Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.30 no.1 Bogotá Jan./Mar. 2015
Report of 21 Cases of Neonatal Cholestasis in a Children's Hospital in Cartagena, Colombia
Jorge José Mendoza H. MD. (1), Rodrigo De Vivero C. MD. (2), Nelson Muñoz A. MD. (3), Amileth Suarez C. PhD. (4), Carlos Moneriz P. PhD. (5)
(1) Pediatrician at the School of Medicine of the Universidad de Cartagena and the Hospital Infantil Napoleón Franco Pareja in Cartagena, Colombia.
(2) Pediatric Gastroenterologist at the School of Medicine of the Universidad de Cartagena and the Hospital Infantil Napoleón Franco Pareja in Cartagena, Colombia.
(3) Pediatrician at the Hospital Infantil Napoleón Franco Pareja in Cartagena and Professor of the Faculty of Medicine at the Universidad de Cartagena in Cartagena, Colombia.
(4) Prometheus Group and Applied Biomedicine for the Clinical Sciences in the Faculty of Medicine at the Universidad de Cartagena in Cartagena, Colombia.
(5) Biochemistry and Disease Group in the Faculty of Medicine at the Universidad de Cartagena in Cartagena, Colombia.
Correspondence: Carlos Moneriz Pretell PhD, Campus de Zaragocilla. Laboratorio de Bioquímica (2°piso), Facultad de Medicina, Universidad de Cartagena (Colombia). Telephone (055) 6698176 ext. 130; Fax: (055) 6698177. Email: cmonerizpretel@yahoo.com
Financing: Financing was received from the office of the Vice-Rector for Research of the University of Cartagena as part of the plan for strengthening and sustainability of the Biochemistry and Disease Group and recognized by Colciencias.
Received: 05-11-14 Accepted: 02-02-15
Abstract
Cholestasis is an alteration in the flow of bile resulting from decreases or cessation of biliary excretion. To date, there have been only a few studies on this topic in Colombia. This article presents twenty-one cases of neonatal cholestasis from a Children's Hospital in Cartagena, Colombia that occurred between 2010 and 2013. The aim of this study is to characterize the etiology and clinical characteristics of the disease. Patients between birth and 3 months old with direct bilirubin levels over 2 mg/dl were selected. By gender, 52.4% of the patients were male, and 47.6% were female. 76.2% of the patients were full term, and 57.1% had no perinatal antecedents. Clinical symptoms presented in the first 30 days after birth in 71% of the patients, and 4 patients were referred for liver transplantation. The most common etiology was infectious (13 patients), and 4 patients had atresia. The most common cause of neonatal cholestasis in this study was infection, but obstructive disorders such as biliary atresia still account for an important proportion of the patients. They require priority study and handling because early intervention results in better prognoses.
Keywords
Neonatal cholestasis, bilirubin, biliary atresia, neonatal jaundice, neonatal sepsis.
INTRODUCTION
Cholestasis is a liver dysfunction in which the flow of bile is impaired. It is analytically defined by either a combination of elevated serum levels of bile acids and direct bilirubin over 2 mg/dl (1), or by direct bilirubin that is more than 20% of total bilirubin. It is caused by a blockage of flow through the intrahepatic or extrahepatic ducts of the biliary tree. This causes an accumulation of substances normally excreted in the bile and usually presents with prolonged jaundice, dark urine, pale stools, hepatomegaly and itching during the first four weeks of life (2, 3). The term neonatal cholestasis covers the first three months of a newborn's life (1, 4).
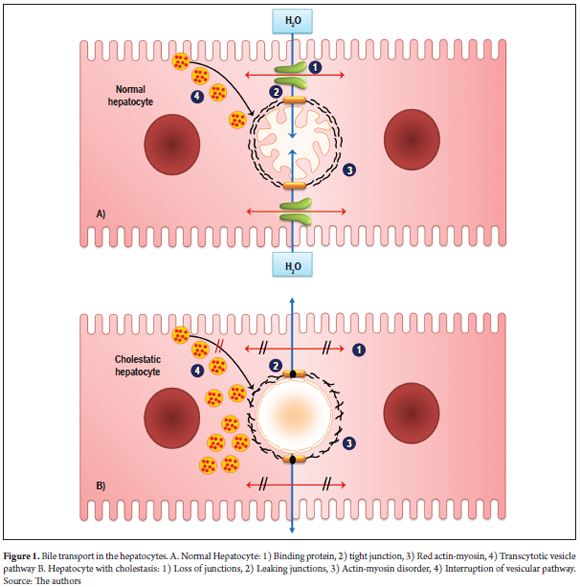
Bile acids are toxic to membranes and are transported through bile ducts which protect tissues from these acids through the action of various proteins and vesicular transport within the hepatocytes (Figure 1) (5). In a cholestatic hepatocyte, the functionality of the carrier proteins is altered in ways that affect vesicular transport by disruption of microtubules in the cell. This causes accumulation of bile components (6, 7).
