Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista colombiana de Gastroenterología
Print version ISSN 0120-9957
Rev Col Gastroenterol vol.31 no.2 Bogotá Apr./June 2016
A Structured Review of Approaches for Establishing and Evaluating Clinical Relevance of Drug Interactions in Patients with Hepatitis C Virus Genotype 1
Daniel Pino-Marín (1,2), Newar Giraldo PhD (1,2), Pedro Amariles PhD. (1,2)
(1) Pharmaceutical promotion and prevention group at the University of Antioquia in Medellín, Colombia.
(2) Corresponding author. Department of Pharmacy of the University of Antioquia in Medellín, Colombia. Mail: pedro.amariles@udea.edu.co
Received: 06-02-15 Accepted: 18-04-16
Abstract
Objective: Our objective was to establish and evaluate the clinical relevance of drug interactions in the treatment of patients with hepatitis C genotype 1.
Method: We searched for articles published in English and Spanish from December 2004 to December 2014 in PubMed/MedLine. We used the following Medical Subject Headings (MESH): Hepatitis C and drug interactions OR herb-drug interactions OR food-drug interactions studies performed in humans. We conducted an additional complementary search for articles published in the same period about interactions of anti-retroviral and hepatitis C in humans using the following MESH: (Anti-retroviral agents AND Hepatitis C and drug interactions OR herb-drug interactions OR food -drug interactions). The clinical relevance of drug interactions was defined and evaluated based on the probability of occurrence and severity of interaction.
Results: We identified 228 articles. Of these, it was possible to read the full text of 212. Of these, 62 contributed interactions which allowed us to identify 128 pairs of drug interactions, of which 120 (93.7%) were pharmacokinetic and 8 (6.3%) pharmacodynamic. Of these 128 pairs, two (1.6%) were rated Level 1: 110 (53.7%) were Level 2, 16 (7.8%) were Level 3, and 0 (0%) were Level 4. In addition, 78 pairs were identified that were grouped as interactions with evidence of absence of clinical significance.
Conclusions: More than 90% of clinically relevant drug interactions are pharmacokinetic interactions associated with hepatic metabolism. Telaprevir has the greatest number of interactions.
Keywords
Drug Interactions, Antiretrovirals, Hepatitis C, boceprevir, telaprevir.
INTRODUCTION
The hepatitis C virus (HCV) has a minimum of six genotypes and about 100 different strains whose prevalences and responses to treatment vary geographically. Of these variants, genotype 1 is responsible for most infections in the Americas and Europe. (1) Sustained viral response (SVR), defined as the absence of any detectable HCV RNA load after 24 weeks of treatment, was used to assess the effectiveness of antiviral treatment. (2) In the past, treatment of HCV genotype was based on the combination of pegylated interferon alpha (INF) and ribavirin (RIB) which was able to reduce SVR to a range of 40% to 50%. More recently, new drugs have come into use, especially important are the protease inhibitors (PIs) such as telaprevir (TLV) and boceprevir (BOC) which achieve responses of 60% to 75% in naive patients. In addition, treatment time has been reduced from 48 weeks to a range of 24 to 28 weeks and in some cases to even 12 weeks depending on the type of polymerase or protease inhibitor used and depending on the behavior of SVR. (1)
What has happened to treatment of HCV infected patients as the result of the use of these drugs can be compared to what happened with highly active antiretroviral therapy (HAART) for HIV patients. (1) As is known, HAART has led to a significant decrease in mortality and morbidity, but it has also been associated with the occurrence of adverse reactions, adhesion problems and drug interactions (DI).
HCV coinfections are common in HIV-infected patients, especially because of the similar modes of transmission: sexual, parenteral and vertical. (3) In addition, the existence of an HCV infection conditions the early treatment of HIV since HIV worsens and accelerates progression of hepatitis. HIV infections cause increases of two to 8 times the HCV viral load and leads to progression of the infection and the appearance of cirrhosis. (4, 5) Antiviral treatment for HCV in patients with HIV is a situation that is be common in clinical practice and which may be associated with DI related to the pharmacological treatment of the two morbidities. These interactions occur in part because of the ability of antiretrovirals to induce or inhibit hepatic metabolism and because of pharmacodynamic interactions that generate or enhance liver damage. (6) This study's objective is to deepen our understanding of how common and clinically relevant these drug interactions are in HCV patients through a structured review, based on the severity and the probability of interactions.
METHOD
A search of articles published in English and Spanish in PubMed/MedLine from December 1, 2004 until December 31, 2014 was conducted using the MESH terms: Hepatitis C and drug interactions OR herb-drug interactions OR food- drug interactions. Because of the possibility of coinfections of HCV and HIV and because in recent years some antiretroviral agents have been used for treatment of hepatitis C (e.g. ritonavir has been used to extend or enhancer some PIs), the search was supplemented with a review of articles in the same period of time about interactions of antiretroviral drugs (ARD) in humans and hepatitis C, using the following MESH terms: anti-retroviral agents AND hepatitis C and drug interactions OR herb-drug interactions OR food-drug interactions.
