedures conducted: High digestive endoscopy, gastric pH measurements and gastric biopsies were performed. Measurements: We evaluated and compared endoscopic and pathological measurements as well as measurement of gastric pH. Results: The case group's 60 % frequency of pangastritis was higher than the control group's 28.8% (OR 3.32, CI 1.92 to 5.74, p < 0.05). There were findings suggestive of atrophy in 19.6% of the case group patients and in 7.7 % of the control group (OR 2.65, CI 1.16 to 6. 04, p < 0.05), findings suggestive of intestinal metaplasia in 12.5% of the case group and 0% of the control group, alkaline pH in 35.7 % of the case group and 7 % of the control group (OR 5.94, CI 2.72 to 12.98, p < 0.05). There were 4 cases of low grade dysplasia, two cases of high grade dysplasia (P <0.005), and two of early GC (NS). Conclusions: FDR-GCs had more achlorhydria, premalignant conditions, dysplasia and GC than control patients. The measurement of gastric pH is sensitive for detecting gastric atrophy. FDR-GC patients should be routinely screened with endoscopy and gastric pH measurement.
Key words
Gastric cancer, relatives, atrophy, metaplasia.
INTRODUCTION
Gastric cancer (GC) is a pathology that has a great impact on global morbidity and mortality, especially in countries such as Japan and Colombia. It is one of the most frequently occurring malignant tumors and is the first cause of death by cancer (1-3). In our country it occupies second place within the neoplastic pathologies and causes 15% of the deaths by cancer (2). Although incidence, diagnosis and therapeutic options have changed greatly in recent decades, GC's prognosis continues to be poor: GC patients have a survival rate below 10% because more than 90% of them are diagnosed when the cancer is in advanced stages (1). In 2012, in the United States alone, there were approximately 21,320 new cases diagnosed. Of these, 10,540 (49%) are expected to die (4). Histologically, there are two types of GC: intestinal and diffuse. The first is more common, and more risk factors have been identified for it. Helicobacter pylori (H. pylori) infections are the etiological agent in at least 90% of cases (5). Diffuse type is less frequent, is observed in young people, and has a worse prognosis (6). The physiopathology of intestinal type cancer has yet been not sufficiently well explained, but it is considered that it does not occur spontaneously but rather occurs as a result of chronic inflammatory changes triggered by H. pylori over 15 to 20 years (7). This carcinogenetic pathway has been hypothesized for intestinal cancer. In 1975 Dr. Pelayo Correa proposed the existence of a sequential progression from chronic gastritis through atrophic gastritis, intestinal metaplasia, dysplasia, early adenocarcinoma and finally to advanced cancer (8). Nevertheless, GC is multifactorial, and it is considered that there are also genetic factors required for its development. These factors interact with environmental factors such as smoking, high salt intake and most importantly - H. pylori infection to lead to the appearance of this tumor. H. pylori infection is a necessary, but not a sufficient factor (5, 9). In 1994 the International Agency for Research on Cancer (IARC) named H. pylori a Type I carcinogenic agent (10). At present it is considered to have a variable effect on carcinogenesis which depends on virulence factors such as cagA and vacA genes (11-14). The genetic component is illustrated by the fact that GC is more frequent in individuals with histories of gastric cancer in first degree relatives (9, 11, 15, 16). According to the literature, between 5% and 10% of gastric cancer patients have family histories of the tumor (17-19). while other authors have established that the risk for those with family histories of GC is two to three times more than that of the general population (19). Nevertheless, family history is much more important for diffuse cancer than for intestinal type GC, especially when the first-degree relative with diffuse cancer was under 40 years of age. In addition, some families have hereditary diffuse GC syndrome (20). Multiple studies have evaluated the relationship of family histories of GC and H. Pylori infections, and one recent study showed an odds ratio of 1.94 (21). Two recently published Colombian studies have shown that one of the main risk factors for patients with family histories of GC is when first-degree relatives have this illness (22, 23). On the other hand, one of the main components in Dr. Correa's carcinogenesis cascade is gastric atrophy which is associated with parietal cells loss due to secondary reduction of acid production. Hypochlorhydria and compensatory increases of serum gastrin act to induce proliferation of gastric epithelial cells and also favor colonization of bacteria that are capable of transforming nitrates from dietary to mutagenic agents such as