This clinical syndrome occurs in 1 out of every 2,500 to 5,000 live births (8). However, this rate varies according to various factors including race and gender. For example, the incidence in Europe is 1 in 18,000 (1, 9, 10).
Since global incidence of neonatal cholestasis is low, the pathology is underestimated. On the other hand, its etiology is multifactorial and can involve genetic, environmental and hormonal factors (11, 12). It may be related to neonatal hepatitis, biliary obstruction, genetic cholestatic syndromes and metabolic diseases (13). For these reasons, it is important that all infants with symptoms such as prolonged jaundice are studied to achieve an accurate diagnosis which allows successful treatment that can ensure their survival (14-16). Some authors claim that it is not always possible to identify the causes of cholestasis because of the urgency of treating the patient (17). Biliary atresia and neonatal hepatitis account for 55% to 65% of all cases of neonatal cholestasis (1, 4). Treatment may be medical or surgical, including occasional liver transplantation, depending on the case. Transplantation is the standard therapy for acute and chronic liver failure, and over 50% of transplants are due to cholestatic diseases (18).
Since newborn babies have immature livers (19), they are at a disadvantage compared with older children. They have lower concentrations of bile acids and express less hepatobiliary transporters (20). Consequently, this condition predisposes the newborn to bleeding, encephalopathy, lack of vitamins A, D, E, and K, and jaundice (4, 21).
Neonatal cholestasis is a problem throughout the world. Its mortality rate is estimated to be between 15% and 50%. It is associated with perinatal factors whose presence can help make a proper diagnosis which can lead to optimal management. This in turn can contribute to decreases in serious consequences such as sepsis, liver failure and death (8, 22).
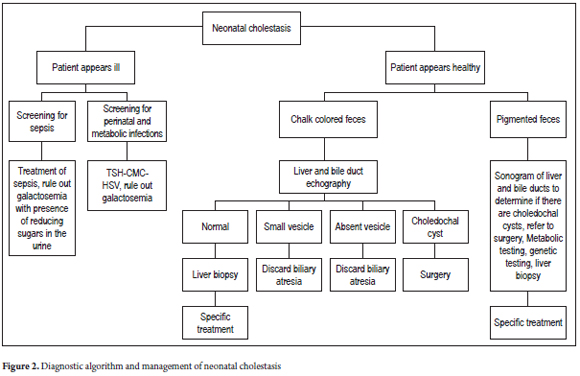
In cases of suspected cholestasis, the most important element of the evaluation is the color of stools. If the stools are clay-colored it is a sign of acholia and biliary atresia must be diagnosed or excluded (Figure 2). If obstruction of the extrahepatic biliary tree is excluded, a diagnosis of biliary atresia is likely. Cholangiography and should be performed and a liver biopsy should be taken. If the bile duct is not permeable, it is necessary to perform a Kasai intervention (portoenterostomy). Conversely, if the bile duct is permeable, another diagnosis such as neonatal sclerosing cholangitis should be considered (12).
The purpose of this article is to identify the major etiological and clinical characteristics of patients with neonatal cholestasis cared for in the Hospital Infantil Napoleón Franco in Cartagena. It is important to note that few studies of this disease have been done in Colombia. Therefore, the aim of this research is to develop new knowledge, determine supporting elements for the most accurate diagnoses and to improve neonatal care.