Inclusion criteria
Only systematic reviews, metaanalyses, multicenter studies, randomized controlled trials, quasi-experimental studies (nonrandomized), observational studies, guidelines, letters and case reports that were made in humans and in either the Spanish or English language and for which access to full text was available were selected for review. Articles about DI between medications used in the treatment of hepatitis C with other drugs were included. Some of the references used in those articles were also included in order to broaden the context or increase support for results.
Exclusion criteria
Publications of in vitro studies, animal models, experimental drugs and those which did not address interactions with drugs for the treatment of hepatitis C were excluded.
Review methods
Items included were independently selected by three researchers. They reviewed the titles and abstracts of all papers identified to determine their eligibility. The articles selected were jointly analyzed by the group which used consensus to determine whether or not an article would be included.
Measurement of results and evaluation of clinical relevance of interactions
The clinical relevance of DI was defined and evaluated using probability of occurrence and severity of interaction. (7, 8) On the basis of the type of study that documented and on whether or not the interaction had been published in indexed, peer-reviewed journals, the probability of interaction was divided into three categories: defined, probable and possible.
- Defined: interaction documented in metaanalyses, systematic reviews, randomized clinical trials or nonrandomized clinical trials.
- Probable: interaction documented in analytical studies or through description of three or more clinical cases.
- Possible: interactions documented by description of less than three cases or expert recommendations.
Severity of interactions was assigned to three additional categories:
- Severe: Interaction may cause damage or injury to the patient. The consequence of negative clinical outcome of drug therapy can cause or generate a patient's death, result in a life-threatening situation or hospitalization, lead to permanent or significant disability, cause congenital anomalies or malformations at birth, or result in other effects that, in the judgment of physicians, may compromise the integrity of the patient and require surgery to prevent death, hospitalization or congenital abnormalities.
- Moderate: Interaction creates the need to track the patient. The consequence of negative clinical outcome of drug therapy can cause a change or interruption in pharmacotherapy or require the use of new drugs to treat the problem related to drugs or can prolong hospitalization.
- Mild: Interaction does not cause harm to the patient. The consequence of the negative result of medication does not require modification, change or suspension of pharmacotherapy or the use of new drugs to treat the problem related to drugs or prolonged hospitalization.
From the possible combinations of severity and probability of occurrence, interactions can be grouped into four categories:
- Level 1 (very high risk): Includes combinations of severe and defined, and severe and probable. The simultaneous use of these drugs is considered to be absolutely contraindicated.
- Level 2 (high risk): Includes combinations of severe and possible, moderate and definite, and moderate and probable. The simultaneous use of these drugs requires adjustment of the dosing regimen, and assessment of signs and symptoms of effectiveness and safety of treatment. Ideally, the evaluation should be quantitative.
- Level 3 (medium risk): Includes combinations of moderate and possible, mild and definite, mild and probable. The simultaneous use of these drugs requires adjustment of the dosing regimen, and assessment of signs and symptoms of effectiveness and safety of treatment. Ideally, the evaluation should be quantitative.
- Level 4 (low risk): Combination of mild and possible categories. The interaction has little clinical relevance.
In addition, a list of pairs of medications for which there is no evidence of clinically relevant interactions was developed.
Data collection form
A form for recording and tabulating data on drug interactions was designed in Excel 2010 for Windows. The form had the following structure:
- Pharmacological group of drug used concomitantly with medication for treatment of HCV
- Kind of interaction (drug-drug, drug- phytotherapeutic agent, drug-food)
- Pair of interacting agents
- Level, severity and probability of interaction
- Bibliography
- Mechanism of interaction (pharmacokinetic or pharmacodynamic)
- Details of mechanism of interaction
- Comments
- Recommendation.