N-nitrous compounds (5, 15). Consequently, detection through panendoscopy (PES) becomes important even though it has poor sensitivity and specificity and biopsies are required to determine the presence of atrophy. Requiring biopsies from all of these patients creates additional expenses and requires the involvement of the pathologist and therefore cannot be very cost effective (15). As previously mentioned, one of the consequences of atrophy is the reduction or elimination of acid (achlorhydria), however detection of atrophy by examining gastric acid is very bothersome and expensive and is not widely used. In a recently published study litmus paper was used to measure the pH of the gastric acid (16). This method effectively identified achlorhydria as an atrophy indicator with excellent correlation to histology. This is a very economic and easy to use method which has been proposed for routine use in our environment to establish the gastric pH of the patients who undergo endoscopy. This method was previously described by Levine and colleagues in 1994 when this author utilized it to measure the pH of vacuumed gastric samples from UCI patients. That study compared the method with pH measurement with an electrode placed on a probe to measure the pH. The excellent correlation between the two methods was excellent (R2 = 0.93, p < 0.001) (24).
Keeping in mind the family component of GC and the possibility of determining the presence of atrophy by measuring gastric pH as an indicator of achlorhydria or hypochlorhydria (pH >5), we decided to undertake this study to estimate the frequency of atrophy and metaplasia which are precursors of GC, premalignant lesions (dysplasia), and hypochlorhydria (pH>5) in first degree relatives of people with gastric cancer (FDRGC). If it can be proven that FDRGC have higher risks of GC (as shown in the literature), and if GC develops over decades from a cascade of inflammatory events, it should be possible to evaluate FDRGC with endoscopy, biopsies and pH measurement and to diagnose lesions opportunely prior to the onset of GC during the early stages of GC when has greater than 90% probability of achieving a cure (25-28). This could prevent us from continuing to face advanced GC with its awful prognosis with a 5 year survival rate of less than 10% (4).
MATERIALS AND METHODS
This is a prospective case and control study undertaken in the gastroenterology service of Hospital El Tunal and the gastroenterology unit at the Universidad Nacional de Colombia. This study was conducted between March 1, 2011 and March 31, 2012. All non-cardiac GC cases confirmed by pathology (37 patients) were identified, and their first-degree relatives were invited to participate. There were 185 potential participants, but only 112 people accepted the invitation to be included in the study. These people became Group I and were labeled first degree relatives of people with gastric cancer (FDRGC). In the structure of this study, the FDRGC group corresponds to cases. They were paired by age and gender with 117 patients who had been diagnosed with functional dyspepsia during the same period of study in our service but who did not have family histories of GC. These patients constitute the control group which was labelled Group II.
All patients signed informed consent forms in order to participate in this study which work was approved by the research committee of the institution in which it was carried out. A gastroenterologist filled out a data collection form for every participant. The document recorded the patient's identification, history, and time of onset of symptoms. Subsequently all patients from both groups underwent panendoscopy without sedation but with topical anesthesia following a minimum 8 hour fast. An Olympus Exera II endoscope was used. All procedures were carried out by the same gastroenterologist (MG) to avoid interobserver bias. As part of the bias control strategy, the endoscopist did not know the data recorded on patient questionnaires. Once the endoscope was in the patient's stomach, the gastric mucous lake was vacuumed into a container. A strip of litmus paper was then dipped into the material collected to measure its pH. Hypochlorhydria was diagnosed when the litmus paper strip showed a pH above 5 according to the method described in the literature (16). The Sydney protocol was followed for staging chronic gastritis. It recommends taking two biopsies from the corpus 8 cm from the cardia, one from the anterior wall and one other from the posterior wall; and two more biopsies from the antrum 2 or 3 cm from the pylorus, one from the greater curve and one from the lesser curve; and one biopsy from the angular incisure (29). An additional biopsy was taken for the rapid urease test for H. pylori infection.
Inclusion Criteria
Group I (cases): Patients older than 18 years, histories of with gastric cancer in first-degree relatives.