PRESENTATION OF CASES
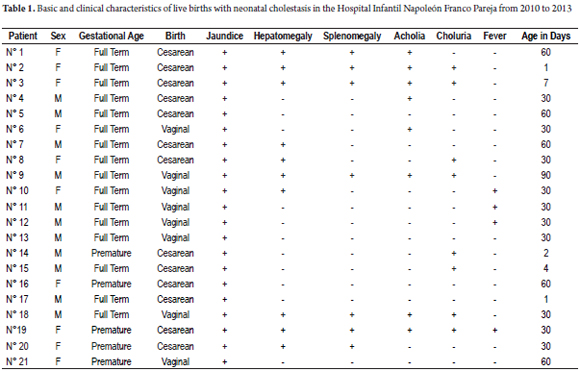
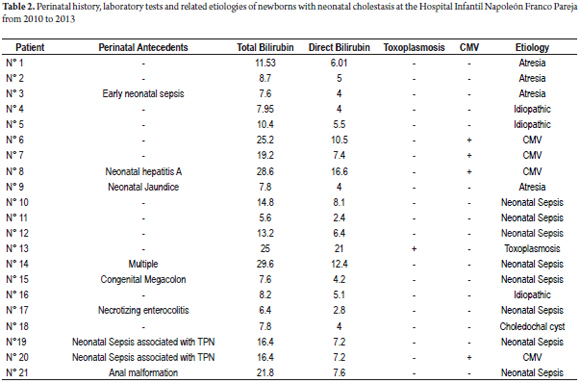
Twenty-one patients with neonatal cholestasis, direct bilirubin higher than 2 mg/dl, were admitted to the Hospital Infantil Napoleón Franco in Cartagena, Colombia between January 2010 and April 2013. Patients ranged in age between zero and three months old. Data were collected from interviews with families responsible for the patients, medical personnel responsible for the patients' care, and reviews of medical records. Parameters evaluated and shown in Table 1 are age, sex and clinical manifestations. Parameters evaluated and shown in Table 2 are perinatal history, lab results such as bilirubin and TORCH Infections (Toxoplasmosis, Other [syphilis, varicella-zoster, parvovirus B19], Rubella, Cytomegalovirus (CMV), and Herpes), and related etiologies. This study was classified in the category of research without risk in accordance with Article 11 of the Resolution No. 1993 008430 of the Ministry of Health.
The study group was comprised of almost equal numbers of male and female patients. Sixteen of the patients (76%) were full-term babies while five patients (24%) were premature (Table 1). The most common clinical findings were jaundice and hepatomegaly, but some presented neither acholia nor choluria. Average age at clinical onset was 33.6 ± 24 days.
It was observed that a significant proportion of these patients (57.1%) had no perinatal history of any kind. Four patients (14.3%) had histories of neonatal sepsis (Table 2). Patient No. 3 had early neonatal sepsis, patient No. 19 and patient No. 20 had neonatal sepsis associated with parenteral nutrition, patient No. 21 had neonatal sepsis associated with intestinal atresia and anorectal malformations. The remaining five patients had various histories: Patient No. 9 has neonatal jaundice, patient No. 17 had necrotizing enterocolitis, patient No. 15 had congenital megacolon, patient No. 8 had neonatal hepatitis, and patient No. 14 had severe Rh alloimmunization, Down syndrome, duodenal atresia, thrombocytopenia, septic shock, and persistent ductus arteriosus.
Most frequently, direct bilirubin levels, were about 4 mg/dl although one patient, No. 13, had a level of 21 mg/dL (Table 2) .The average direct bilirubin of all the patients was 7.2 ± 4.6 mg/dL. The liver profiles of all patients were altered as measured by transaminases and prothrombin time (PT). In addition, most patients had decreased hemoglobin and hematocrit.
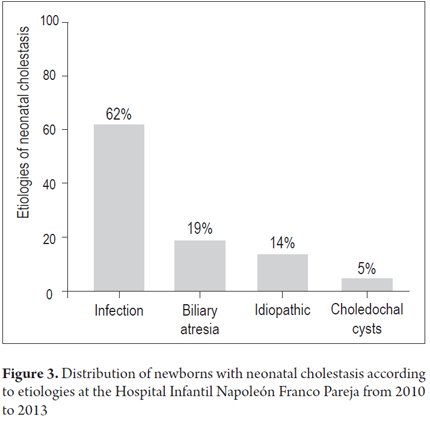
Etiologies included thirteen cases associated with infectious causes, four cases of biliary atresia, three with idiopathic causes, and one patient with a biliary duct cysts (Figure 3). Significantly, the direct bilirubin level of 21 mg/dl was associated with toxoplasmosis and not biliary atresia. It was also noted that the leading infectious cause was neonatal sepsis which affected eight patients (62%). Four patients (31%) had cytomegalovirus infections, and one patient (8%) had toxoplasmosis.