RESULTS
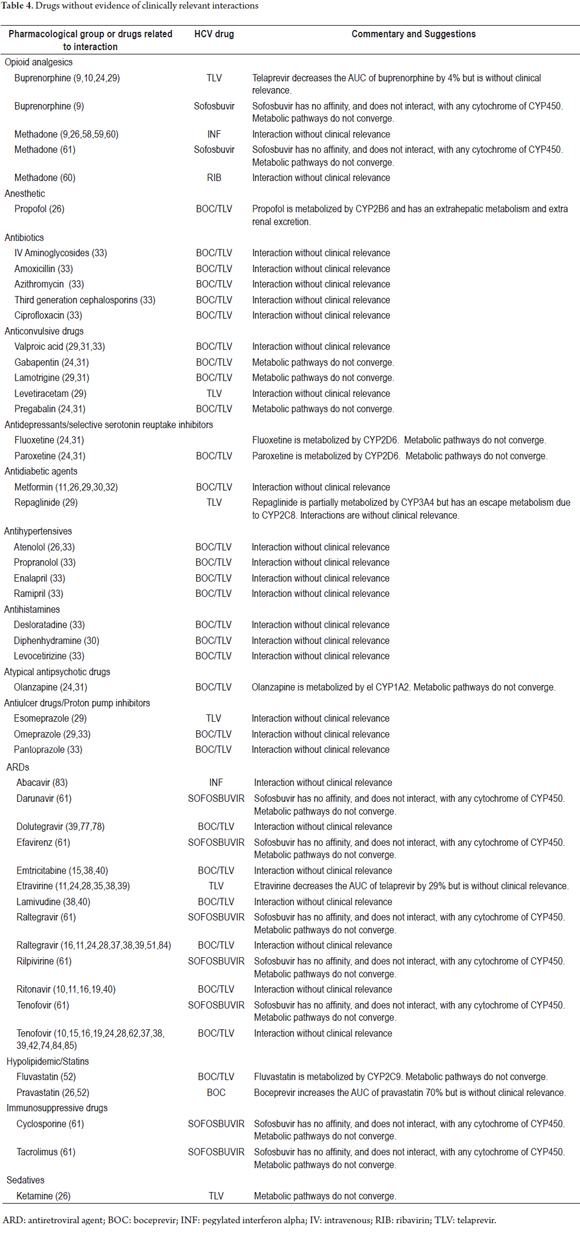
The search terms "Hepatitis C AND drug interactions OR herb-drug interactions OR food-drug interactions" yielded 193 articles, of which the full text was available for 178. Of these, 56 reported DIs. Thirty-five articles were identified with the second set of search terms. Of these, the full text was available for 34. Six of these reported HCV treatment-related DIs. In total, 228 articles were identified. The full text was available for 212 and of these 62 articles met the inclusion criteria (Figure 1). Two articles (1.6%) were Level 1, 110 articles (85.9%) were Level 2 and 16 articles (12.5%) were level 3 (Tables 2 and 3). There were 206 DI pairs of which 128 were determined to be clinically significant (Table 1). Of these 128 pairs, 120 have pharmacokinetic interaction mechanisms. These mechanisms included enzyme inhibition in 82 pairs (64.0%), enzyme induction in 35 pairs (27.3%) and change in bioavailability in three pairs (2.4%). Eight pairs had pharmacodynamic interaction mechanisms. Seventy-eight drug pairs for which there was no evidence of clinically relevant interactions were identified. Of these pairs, thirty-six included telaprevir, thirty included boceprevir, nine included sofosbuvir, one included ribavirin and two included interferon (Table 4).
Of the 8 pairs of pharmacodynamic interactions, three were due to antagonisms between ribavirin and nucleoside analog reverse-transcriptase inhibitors (NRTIs: abacavir, didanosine and stavudine) resulting from nuclear phosphorylation and associated with increased mitochondrial toxicity and especially fatal lactic acidosis. (63.71, 86-89) One was due to synergism of the antiviral effect of acyclovir and ribavirin. (21) Four were synergisms of adverse effects between zidovudine and TLV, BOC, RIB or INF which were associated with increased probability of complications related to anemia and hematological toxicity. (63-68, 70, 71- 73,75, 86, 88 , 89)
DISCUSSION
HCV is a chronic condition that can be associated with co-infections with HIV and other diseases. Coinfections may lead to therapeutic need to jointly use ARD drugs for HCV and other comorbidities, a situation that increases the probability of clinically relevant drug interactions. This structured review of interactions specific to treatment of HCV was complemented by a search of interactions between antiretrovirals and drugs for treating HCV. The review identified a total of 128 pairs of clinically relevant drug interactions: two were Level 1 (1.6 %); 110 were Level 2 (85.9%); sixteen were Level 3 (12.5%) and there were no Level 4 interactions (0.0%). The assessment of clinical relevance was based on a model proposed by the authors based on probability of occurrence and severity of interaction. (7) This method is one of the strengths of this study when it is compared to other similar studies. (1) In addition, 78 other pairs of drugs were found which had no evidence of clinically relevant interactions.
This reviews findings were similar to those of other structured reviews about interactions in patients infected with HIV and dyslipidemia. (8, 80) Clinically relevant DIs are those with pharmacokinetic mechanisms, especially inhibition or enzyme induction. This indicates that physicians in clinical practice need to evaluate concomitant drug therapy in cases in which drugs that have the ability to affect CYP450 enzyme complex are used.
In general, a CYP450 inducer generates increased enzyme activity and a resulting decrease in plasma levels of substrates. An inhibitor decreases the activity of the enzyme and thereby increases plasma concentrations of substrates. However, boceprevir and telaprevir do not always behave in this manner. One example was found in a study conducted among healthy volunteers. Rather than increasing their levels as would be expected because of the b inhibiting effect of ritonavir on CYP3A4, TLV levels decreased when it was administered with ritonavir-boosted protease inhibitors. (81, 82) The importance of structured reviews like this one which aim at identifying articles with results from human studies surpasses that of theoretical projections of drug interactions based on similarities or differences of metabolic pathways of related drugs that may possibly interact.