Group II (controls): Patients older than 18 years, with functional dyspepsia, without histories of with gastric cancer in first-degree relatives.
Exclusion criteria for both groups
Presence of a gastric bile lake that could give false negative results for hypochlorhydria because of alkalinity of the gastric acid, use of PPIs in the two weeks prior to examination, previous treatments for H. pylori eradication.
STATISTICAL ANALYSIS
Information was entered into Excel 2007 and analyzed with Stata 10.0. Descriptive statistics were used to describe variables in the study. Frequency distributions and percentages were used to analyze nominal and ordinal variables. Numerical variables were expressed with central tendencies and dispersions. Statistical tests were evaluated to a degree of significance of 5% (p<0.05). Continuous variables were informed as frequencies and averages. The differences between averages were determined with the Student's t test.
RESULTS
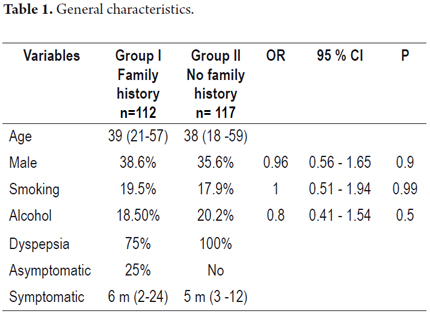
In our work two groups of patients were compared: a group without family histories of gastric cancer and one with family histories of gastric cancer. The number of patients in each group was similar (117 and 112 respectively) as were age ranges, gender distributions, and distributions of smoking habits and alcohol intake (Table 1).
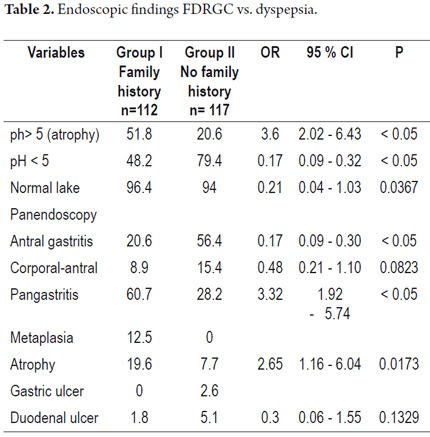
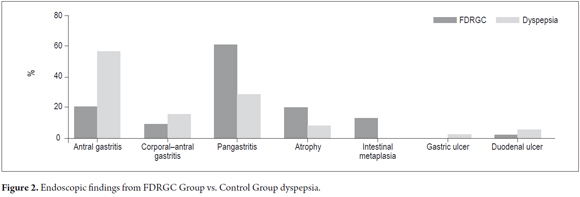
A comparison of endoscopic findings from the two groups (Table 2) showed that the FDRGC group had a greater frequency of pangastritis (60%) than the control group (28.8%) OR 3.32 (CI 1.92 -5.74 p< 0.05), more frequent findings suggestive of atrophy such as increased vascular indications and thin mucosa (19.6) than the control group (7.7%) OR 2.65 (CI 1.16 6. 04 p < 0.05), more indications of intestinal metaplasia such as dental plaque (12.5%) than the control group (0%), and had alkaline pH (35.7 %) more often than the control group (7%), OR 5.94 (CI 2.72 12.98 p < 0.05) (Figures 1 and 2).
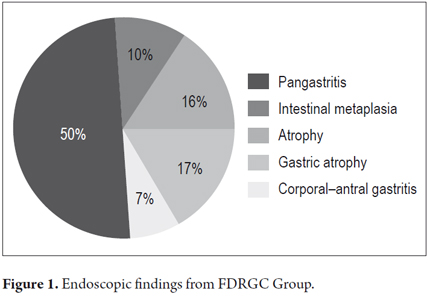
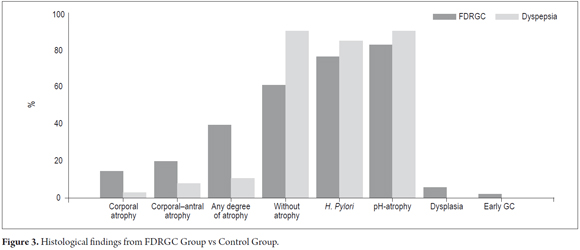
Histologic findings (Table 3) showed a higher frequency of atrophy among FDRGC patients (39.3 %) than in control patients (10.3%) OR 5.02 (CI 2.47 10, 17 p < 0.05). Six cases of dysplasia cases were found in the FDRGC group: four were low grade and two were high grade but without any apparent focal lesion. In addition, in this group two cases of early GC were found. They were treated endoscopically. No cases of advanced GC were found (Figure 3).