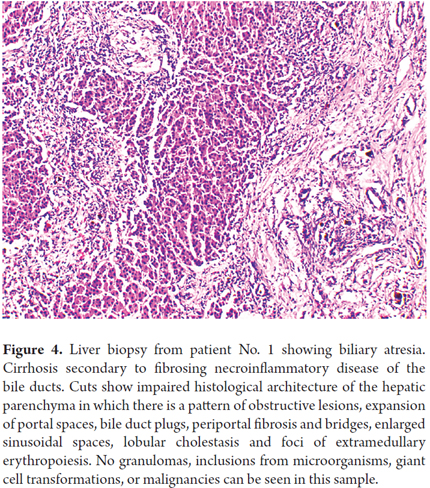
Infants who were referred for transplantation were characterized by being full term babies who had perinatal histories, acholia, dark urine, increased AST and ALT levels, direct bilirubin from 4 to 16.6 mg/dL, and prolonged prothrombin times. Finally, it is noteworthy that 75% of patients in the group with atresia (Figure 4) were referred for transplantation compared while only 8% of the group whose conditions were due to infectious causes were referred for transplantation.
DISCUSSION
Worldwide, the incidence of cholestasis is very low but may vary by race and gender (23). In this study 21 patients with neonatal cholestasis were diagnosed over a period of three years. These data are representative of the newborn population of the city of Cartagena since the children's hospital in this study is the largest in the area and it receives the laergest number of pediatric referrals.
This study found no significant differences in genders, but the large majority of patients were term rather than premature. These data are similar to those in a study by Alvarez et al. in the William Soler Pediatric Hospital in Cuba (9), although a predominance of male patients with neonatal cholestasis was found in a retrospective study by Jacquemin et al. (24). In addition, most cases have no perinatal histories although those with antecedent symptoms and diagnoses including referrals for transplantation.
The results of this study demonstrate b associations between some etiologies such as the neonatal sepsis and biliary atresia with this pathology. Our results show etiologies that are different than those in other studies in other parts of the world. For example, the main etiologies found in a study by Mendez et al. in Montevideo were hepatitis (16 patients) and atresia (13 patients) (25).
Infectious causes associated with cholestasis include cytomegalovirus, toxoplasmosis, rubella, syphilis, herpes, and bacterial sepsis (26-28). This information coincides with the result of this study. Congenital infections usually share clinical similarities including an enlarged liver and spleen, jaundice, pneumonitis and a tendency to prematurity or poor intrauterine growth (27). A clinical presentation of neonatal liver failure is possible with any of these agents, but in this study it was mostly evidenced with cytomegalovirus infections. It has been reported to be more common with herpes simplex infections.
Four patients (19% of total) with neonatal cholestasis associated with cytomegalovirus infections were found in this study. Cytomegalovirus (CMV) is the most common cause of congenital infections. They affect one to two percent of newborns most of whom are asymptomatic (27). Clinical findings include petechiae, hepatosplenomegaly and jaundice in 60% to 80% of patients. (27) Although the literature reports that it is unusual for CMV infections to cause neonatal liver failure, in this study a high proportion of patients were found with neonatal cholestasis associated with CMV.
Our finding of congenital toxoplasmosis in 5% of the patients in the study is a warning to others especially since our findings match those reported by other investigators (29, 30). A study in Brazil showed an incidence of toxoplasmosis in that country ranging from 2-20 per 10,000 births. That case study presented direct bilirubin of 35.6 mg/dl and concluded that toxoplasmosis is a disease with high morbidity which must be prevented by treatment while the patient is still alive (31).
Nevertheless, our study showed that in contrast to other reports in the literature, the most frequent etiologies encountered were those of infectious natures. Reports in the literature show that atresia of the bile ducts is the most frequent etiology (15, 18, 32), while in our population there are more cases of sepsis and CMV infections than cases of biliary atresia. This finding makes us think that the coincidence of this type of infectious etiology in cases of neonatal cholestasis is an important factor for further study in our population.