Antiarrhythmic drugs, immunosuppressants, statins and ergot derivatives are the drugs that most commonly interact. They account for 26 of the 128 pairs of relevant drug interactions. This is due to the ability of protease inhibitors, in this case boceprevir and telaprevir, to inhibit CYP3A4 and P-glycoprotein (TLV). Pegylated interferon alpha and ribavirin have lower frequencies of pharmacokinetic interactions because they are primarily eliminated by the kidneys. D. Back et al. have established that pharmacological groups such as diuretics, angiotensin converting enzyme inhibitors and angiotensin II receptor blockers are not significantly metabolized by CYP3A4. For this reason, it would be expected that they would not generate drug interactions with telaprevir. (29) This hypothesis is supported by the results of our review. However, absence of interactions were only evident with enalapril and ramipril. Back et al. also established that β-blockers have problems when used with PIs because some of them such as atenolol and sotalol are primarily eliminated by the kidneys and others such as metoprolol and carvedilol are metabolised by CYP2D6. (29) This generally coincides with the absence of clinically relevant interactions of telaprevir and boceprevir with atenolol identified in our study, but it departs from the finding of increased plasma levels of sotalol when combined with TLV. This effect may be because sotalol is a substrate for P-glycoprotein and this glycoprotein inhibits TLV. (76)
Our review has documented information about the absence of interactions of clinical relevance for sofosbuvir (61), a polymerase inhibitor recently approved by the Food and Drug Administration and available in the market. This drug is apparently not a substrate, inhibitor and inducer of some isoenzymes of CYP450 which limits the possibility of relevant interactions with drugs such as tacrolimus, cyclosporine, rilpivirine and efavirenz. This could be clinically beneficial for patients with HCV and other comorbidities. The main limitation of this study is that the search was restricted to the databases of PubMed/Medline. Nevertheless, this limitation was minimized by the complementary search for references to DIs in HIV-infected patients on antiretroviral therapy.
CONCLUSIONS
More than 90% of clinically relevant drug interactions in patients infected with HCV who receive drug therapy are pharmacokinetic. They are associated with induction or inhibition of hepatic metabolism. When antiviral drugs such as boceprevir and telaprevir are used to treat patients infected with HCV and other associated diseases, clinically relevant interactions are likely to occur. The largest number of drug interaction pairs identified include telaprevir. There were sixty-three pairs including telaprevir in total: two in Level 1 (1.6%), 56 in Level 2 (43.7%), and five in Level 3 (3.9 %). There were fifty-four pairs that included boceprevir: 44 in Level 2 (34.4%) and 10 in Level 3 (7.8%). Clearly, HCV patients who have other associated diseases and who are being treated with three drugs may have altered plasma concentrations of concomitant medications. This is more probable in cases in which TLV or BOC are used concomitantly with antiarrhythmic drugs, immunosuppressants, statins and ergot derivatives. When INF, RIB or sofosbuvir are used, the probability of relevant interactions is lower. This is primarily due to the fact that CYP450 isoenzymes are not involved in their elimination. This feature enhances their choice for the treatment of HCV in patients who have other associated diseases, especially HIV-infected patients and patients who have solid organ transplants.
Source of financial support
The Pharmaceutical Promotion and Prevention Group received funding from the 2014-2015 sustainability call of the Committee for Development of Research (CODI) of the University of Antioquia in Medellin, Colombia.
REFERENCES
1. Izquierdo-García E, Escobar-Rodríguez I. Systematic review of new protease inhibitors interactions: telaprevir and boceprevir. Farm Hosp. 2012;36:469-82. [ Links ]
2. Morillo Verdugo R, Romero Gómez M. A new therapeutic scene in the treatment of the chronic hepatitis for virus C. Farm Hosp. 2012;36:466-8. [ Links ]