DISCUSSION
This study found that the FDRGC group had greater risks of pangastritis than did the control group (OR 3.32, CI 95% 1.925.74). Antral gastritis, which is not related to GC, was predominant among control group patients (5). Pangastritis is the type of gastritis present in patients with GC (5,30). Atrophy occurred almost four times more often in group I than in the control group (OR 5.02 (CI95%2.47-10.17). Atrophic gastritis with or without metaplasia is considered to be the main precursor lesion to GC and the field upon which cancer forms (31). The correlation between endoscopic suspicion of atrophy and histological confirmation was 19.6% which proves once again that diagnosis requires histological study as expert researchers have already said (17). Histology is also the only means for staging GC risk in accordance with the severity of atrophy, so it is a necessity for planning strategy for monitoring these patients (32). After evaluating the pH with litmus paper, we found that the probability of reversing hypochlorhydria was greater in Group I than in the Group II (OR 3.6, 95 % CI 2.02 6.43) and that this measurement had a b linear correlation with histologic findings of atrophy with an r2 of 0.82. In contrast, most control group patients had pH below 5 indicating that acidity correlates with low frequencies of atrophy (10%). Over 10 years ago a study by El-Omar and colleagues with a design to that of this study evaluated gastric pH with a more complex and more expensive method (33). People with family histories of GC history were compared to patients with dyspepsia without family histories of GC. They found that the frequency of hypochlorhydria was 27% in the group with family histories of GC, but only 3% in the group without such histories. Similarly they found a rate of atrophy of 34% in the FDRGC group, but only 5% in the control group. Not surprisingly given the high frequency of H. pylori infections in our population, both groups had high incidences of H. pylori infections: 82.1% for FDRGC and 89.7% for the control group (22). The fact that the frequency of this microorganism was similar in the two groups reinforces the idea that this infection alone is not sufficient to cause GC. Its appearance requires additional factors such as the environment, the diet, and genetics (family history). Among the conditions that are precursors for GC, intestinal metaplasia (IM) was found in 27.7% of the biopsies from the FDRGC group but only in 7.5% of the control group (p<0.05). The high frequency of IM in Group I further corroborates that Group I patients have higher GC risks than do Group II patients since IM is more advanced than atrophy in the chain of carcinogenic events (8, 9, 15). Another advantage of histology is that its diagnostic has less inter-observer variability (34, 35). Poor correlation (12.5%) between endoscopic suspicion of IM and histological diagnosis of IM confirms that endoscopy alone is not reliable for a definite diagnosis of these premalignant conditions. In addition to the greater frequency of atrophy and metaplasia in Group I, dysplasia was found in six of these patients (four low grade cases and two high grade cases). Since the two high grade cases were similar to early GC in situ, these lesions were endoscopically removed in accordance with the current recommendation for such alterations (28). Patients with low degree dysplasia are currently being monitored endoscopically every six months. None of these types of lesions were found in the control group. Taking these findings in the patients of the FDRGC group together and comparing them with the findings from the group of patients with dyspepsia without GC family histories of GC shows that an accelerated process of carcinogenesis exists among FDRGC group patients. In addition to H. pylori infections, risk factors may include genetic susceptibility and environments favorable to GC which are similar to the environments of relatives and which include factors such as diet, habits of life and overcrowding. The results of this study corroborate findings of a previous study which showed that patients with GC family histories had higher risks for developing this tumor than did patients with duodenal ulcers (22).