A study conducted in Medellín, Colombia coincides with our study in terms of the presence of clinical symptoms such as jaundice, dark urine, and acholia. Nevertheless, that study found atresia to be the cause in eighteen children (28%) which made it the most common cause there and which differs from the results obtained in our investigation (33).
Biliary atresia was the most common reason for pediatric liver transplantation in our study which is consistent with the literature. Nevertheless, may be other liver diseases that require transplantation (34). Up to 60% of patients with biliary atresia may require a transplant in the first 10 years of life, and even those who undergo surgery to restore bile flow may need a transplant in the second or third decade of life (18).
This study found one patient with a choledochal cyst. This type of cyst is related to a group of congenital malformations in the biliary system. The most common type is a fusiform, sometimes sausage-shaped, dilated extrahepatic bile duct. Choledochal cysts are increasingly being identified in the fetus by prenatal ultrasound and have even been found at 15 weeks of gestation (27). The diagnosis should be confirmed immediately after birth. Most of these children have conjugated hyperbilirubinemia, and surgery should be performed promptly.
Finally, the diagnostic evaluation of a newborn with cholestasis should be systematic, logical and cost-effective. A coordinated team of clinicians, surgeons, radiologists and biochemists can minimize unnecessary tests and achieve a correct diagnosis in the shortest possible period of time.
CONCLUSION
This case report indicates that the etiologic features of cholestasis reported in the literature are not accurate because there is more diversity in terms of classification. In some regions the most common cause may be biliary atresia, but elsewhere infectious causes may be more common as in this study. Diagnostic procedures for infants should include all necessary studies to determine the most prevalent cause and address treatment in the most successful way, taking into account gender, race, and geographic location.
Acknowledgements
The authors wish to thank the Faculty of Medicine of the University of Cartagena and the Hospital Infantil Napoleón Franco Pareja for facilitating access to information.
Conflict of interest
The authors declare that they have no conflicts of interest with respect to this research.
Financing
This study was financed by the office of the Vice-Rector for Research of the University of Cartagena in the plan of strengthening and sustainability of the Biochemistry and Disease Group and was recognized by Colciencias.
REFERENCES
1. Frauca E, Muñoz G. Colestasis en el lactante. En: Asociación Española de Pediatría (AEP). Protocolos de gastroenterología, hepatología y nutrición. 2.a edición. España: Ergón S.A.; 2010. p. 177-87. [ Links ]
2. Samyn M, Mieli-Vergani G. Liver and biliary disease in infancy. Medicine 2007;35(2):61-6. [ Links ]
3. Bellomo-Brandao MA, Porta G, Hessel G. Clinical and laboratory evaluation of 101 patients with intrahepatic neonatal cholestasis. Arq Gastroenterol 2008;45(2):152-5. [ Links ]
4. Heller S. Colestasis neonatal. MedUNAB 2005;8(1):S18-25. [ Links ]
5. Wagner M, Zollner G, Trauner M. New molecular insights into the mechanisms of cholestasis. J Hepatol 2009;51(3):565-80. [ Links ]
6. Trauner M, Meier PJ, Boyer JL. Molecular pathogenesis of cholestasis. N Engl J Med. 1998 Oct 22;339(17):1217-27. [ Links ]
7. McKiernan PJ. Neonatal cholestasis. Semin Neonatol 2002;7(2):153-65. [ Links ]
8. De Bruyne R, Van Biervliet S, Vande Velde S, Van Winckel M. Clinical practice: neonatal cholestasis. Eur J Pediatr 2011;170(3):279-84. [ Links ]
9. Hondal Alvarez N, Silverio García C. Comportamiento de la colestasis del recién nacido y del lactante en el Hospital Pediátrico "William Soler". Rev Cub Pediatr 2010;82:13-9. [ Links ]