3. Mallolas Masferrer J, Martínez-Rebollar M, Laguno Centeno M. Treatment of hepatitis C virus in HIV-positive patients. Gastroenterol Hepatol. 2011;34:558-67 [ Links ]
4. Benhamou Y, Bochet M, Di Martino V, Charlotte F, Azria F, Coutellier A, Vidaud M, Bricaire F, Opolon P, Katlama C, Poynard T. Liver fibrosis progression in human immunodeficiency virus and hepatitis C virus coinfected patients. The Multivirc Group. Hepatology. 1999;30:1054-8. [ Links ]
5. Valencia ME, Moreno V. Coinfection with HIV and HCV: abacavir and ribavirine, why not?. Rev Clin Esp. 2012;212:26-30. [ Links ]
6. Soriano V, Martin-Carbonero L, Vispo E, Labarga P, Barreiro P. Human immunodeficiency virus infection and viral hepatitis. Enferm Infecc Microbiol Clin. 2011;29:691-701. [ Links ]
7. Amariles P, Giraldo NA, Faus MJ. Interacciones Medicamentosas: aproximación para Establecer y evaluar su relevancia clínica. Med Clin Barc 2007; 129:27-35. [ Links ]
8. Giraldo NA, Amariles P, Gutiérrez FJ, Monsalbe M, Faus MJ. Aproximación para establecer y evaluar la relevancia clínica de las interacciones medicamentosas en pacientes infectados con virus de la inmunodeficiencia humana: actualización 2009. Farm Hosp 2010;34:90-93 [ Links ]
9. Bruce RD, Moody DE, Altice FL, Gourevitch MN, Friedland GH. A review of pharmacological interactions between HIV or hepatitis C virus medications and opioid agonist therapy: implications and management for clinical practice. Expert Rev Clin Pharmacol. 2013;6:249-69. [ Links ]
10. Seden K, Back D. Directly acting antivirals for hepatitis C and antiretrovirals: potential for drug-drug interactions. Curr Opin HIV AIDS. 2011;6:514-26. [ Links ]
11. Rodríguez-Torres M. Challenges in the treatment of chronic hepatitis C in the HIV/HCV-coinfected patient. Expert Rev Anti Infect Ther. 2012;10:1117-28. [ Links ]
12. Varun Garg, Gurudatt Chandorkar, Yijun Yang, Nathalie Adda,et al :The effect of CYP3A inhibitors and inducers on the pharmacokinetics of telaprevir in healthy volunteers, Br J Clin Pharmacol 2012;75:431–39 [ Links ]
13. Hulskotte EG, Feng HP, Xuan F, van Zutven MG, Treitel MA, Hughes EA, O'Mara E, Youngberg SP, Wagner JA, Butterton JR. Pharmacokinetic interactions between the hepatitis C virus protease inhibitor boceprevir and ritonavir-boosted HIV-1 protease inhibitors atazanavir, darunavir, and lopinavir. Clin Infect Dis. 2013;56:718-26. [ Links ]
14. Ceballos ME. Considerations in first line antiretroviral selection for adults. Rev Chilena Infectol. 2013;30:522-37. [ Links ]
15. Kiser JJ, Burton JR, Anderson PL, Everson GT. Review and management of drug interactions with boceprevir and telaprevir. Hepatology. 2012;55:1620-8. [ Links ]
16. Sulkowski MS. HCV therapy in HIV-infected patients. Liver Int. 2013;33 Suppl 1:63-7. [ Links ]
17. Schwarze-Zander C, Rockstroh JK. HIV protease inhibitors in combination with boceprevir: are drug-drug interactions the same for all patients? AIDS. 201210;26:1845-6. [ Links ]
18. Tseng A, Foisy M, Hughes CA, Kelly D, Chan S, Dayneka N, et al. Role of the Pharmacist in Caring for Patients with HIV/AIDS: Clinical Practice Guidelines. Can J Hosp Pharm. 2012;65:125-45. [ Links ]
19. Rodríguez-Torres M. Focus on drug interactions: the challenge of treating hepatitis C virus infection with direct-acting antiviral drugs in the HIV-positive patient. Curr Opin Infect Dis. 2013;26:50-7. [ Links ]
20. Deeks D. E. Darunavir: A Review of Its Use in the Management of HIV-1 Infection, Drugs (2014) 74:99–125 [ Links ]
21. Lauren J Gleason, Amneris E Luque, Krupa Shah; Polypharmacy in the HIV-infected older adult population Clinical Interventions in Aging 2013:8 749–763 [ Links ]
22. M. Rabanal Tornero; Terapia de sustitución con metadona: interacciones farmacológicas y no farmacológicas; Butlletí d'informació terapéutica. Generalitat de Catalunya, Departament de salut, Vol 24, num 6. 2013. [ Links ]
23. Vanpouille C, Lisco A, Introini A, Grivel JC, Munawwar A, Merbah M, et al.Exploiting the anti-HIV-1 activity of acyclovir: suppression of primary and drug-resistant HIV isolates and potentiation of the activity by ribavirin. Antimicrob Agents Chemother. 2012;56:2604-11. [ Links ]