Atrophy, intestinal metaplasia, dysplasia and early GC findings from Group I patients, support Doctor Correa's gastric carcinogenesis hypothesis (8). In addition, they imply that conducting endoscopies in FDRGC patients should have a high probability of timely detection of lesions and premalignant conditions. Moreover, it implies that these patients should continue in an endoscopic surveillance program in which early detection of GC would result in timely treatment through endoscopic resection. This would allow it to heal itself as has been shown by diverse researchers (23, 28). Finally, it would end the tragedy in which cancer is usually detected in our country in advanced stages in which surgery or palliative treatments - with their high human and financial costs - are the only alternatives that can be offered to patients.
With the results of this study, we want to highlight that measuring gastric acid with litmus paper is easy to execute and correlates extremely well with histological findings. It requires only a minute to determine whether the vacuumed sample's pH is alkaline indicating atrophy, or acid indicating absence of atrophy. This corroborates recently published findings (9). As far as we know, this is the first study that measures pH through endoscopy and which correlates these results with the results of histology for people with family histories of gastric cancer. With this methodology we have shown that atrophy and hypochlorhydria occur more frequently among this group of people than they occur among patients with functional dyspepsia but no family histories of GC. Atrophy and hypochlorhydria are two conditions that have been clearly identified by diverse authors as risk factors for GC which explains so many more cancer cases are found in this population. (7, 8, 36, 37, 38). The higher than 80% correlation above between histology and pH measurements that this study found allows us to recommend this innovative method as a histologic alternative for establishing or dismissing gastric atrophy. It has the additional advantages of being simple and very economical when compared to other methods used to measure gastric acidity with probes, complex devices and expensive medicine such as pentagastrin (39).
One weakness of our work was that we did not use the OLGA staging system to rank results from pathology reports and to correlate them with our endoscopic findings. Some studies (40) have shown that patients classified as OLGA III/IV have a higher risk of atrophy and therefore of cancer (41). Nevertheless, this classification requires five biopsies and a pathologist to evaluate them. For these reasons our group decided to classify atrophy into only two categories: absent or present. In addition, evaluation of atrophy has great inter-observer variability and does not take into account. Even though atrophy has an inherent GC risk, recent reports have shown that metaplasia is a premalignant condition which is much more relevant in the process of gastric carcinogenesis (42, 43). For this reason, it has recently been proposed to stage intestinal metaplasia using the OLGIM scale (44).
CONCLUSIONS
1. Patients with dysplasia who have first-degree relatives of patients with gastric cancer more frequently develop hypochlorhydria, dysplasia, and gastric cancer and have more premalignant conditions than do control patients with dysplasia.
2. The litmus paper method of measuring acidity of a vacuumed gastric sample is innovative, simple, very economic and highly sensitive for detecting gastric atrophy (hypochlorhydria).
3. Patients with family histories of GC must be screened with endoscopy including measurement of the gastric pH because of the high risks they run of developing GC precursor conditions such as atrophy, intestinal metaplasia, dysplasia, and early GC which are susceptible to endoscopic treatment.
4. It would be interesting to continue this research about pH measurement with litmus paper by correlating it with the recently described OLGA system that stages GC risks in accordance with atrophy.