10. Ciocca M, Alvarez F. Transient neonatal cholestasis. Arch Argent Pediatr 2011;109(2):163-6. [ Links ]
11. Lorente S, Montoro MA. Cholestasis of pregnancy. Gastroenterol Hepatol 2007;30(9):541-7. [ Links ]
12. Girard M, Lacaille F. Diagnosis of neonatal cholestasis. Annales Nestlé (English ed). 2008;66(3):109-20. [ Links ]
13. López Panqueva R. Aproximación al diagnóstico patológico de las enfermedades colestásicas. Rev Col Gastroenterol 2014;29:189-99. [ Links ]
14. Balistreri WF. Neonatal cholestasis: lessons from the past, issues for the future. Semin Liver Dis 1987;7(2):61-6. [ Links ]
15. Fischler B, Papadogiannakis N, Nemeth A. Aetiological factors in neonatal cholestasis. Acta Paediatr 2001;90(1):88-92. [ Links ]
16. Venigalla S, Gourley GR. Neonatal cholestasis. Semin Perinatol 2004;28(5):348-55. [ Links ]
17. Balistreri WF. Neonatal cholestasis. J Pediatr 1985;106(2):171-84. [ Links ]
18. Donat Aliaga E, Polo Miquel B, Ribes-Koninckx C. Atresia de vías biliares. An Pediatr (Barc) 2003;79(02):168-73. [ Links ]
19. Balistreri WF, Heubi JE, Suchy FJ. Immaturity of the enterohepatic circulation in early life: factors predisposing to "physiologic" maldigestion and cholestasis. J Pediatr Gastroenterol Nutr 1983;2(2):346-54. [ Links ]
20. Hierro L, Jara P. Childhood cholestasis and bile transporters. Gastroenterol Hepatol 2005;28(7):388-95. [ Links ]
21. López-Manzanares J, Medina Benítez E. Colestasis en el recién nacido y lactante. Orientación diagnóstica. An Pediatr 2003;58(02):162-7. [ Links ]
22. Moyer V, Freese DK, Whitington PF, Olson AD, Brewer F, Colletti RB, et al. Guideline for the evaluation of cholestatic jaundice in infants: recommendations of the North American Society for Pediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr 2004;39(2):115-28. [ Links ]
23. Suchy FJ. Neonatal cholestasis. Pediatr Rev 2004;25(11):388-96. [ Links ]
24. Jacquemin E, Lykavieris P, Chaoui N, Hadchouel M, Bernard O. Transient neonatal cholestasis: origin and outcome. J Pediatr 1998;133(4):563-7. [ Links ]
25. Méndez V, Burastero M, Cuadro MN, Casamayou L, GutiéRrez C, Montano A, et al. Colestasis neonatal: evolución de los pacientes diagnosticados entre 1983 y 2000 en dos servicios gastroenterológicos de referencia de Montevideo. Arch Pediat Urug 2005;76:210-21. [ Links ]
26. Mowat AP, Davidson LL, Dick MC. Earlier identification of biliary atresia and hepatobiliary disease: selective screening in the third week of life. Arch Dis Child 1995;72(1):90-2. [ Links ]
27. Roberts EA. Neonatal hepatitis syndrome. Semin Neonatol 2003;8(5):357-74. [ Links ]
28. Vidal Duran C, Ureche Aguarán G. Hepatitis A colestásica. A propósito de un caso. Rev Col Gastroenterol 2006;21:225-9. [ Links ]
29. Glassman MS, Dellalzedah S, Beneck D, Seashore JH. Coincidence of congenital toxoplasmosis and biliary atresia in an infant. J Pediatr Gastroenterol Nutr 1991;13(3):298-300. [ Links ]
30. Robino L, Machado K, Montano A. Neonatal cholestasis due to congenital toxoplasmosis: Case report. Arch Argent Pediatr 2013;111(4):e105-8. [ Links ]
31. Tavares LA, Bittar D, Teixeira S. Relato de caso de colestase neonatal ocasionada por Toxoplasma gondii. HU Revista, Juiz de Fora 2009;35(4):324-7. [ Links ]
32. Donat Aliaga E, Polo Miquel B, Vila Carbó JJ, Sanguesa Nebot C, García-Sala Viguer C, Hernández Martí M, et al. Atresia de vías biliares: estudio clínico retrospectivo. An Pediatr (Barc) 2004;79(02):323-9. [ Links ]
33. Sepulveda Hincapie ME, Yepes Palacio NL, Gutierrez Mendoza FA. Hepatopatía crónica en niños. Justificación para un programa de transplante hepático infantil. Iatreia 2002;15(1):35-42. [ Links ]
34. Rosenthal P, Podesta L, Sher L, Makowka L. Liver transplantation in children. Am J Gastroenterol 1994;89(4):480-92. [ Links ]











 text in
text in