24. Mauss S, Klinker H. Drug-drug interactions in the treatment of HCV among people who inject drugs. Clin Infect Dis. 2013;57 Suppl 2:125-8. [ Links ]
25. Ingiliz P, Rockstroh JK. HIV-HCV co-infection facing HCV protease inhibitor licensing: implications for clinicians. Liver Int. 2012;32:1194-9. [ Links ]
26. Burger D, Back D, Buggisch P, Buti M, Craxí A, Foster G, et al. Clinical management of drug-drug interactions in HCV therapy: challenges and solutions. J Hepatol. 2013;58:792-800. [ Links ]
27. Teixeira R, Nascimento Yde A, Crespo D. Safety aspects of protease inhibitors for chronic hepatitis C: adverse events and drug-to-drug interactions. Braz J Infect Dis. 2013;17:194-204. [ Links ]
28. Kiser JJ, Burton JR Jr, Everson GT. Drug-drug interactions during antiviral therapy for chronic hepatitis C. Nat Rev Gastroenterol Hepatol. 2013;10:596-606. [ Links ]
29. Berenguer Berenguer J, González-García J. Pharmacokinetic interactions of telaprevir with other drugs. Enferm Infecc Microbiol Clin. 2013;31 Suppl 3:37-48. [ Links ]
30. Back D, Else L. The importance of drug-drug interactions in the DAA era. Dig Liver Dis. 2013;30:45 Suppl 5:S343-8. [ Links ]
31. Sockalingam S, Tseng A, Giguere P, Wong D. Psychiatric treatment considerations with direct acting antivirals in hepatitis C. BMC Gastroenterol. 201314;13:86. [ Links ]
32. Barritt AS 4th, Fried MW. Maximizing opportunities and avoiding mistakes in triple therapy for hepatitis C virus. Gastroenterology. 2012;142:1314-1323. [ Links ]
33. Kozielewicz D, Halota W, Dybowska D. Preparation of HCV infected patients to the triple therapy with first generation protease inhibitors. Przegl Epidemiol. 2013;67:623-8,713-6. [ Links ]
34. Lamers MH, Broekman MM, Boucher CA, Brouwer JT, Burger DM, van Hoek B, Hoepelman AI, de Knegt RJ, Reesink HW, Drenth JP et al. Treatment of hepatitis C monoinfection in adults--Dutch national guidelines. Neth J Med. 2013;71:377-85. [ Links ]
35. Sulkowski MS. HCV therapy in HIV-infected patients. Liver Int. 2013;33 Suppl 1:63-7. [ Links ]
36. Jennings CL, Sherman KE. Hepatitis C and HIV co-infection: new drugs in practice and in the pipeline. Curr HIV/AIDS Rep. 2012;9(3):231-7. [ Links ]
37. Luetkemeyer AF, Havlir DV, Currier JS. Complications of HIV disease and antiretroviral therapy. Top Antivir Med. 2012;20:48-60. [ Links ]
38. Puoti M, Rossotti R, Travi G, Panzeri C, Morreale M, Chiari E, et al. Optimizing treatment in HIV/HCV coinfection. Dig Liver Dis. 2013;45 Suppl 5:S355-62. [ Links ]
39. Karageorgopoulos DE, El-Sherif O, Bhagani S, Khoo SH. Drug interactions between antiretrovirals and new or emerging direct-acting antivirals in HIV/hepatitis C virus coinfection. Curr Opin Infect Dis. 2014;27:36-45. [ Links ]
40. Vachon ML, Dieterich DT. The HIV/HCV-coinfected patient and new treatment options. Clin Liver Dis. 2011;15:585-96. [ Links ]
41. Wilby KJ, Partovi N, Ford JA, Greanya E, Yoshida EM. Review of boceprevir and telaprevir for the treatment of chronic hepatitis C. Can J Gastroenterol. 2012;26:205-10. [ Links ]
42. Bruno R, Fagiuoli S, Sacchi P. Will direct-acting antivirals make a difference in HIV-HCV coinfected patients? Expert Rev Anti Infect Ther. 2011;9:699-701. [ Links ]
43. Sulkowski MS. Hepatitis C virus-human immunodeficiency virus coinfection. Liver Int. 2012;32 Suppl 1:129-34. [ Links ]
44. Kim JJ, Culley CM, Mohammad RA. Telaprevir: an oral protease inhibitor for hepatitis C virus infection. Am J Health Syst Pharm. 20121;69(1):19-33. [ Links ]
45. Schaefer M, Capuron L, Friebe A, Diez-Quevedo C, Robaeys G, Neri S, Foster GR, Kautz A, Forton D, Pariante CM. Hepatitis C infection, antiviral treatment and mental health: a European expert consensus statement. J Hepatol. 2012;57:1379-90. [ Links ]
46. Chaparro M, Trapero-Marugán M, Moreno-Otero R, Gisbert JP. Azathioprine plus ribavirin treatment and pancytopenia. Aliment Pharmacol Ther. 2009;30:962-3. [ Links ]
47. Sockalingam S, Tseng A, Giguere P, Wong D. Psychiatric treatment considerations with direct acting antivirals in hepatitis C. BMC Gastroenterol. 2013;14;13-86. [ Links ]
48. Gutierrez-Valencia A, Ruiz-Valderas R, Torres-Cornejo A, Viciana P, Espinosa N, Castillo-Ferrando JR, Lopez-Cortes LF. Role of ritonavir in the drug interactions between telaprevir and ritonavir-boosted atazanavir. Clin Infect Dis. 2014;58:268-73. [ Links ]