REFERENCES
1. World Health Organization. The Global Burden of Disease: 2004 Update. Geneva: World Health Organization; 2008. [ Links ]
2. Ferlay J, Shin HR, Bray F, Forman D, Mathers C and Parkin DM. GLOBOCAN 2008 v1.2, Cancer Incidence and Mortality Worldwide: IARC CancerBase No.10 [Internet]. Lyon, France: International Agency for Research on Cancer; 2010. Disponible en: http://globocan.iarc.fr. Accesado en junio 15 de 2012. [ Links ]
3. Martínez J, Garzón M, Lizarazo I, Marulanda J, Molano. Características de los pacientes con cáncer gástrico del departamento de Cundinamarca, remitidos al Hospital Universitario de la Samaritana entre los años 2004 y 2009. Rev Col Gastroenterol. 2010;5:344-348. [ Links ]
4. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69-90. [ Links ]
5. Otero W, Gómez M, Castro D. Carcinogénesis gástrica. Rev Col Gastroenterol. 2009;24: 314-329. [ Links ]
6. Sugano K. Gastric cancer: pathogenesis, screening and treatment. Gastrointest Endosc Clin N Am. 2008;18(3):513-22. [ Links ]
7. Mueller A, Falkow S, Amieva MR. Helicobacter pylori and gastric cancer: what can be learned by studying the response of gastric epithelial cells to the infection? Cancer Epidemiol Biomarkers Prev. 2005;14:1859-64. [ Links ]
8. Correa P. Human Gastric carcinogenesis: a multistep and multifactorial process. Cancer Res. 1992;52:6735-40. [ Links ]
9. McNamara D, El-Omar E. Helicobacter pylori infection and the pathogenesis of gastric cancer: A paradigm for host-bacterial interactions. Dig Liv Dis. 2008;40:504-9. [ Links ]
10. IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Lyon, 7–14 June 1994. IARC Monogr Eval Carcinog Risks Hum. 1994;61:1–241. [ Links ]
11. Wang C, Hunt RH. The association between Helicobacter pylori infection and early gastric cancer: a meta-analysis. Am J Gastroenterol 2007; 102: 1789-98. [ Links ]
12. Figueiredo C, Machado JC, Pharoah P, et al. Helicobacter pylori and interleukin 1 genotyping: an opportunity to identify high-risk individuals for gastric carcinoma. J Natl Cancer Inst. 2002;94:1680-7. [ Links ]
13. Amieva MR, El-Omar EM. Hostbacterial interactions in Helicobacter pylori infection. Gastroenterology. 2008;134:306-23. [ Links ]
14. Machado JC, Figueiredo C, CanedoP, et al. A proinflammatory genetic profile increases the risk for chronic atrophic gastritis and gastric carcinoma. Gastroenterology. 2003;125:364-71. [ Links ]
15. Kelley J, Dugan J. Gastric cancer epidemiology and risk factor. J ClinEpidemiol. 2003;56:1-9. [ Links ]
16. Gomez M. Accuracy of Litmus Paper Determined Intragastric pH in the Evaluation of Gastric Atrophy. Gastroenterology. 2011;140(5):S-564. [ Links ]
17. Li; Genta, R, Mae, F; Gutiérrez, O; Kim, J; Graham D. Helicobacter pylori Strain and the Pattern of Gastritis among Fisrt-degree Relatives of Patients with Gastric Carcinoma. Helicobacter. 2003;7(6):349-65. [ Links ]
18. Barber M, Fitzgerald RC, Caldas C. Familial gastric cánceraetiology and pathogenesis. Best Pract Res ClinGastroenterol 2006;20:721-34. [ Links ]
19. Brenner H, Arndt V, Sturmer T, et al. Individual and joint contribution of family history and Helicobacter pylori infection to the risk of gastric carcinoma. Cancer 2000;88:274-279. [ Links ]
20. Blair V, Martin I, Shaw D, et al. Hereditary diffuse gastric cancer: diagnosis and management. Clin Gastroenterol Hepatol. 2006;4:262-75. [ Links ]
21. Correa P, Houghton J. Carcinogenesis of Helicobacter pylori. Gastroenterology. 2007;133:659-72. [ Links ]
22. Gómez Zuleta M, Otero William, Ruiz X. Factores de riesgo para cáncer gástrico en pacientes colombianos, Rev Colomb Gastroenterol. 2009;24:134-43. [ Links ]
23. Gómez M, Concha A, Otero W. Diferenciación de cáncer gástrico temprano vs linitis plástica; ¿los síntomas hacen la diferencia? Act Med Colomb. 2012;37:62-65. [ Links ]