49. O'Leary JG, McKenna GJ, Klintmalm GB, Davis GL. Effect of telaprevir on the pharmacokinetics of sirolimus in liver transplant recipients. Liver Transpl. 2013;19:463-5. [ Links ]
50. Lê MP, Gervais A, Le Beller C, Long K, Larrouy L, Papy E, Mal H, et al. Serious neuropsychiatric adverse effects in a hepatitis C virus/hepatitis B virus/HIV-coinfected patient receiving bosentan and telaprevir. J Antimicrob Chemother. 2013;68:1208-9. [ Links ]
51. Luetkemeyer AF, Havlir DV, Currier JS. CROI 2013: Complications of HIV disease, viral hepatitis, and antiretroviral therapy. Top Antivir Med. 2013;21:62-74. [ Links ]
52. Chauvin B, Drouot S, Barrail-Tran A, Taburet AM. Drug-drug interactions between HMG-CoA reductase inhibitors (statins) and antiviral protease inhibitors. Clin Pharmacokinet. 2013;52:815-31. [ Links ]
53. Charlton M. Telaprevir, boceprevir, cytochrome P450 and immunosuppressive agents--a potentially lethal cocktail. Hepatology. 2011;54:3-5. [ Links ]
54. Terrault N. Liver transplantation in the setting of chronic HCV. Best Pract Res Clin Gastroenterol. 2012;26:531-48. [ Links ]
55. Werner CR, Egetemeyr DP, Lauer UM, Nadalin S, Königsrainer A, Malek NP, Berg CP. Telaprevir-based triple therapy in liver transplant patients with hepatitis C virus: a 12-week pilot study providing safety and efficacy data. Liver Transpl. 2012;18:1464-70. [ Links ]
56. Garg V, van Heeswijk R, Lee JE, Alves K, Nadkarni P, Luo X. Effect of telaprevir on the pharmacokinetics of cyclosporine and tacrolimus. Hepatology. 2011;54:20-7. [ Links ]
57. Hulskotte E, Gupta S, Xuan F, van Zutven M, O'Mara E, Feng HP, Wagner J, Butterton J. Pharmacokinetic interaction between the hepatitis C virus protease inhibitor boceprevir and cyclosporine and tacrolimus in healthy volunteers. Hepatology. 2012;56:1622-30. [ Links ]
58. Gupta SK, Sellers E, Somoza E, Angles L, Kolz K, Cutler DL. The effect of multiple doses of peginterferon alfa-2b on the steady-state pharmacokinetics of methadone in patients with chronic hepatitis C undergoing methadone maintenance therapy. J Clin Pharmacol. 2007;47:604-12. [ Links ]
59. Berk SI, Litwin AH, Arnsten JH, Du E, Soloway I, Gourevitch MN. Effects of pegylated interferon alfa-2b on the pharmacokinetic and pharmacodynamic properties of methadone: a prospective, nonrandomized, crossover study in patients coinfected with hepatitis C and HIV receiving methadone maintenance treatment. Clin Ther. 2007;29:131-8. [ Links ]
60. Neuman MG, Monteiro M, Rehm J. Drug interactions between psychoactive substances and antiretroviral therapy in individuals infected with human immunodeficiency and hepatitis viruses. Subst Use Misuse. 2006;41:1395-463. [ Links ]
61. Rodríguez-Torres M. Sofosbuvir (GS-7977), a pan-genotype, direct-acting antiviral for hepatitis C virus infection. Expert Rev Anti Infect Ther. 2013;11:1269-79. [ Links ]
62. Fowell AJ, Nash KL. Telaprevir: a new hope in the treatment of chronic hepatitis C? Adv Ther. 2010;27(8):512-22. [ Links ]
63. Seden K, Back D, Khoo S. New directly acting antivirals for hepatitis C: potential for interaction with antiretrovirals. J Antimicrob Chemother. 2010;65(6):1079-85. [ Links ]
64. Soriano V, Puoti M, Sulkowski M, Cargnel A, Benhamou Y, Peters M, Mauss S, Bräu N, Hatzakis A, Pol S, Rockstroh J. Care of patients coinfected with HIV and hepatitis C virus: 2007 updated recommendations from the HCV-HIV International Panel. AIDS. 2007;21:1073-89. [ Links ]
65. Lai AR, Tashima KT, Taylor LE. Antiretroviral medication considerations for individuals coinfected with HIV and hepatitis C virus. AIDS Patient Care STDS. 2006;20:678-92. [ Links ]
66. Nelson M, Lipman M. Management of advanced HIV disease in patients with tuberculosis or hepatitis co-infection. Int J Clin Pract. 2006;60:976-83. [ Links ]
67. Soriano V, Nuñez M, Garcia-Samaniego J, Labarga P, Simarro N, Martín-Carbonero L, Romero M, Ramos B, Barreiro P. Chronic hepatitis C in HIV-infected patients: those who more need therapy are those who respond less. Infect Disord Drug Targets. 2006;6:57-88. [ Links ]
68. Park JS, Saraf N, Dieterich DT. HBV plus HCV, HCV plus HIV, HBV plus HIV. Curr Gastroenterol Rep. 2006;8:67-74. [ Links ]
69. Sherman KE. Treatment of hepatitis C virus and human immunodeficiency virus co-infection. Clin Gastroenterol Hepatol. 2005;3:S118-21. [ Links ]
70. Mallolas Masferrer J, Martínez-Rebollar M, Laguno Centeno M. Treatment of hepatitis C virus in HIV-positive patients. Gastroenterol Hepatol. 2011;34:558-67. [ Links ]
71. Miró JM, Torre-Cisnero J, Moreno A, Tuset M, Quereda C, Laguno M, et al. GESIDA/GESITRA-SEIMC, PNS and ONT consensus document on solid organ transplant (SOT) in HIV-infected patients in Spain (March, 2005). Enferm Infecc Microbiol Clin. 2005;23:353-62. [ Links ]
72. Nogueira JB, Sena LC, Quintans Jde S, Almeida JR, França AV, Júnior LJ. Side effects of the therapy with peginterferon and ribavirin in chronic hepatitis C: a small audit. J Pharm Pract. 2012;25:85-8. [ Links ]
73. Simón-Talero M, Buti M, Esteban R. Severe anaemia related to oseltamivir during treatment of chronic hepatitis C: a new drug interaction? J Viral Hepat. 2012;19:14-7. [ Links ]
74. Ramanathan S, Cheng A, Mittan A, Ebrahimi R, Kearney BP. Absence of clinically relevant pharmacokinetic interaction between ribavirin and tenofovir in healthy subjects. J Clin Pharmacol. 2006;46:559-66. [ Links ]
75. Matthews GV, Dore GJ. HIV and hepatitis C coinfection. J Gastroenterol Hepatol. 2008;23:1000-8. [ Links ]
76. Bachmakov I, Werner U, Endress B, Auge D, Fromm MF. Characterization of beta-adrenoceptor antagonists as substrates and inhibitors of the drug transporter P-glycoprotein. Fundam Clin Pharmacol. 2006;20:273-82. [ Links ]
77. Cottrell ML, Hadzic T, Kashuba AD. Clinical pharmacokinetic, pharmacodynamic and drug-interaction profile of the integrase inhibitor dolutegravir. Clin Pharmacokinet. 2013;52:981-94 [ Links ]
78. Shah BM, Schafer JJ, Desimone JA Jr. Dolutegravir: a new integrase strand transfer inhibitor for the treatment of HIV. Pharmacotherapy. 2014;34:506-20. [ Links ]
79. González-Colominas E, Solà R, Barrantes-González M, Salas E. Prevalence of potential interactions between antiretroviral therapy and boceprevir or telaprevir in human immunodeficiency virus-hepatitis C virus coinfected patients. Enferm Infecc Microbiol Clin. 2013;31:350-1. [ Links ]
80. Franco D, Henao Y, Monsalve M, Gutiérrez F, Hincapie J, Amariles P. Hypolipidemic agents drug interactions: approach to establish and assess its clinical significance. Structured review. Farm Hosp. 2013;37:539-57. [ Links ]
81. Kasserra C, Hughes E, Treitel M, et al. Clinical pharmacology of boceprevir: metabolism, excretion, and drug-drug interactions abstract 118. 18th Conference on Retroviruses and Opportunistic Infections, Feb 27-Mar 2, 2011, Boston, USA. [ Links ]
82. Garg V, Luo X, McNair L, et al. Low-dose ritonavir and the pharmacokinetics of the investigational HCV protease inhibitor telaprevir in healthy volunteers abstract 629. 18th Conference on Retroviruses and Opportunistic Infections, Feb 27-Mar 2, 2011, Boston, USA. [ Links ]
83. Solas C, Pambrun E, Winnock M, Salmon D, Poizot-Martin I, Dominguez S, Bani-Sadr F, Izopet J, Garraffo R, Peytavin G; ANRS CO-13 HEPAVIH Study Group. Ribavirin and abacavir drug interaction in HIV-HCV coinfected patients: fact or fiction? AIDS. 2012;26:2193-9. [ Links ]
84. Naggie S, Sulkowski MS. Management of patients coinfected with HCV and HIV: a close look at the role for direct-acting antivirals. Gastroenterology. 2012;142:1324-1334.e3 [ Links ]
85. Wilby KJ, Greanya ED, Ford JA, Yoshida EM, Partovi N. A review of drug interactions with boceprevir and telaprevir: implications for HIV and transplant patients. Ann Hepatol. 2012;11:179-85. [ Links ]
86. Tuma P, Vispo E, Barreiro P, Soriano V. Role of tenofovir in HIV and hepatitis C virus coinfection. Enferm Infecc Microbiol Clin. 2008;26 Suppl 8:31-7. [ Links ]
87. Sherman KE. New paradigms in the management of hepatitis C virus co-infections. Nat Clin Pract Gastroenterol Hepatol. 2007;4 Suppl 1:S10-6. [ Links ]
88. Ruiz-Sancho A, Soriano V. HIV and HCV coinfection. Enferm Infecc Microbiol Clin. 2006;24:335-45; quiz 346. [ Links ]
89. Dieterich DT. Hepatitis C virus and human immunodeficiency virus: clinical issues in coinfection. Am J Med. 199927;107:79S-84S. [ Links ]











 text in
text in