24. Levine RL, Fromm RE Jr, Mojtahedzadeh M, Baghaie AA, Opekun AR Jr. Equivalence of litmus paper and intragastric pH probes for intragastric pH monitoring in the intensive care unit. Crit Care Med. 1994 Jun;22(6):945-8. [ Links ]
25. Rembacken BJ, Gotoda T, Fujii T, et al. Endoscopic mucosal resection. Endoscopy. 2001;33:709-718. [ Links ]
26. Soetikno R, Gotoda T, Nakanishi Y, et al. Endoscopic mucosal resection. Gastrointest Endosc. 2003:57:567-579. [ Links ]
27. Soetikno R, Kaltenbach T, Yeh R, et al. Endoscopic mucosal resection for early cancers of the upper gastrointestinal tract. J Clin Oncol. 2005:4490-98. [ Links ]
28. Gotoda T: Endoscopic resection of early gastric cancer. Gastric Cancer. 2007;10:1-11. [ Links ]
29. Dixon MF, Genta RM, Jardley JH, Correa P. Classification and Grading of Gastritis-The Updated Sydney System". Am J Surg Pathol. 1996;20:1161-81. [ Links ]
30. Sepulveda AR,Peterson LE, Shelton J, et al. Histological patterns of gastritis in H.pylori-infected individuals with a family history of gastric cancer. Am J Gastroenterol. 2002;97:1365-70. [ Links ]
31. Rugge M, Pennelli G, Pilozzi E, Fassan M, et al. Gastritis: The histology report. Dig Liv Dis. 2011;43S:S373-S384. [ Links ]
32. Rugge M, Correa P, Di Mario F, El-Omar E, Fiocca R, Geboes K, et al. OLGA staging for gastritis: A tutorial. Dig Liv Dis. 2008;40:650-8. [ Links ]
33. El-Omar E, Obrien K, Murray LS, et al. Increased frequency of precancerous changes in relatives of gastric cancer patients: critical role of H. pylori. Gastroenterology. 2000;118:22-2. [ Links ]
34. El-Zimaity HM, Graham DY, Al-Assi MT, et al. Interobserver variation in the histopathological assessment of Helicobacter pylori gastritis. HumPathol. 1996;27:35–41. [ Links ]
35. Chen XY, van der Hulst RW, BrunoMJ, et al. Interobserver variation in the histopathological scoring of Helicobacterpylori related gastritis. J Clin Pathol. 1999;52:612-5. [ Links ]
36. Correa P, Schneider BG. Etiology of gastric cancer: what is new? Cancer Epidemiol Biomarkers Prev. 2005;14:1865-8. [ Links ]
37. Guillen D, McColl KEL. Gastroduodenal disease, Helicobacter pylori, and genetic polymorphisms. Clinical Gastroenterol Hepatol. 2005;3:1180-86. [ Links ]
38. El Omar EM, Penman I, Ardill JE, et al. Helicobacter pylori infection and abnormaties of acid secretion in patients with duodenal ulcer disease. Gastroenterology. 1995;109:681-91. [ Links ]
39. Calam J, Baron J. Pathophysiology of duodenal and gastric ulcer and gastric cancer. ABC of the upper gastrointestinal tract. BMJ. 2001;2323:980-82. [ Links ]
40. Rugge M, Fassan M, Pizzi M, PennelliG, Nitti D, Farinati F. Operative Link for Gastritis Assessment gastritis staging incorporates intestinal metaplasia subtyping. Hum Pathol. 2011;42:1539-44. [ Links ]
41. Rugge M, De Boni M, Pennelli G, De Bona M, Giacomellis L, Fassan M, et al. Gastritis OLGA-staging and gastric cáncer risk: a twelve-year clinico-pathological follow-up study. Aliment Phramacol Ther. 2010;31:1104-11. [ Links ]
42. Yeh JM, Hur C, Kuntz KM, Ezzati M, Goldie SJ. Cost effectiveness of treatment and endoscopic surveillance of precancerous lesions to prevent gastric cancer. Cancer. 2010;116:2941-53. [ Links ]
43. deVries AC, Kuipers EJ. Epidemiology of premalignant gastric lesions: implications for the development of screening and surveillance strategies. Helicobacter. 2007;12(Suppl 2):22-31. [ Links ]
44. Capelle LG, de Vries AC, Haringsma J, Borg FT, de Vries RA, Bruno MJ, et al. The staging of gastritis with the OLGA system by using intestinal metaplasia as an accurate alternative for atrophic gastritis. Gastrointest Endosc. 2010;71:1150-8. [ Links ]











 text in
text in